Abstract
Background:
The single-arm phase II APT trial established trastuzumab and paclitaxel (TH) as the standard adjuvant regimen for small human epidermal growth factor receptor 2 (HER2+) tumors. However, paclitaxel causes alopecia and has high rates of neuropathy and hypersensitivity reactions. In patients with metastatic HER2+ breast cancer (BC), the combination of trastuzumab and vinorelbine (TV) is effective and well tolerated. There is a need for alternative non-anthracycline/taxane-based regimens for patients with HER2+ early-stage BC, especially for those with contraindications or who wish to avoid side effects of taxane-based regimens. Here we describe our institutional experience with adjuvant TV for patients with early-stage HER2+ BC.
Methods:
Clinicopathological characteristics, treatment details, and outcomes of patients with localized HER2+ BC treated with adjuvant TV from 2007 to 2021 at a large academic medical institution were collected. Study endpoints included invasive disease-free survival (IDFS), overall survival (OS), and safety/tolerability. IDFS and OS were measured from start date of TV treatment to date of event/last follow-up and date of death/last follow-up, respectively.
Results:
A total of 30 patients were treated with TV. All patients received trastuzumab at standard dosing and vinorelbine at a starting dose of 25 mg/m2 either on days 1/8 or on days 1/8/21 (weekly) of a 21-day cycle with four planned cycles. Median age at diagnosis was 59 years (range: 36–81). 90.3% of patients had anatomic pathologic stage IA BC and 9.7% stage IIA BC. Of the 30 patients, 24 of them opted to pursue TV due to concerns related to alopecia, neuropathy, and other toxicities, and 6 switched from treatment with TH to TV due to toxicities. Eight patients experienced neutropenia with no cases of febrile neutropenia. No patients experienced alopecia or long-term neuropathy. With a median follow-up of 68 months (5.7 years), the 5-year IDFS rate was 90.9%, with one local and one distant recurrence. The 5-year OS was 100%.
Conclusions:
Trastuzumab in combination with vinorelbine in the adjuvant, early-stage setting for low-risk HER2+ BC demonstrated clinical efficacy and appeared to be well tolerated. TV warrants further evaluation as an alternative regimen to TH for patients with early-stage HER2+ BC.
Introduction
The overexpression of human epidermal growth factor receptor 2 (HER2, also known as ERBB2) occurs in about 20% of breast cancer (BC) tumors and is associated with poor prognosis and clinical outcomes.1–3 However, anti-HER2 therapy has revolutionized the treatment of HER2+ BC. Trastuzumab, a humanized monoclonal antibody against HER2, significantly improves outcomes in patients with early or metastatic HER2-positive BC when combined with anthracycline and taxane-based chemotherapy regimens.1–3 However, many studies have shown that pairing anthracycline-based chemotherapies with trastuzumab can result in significant adverse events (AEs), namely cardiotoxicity.4,5 Several clinical trials in the adjuvant and neoadjuvant setting of early-stage HER2+ BC demonstrated that anti-HER2 therapies paired with anthracycline-free chemotherapy regimens were equally effective but with fewer cardiotoxic and myelosuppressive side effects compared with the anthracycline-based regimens.4–6 These studies resulted in a shift away from anthracycline-based regimens toward taxane-based regimens for treatment of early HER2+ BC.
Since these clinical trials were largely focused on stage II and III HER2+ BC, other studies turned their focus to understanding the relative risks and benefits of trastuzumab in combination with chemotherapy for lower-risk HER2+ BC. The APT trial found that treatment of stage I (and T2 tumors 3 cm or smaller) HER2+ BC with adjuvant paclitaxel and trastuzumab (TH) resulted in remarkably high survival rate and low risk of recurrence [7-year overall survival (OS) and disease-free survival (DFS) were 95% and 93%, respectively] as well as a low rate of toxicity.7,8 However, 13.1% of patients in the study had grade 2–3 neuropathy and the vast majority of the patients had alopecia. 7 The ATEMPT trial compared adjuvant TH to adjuvant trastuzumab emtansine (TDM-1), an antibody–drug conjugate (ADC), for stage 1 HER2+ BC. The co-primary endpoints were to compare the incidence of clinically relevant toxicities and to evaluate invasive disease-free survival (IDFS) in patients receiving TDM-1. The study found that treatment with TDM-1 had an excellent 3-year IDFS (97.8%), but no significant difference in overall levels of clinically relevant toxicities compared to TH were found. 9
In metastatic HER2+ BC, the combination of pertuzumab, trastuzumab, and taxane is the standard first-line regimen based on the results from the pivotal CLEOPATRA trial. 10 However in the pre-pertuzumab era, trastuzumab and vinorelbine (TV regimen) had shown promise, with comparable efficacy and significantly less toxicity (including neuropathy, alopecia, and allergic reactions) compared to the then-standard trastuzumab/paclitaxel.11,12 A more recent study showed reduced neuropathy and alopecia rates with the use of pertuzumab, and TV compared to the ones observed with taxane in the CLEOPATRA trial.10,13 In addition, the TV regimen was evaluated in two retrospective studies in the adjuvant and neoadjuvant setting of early HER2+ BC.14,15 However, since the time of these publications, clinical guidelines have evolved to increasingly recommend neoadjuvant therapy for many patients with localized HER2+ BC and therefore several of the patients included in the prior studies of the TV regimen would now have received neoadjuvant therapy. Therefore, there is a need to examine contemporary outcomes for patients with low-risk HER2-positive tumors for whom neoadjuvant therapy would not be offered and evaluate whether TV provides a suitable alternative to TH in select patients.
On the basis of all of the above, we hypothesized that the TV regimen could be an effective and well-tolerated regimen, with a lower incidence of allergic reactions, neuropathy, and alopecia, for the adjuvant treatment of early-stage HER2+ BC. Here we describe our institutional experience using adjuvant TV in the early-stage setting as well as a subset of patients who switched to the TV regimen after complications with paclitaxel.
Methods
Eligibility and patient selection
Patients who received the combination of TV for HER2-positive early-stage BC in the adjuvant setting at a large academic institution were identified from an IRB-approved Research Patient Data Registry. The registry search initially identified a total of 267 patients who were then systematically excluded based on a variety of clinical factors. Patients were excluded if they had metastatic disease, received TV for treatment of a different type of cancer, or if they received neoadjuvant chemotherapy. Eligible patients were those who received trastuzumab in combination with vinorelbine for early-stage HER2-positive BC in the adjuvant setting. Both HR-positive and HR-negative patients were included. HER2 positivity was determined with immunohistochemistry or fluorescent in situ hybridization based on local testing as per ASCO/CAP guidelines. In addition, two cohorts of patients were considered to be eligible. Cohort one included patients who received TV as their first treatment in the adjuvant setting. Cohort two included patients who switched to TV after complications using adjuvant TH. All patients were made aware that this regimen was not the standard of care in the adjuvant setting, though it had performed well in the metastatic and neoadjuvant settings, and all made an informed decision to accept treatment with this combination.
Treatment regimen
All patients underwent surgery as their first oncologic intervention (either breast conserving surgery or mastectomy) prior to initiating adjuvant treatment with TV. For patients in cohort one, adjuvant treatment consisted of four 21-day cycles with TV followed by trastuzumab alone for a total of 1 year of anti-HER2 therapy. Patients in cohort two switched to treatment with adjuvant TV after experiencing complications with paclitaxel and were able to complete adjuvant chemotherapy with TV. On average, patients in cohort two switched to TV after 2.3 weeks of treatment with paclitaxel. Patients in cohort two also continued trastuzumab to complete a year of treatment. For both cohorts, trastuzumab was given on Day 1 and vinorelbine was given either on Days 1 and 8 or on Days 1, 8, and 21 (weekly) of a 21-day cycle.
Data collection and study endpoints
A systematic chart review was conducted for each of the eligible patients using the electronic medical record. Data points collected for each subject included patient demographics, BC stage, pathology results, comorbidities, hematologic data, treatment details, and other notable toxicities. Left ventricular ejection fraction (LVEF) was assessed prior to treatment and at regular intervals during treatment. Treatment tolerability was examined using the number of treatment delays, dose reductions, discontinuations, and hospitalizations due to toxicity. The study endpoints were IDFS, either recurrence or new invasive disease, distant disease-free survival (DDFS) and OS as defined by the standardized efficacy endpoints criteria, 16 as well as toxicity and tolerability of the TV regimen.
Statistical analysis
IDFS and OS were defined as the time from start date of TV treatment to date of recurrence/last follow-up or death/last follow-up, respectively. DDFS was defined as the time from start date of TV treatment to date of recurrence at distant site or last follow-up. Estimation of the 5-year survival and survival free from invasive disease were performed using the Kaplan–Meier product-limit method and survival curves were generated in GraphPad Prism.
Results
Patients
A total of 30 women treated with adjuvant TV for HER2+ early-stage BC from 2007 to 2021 were included in the dataset. Baseline characteristics are shown in Table 1. The median age at diagnosis in cohort one was 61 years, ranging from 36 to 81 with 16% being above the age of 70. 88% of patients had anatomic pathologic stage IA BC and 12% stage IIA BC. None of the patients had positive lymph node involvement. All 24 patients in cohort one opted to pursue TV due to concerns over alopecia, neuropathy, and other toxicities. Most patients in cohort one (N = 21, 88%) received vinorelbine on Days 1 and 8 of each chemotherapy cycle, while the remaining three patients (12%) received vinorelbine on a weekly basis. In cohort two, the median age at diagnosis was 56, ranging from 37 to 81. All patients in cohort two had anatomic pathologic stage IA BC. Among the 30 patients, 17 had estrogen receptor (ER)+/progesterone receptor (PR)+ tumors and 8 had ER+/PR− tumors. In addition, a majority of patients were postmenopausal at the time of diagnosis. In all, 18 women had tumors that measured 1 cm or less in greatest dimension and 12 had tumors with a greatest dimension larger than 1 cm (range of tumor size in greatest dimension was from less than 0.1 to 2.5 cm). Two patients in cohort two (33%) received vinorelbine on Days 1 and 8 of each cycle, while the remaining four patients (67%) received vinorelbine on a weekly basis.
Patient characteristics separated by cohort. Cohort 1 represents patients who received TV as their first-line therapy in the adjuvant setting. Cohort 2 represents patients who switched to TV after complications with adjuvant trastuzumab and paclitaxel.

ALND, axillary lymph node dissection; FISH, fluorescent in situ hybridization; HER2, human epidermal growth factor receptor 2; IDC, invasive ductal carcinoma; IHC, immunohistochemistry; ILC, invasive lobular carcinoma; ND, node dissection; SLNB, sentinel lymph node biopsy.
Treatment tolerability and AEs
A breakdown of treatment tolerability is shown in Figure 1. Among the 30 patients, 27 (90%) completed the intended four cycles of therapy. Discontinuation of treatment all together with TV occurred in two patients (7%) due to abdominal discomfort and neutropenia, and one additional patient (3%) discontinued vinorelbine only due to worsening neuropathy (related to ankle injury). In total, 26 patients continued with trastuzumab dosing to complete a year of treatment. One patient discontinued trastuzumab after four cycles due to development of rash, three patients opted to discontinue per personal preference, two after completing four cycles, and one after six cycles.

Breakdown of patient tolerability of TV treatment.
Treatment delays due to a treatment-related toxicity occurred in six (20%) patients while dose reductions occurred in seven (23%) patients. Of the seven patients who had a dose reduction, one patient had a 17% dose reduction, three patients had a 20% dose reduction, and three patients had a 40% dose reduction of vinorelbine. Overall, 17 (57%) of patients completed four cycles of TV without dose holds or delays. Among patients that completed their intended cycles of TV, the most common toxicities leading to dose reduction or delay were mucositis (2 patients, 7%), elevated transaminases (3 patients, 10%), and neutropenia (3 patients, 10%). Of note, some patients in our cohort received vinorelbine only on Days 1 and 8 of each chemotherapy cycle while other patients received vinorelbine on a weekly basis. Among the patients that experienced neutropenia-related dose delays, reductions, or holds, all were receiving vinorelbine on a weekly basis (as opposed on only on Days 1 and 8). Additional eight patients experienced documented neutropenia on a treatment day, but still met criteria for treatment. There were no cases of febrile neutropenia. A breakdown of laboratory-based AEs is presented in Table 2.
Breakdown of hematologic AEs, including grades according to CTCAE criteria.
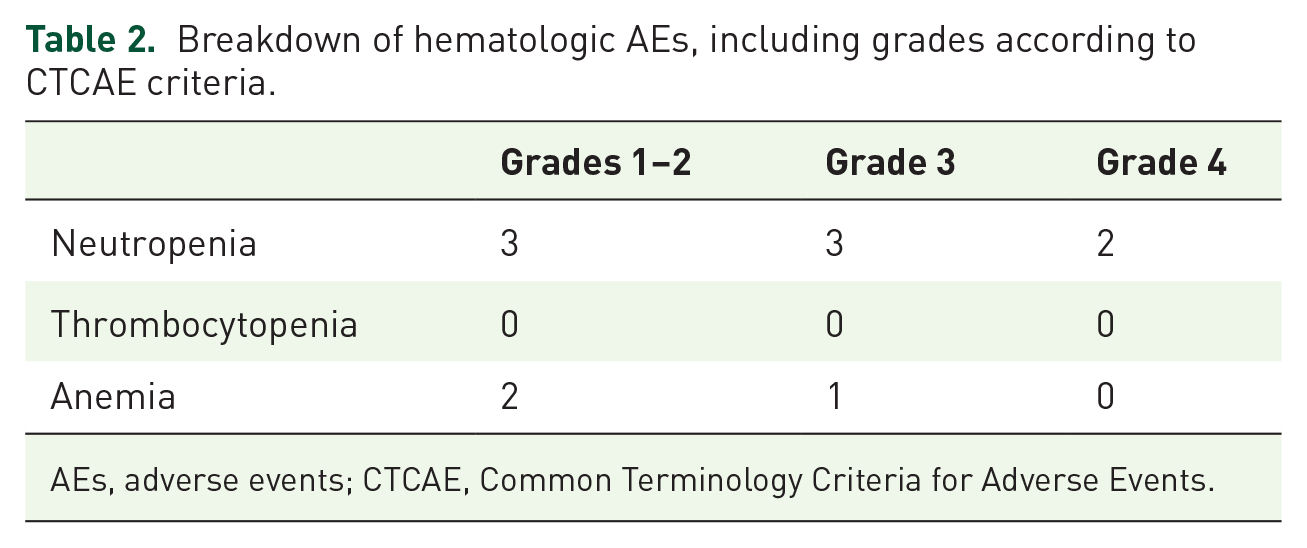
AEs, adverse events; CTCAE, Common Terminology Criteria for Adverse Events.
No alopecia was reported. One patient reported worsening of baseline neuropathy (from a prior ankle fracture) but no long-term neuropathy was reported. Routine anti-emetic prophylaxis was not given. Only two patients (7%) complained of nausea requiring an anti-emetic (both after initial trastuzumab infusion accompanied by fever). Two patients (7%) required hospitalization during treatment with TV due to toxicity. One had bloody diarrhea due to a flare of ulcerative colitis while off mercaptopurine, the other rigors/fever attributed to first dose of trastuzumab. There were no significant decreases noted in LVEF on serial echocardiography.
Efficacy
With a median follow-up time of 68 months (5.7 years), the overall 5-year rate of IDFS for both cohorts combined was 92.6%, with one local and one distant recurrence, both in cohort one. Of note, the local recurrence had a different hormone receptor profile, with original tumor profile being ER+, PR+, HER2+ and the local recurrence was ER negative, PR negative and HER2+, and may represent a new primary tumor or tumor heterogeneity. For cohort one alone, the 5-year IDFS rate was 90.0% (median follow-up time: 64 months) and for cohort two was 100% (median follow-up time: 97.5 months). The 5-year OS for both cohorts was 100%, Figure 2(b). The IDFS curve for both cohorts is presented in Figure 2(a). The 5-year DDFS rate for both cohorts combined was 95.2%.
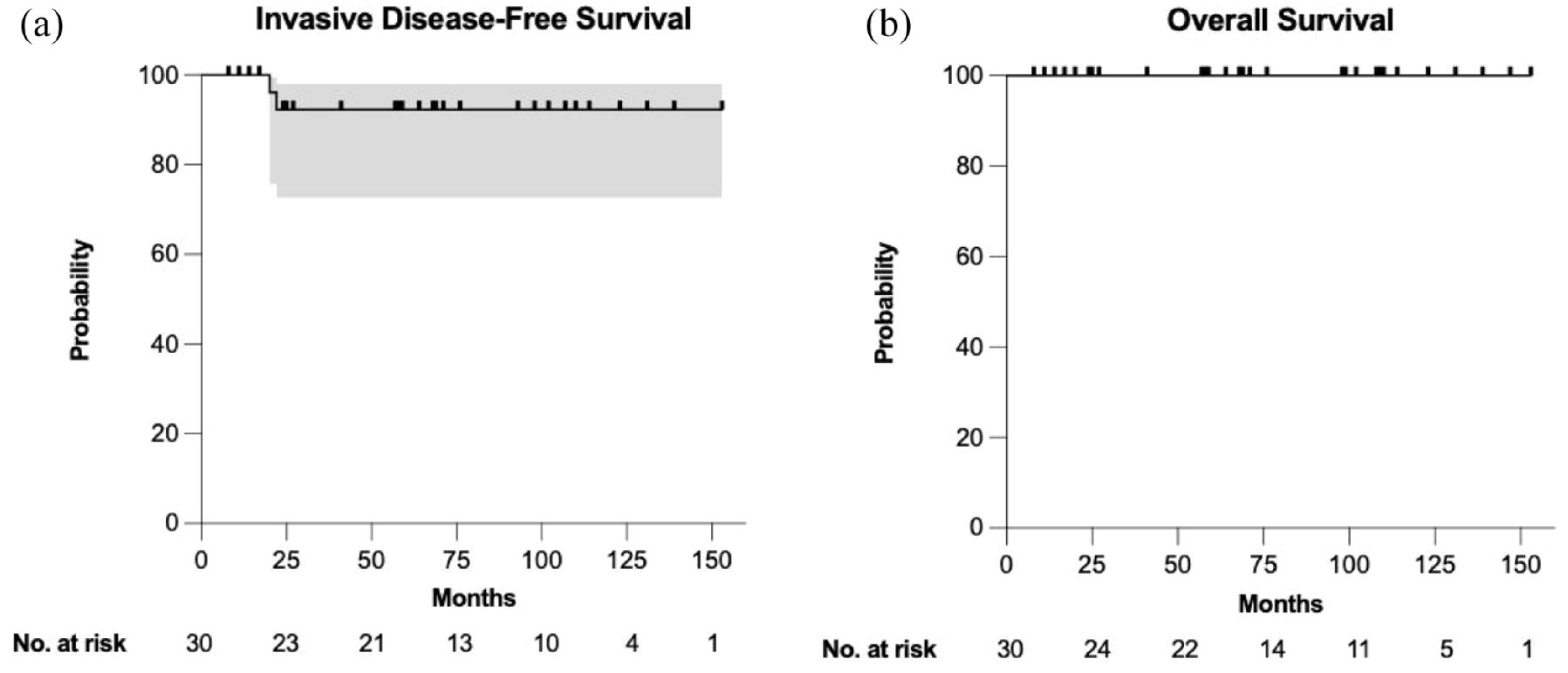
(a, b) IDFS and OS for combined cohorts (N = 30). 95% CI and no. at risk are included.
Discussion
This single institution retrospective study with a long follow-up period demonstrates that adjuvant TV appears to be a well-tolerated treatment for low-risk early-stage HER2+ BC. The IDFS (92.6% for combined cohorts), OS (100% for combined cohorts), and DDFS (95.2% for combined cohorts) were all favorable and no patients in our cohort experienced alopecia or long-term neuropathy, common AEs experienced with the TH regimen. The major adverse effect reported in our cohort was neutropenia, but there were no occurrences of febrile neutropenia. Of note, some patients in our cohort received vinorelbine on Days 1 and 8 of each chemotherapy cycle while other patients received vinorelbine on a weekly basis. All the patients that experienced neutropenia-related dose delays, reductions, or holds received vinorelbine on a weekly basis. Although in our cohort the weekly schedule was more toxic, further research with larger number of patients is needed to determine the recommended adjuvant TV schedule based on efficacy and tolerability.
One of the challenges in the management of early-stage HER2+ BC is defining the threshold to initiate systemic therapy. 7 Recommendations from the National Comprehensive Cancer Network note that adjuvant trastuzumab in combination with chemotherapy should be considered in patients with small, node negative tumors. Management is further complicated by determining the safest and most effective systemic regimen. Standard regimens with trastuzumab in combination with chemotherapy can have considerable toxic effects for some patients. The APT7,8 and ATEMPT 9 trials were major advances for the treatment of small, node-negative HER2+ breast tumors. They demonstrated the success of de-escalation strategies to treat low-risk HER2+ BC with a single chemotherapy agent (paclitaxel, APT trial) or an ADC (TDM-1, ATEMPT trial). The TH regimen has thus become the standard of care for low-risk early-stage HER2+ BC because of its efficacy and relatively lower toxicity profile compared to previous chemotherapy regimens that are commonly used to treat higher stage HER2+ breast tumors. However, there is a need to develop alternative treatment options for low-risk early-stage HER2+ BC as paclitaxel still has notable side effects, namely neuropathy and alopecia. TV has been suggested as a viable alternative treatment due to its milder toxicity profile. In this retrospective study, we found that TV in the adjuvant setting had encouraging efficacy, but a milder toxicity profile (no long-term neuropathy or alopecia). Our results are consistent with other studies supporting a favorable toxicity profile and clinical activity of the TV regimen in treatment of metastatic11,12 and early-stage14,15 HER2+ BC. Our work supports the findings from important earlier works that evaluated the TV regimen in early HER2-positive BC14,15 with the advantage of longer follow-up, and data from a more recent follow-up period, reflecting contemporary clinical guidelines for that patient population. In addition, our work included a more uniform cohort of low-risk early HER2+ patients who received the TV regimen in the adjuvant treatment instead of the accepted TH regimen, unlike other studies which included patients in the neoadjuvant setting that were likely higher-risk patients who would have received a more aggressive regimen according to current guidelines, if treated today.
Given the favorable prognosis of early-stage HER2+ BC, it is important to consider the quality of life (QOL) of patients in this population. Alopecia consistently ranks among one of the most distressing side effects in patients receiving treatment for BC and has been associated with lower components of QOL such as self-esteem, social functioning, and willingness to continue or return to work. While scalp cooling is effective for alopecia from the TH regimen, it is costly, requires significant resources, and is not available to all patients.17,18 While treatment of early-stage HER2+ BC has made great strides in reducing adverse effects of treatments, there is still room to continue finding treatment regimens that reduce these adverse effects. The ATEMPT 2.0 (NCT04893109) study is currently recruiting patients to further assess the efficacy and tolerability of a shorter course of TDM-1. However, there is also some concern that TDM-1 may not be sufficient for HER2 heterogeneous tumors, and likewise the chemotherapy backbone remains important. 19
Our study has several limitations. First, the study was limited by its small sample size and single institutional experience. Second, due to high financial and time costs associated with prospective clinical trials, this was a retrospective non-randomized study, and it is likely that patient selection bias was present. Third, given the retrospective nature, it was not possible to ascertain the grade of subjective AEs such as nausea and fatigue. Collection of AE data was done through documentation in chart notes and through laboratory data and lacks collection of AEs using established reporting mechanisms, such as the Common Terminology Criteria for Adverse Events. Therefore, the toxicity data reported here are likely underestimated. Finally, efficacy of TV with other anti-HER2 agents such as pertuzumab requires further research. Of note, the single-arm phase II VELVET trial investigated the efficacy and tolerability of the pertuzumab-TV regimen in the first-line setting of metastatic HER2+ BC. This trial demonstrated high response rates (74%) and good tolerability for the addition of pertuzumab to the TV regimen. 13
Overall, our institutional experience with TV in the adjuvant setting suggests TV is clinically active in addition to featuring a favorable toxicity profile. Although the use of trastuzumab in combination with vinorelbine for the treatment of low-risk early-stage HER2-positive BC is unlikely to be evaluated in a prospective clinical trial due to the great efficacy of the current approach, it can be highly useful in select cases and therefore warrants further evaluation in larger multi-institutional data sets.
