Abstract
Due to occupational asbestosis exposure, the incidence of malignant pleural mesothelioma (MPM) has continuously increased over the last 30 years, with a plateau anticipated around the year 2030 in Western countries. Molecular MPM carcinogenesis involves alterations of NF2, RASSF1, LATS2WT1, p16, as well as BAP-1tumor-suppressor genes, which usually regulate apoptosis, cell invasion, motility, cell division, chromatin remodeling, as well as control of DNA repair. In few selected patients, debulking surgery consisting of pleurectomy-decortication is carried out, resulting in unsatisfactory long-term results. For about 15 years, first-line chemotherapy has been primarily based on a doublet of pemetrexed and cisplatin. Adding the monoclonal antibody bevacizumab (Avastin®), which targets vascular endothelial growth factor (VEGF), has been shown to improve overall survival (OS) by nearly 19 months. The emergence of immune check-point inhibitors (ICIs) in MPM treatment has recently been associated with substantial survival improvements in both second- and first-line settings. Similarly to non-small-cell lung cancer (NSCLC) patients, on-going trials are presently exploring the chemotherapy-ICI combination in MPM management, and depending on their results, this combination could represent a further major advance in this previously orphan disease. The current article reviews recent clinical trial results, as well as future clinical developments in this moving field.
Introduction
In 2016, the randomized phase-3 trial ‘MAPS’ assessing pemetrexed–cisplatin–bevacizumab triplet in malignant pleural mesothelioma (MPM) demonstrated the superiority of such triplet over cisplatin and pemetrexed doublet.
1
However, median overall survival (OS) of the experimental arm was only 18.8 months, emphasizing the need of new therapeutic standards. Immune system targeting drugs then emerged as potentially interesting for MPM treatment.
2
Owing to chronic inflammation due to asbestosis fiber deposits in pleural space or deep lung, the immune system has been suspected to play a major role in the MPM pathogenesis, which is still imperfectly understood. Indeed, improved outcomes were reported to correlate with higher intra-tumor infiltration by cytotoxic CD8+ T-cells.
3
Conversely, high tumor expression of programmed cell death-ligand 1 (PD-L1, aka B7-homolog 1, B7-H1), which inhibits T-cell function
Biological background
Clinical ICI efficacy has been claimed to correlate with high tumor mutational burden, as in the case of melanoma or NSCLC patients, whereas mesothelioma was consistently reported to harbor low mutation numbers per megabase of genomic DNA. 5 However, instead of the mutational burden by itself, it is probably rather the type of mutated genes that enhances the mutational burden and plays a major role. We know very little about which genes could drive ICIs’ potential efficacy in MPM. Yet, it may be assumed that p16 (somatic mutations, deletions, or gene promoter methylation in 22% of MPM samples6,7) and BAP1(somatic mutation in 23% of MPM samples 5 ) could possibly drive such effect, given that they both regulate cell cycle arrest, DNA repair, or chromatin remodeling. Hippo gene pathway alterations (RASSF1A promoter gene methylation in 30% of MPM samples,8,9 NF2 truncation mutations in 19% of MPM cases, 5 MST1/hippo promoter gene methylation in 8.5% 10 of MPM cases, or LATS2 mutations in 11% or MPM)5,11 could influence anti-tumor immune responses by governing YAP transcriptional co-activator activity state. YAP actually controls transcription of multiple immune genes including cytokine CXLC5 able to attract CXRC2-expressing myeloid-derived suppressor cells (MDSCs), 12 while cross-talks between Hippo/YAP pathway and TGF-β or JAK-STAT pathways that are involved in immune response regulation have similarly been described. 13 However, based on the rich inflammatory stromal component of these tumors, especially in the sarcomatoid or biphasic subtypes, the common view of a so-called ‘hot’ tumor has emerged, with tumor stromal infiltration by mono-macrophage cells, T-lymphocytes, or even neutrophils.
Several retrospective series paved the way for our knowledge on PD-L1 mesothelioma cell expression. However, the use of different antibodies and immune-staining automats yielded contradictory results. Almost all pharmaceutical companies that have developed therapeutic anti-PD-1 or anti PD-L1 antibodies have also developed their own associated diagnosis assay, rendering cross-comparisons rather difficult. Indeed, Roche promoted the SP142 clone for Ventana autostainer pertaining to atezolizumab trials, providing tumor staining of lower intensity than the other antibodies, yet able to stain PD-L1 expressed by infiltrating immune cells. To develop pembrolizumab, Merck used the 22 C3 clone from Dako, along with Dako autostainer. Bristol Meyers Squibb (BMS) used either the SP263 clone from Roche-Ventana or 28.8 clone from Dako in the nivolumab trials, while Astra-Zeneca used only the SP263 clone. All these antibodies have been extensively compared in cross-comparison studies based on NSCLC samples.14–16 To summarize these studies, the only antibody with diverging quantitative and qualitative results is the SP142, whereas all other antibodies provide rather concordant results. For mesothelioma tissue samples, such systematic comparison has not yet been published, with tumor PD-L1 staining being overall weaker than in NSCLC samples. Nevertheless, the generalized use of SP263 and E1L3N antibodies for routine practice, whatever the histological tumor type, has actually smoothened the discrepancies among laboratories and studies, 16 along with protocols adapted to the different staining automats.
Mansfield
In the MAPS Phase-3 study,
1
E1L3N clone was employed in diagnostic specimens from 214 patients with remaining available tissue that were accrued. In this study, with a cutoff set at 1% of membranous staining regardless of intensity, only 36% of patients turned out to be positive. Likewise, there was a significantly higher rate of positivity in sarcomatoid or biphasic tumors (68% of positive specimens
Raffit Hassan’s group studied PD-L1 expression on tumor cells and infiltrating lymphocytes using samples from 65 patients with malignant effusions from pleural and peritoneal mesotheliomas.
20
The authors found 41 (63%) patients to be PD-L1 positive based on a 5% cutoff for positivity, yet they provided no details on the antibody used. These patients were reported to exhibit a worse OS (median OS: 23.0 months
Finally, a series of primary diffuse pleural mesotheliomas including the epithelioid (
Second-line trials using single-therapy with anti-CTLA-4 monoclonal antibodies
CTLA-4 was the first checkpoint targeted by clinical trials involving MPM patients (Table 1). First encouraging results were reported in patients with chemotherapy-resistant advanced malignant mesothelioma. Tremelimumab was employed in these trials. The open-label, single-arm, phase-2 trial (MESOTTREM- 2008) 24 assessed this selective human immunoglobulin IgG-2 monoclonal antibody directed against CTLA-4 in MPM patients. Performance status (PS) 0–2 patients with measurable lesions, received second-line tremelimumab 15 mg/kg intravenously once every 90 days until progressive disease or severe toxicity. The trial’s primary endpoint was overall response rate (ORR), aiming a 17% target ORR, yet without a central independent response assessment. Most patients (25/29, 86%) received a standard first-line treatment with platinum combined with pemetrexed. Overall, 23 (79%) of 29 patients had documented disease progression within 6 months of first-line platinum-based chemotherapy, and 15 (68%) of them progressed during chemotherapy. Thus, one-fifth of these patients (21%) displayed indolent disease, progressing beyond 6 months of platinum-based first-line treatment. Given that only two therapeutic responses were observed, this trial should have been considered negative. In seven patients, more stable disease was observed, resulting in a 31% of disease control rate (DCR). The trial’s authors interpreted these data as encouraging, in spite of recruiting patients with possibly slowly growing tumors. Median duration of disease control was estimated at 12.4 months. Notably, the two responder patients displayed long-lasting response over 6 months, which grabbed the investigators’ attention. Median progression-free survival (PFS) of the study’s whole cohort was 6.2 months, median OS was 10.7 months, with a 2-year survival rate of 37%. However, no data on subsequent therapies were reported that could have helped interpret the OS data. Tremelimumab’s safety profile was found to be favorable, with only 14% of patients exhibiting grade 3–4 toxicities.
Clinical trials assessing anti-CTLA-4 monoclonal antibody single-therapy in mesothelioma patients.
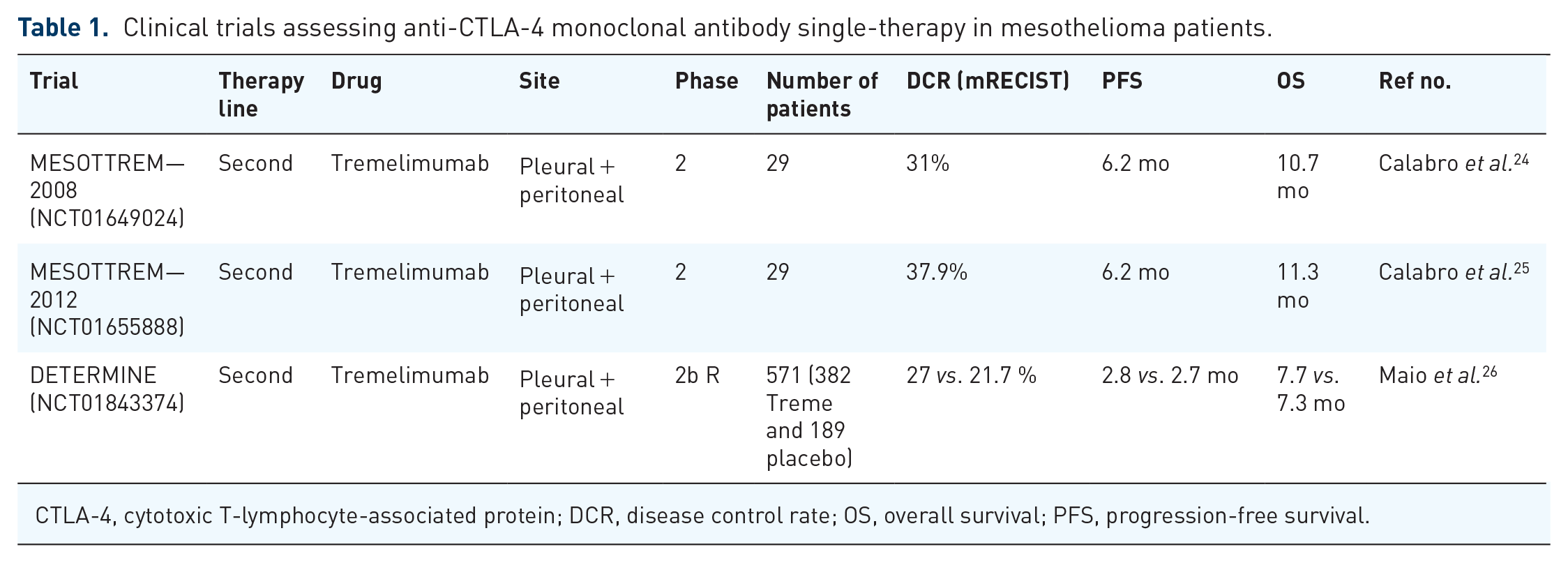
CTLA-4, cytotoxic T-lymphocyte-associated protein; DCR, disease control rate; OS, overall survival; PFS, progression-free survival.
The same investigators reported a second academic open-label, single-arm, phase-2 study (MESOTTREM- 2012), 25 using an ‘intensified’ tremelimumab schedule, at 10 mg/kg every 4 weeks for six doses, then every 12 weeks thereafter, until progression or toxicity. The primary endpoint was ORR based on iRECIST with a 17% ORR target rate. In this trial, patients with progressive disease at the first tumor assessment continued to receive tremelimumab if they did not suffer from deleterious clinical signs or progression symptoms. A pre-specified interim safety analysis did not detect any clinically relevant toxicity in the first 11 patients treated. Four patients displayed an immune-related partial response, including one at the first tumor assessment (12 weeks) and three others at the second tumor assessment (24 weeks). This resulted in only 13.8% ORR based on iRECIST, not clearly indicating significant activity. Even more puzzling, when the classical mRECIST for mesothelioma was applied, only one partial response was found. Using iRECIST, the proportion of patients with disease control was 51.7% (15 of 29 patients), with a consistent 10.9-month median duration of disease control. However, again, DCR was only 37.9% when applying mRECIST. Median immune-related PFS was 6.2 months, median OS was 11.3 months, and 1-year survival was 48.3%, all values being strictly comparable with the previous trial. Nevertheless, the seven patients with biphasic or sarcomatoid malignant mesotheliomas displayed a median OS of 15.8 months, which compared favorably with that observed in the seminal pemetrexed registration trial. Again, the safety profile was favorable, without any toxic death reported, and only a few and expected Grade 3–4 treatment-related adverse events (TRAEs).
Based on these data, tremelimumab was tested alone
Second-line trials using single therapy with anti-PD-1 or PD-L1 monoclonal antibodies
As different anti-PD-L1 commercially available antibodies were used in the different studies listed below, the Tables 2 and 3 present the monoclonal antibody (mAb) clones employed to either select or stratify patients, with such anti-PD-L1 staining appearing more versatile in MPM than in NSCLC samples, with subsequently a debatable predictive impact.
Clinical trials assessing anti-PD-1 or PD-L1 monoclonal single antibody therapy in mesothelioma patients.

DCR, disease control rate; MPM, malignant pleural mesothelioma; NA, not available; OS, overall survival; PD-1, programmed cell death-1; PD-L1, programmed cell death-ligand 1; PFS, progression-free survival.
Keynote-158 is a large basket trial in which 118 MPM patients were enrolled.
Companion diagnosis assays for PD-L1 IHC in anti-PD-1 single-drug second-line trials.

IHC, immunohistochemistry; PD-1, programmed cell death-1; PD-L1, programmed cell death-ligand 1; UNK, unknown.
The first data originated from a Phase-Ib large multicenter, non-randomized, open-label, and multi-cohort ‘basket’ trial (Keynote 028), with an MPM stratum including 25 patients with PD-L1-expressing MPM; these patients were treated with the anti-PD-1 antibody pembrolizumab from Merck, at 10 mg/kg intravenously every 2 weeks. 27 To be included into the study, patients had to be PS 0-1, have measurable disease and histological MPM diagnosis, have failed to respond to standard therapy, or be considered ‘unable to receive chemotherapy’. PD-L1 expression was assessed using the 22C3 antibody (Merck, Kenilworth, NJ, USA) assay, with a cutoff set at ⩾1% of tumor cells or associated inflammatory cells upon using membranous staining, regardless of intensity. Among 83 patients with evaluable histological specimen, median age was 65.0 years, two-thirds were men, 72% displayed epithelioid histology, and one-third had received at least two previous therapy lines, with two patients entirely naive of any treatment. About 84% had previously been treated with pemetrexed and 88% been exposed to a platinum salts. Primary endpoint was an ORR exceeding 10% at an overall one-sided 8% alpha-level. Overall, 25 patients were eventually accrued, receiving at least one pembrolizumab dose. Safety was considered manageable, with classical immune-related adverse events (IRAEs) in 12% of patients, with a dose reduction required in only one patient. Moreover, Grade-3 AEs were recorded in only 20% of patients, and there was no Grade 4 or 5 at all.
The results turned out to be encouraging, reflected by 20% ORR, with 52% of patients exhibiting stable disease. Even more convincing was the durability of responses, given that the median duration of response (DOR) was 12 months (95% CI: 3·7-not reached (NR)), while the durability of stable disease (5.6 months) was in accordance with the duration observed in the DETERMINE placebo arm. Median OS was 18 months, with 62.6% of patients being alive at 1 year and two patients having received the maximal treatment length of 24 months, whereas two other responders were still under treatment at 22 months. Even more strikingly, four patients exhibited ⩾70% decrease in their tumor burden, clearly supporting drug activity, which had not be reported in previous anti-CTLA-4 trials.
Final results from a U.S. Phase-2 single-arm trial assessing the activity of fixed-dose pembrolizumab (200 mg every 3 weeks) in second-line setting (KEYNOTE-139, NCT02399371) confirmed both the activity and tolerability of such anti-PD-1 agent in pre-treated MPM patients.
28
Overall, 35 MPM patients, PS = 0–1, with disease progression after 1–2 prior regimens, one of which including pemetrexed-platinum doublet, and measurable disease and were treated using pembrolizumab 200 mg intravenously q21 days with first computer tomography (CT)-scan evaluation at 9 weeks. Their median age was 68 years, while 53% were PS = 0; epithelioid histology was revealed in 77%, 12% had peritoneal while 61% of patients had received only one prior treatment. Partial response according to mRECIST was observed in 14 patients (22%), while 26 displayed stable disease (56%), providing a 63% DCR at 9 weeks. Toxicity was estimated manageable using a centrally assessed 22C3 antibody-based immunohistochemistry assay, PDL1 expression was available for 62 patients: 45% were found negative (<1%) and 32% displayed low PD-L1 expression (1%–49%), whereas 23% presented with high expression (⩾50%). When used as a continuous marker, higher PD-L1 expression was associated with a higher response rate (ROC area under the curve = 0.69; 95% CI: 0.53–0.84). In patients with high PD-L1 expression, ORR was higher and PFS longer compared with those comprising no or low PD-L1 expression (0.021 and 0.034, respectively). Nevertheless, OS was not influenced by PD-L1 tumor content. Overall, 1-year PFS was 40.2% in high PD-L1 expression patients
Keynote-158 trial was another large basket, pembrolizumab second- or further-line trial, which accrued 118 MPM patients, with results published in 2021. 35 In these heavily pretreated patients (52% of patients with ⩾ two previous lines), only 10 (8%) exhibited an objective response with a median duration of 14.3 months, while nearly two-thirds were still on-going at 12 months. Median OS was encouraging at 10.0 months (95% CI: 7.6–13.4), yet median PFS was only 2.1 months (2.1–3.9). In total, TRAEs occurred in 14 (12%) patients. Overall, 19 (16%) patients suffered from Grade 3–4 TRAEs, the most common of which were colitis reported by three patients and pneumonitis displayed by two.
Finally, a Swiss registry analysis involving 48 MPM patients, PS 0-2, having progressed after pemetrexed-based single-line treatment, who were then treated with pembrolizumab off-label, was presented at 2017 ESMO meeting.
29
An investigator-based response analysis according to mRECIST found a 25% ORR and 53% DCR (11% for PS 2 patients
Convinced by these preliminary data, the ETOP group launched the ETOP 9–14 PROMISE-Meso trial. This study sought to compare, following one previous chemotherapy line, pembrolizumab 200 mg fixed-dose intravenously, given at Day 1 of each 3-week cycle, until progressive disease by iRECIST, for maximally 2 years,
Another anti-PD-1 antibody, nivolumab (Nivo), was evaluated in second- or third-line settings in the NivoMes study, by a group from the Netherlands.
30
To be included in that trial, patients should have progressed after ⩾1 line therapy. Primary endpoint was DCR at 12 weeks, and the study sought to increase DCR from 20% to 40%. Patients received nivolumab 3 mg/kg, q2weeks, and were to undergo repeated biopsy and PBMC sampling at 6 weeks. Accrual was fast, with 34 patients, exhibiting 28 epithelioid and six sarcomatoid/biphasic types, enrolled in less than 1 year. Patients displayed standard epidemiological features, with a median age of 68 years, and 28 males. The trial was clearly positive, given that DCR was 29% at 6 months including two pseudo-progression cases, with an 23.5% ORR (
A Japanese multicenter trial, MERIT (ONO-4538-41/JapicCTI-No.163247), assessed nivolumab in second- or third-line settings, in 34 advanced or metastatic MPM patients, with resistance or intolerance to pemetrexed and platinum-based combination therapy.
31
Primary endpoint was OR, and nivolumab was used at 240 mg flat dose q2weeks. The expected response rate was 19.2%, giving an 80% power at 5% alpha-level. Median age was 68.0 years, 29.4% of patients received two previous treatment lines, 79.4% exhibited epithelioid MPM, and 61% were PS1. The trial was positive, with 29.4% ORR and 67.6% DCR at 6 months, without any unexpected safety concerns. With a median follow-up of 16.8 months, median PFS was 6.1 months and median OS 17.3 months. PD-L1 status, which was determined using the Dako PD-L1 IHC 28-8 pharmDx test, clearly influenced survival. Indeed, median PFS was 7.2 months in PD-L1 tumor expression >1% patients, compared with 2.9 months for PD-L1-negative patients; median OS was 17.3 months in PD-L1-positive patients
Results from the MPM strata of the multicenter non-randomized, open-label, and multicohort ‘basket’ Phase-Ib trial ‘JAVELIN’ (NCT01772004) were presented as a poster session at 2018 ASCO meeting.
32
This trial assessed the anti-PD-L1 fully humanized IgG1 avelumab in around 2,200 patients suffering from more than 15 cancer types. Overall, 53 patients exhibited unresectable pleural or peritoneal mesotheliomas, having progressed after platinum-pemetrexed combination therapy. These patients were then treated using avelumab, 10 mg/kg intravenously Q2W, until progression, unacceptable toxicity, or withdrawal. Patients had previously received one (
All these data supported the nivolumab
Trials using combination therapy consisting of anti-PD-1 or PD-L1 and anti-CTLA-4 monoclonal antibodies
As in NSCLC or melanoma, a putative synergy was expected between anti-CTA-4 antibodies, acting at priming phase by impairing dendritic and T-cells interaction, and anti-PD-1 antibodies, acting at the effector phase by impairing T-cell inactivation by tumor cells. With this background in mind, IFCT 1501 MAPS2 trial (NCT 02716272) was launched. This randomized, non-comparative Phase-2 trial assessed the usefulness of anti-PD-1 mAb nivolumab, either as single therapy or in combination with the anti-CTLA-4 mAb ipilimumab (ipi) from BMS, in second- or third-line settings in MPM patients (Table 4).
36
These were MPM patients, PS 0-1, with histologically confirmed pleural unresectable mesothelioma, and measurable, documented progression (all CT-scanners were centrally reviewed). The patients were randomized according to 1:1 ratio between nivolumab (nivo) 3 mg/kg, q2weeks and nivolumab 3 mg/kg, q2weeks plus ipilumumab (ipi) at 1 g/kg q6 weeks. They were treated until progression or unacceptable toxicity for up to 2 years. Patient accrual was impressively fast, resulting in 125 patients recruited within 5 months in 25 French centers. Overall, 63 patients were allocated to nivolumab and 62 to the combination. Median age was slightly higher than in previous studies (72.3 and 71.1 years, respectively), likely reflecting less patient selection. Overall, 84% patients exhibited epithelioid histology, two-thirds were PS = 1, with slightly more males in the combination arm (85
Clinical trials assessing combination therapy of anti-PD-1 or PD-L1 plus anti-CTLA-4 monoclonal antibody in mesothelioma patients.

DCR, disease control rate; IHC, immunohistochemistry; NA, not available; OS, overall survival; PD-1, programmed cell death-1; PDL1, programmed cell death-ligand 1; PFS, progression-free survival.
The trial met its primary endpoint in both arms, with 44.4% DCR in the first 54 eligible patients in the nivolumab arm,
Strikingly, waterfall plots of percentage change in tumor size from baseline at 12 weeks clearly confirmed a major activity in three and 10 patients exhibiting tumor shrinking of more than 60%, in the nivolumab and combination groups, respectively, irrespective of the histological subset. 40 Conversely, 12 and five patients, respectively, exhibited more than 60% of tumor size increase, with major and rapid tumor burden progression exceeding 80% observed in a few patients (six and two, respectively). This suggested some patients could have experienced ‘hyper-progression’ an aggressive pattern of progression, while MPM is a rather indolent disease, in line with observations by other authors in NSCLC settings.41–43
After a median 32.5-month follow-up, median response duration was 7.4 months in the nivolumab group
Another smaller, yet non-randomized trial (‘INITIATE’, NCT03048474) using the same design than the NIVOMES trial, carried-out by P. Baas
A third single-arm Phase-II trial (‘NIBIT-MESO-1’; NCT02588131) evaluated combined 1 mg/kg tremelimumab and 20 mg/kg durvalumab given in four intravenous doses every 4 weeks, which was followed by durvalumab maintenance at the same dose and schedule for nine doses, as first- or second-line treatment for unresectable malignant mesothelioma patients. 38 This trial met its primary endpoint, with 11/40 (27·5%) patients exhibiting immune-related (ir)-partial response (median DOS: 16.1 months) and 25/40 (65%) ir-disease control, resulting in a median ir-PFS of 8 months (median PFS = 5.7 months) and median OS of 16.6 months (95% CI: 13·1–20·1). In this specific trial, baseline tumor PD-L1 expression displayed no predictive or prognostic value.
Finally, immunotherapy-based combinations were recently completed by the BMS-sponsored large randomized Phase-III CheckMate CA209-743 trial. This trial assessed the benefit of nivolumab + ipi combination
Trials using combination therapy with anti-PD-1 or PD-L1 monoclonal antibodies and pemetrexed-platinum-based chemotherapy
The well-documented activity of immunotherapy in MPM patients in second-line and now even first-line settings led some investigators to assess the chemo-immunotherapy strategy, being inspired by the results of such combination in stage-IV non-small-cell cancer patients (Table 5).
Trials using combination therapy with anti-PD-1 or PD-L1 monoclonal antibodies and pemetrexed–platinum based chemotherapy.

DCR, disease control rate; IHC, immunohistochemistry; OS, overall survival; PD-1, programmed cell death-1; PDL1, programmed cell death-ligand 1; PFS, progression-free survival.
Two single-arm Phase-II trials assessed durvalumab, an anti-PD-L1 monoclonal antibody form Astra-Zeneca, combined with the cisplatin-pemetrexed doublet, in the US (PrE0505; NCT 02899195;
These results supported the launching of a Phase-3 trial titled ‘DREAM3R’’ (NCT04334759) in the first-line setting, with OS as primary endpoint, sponsored by PrECOG and the ALTG Australian group. This trial is designed to recruit 480 patients until 2024.
In the meantime, the results of the IND227-IFCT1901 (NCT02784171) Phase 2–3 trial, which was initiated by the Canadian Cancer Trials Group along with the French Intergroup (IFCT) will be made available. This trial seeks to explore the efficacy of a frontline therapy combining pemetrexed-cisplatin with pembrolizumab
Therefore, we should know very soon whether chemo-immunotherapy combination is as efficacious as double immunotherapy for MPM treatment. Should this be the case, this would represent a major advance in the care of this previously orphan disease, for which sensitivity to checkpoint inhibitors was put in the forefront.
Conclusion
MPM is a low mutational burden tumor, with only moderate PD-L1 expression levels in MPM tissue samples. Nevertheless, data are accumulating supporting the use of modern immunotherapy in MPM patients, using anti-PD-1 or anti-PD-L1 antibodies, in combination with (first-line setting) or without (second-/third-line settings) an anti-CTLA-4 antibody (yet not anti-CTLA-4 antibody single-therapy).
While available data actually support the adverse prognostic effect of PD-L1 expression in MPM, more and more data are accumulating, suggesting a favorable predictive effect of such PD-L1 tumor expression in MPM patients treated with anti-PD-1 or PD-L1 single-drug antibodies. However, this predictive effect is often obscured in MPM, as compared with NSCLC, by the lower and overall heterogeneous expression of PD-L1 by mesothelial tumor cells.
Phase-2 results with pembrolizumab or nivolumab-based therapy appear to be concordant when considering fit, not-refractory, and pre-treated MPM patients, given than more than 300 patients have now been treated in clinical trials, with remarkable PFS and long-term OS data. In the literature, similar data had never been reported with previously available cytotoxic drugs, which rarely resulted in more than 25% ORR and significant PFS data exceeding 3 months. In particular, MAPS2 Phase-2 randomized trial, though not comparative, could be considered as having generated sufficient data on the drugs’ tolerability and efficacy to justify nivolumab single-therapy, or nivolumab plus ipilimumab doublet, in second- or third-line settings in MPM patient, PS 0–1. With this reasoning in mind, which appears particularly sound in an orphan disease like MPM, NCCN guidelines have actually integrated this second-line therapeutic option, without waiting for putative Phase-3 results.
In first-line setting, positive results of the anti-PD-1 and anti-CTLA-4 antibody combination in the CheckMate-743 Phase-3 trial supported the superiority of i.o. combination over standard pemetrexed-platinum-based doublet, yet with lower impact of PD-L1 tumor expression. However, the control arm of this trial could be a matter of debate, and the triplet bevacizumab-pemetrexed-cisplatin should certainly have been chosen as control arm in patients eligible for bevacizumab or, alternatively, the nivolumab plus ipilimumab combination. Whether PD-L1 expression remains important for patient selection remains debatable, and clearly of less importance in double i.o combination, as also observed in NSCLC. More complex immune signature should be further explored as suggested in CheckMate-743 trial to better select patients deriving the most important effect from immunotherapy combinations.
Thus, a future challenge would be to assess a 4-drug combination with bevacizumab, anti-PD-1/PDL1 antibody, pemetrexed, and platinum, as such a combination has recently been proven efficient in NSCLC patients, and given that there is a biological rationale supporting the synergy anti-VEGF therapy and immuno-therapeutics. The BEAT-meso Phase-3 trial, sponsored by ETOP, has actually chosen such a design. This study is designed to compare atezolizumab plus bevacizumab and standard chemotherapy
Further clinical trial results are also eagerly awaited (Table 6), such as those assessing frontline chemo-immunotherapy regimens, and future years will clearly see a major improvement in the care of MPM patients, after years of stagnation and therapeutic failures.
On-going immunotherapy-based trials in pleural mesothelioma with pending results.

EPP, extrapleural pleuro-pneumonectomy; IFN, interferon; OS, overall survival; VEGF, vascular endothelial growth factor.
Footnotes
Author contributions
Valerie Gounant and Gérard Zalcman conceived the review of Literature, draw the first draft. All authors revuewed the final version
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GZ participated in advisory boards for Roche, Bristol-Myers-Squibb (BMS), Merck & Co, Boehringer Ingelheim, and Astra-Zeneca, and received reimbursements for attendance at 2016 and 2017 ASCO annual meetings from Roche;2018 ASCO annual meeting from Abbvie, 2017 ESMO annual meeting from Astra-Zeneca; 2016 WCLC meeting from BMS. He is the co-principal investigator of the MAPS2 trial with nivolumab and ipilimumab, sponsored by the French IntergReviewroup (IFCT), with the support of BMS. His research group received a research grant from Roche for funding a PhD student. VG received reimbursements for attendance at 2016 ASCO annual meeting from Roche. She is the co-principal investigator of the SAVIMMUNE Phase 2 trial with durvalumab, sponsored by the French Intergroup (IFCT), with the support of Astra-Zeneca. SB has no potential conflict of interest to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
