Abstract
Background:
Trastuzumab (Herceptin) can be administered intravenously (IV Herceptin) and subcutaneously, with similar efficacy and safety, but with differences in dosage and costs. Previous studies have evaluated the costs of both treatment approaches in the outpatient settings, but no study has compared the costs of IV Herceptin administered in outpatients with subcutaneous Herceptin administered at patients’ homes (Homecare SC Herceptin).
Objectives:
This study aimed to compare the per-patient costs of Homecare SC Herceptin versus IV Herceptin administered in a healthcare institution’s outpatient setting in Singapore.
Designs:
We performed a model-based cost-minimization analysis to estimate and compare the per-patient annual costs associated with each treatment modality from a societal perspective.
Methods:
Direct cost comprised healthcare resources utilization: drug, consumables, manpower, facility and cardiac assessment. Indirect cost was valued using a human capital approach to account for productivity lost by patients. Monte Carlo simulations with 1000 iterations were performed to account for parameter uncertainties. Costs were reported in 2023 Singapore dollars.
Results:
The annual societal cost per patient receiving IV Herceptin ranged from S$64,194 to S$65,135, while for Homecare SC Herceptin, it ranged from S$25,865 to S$26,807. Homecare SC Herceptin reduced the annual cost burden by 58.8% and 59.7%, per non-metastatic and metastatic breast cancer patient, respectively. The primary cost contributor was drug therapy, comprising more than 90% of the total cost. Even when excluding the cost of drugs, Homecare SC Herceptin remained cheaper by S$1912 annually. The cost reduction is approximately 60% compared to IV Herceptin regardless of disease status, with a 100% probability that the decision to adopt Homecare SC Herceptin leads to cost savings in Singapore.
Conclusion:
Treatment of breast cancer with Homecare SC Herceptin is a cost-saving option compared to IV Herceptin.
Keywords
Introduction
Breast cancer is the most frequently diagnosed cancer in females worldwide, with an estimated 2.3 million new cases in 2022. 1 The rising trend of breast cancer in Singapore through the decades culminated in its overtaking of colorectal cancer for the top spot in the incidence rankings for the period 2017–2021, becoming the most common cause of cancer death in women according to the Singapore Cancer Registry. 2 The human epidermal growth factor receptor 2 (HER2) 3 is over-expressed or over-amplified in 15%–20% of breast cancers. The advent of Trastuzumab (Herceptin®, Genentech, USA) revolutionized the treatment landscape of HER2+ breast cancer, changing its prognosis from one of the worst to one of the best. Trastuzumab has demonstrated high efficacy in both early and metastatic breast cancer (MBC), significantly prolonging survival by reducing the risk of disease recurrence and progression.4–6
Herceptin was approved by the Food and Drug Administration (FDA) 7 in 1998 as an intravenous (IV) infusion administered weekly or 3-weekly over 1 year for non-metastatic (early stage) patients, and indefinitely (till disease progression) for metastatic (advanced stage) patients. It is dosed according to the patient’s weight. Subsequently, subcutaneous (SC) Herceptin was approved by the FDA in 2019, 8 based on the findings of two large randomized trials, HannaH 9 and SafeHER, 10 which demonstrated comparable efficacy and safety between SC and IV formulations. SC Herceptin is administered 3-weekly as a fixed dose of 600 mg via syringe injection under the skin, thereby minimizing drug wastage and dosing mistakes. It appears advantageous over IV Herceptin as a time-saving option, given the shorter administration time, 2–5 11 versus 30–90 min. 12 Moreover, the long-term administration of IV Herceptin usually requires an indwelling IV catheter, presenting its risks (infection, blood clots) and financial costs (line insertion/removal, hospitalizations).
The National Cancer Centre Singapore (NCCS) established NCCS Home Care in July 2000. Gearing towards patient-centric care, NCCS Home Care was originally intended for elderly patients who were less mobile, requiring special transport arrangements (ambulance) and/or carer supervision during visits to NCCS. This was in tandem with a national movement to expand clinical services beyond the hospital to the community. During the COVID-19 pandemic, NCCS Home Care dovetailed neatly with measures to reduce people clustering at healthcare institutions, driving up its take-up rate. This initiative enabled selected cancer treatments to continue at home, thereby allowing the delivery of personalized care in the comfort and familiarity of the patient’s surroundings, enhancing his/her well-being and convenience. This approach potentially streamlines the patient’s treatment journey by reducing the burden of frequent hospital visits (travel expenses, long waiting times in high-volume healthcare institutions, alternative childcare expenses) and offering savings in opportunity costs, such as the patient’s and caregiver’s productivity loss by taking time off work to attend hospital appointments. There is a capacity gain for the healthcare institution in terms of space and infusion chair time to treat other patients, where their treatment can only be administered on the hospital premises.
Previously, multiple countries have explored how much money can be saved by switching from the IV to SC formulation. Despite differences in healthcare system structures and study designs, economic evaluations from Switzerland, 13 Spain, 14 Chile, 15 Malaysia 16 and Hong Kong 17 have consistently yielded a shared finding: adopting SC Herceptin is more cost-saving within the respective hospital contexts. However, none reported the benefits of adopting SC Herceptin treatment at the patient’s home. To date, there has been no similar economic evaluation done in Singapore.
In this study, our objective was to compare the per-patient costs of SC Herceptin administered at home (Homecare SC Herceptin) versus IV Herceptin administered in a healthcare institution’s outpatient setting in Singapore (NCCS).
Methods
Target population
The target population for this study was female patients from NCCS with early (non-metastatic) and advanced (metastatic) stages of breast cancer who were prescribed Herceptin treatment with curative and palliative intent, respectively. NCCS is the national specialty centre in Singapore which sees the majority of cancer cases in the public sector, amounting to over 150,000 patient visits every year. 18
Model overview, perspective and time horizon
The reporting of this study conforms to the CHEERS guidelines for economic evaluations (Appendix A). 19 A model-based economic evaluation was developed to estimate and compare the per-patient annual costs associated with Homecare SC Herceptin versus IV Herceptin treatment at NCCS outpatient setting, from a societal perspective. It considered costs incurred by health resources and productivity losses involved in both treatment modalities, incorporating data from cross-sectional surveys, clinical practice protocol, as well as expert opinions. The time horizon for the analysis is 1 year, as a complete regime of Herceptin consists of tri-weekly administration for 18 cycles. No discounting was applied due to the short time frame. As non-MBC and MBC patients received different treatment regimens, individual costs were modelled and stratified by treatment modality and disease status. In view of comparable efficacy and safety between SC and IV Herceptin in the HannaH 9 and MetaspHER 20 trials, using cost-minimization for decision-making is justified.
Measurement and valuation of resources and costs
Direct costs
To gauge direct costs, the NCCS Homecare programme utilized data collected through surveys administered to healthcare professionals and patients. Five major medical resource components were included: drug, consumables, manpower, facility and cardiac assessment. As the dosage of IV Herceptin is weight-dependent, retrospective data on the initial weight of 100 patients who started IV Herceptin treatment between 2018 and 2021 were extracted from a hospital database. We assumed that there are no meaningful weight changes to the patient during the treatment period, and no sharing of vials between patients is allowed once opened. Only major consumables involved in treatment were included: an IV infusion line set (IV Herceptin) and a syringe-and-needle set (Homecare SC Herceptin). Other consumables, such as disposable gauze bandages for hygienic application, were too minimal to consider in this analysis. A questionnaire was designed and conducted with 20 healthcare professionals to specifically gather information on the frequency and volume of health services: nurse time, pharmacist time, infusion chair time as well as cardiac assessment via two-dimensional echocardiogram (2D-Echo). Corresponding costs were computed by multiplying the volume used per year with its unit costs.
Indirect costs
Productivity losses due to absenteeism were considered as an indirect cost to the economy and were valued using the human capital approach. 21 Absenteeism refers to time lost from work due to treatment. A questionnaire was administered to 100 patients who received IV Herceptin and 104 patients who received Homecare SC Herceptin to capture information about time taken to travel to NCCS, time spent at NCCS as well as time duration receiving Homecare SC Herceptin. We assumed the patient was a working employee since the average age of patients enrolled in the survey (mean = 59.64 years old) was below 63 years, which is the minimum retirement age in Singapore. 22 Gender- and age-specific median gross monthly income was retrieved from the Ministry of Manpower Labour Force 2023 report. 23
Analytical approach
All parameters were assigned an appropriate distribution to allow for probabilistic sensitivity analysis, see Table 1 for the most important parameters. Monte Carlo simulations with 1000 iterations were performed to account for parameter uncertainties. The output distribution for the expected change to total costs was summarized by the mean, and a 95% uncertainty interval (UI) was reported. The probability of one option being cost-saving was reported as a percentage. The economic evaluation was performed using Microsoft Excel 2019 (Microsoft Corporate, Redmond, Washington, USA) with visual basic code used for simulation. Details for all parameters are reported in Appendix B, Table A2. All costs were reported in 2023 Singapore dollars.
Major model input parameters.

HCP, healthcare professionals; HR, human resource; IV, intravenous; MOM, Ministry of Manpower; NCCS, National Cancer Centre Singapore; CPF, Central Provident Fund; SC, subcutaneous.
Results
Base-case results
Table 2 reveals the annual healthcare resource utilization and absenteeism for IV Herceptin and Homecare SC Herceptin. Each year, each patient receiving IV Herceptin required an additional 705 min for infusion chair time and 340 min for pharmacist time in drug preparation. They also spent another 15.30 h travelling to NCCS, and an extra 31 h in NCCS for treatment. By contrast, the total nurse time required for Homecare SC Herceptin was longer by 579 min per patient (797 vs 218 min), involving more nurse activities: drug reconstitution, injection and travel between house visits. The model was further categorized by the patient’s disease status. The key difference between non-MBC and MBC is the frequency of 2D-Echo (4 vs 2 times per year). All these translate into relevant costs and are summarized in Tables 3 and 4 as the base case result of this analysis.
Healthcare resources utilization and absenteeism per year per patient.

2D-Echo, two-dimensional echocardiogram; IV, intravenous infusion; MBC, metastatic breast cancer; NA, not applicable; SC, Homecare subcutaneous administration.
Cost breakdown and distribution.
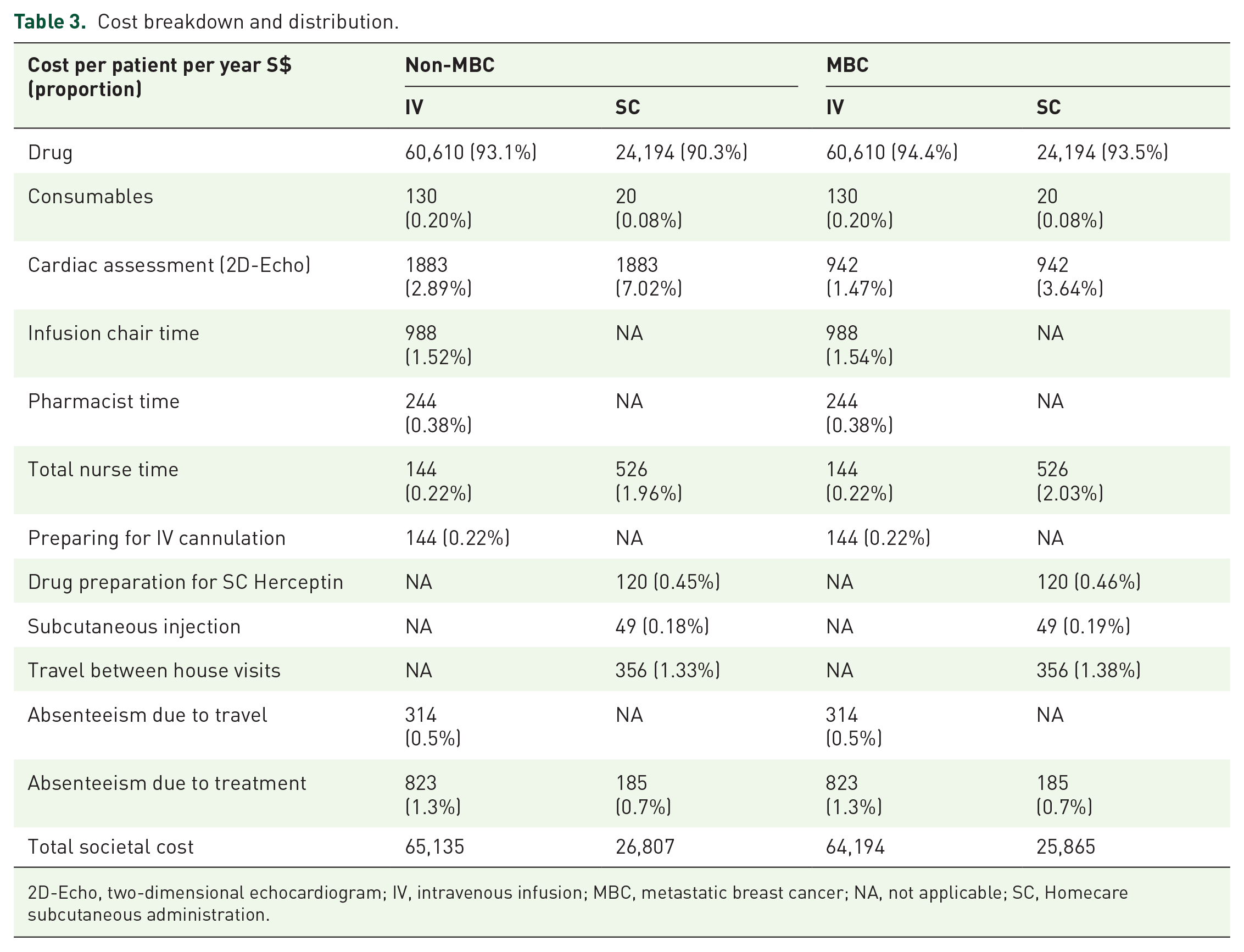
2D-Echo, two-dimensional echocardiogram; IV, intravenous infusion; MBC, metastatic breast cancer; NA, not applicable; SC, Homecare subcutaneous administration.
Costs per patient per year.

IV, intravenous infusion; MBC, metastatic breast cancer; NA, not applicable; SC, Homecare subcutaneous administration.
The detailed cost breakdown is illustrated in Table 3. The primary contributor to cost outcomes was the drugs, comprising more than 90% of the total cost. Next was infusion chair time and pharmacist time, with patients who received IV Herceptin estimated to cost an additional S$988 and S$244 every year. For nurse time, patients receiving Homecare SC Herceptin were expected to incur higher costs at S$526 per year, compared to S$144 for IV Herceptin. The cost of absenteeism in the workplace due to travelling to the cancer centre was estimated at S$314 per patient receiving IV Herceptin. Costs of absenteeism due to receiving treatment were estimated at S$823 and S$185 per patient receiving IV and Homecare SC Herceptin, respectively. In Table 4, we show the annual societal cost to be S$64,194–S$65,135 and S$25,865–S$26,807 per patient receiving IV and Homecare SC Herceptin, respectively. Homecare SC Herceptin reduced the annual cost burden by 58.8% and 59.7%, per non-MBC and MBC patient, respectively. Nevertheless, even excluding the cost of the drug, Homecare SC Herceptin remained cheaper by S$1912 annually, representing a cost reduction of 42.2% for non-MBC patients and 53.3% for MBC patients.
Probabilistic results
The outcomes of the Monte Carlo simulation are presented in Table 5. For non-MBC patients, the annual cost of Homecare SC Herceptin was S$27,013 (95% UI: S$26,997–S$27,028), while IV Herceptin incurred S$69,133 (95% UI: S$68,090–S$70,177) annually. Similarly, for one MBC patient, Homecare SC Herceptin costs S$26,067 (95% UI: S$26,057–S$26,077) per year, while IV Herceptin costs S$66,426 (95% UI: S$65,527–S$67,326) per year.
Results of probabilistic sensitivity analysis.

IV, intravenous infusion; MBC, metastatic breast cancer; NA, not applicable; SC, Homecare subcutaneous administration; UI, uncertainty interval.
In Figure 1, the boxplots illustrate that adopting Homecare SC Herceptin should reduce costs by S$42,121 (95% UI: S$41,077–S$43,165) per non-MBC patient and S$40,360 (95% UI: S$39,460–S$41,259) per MBC patient annually. Over 1000 iterations, the difference in costs could potentially exceed S$90,000 per year, as shown by outliers in Figure 1. This is particularly substantial for patients with higher body weight, given the weight-dependent nature of IV Herceptin administration, where no vial sharing is assumed.

Boxplots of expected changes to cost by adopting Homecare SC Herceptin.
On average, the cost reduction is approximately 60%, with a 100% probability that the decision to adopt Homecare SC Herceptin leads to cost savings from a societal perspective.
Discussion
Singapore is a small, densely populated country (population: 5.6 million; land area: 734 km 2 ) with highly developed infrastructure for roads and public transport. In the past 50 years, in tandem with the country’s economic development, the pattern of diseases in Singapore shifted from infectious diseases to chronic and non-communicable diseases, such as cancer and cardiovascular diseases. Correspondingly, Singapore’s disease burden changed. Measured by disability-adjusted life years (DALYs), the Global Burden of Disease 2019 study showed that cancer took the top spot, accounting for 15.5% of DALYs, alongside cardiovascular diseases (13.9%) and musculoskeletal disorders (14.4%). 24 It is of paramount importance that cancer patients receive standard-of-care treatment in a timely fashion, to reduce the risk of disease recurrence or progression, as this would naturally add to the nation’s disease burden if they were to occur.
Recognizing the resource-intensive nature of administering IV treatment in cancer centres, pharmaceutical companies have explored reducing drug administration durations by developing alternative formulations for SC injection. This simultaneously and conveniently provides greater flexibility in terms of the location where the drug can be administered, such as an outpatient procedure room, or the patient’s home, and not necessarily the outpatient cancer treatment facility. Trastuzumab (Herceptin), Trastuzumab + Pertuzumab (Phesgo®), Rituximab (MabThera®/Rituxan®) and Atezolizumab (Tecentriq®) are examples of molecularly targeted therapies that have corresponding SC formulations. Another consequence of adopting SC drug administration is off-loading the outpatient cancer treatment facility where IV drugs are administered, thereby increasing the healthcare institution’s capacity to administer treatment that can only be given on the premises. For a busy cancer centre with a heavy outpatient clinical load, this is an operational advantage. In NCCS, switching breast cancer patients from IV Herceptin to SC Herceptin initially meant diverting those on Herceptin monotherapy to the outpatient procedure room. However, the queue for the outpatient procedure room for SC Herceptin administration quickly reached capacity – the slack was taken up by NCCS Home Care. In fact, given that the outpatient procedure room handles a myriad of different procedures that can only be performed on the premises, the vast majority of patients on SC Herceptin have their treatment administered at home.
This study represents the first analysis of the economic value of Homecare SC Herceptin versus IV Herceptin in NCCS in the Singapore setting. Our findings highlight cost-savings for consumables, infusion chair time, pharmacist time and reduced absenteeism attributed to travelling or treatment time incurred. Increased costs associated with nursing time were observed, primarily due to additional travel time for nurses.
The substantial decrease in chair time in the chemotherapy unit is particularly valuable. This is important in a resource-limited setting such as the NCCS, where wait times for chemotherapy unit chairs can extend to 4 weeks. Changing to home administration of SC Herceptin allows for chemotherapy chairs to be allocated to patients in need of lengthier infusions for medications carrying a higher risk of hypersensitivity or acute toxicities.
Homecare services are patient-centric but do require additional costs for nurses to travel to patients’ homes. Although travel time was based on expert opinions, the proximity and ease of travel within a small densely populated country with high transport connectivity suggest these are likely reasonable. Moreover, as homecare expands to encompass more patients, the potential for optimizing and clustering services emerges, leading to a reduction in both travel time and nursing costs. Further data collection and analysis for service clustering allows for more efficiency gains.
The cost savings likely extend beyond travel and work absenteeism for patients. Often, individuals seeking cancer treatment are accompanied by caregivers or family members who must also navigate work commitments and logistical challenges. The actual cost-savings might surpass the estimates presented here. The results of this study may also be applied to other molecularly targeted agents that can be administered subcutaneously, broadening the benefits of Homecare SC drug administration.
Home-based treatments, such as SC Herceptin administration, offer patients the comfort of receiving care in familiar surroundings, particularly beneficial for those facing mobility challenges, such as the elderly or disabled. However, these intangible benefits are unable to be accounted for in a traditional economic evaluation study like this.
While our study provides valuable insights, it is essential to acknowledge its limitations, including the evolving landscape of treatment options. The availability of biosimilar IV trastuzumab has reduced cost. Considering the substantial cost disparities between conventional IV Herceptin and biosimilar trastuzumab, future investigations could focus on comparing the cost-effectiveness of Homecare SC Herceptin with IV biosimilar trastuzumab, offering a more comprehensive understanding of treatment dynamics and financial implications. In addition, we acknowledge that the overall costs may vary due to different adverse events, particularly at the individual level. However, due to a lack of data in this study to explore this aspect, future research could focus on collecting extensive data on adverse events and associated costs through a large prospective cohort study to investigate the economic impact.
Conclusion
In summary, treatment of HER2+ breast cancer with Homecare SC Herceptin proves to be a cost-saving option compared to IV Herceptin. The additional benefit of personalized care where patients can receive treatment in the comfort of their own homes makes this initiative a valuable addition to the list of services provided by NCCS.
Footnotes
Appendix A
CHEERS 2022 checklist.

| Item | Guidance for reporting | Reported in section | |
|---|---|---|---|
| Title | |||
| Title | 1 | Identify the study as an economic evaluation and specify the interventions being compared | Title |
| Abstract | |||
| Abstract | 2 | Provide a structured summary that highlights context, key methods, results and alternative analyses | Abstract |
| Introduction | |||
| Background and objectives | 3 | Give the context for the study, the study question and its practical relevance for decision-making in policy or practice | Introduction |
| Methods | |||
| Health economic analysis plan | 4 | Indicate whether a health economic analysis plan was developed and where available | Methods para 2 |
| Study population | 5 | Describe characteristics of the study population (such as age range, demographics, socioeconomic or clinical characteristics) | Methods para 1,4 |
| Setting and location | 6 | Provide relevant contextual information that may influence findings | Introduction para 3; Methods para 1 |
| Comparators | 7 | Describe the interventions or strategies being compared and why chosen | Introduction para 2,3, Methods 2 |
| Perspective | 8 | State the perspective(s) adopted by the study and why chosen | Methods para 2 |
| Time horizon | 9 | State the time horizon for the study and why appropriate | Methods para 2 |
| Discount rate | 10 | Report the discount rate(s) and reason chosen | Methods para 2 |
| Selection of outcomes | 11 | Describe what outcomes were used as the measure(s) of benefit(s) and harm(s) | Not applicable |
| Measurement of outcomes | 12 | Describe how outcomes used to capture benefit(s) and harm(s) were measured | Not applicable |
| Valuation of outcomes | 13 | Describe the population and methods used to measure and value outcomes | Not applicable |
| Measurement and valuation of resources and costs | 14 | Describe how costs were valued | Methods para 3,4; Table 1 |
| Currency, price date and conversion | 15 | Report the dates of the estimated resource quantities and unit costs, plus the currency and year of conversion | Methods para 5; Table 1 |
| Rationale and description of the model | 16 | If modelling is used, describe in detail and why used. Report if the model is publicly available and where it can be accessed | Methods para 2 |
| Analytics and assumptions | 17 | Describe any methods for analysing or statistically transforming data, any extrapolation methods, and approaches for validating any model used | Methods para 5 |
| Characterizing heterogeneity | 18 | Describe any methods used for estimating how the results of the study vary for sub-groups | Not applicable |
| Characterizing distributional effects | 19 | Describe how impacts are distributed across different individuals or adjustments made to reflect priority populations | Not applicable |
| Characterizing uncertainty | 20 | Describe methods to characterize any sources of uncertainty in the analysis | Methods para 5 |
| Approach to engagement with patients and others affected by the study | 21 | Describe any approaches to engage patients or service recipients, the general public, communities or stakeholders (e.g. clinicians or payers) in the design of the study | Not applicable |
| Results | |||
| Study parameters | 22 | Report all analytic inputs (e.g. values, ranges, references) including uncertainty or distributional assumptions | Table 1; Appendix Table A |
| Summary of main results | 23 | Report the mean values for the main categories of costs and outcomes of interest and summarise them in the most appropriate overall measure | Results para 1,2; Tables 2–4 |
| Effect of uncertainty | 24 | Describe how uncertainty about analytic judgements, inputs or projections affect findings. Report the effect of choice of discount rate and time horizon, if applicable |
Results para 3,4; Table 5; Figure 1 |
| Effect of engagement with patients and others affected by the study | 25 | Report on any difference patient/service recipient, the general public, community or stakeholder involvement made to the approach or findings of the study | Not applicable |
| Discussion | |||
| Study findings, limitations, generalizability and current knowledge | 26 | Report key findings, limitations, ethical or equity considerations not captured, and how these could impact patients, policy or practice | Discussion |
| Other relevant information | |||
| Source of funding | 27 | Describe how the study was funded and any role of the funder in the identification, design, conduct and reporting of the analysis | Funding |
| Conflicts of interest | 28 | Report authors’ conflicts of interest according to journal or International Committee of Medical Journal Editors’ requirements | Declaration of interests |
Source: Husereau et al. 25
Appendix B
Model input parameters.

| Parameters | Value at base case (SD) | Distribution | Source |
|---|---|---|---|
| Unit costs in 2023 SGD (S$) | |||
| Drug | |||
| SC Herceptin per vial (600 mg), ±20% | 1344 | Uniform |
NCCS Finance |
| IV Herceptin per vial (440 mg), ±20% | 3190 | Uniform |
|
| Consumables | |||
| SC consumables: syringe and needle, ±20% | 1.12 | Uniform |
Market price26–28 |
| IV infusion set, ±20% | 7.23 | Uniform |
|
| Cardiac assessment | |||
| 2D-Echo, ±20% | 471 | Uniform |
Hospital bill data |
| Healthcare professionals | |||
| Pharmacist wage per hour, ±20% | 43.0 | Uniform |
SingHealth HR |
| Nurse wage per hour, ±20% | 39.6 | Uniform |
|
| Infusion chair time (within 2 h), ±20% | 168 | Uniform |
NCCS Finance |
| Absenteeism | |||
| Median gross monthly income (inc. employer CPF), ±20% | 3608 | Uniform |
MOM Labor Force Report 2023, Section C6 group 55–64, Female (22) |
| Healthcare resource utilization per cycle | |||
| Homecare SC Herceptin-related | |||
| Syringe and needle set | 1 | Uniform (1–2) | Assumption |
| Drug preparation by a nurse, min | 10.10 (7.50) | Gamma (1.81, 5.57) | HCP survey |
| Drug administration by a nurse, min | 4.15 (2.59) | Gamma (2.57, 1.62) | HCP survey |
| Travel time by nurse, min (±20%) | 30.00 | Uniform (24–36) | Expert opinion |
| Time spent on Herceptin treatment, hour (±20%) | 0.5 | Uniform (0.4–0.6) | Expert opinion |
| IV Herceptin-related | |||
| Weight (non-metastatic breast cancer patient), kg | 57.4 (11.5) | Gamma (24.9, 2.30) | Hospital administrative database |
| Weight (metastatic breast cancer patient), kg | 55.4 (11.6) | Gamma (23.0, 2.41) | Hospital administrative database |
| Loading dose, 8 mg/kg | 8 | Fixed | Treatment protocol |
| Maintenance dose, 6 mg/kg | 6 | Fixed | Treatment protocol |
| Infusion set | 1 | Uniform (1–2) | Assumption |
| Drug preparation by a pharmacist, min | 18.9 (6.47) | Gamma (8.54, 2.21) | HCP survey |
| IV cannulation by nurse, min | 12.1 (4.12) | Gamma (8.61, 1.40) | HCP survey |
| Infusion time (loading dose), min | 90 | Fixed | Treatment protocol |
| Infusion time (maintenance dose), min | 36.2 (13.6) | Gamma (7.09, 5.10) | HCP survey |
| Travel time to NCCS, hour | 0.85 (0.52) | Gamma (2.67, 0.32) | Patient survey |
| Time spent at NCCS (enter to exit), hour | 2.23 (1.22) | Gamma (3.34, 0.67) | Patient survey |
| Others | |||
| 2D-Echo (non-metastatic breast cancer patient: every 3 months) per year | 4 | Uniform (3–5) | Expert opinion |
| 2D-Echo (metastatic breast cancer patient: every 6 months) per year | 2 | Uniform (1–3) | Expert opinion |
| Cycle number per course of treatment | 18 | Fixed | Treatment protocol |
2D-Echo, two-dimensional echocardiogram; HCP, healthcare professionals; HR, human resource; IV, intravenous; MOM, Ministry of Manpower; NCCS, National Cancer Centre Singapore; SC, subcutaneous.
Acknowledgements
None.
