Abstract
Background:
Changes in human epidermal growth factor receptor 2 (HER2) status following neoadjuvant chemotherapy (NAC) indicate tumor heterogeneity.
Objective:
Investigating the prognostic impact of the transition between HER2-low and HER2-zero may help eliminate the confounding effects of heterogeneity, thereby clarifying the prognostic significance of HER2-low status.
Design:
Retrospective analysis.
Methods:
Data were collected from patients with HER2-negative, early-stage breast cancer who did not achieve a pathological complete response after NAC. Cox regression models and Kaplan–Meier survival curves were employed to analyze disease-free survival (DFS) and overall survival (OS).
Results:
A total of 744 patients were included in the analysis, including 207 with HER2-zero and 537 with HER2-low pre-NAC. Among these, 46.9% (97/207) of the patients with HER2-zero transitioned to HER2-low, whereas 14.7% (79/537) of those with HER2-low transitioned to HER2-zero. Based on the HER2 status pre-NAC, there was no difference in prognosis between the HER2-zero and HER2-low groups. Patients with constant HER2-zero status had poorer OS than those who transitioned from HER2-zero to HER2-low (p = 0.025) in the hormone receptor-negative population, albeit no such result was observed in the hormone receptor-positive population. No significant difference in OS was observed between patients with constant HER2-low status and those who transitioned from HER2-low to HER2-zero. In addition, no significant differences were noted in the DFS across the groups. Multivariate analysis revealed that the constant HER2-zero status was associated with worse OS when compared with other HER2 statuses.
Conclusion:
HER2 transitions between low and zero expressions were frequently observed after NAC, exhibiting heterogeneity of the HER2 expression. Except for the worst OS in the constant HER2-zero, hormone receptor-negative subgroup, no significant differences were observed in the DFS and OS with respect to the changes of HER2-zero and HER2-low groups. The prognostic significance of HER2-zero and HER2-low changes after NAC requires further exploration through prospective studies.
Introduction
The prognosis for patients with triple-negative or human epidermal growth factor receptor 2 (HER2)-positive breast cancer who do not achieve a pathological complete response (pCR) after neoadjuvant chemotherapy (NAC) is generally poor. 1 Although treatments with drugs such as capecitabine 2 or trastuzumab emtansine 3 have improved survival rates in patients with triple-negative or HER2-positive breast cancer who did not achieve a pCR (non-pCR), these intensive postoperative therapies are not effective for all patients. Patients with poorer prognoses after standard therapy need more effective treatment options. The HER2 status in breast cancer can change following NAC.4–7 In our previous study, we demonstrated that patients whose HER2 status transitioned from negative to positive after NAC and who subsequently received anti-HER2 targeted therapy experienced better disease-free survival (DFS) than those with a constant HER2-negative status. 8 The DESTINY-Breast04 trial highlighted the efficacy of trastuzumab deruxtecan, a novel antibody–drug conjugate (ADC), in HER2-low advanced breast cancer. 9 A significant proportion of patients experience transitions between HER2-low and HER2-zero after NAC,5,6,8 potentially offering new therapeutic opportunities with ADCs such as trastuzumab deruxtecan.
However, the prognostic significance of HER2-low expression remains controversial,10–13 and the impact of changes between HER2-zero and HER2-low status following NAC is not fully understood. These transitions may reflect the spatial and temporal heterogeneity of the HER2 expression in breast cancer,14–19 which could influence the overall prognosis. In the past, relying on the HER2 expression data from a single time point, such as a biopsy or postoperative specimen, could not fully capture the tumor’s HER2 status, yielding inconsistent findings. Therefore, multiple assessments of HER2 status during NAC and subsequent surgical procedures are expected to provide a more accurate understanding of HER2 evolution and its prognostic implications. Analyzing the results from multiple HER2 assessments may thus offer a clearer picture of the true prognostic value of HER2-low expression.
Currently, there is limited research on the prognostic value of transitions between HER2-low and HER2-zero status after NAC. This study aimed to investigate the factors influencing these transitions and their prognostic significance in patients with HER2-negative breast cancer and non-pCR after NAC.
Patients and methods
Study population
This study analyzed data from patients with HER2-negative, early-stage breast cancer, and non-pCR after NAC, who were treated at The Affiliated Cancer Hospital of Zhengzhou University from January 2015 to December 2021. The reporting of this study conforms to the reporting recommendations for tumor marker prognostic studies 20 (Supplemental Table). The inclusion criteria for the subjects were as follows: female patients with invasive breast cancer who had received at least four cycles of NAC, underwent surgery after NAC, had residual invasive cancer in the breast (non-pCR), and had available HER2 status data both pre- and post-NAC, as well as follow-up information. The exclusion criteria included the presence of bilateral breast cancer, advanced breast cancer, or other concomitant tumors at diagnosis, and progressive disease during NAC.
Data collection
Clinicopathological characteristics and therapy-related information were collected from 814 patients. The data included age at the time of diagnosis, menopausal status, estrogen receptor (ER) status, progesterone receptor (PR) status, hormone receptor (HR) status, androgen receptor (AR) status, clinical T stage, clinical N stage, Ki-67 index pre-NAC, NAC regimens, date and type of surgery, Miller–Payne (MP) grade, and HER2 expression pre- and post-NAC. These data were extracted from the clinical medical records and pathological reports. The cutoff value for the positive expression of ER, PR, and AR was set at 1%. HR-positive was defined as ER-positive and/or PR-positive, whereas HR-negative was defined as ER-negative and PR-negative. Clinical TNM staging was performed in accordance with the American Joint Committee on Cancer TNM staging system for breast cancer, 7th edition. The HER2 status was interpreted based on the American Society of Clinical Oncology/College of American Pathologists HER2 testing guidelines. HER2-low was defined as immunohistochemistry (IHC) 1+ or IHC 2+ without HER2 gene amplification in fluorescence in situ hybridization, whereas HER2-zero was defined as IHC 0. HER2-low and HER2-zero were both HER2-negative. The transitions in the HER2 status after NAC were examined, and factors influencing the transition between HER2-low and HER2-zero expression were analyzed.
Survival analysis
DFS was defined as the time from radical surgery for breast cancer to the date of relapse, diagnosis of a second primary cancer, death, or the last follow-up. Overall survival (OS) was defined as the time from diagnosis to the date of death from any cause or the last follow-up. The DFS and OS of the overall population were analyzed based on the HER2 status pre-NAC. Comparisons were made between the DFS and OS of subgroups with constant HER2-zero status and those transitioning from HER2-zero to HER2-low expression after NAC based on HR status. In addition, DFS and OS were compared between the subgroups with constant HER2-low status and those transitioning from HER2-low to HER2-zero expression after NAC based on HR status.
Statistical analysis
Statistical analyses were conducted using SPSS version 23.0. Factors influencing HER2 transitions (from zero to low and from low to zero) were analyzed using the chi-square test and logistic regression models. Kaplan–Meier survival curves were used for unadjusted survival comparisons, whereas Cox proportional hazards regression was employed to assess the independent effect of HER2 status and transitions, adjusting for clinical covariates. Independent prognostic factors for DFS and OS were initially identified through univariate analysis, with variables achieving p < 0.05 subsequently included in the multivariate analysis. Variables with p < 0.05 in the multivariate analysis were considered independent prognostic factors.
Results
Patient features and HER2 transitions
A total of 814 patients were included in this study. The median age at diagnosis was 47 (range: 19–77) years. Among these patients, 70 (8.6%) exhibited a change in the HER2 status from negative to positive following NAC, whereas 744 (91.4%) maintained HER2-negative expression after NAC. Of the 744 patients with constant HER2-negative status, 207 initially presented with HER2-zero status and 537 with HER2-low status before NAC.
Among the 207 patients with HER2-zero status pre-NAC, 110 (53.1%) maintained a constant HER2-zero expression, whereas 97 (46.9%) experienced a transition from HER2-zero to HER2-low following NAC. The HER2 transition subgroup demonstrated higher rates of HR positivity, AR positivity, and Ki-67 index (⩽30%) compared to the constant HER2-zero subgroup. However, no significant differences were observed between the two subgroups in multivariate analysis. In addition, no significant differences were noted in age at the time of diagnosis, menopausal status, clinical T and N stages, TNM stage, or NAC regimens between the two subgroups (Table 1).
Clinicopathological characteristics associated with HER2 transitions from zero to low.
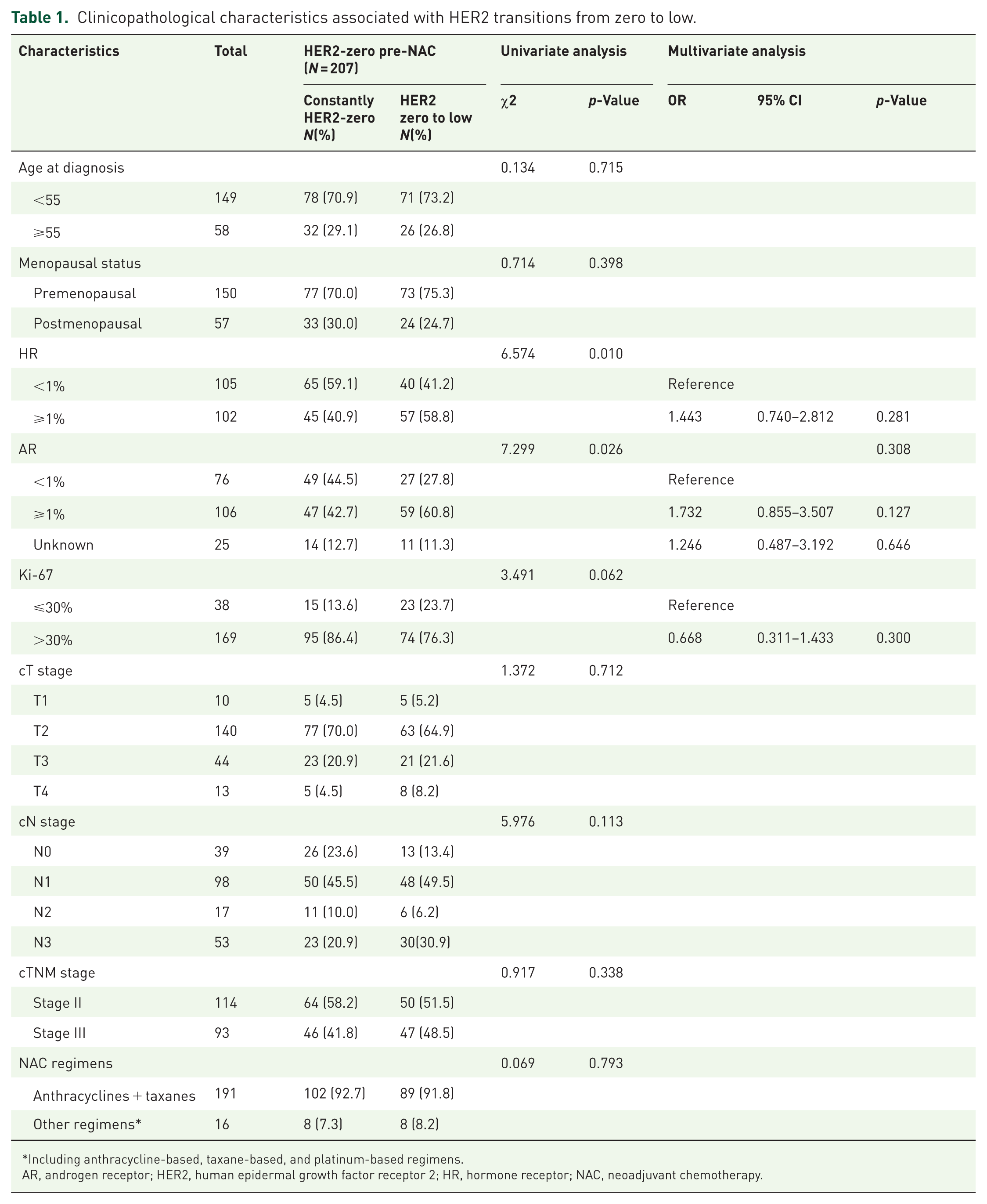
Including anthracycline-based, taxane-based, and platinum-based regimens.
AR, androgen receptor; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; NAC, neoadjuvant chemotherapy.
Among the 537 patients with HER2-low status pre-NAC, 458 (85.3%) maintained a constant HER2-low expression, whereas 79 (14.7%) transitioned from HER2-low to HER2-zero after NAC. The constant HER2-low subgroup showed a higher rate of HR positivity compared to the HER2 transition subgroup (80.3% vs 63.3%, p = 0.022). No significant differences were observed between the two subgroups regarding age at the time of diagnosis, menopausal status, AR status (p = 0.280), Ki-67 index, clinical T and N stages, TNM stage, or NAC regimens (Table 2).
Clinicopathological characteristics associated with HER2 transitions from low to zero.

Including anthracycline-based, taxane-based, and platinum-based regimens.
AR, androgen receptor; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; NAC, neoadjuvant chemotherapy.
Survival analysis based on HER2 status pre-NAC
The follow-up period concluded on June 30, 2024, with a median duration of 46.2 months (95% confidence interval: 44.1–48.4 months). The 3-year DFS rate for the entire cohort was 79.8%, whereas the 3-year OS rate was 93.7%.
Based on the HER2 status before NAC, Kaplan–Meier survival analysis indicated no significant difference in DFS between the HER2-zero and HER2-low groups in HR-negative (log-rank p = 0.655, Figure 1(a) and HR-positive populations (log-rank p = 0.661, Figure 1(b)). The OS of the HER2-low group was numerically superior to that of the HER2-zero group in the HR-negative population; however, the difference was not significant (log-rank p = 0.150, Figure 1(c)). Similarly, no significant differences were observed in OS when comparing the HER2-zero and HER2-low groups in the HR-positive population (log-rank p = 0.061, Figure 1(d)).

Survival analysis based on HER2 status pre-NAC. Disease-free survival analysis based on HER2 status in HR-negative (a) and HR-positive (b) populations; Overall survival analysis based on HER2 status in HR-negative (c) and HR-positive (d) populations.
Survival analysis based on the transition in the HER2 status
A total of 744 patients who maintained the HER2-negative status after NAC were included in this analysis. Among patients with HER2-zero status pre-NAC, no significant difference was noted in the DFS between the subgroup that transitioned from HER2-zero to HER2-low and the subgroup that remained HER2-zero across both HR-negative (HR−, p = 0.801) and HR-positive (HR+, p = 0.520) populations (Figure 2(a) and (b)). However, in OS analysis, patients whose HER2 status transitioned from zero to low demonstrated better OS than those who remained constant HER2-zero in the HR-negative group (log-rank p = 0.025, Figure 2(c)), albeit this survival advantage was not observed in the HR-positive group (log-rank p = 0.548, Figure 2(d)).
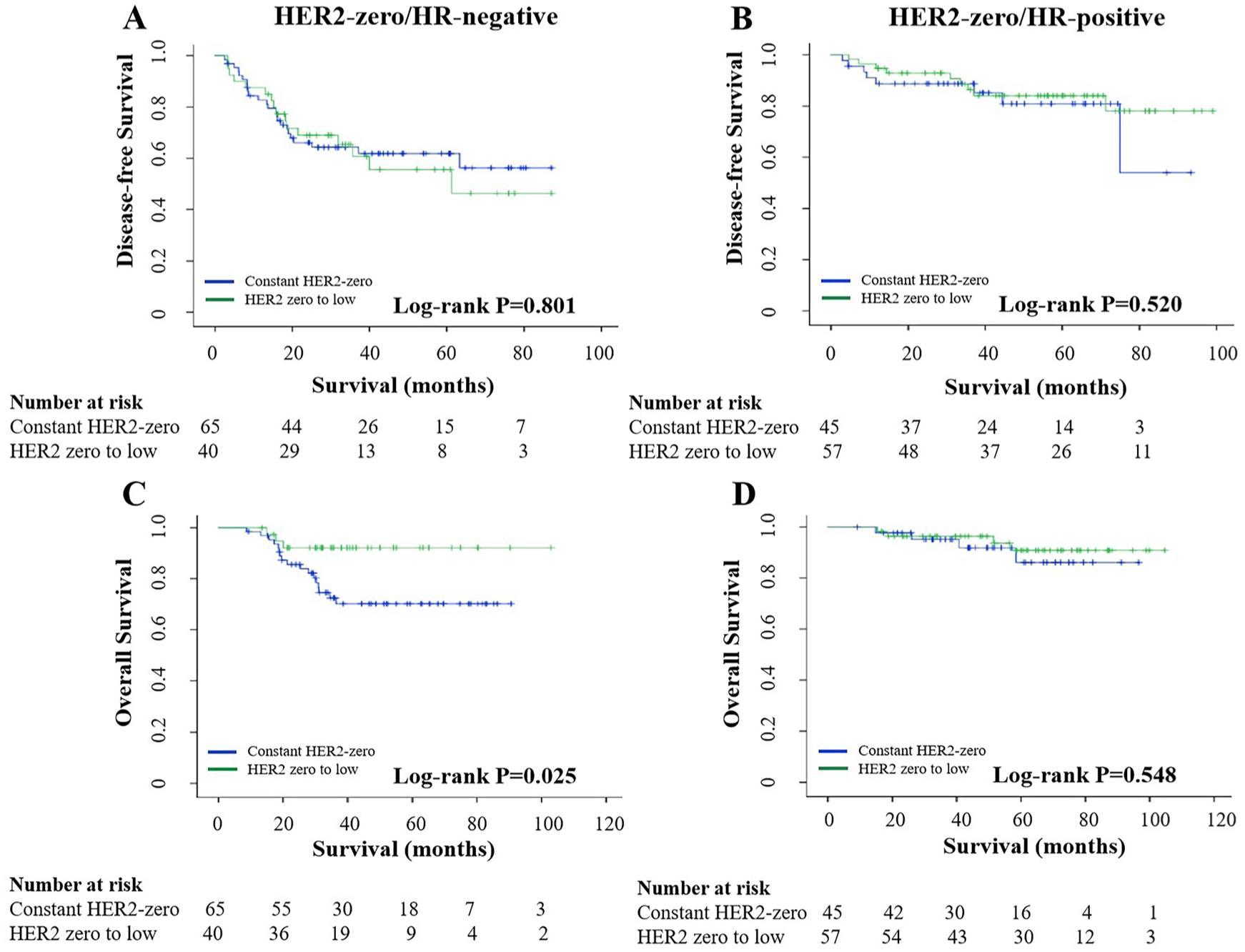
Survival analysis based on HER2 transitions in patients with HER2-zero status before NAC. Disease-free survival analysis in HR-negative (a) and HR-positive (b) populations; overall survival analysis in HR-negative (c) and HR-positive (d) populations.
For patients with HER2-low status pre-NAC, no significant differences were detected in DFS or OS between those who transitioned from HER2-low to HER2-zero and those who maintained HER2-low status in both HR-negative and HR-positive populations (DFS, HR–, p = 0.110, HR+, p = 0.743, Figure 3(a) and (b); OS, HR−, p = 0.647, HR+, p = 0.831, Figure 3(c) and (d)).

Survival analysis based on HER2 transitions in patients with HER2-low status before NAC. Disease-free survival analysis in HR-negative (a) and HR-positive (b) populations; overall survival analysis in HR-negative (c) and HR-positive (d) populations.
Factors associated with DFS and OS
In univariate analysis, several factors were associated with improved DFS, including HR positivity (p < 0.001), lower T stage (p = 0.002), lower N stage (p < 0.001), Ki-67 index ⩽ 30% (p < 0.001), higher MP grade (p < 0.001), and breast-conserving surgery (p = 0.037). Similarly, factors associated with improved OS included premenopausal status (p = 0.041), HR positivity (p < 0.001), HER2-low status pre-NAC (p < 0.001), lower T stage (p < 0.001), lower N stage (p < 0.001), Ki-67 index ⩽ 30% (p = 0.003), HER2-low status post-NAC (p < 0.001), HER2-low status pre- or post-NAC (vs constant HER2-zero, p < 0.001), higher MP grade (p = 0.005), and breast-conserving surgery (p = 0.049; Table 3).
Univariate analysis for clinicopathological characteristics associated with DFS and OS.

Including anthracycline-based, taxane-based, and platinum-based regimens.
DFS, disease-free survival; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; MP, Miller-Payne; NAC, neoadjuvant chemotherapy; OS, overall survival.
In multivariate analysis, DFS was independently associated with the HR status (p < 0.001), clinical T stage (p = 0.005), clinical N stage (p < 0.001), Ki-67 index (p = 0.001), and MP grade (p < 0.001). OS was independently associated with menopausal status (p = 0.010), HR status (p = 0.001), clinical T stage (p = 0.001), clinical N stage (p < 0.001), and HER2 transitions (p = 0.029). Specifically, patients with constant HER2-zero expression pre- and post-NAC showed worse OS than those whose HER2 status transitioned from zero to low (p = 0.030), from low to zero (p = 0.027), or who maintained constant HER2-low expression (p = 0.015; Table 4).
Multivariate analysis for clinicopathological characteristics associated with DFS and OS.

DFS, disease-free survival; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; MP, Miller-Payne; OS, overall survival.
Discussion
This study’s findings demonstrated that the HER2 status can change significantly after NAC, particularly with a notable transition between HER2-low and HER2-zero expressions. No significant differences were observed in DFS and OS between the HER2-zero and HER2-low groups in the HR-negative and HR-positive populations. The effect of transitions between HER2-zero and HER2-low on survival remains unclear, considering the limited relevant literature. Our analysis indicated that patients with HER2 transitioning from zero to low had better OS than those with constant HER2-zero status in the HR-negative population. Multivariate Cox analysis also highlighted that constant HER2-zero expression pre- and post-NAC was associated with poorer OS. Except for the worst OS noted in the constant HER2-zero subgroup, no significant differences in DFS and OS were observed in the changes in HER2-zero and HER2-low.
Consistent with previous research, our study supports the notion that the HER2 status can vary before and after NAC.4–7 Niikura et al. and Kang et al. reported that the proportion of HER2-negative patients who transitioned to HER2-positive status after NAC was 3.4% 4 and 6.1%, 6 respectively, which is slightly lower than the 8.6% observed in our study. The transition between HER2-zero and HER2-low was notably more frequent. Previous studies have reported that 32.0% 6 to 37.7% 5 of HER2-low cases transitioned to HER2-zero after NAC, whereas 29.0% 6 to 33.8% 5 of HER2-zero cases transitioned to HER2-low. In our study, the proportion of HER2-low transforming into HER2-zero was lower than HER2-zero transitioning to HER2-low, indicating a higher frequency of transitions from HER2-zero to HER2-low status. This discrepancy may reflect temporal and spatial heterogeneities in HER2 expression in breast cancer. In addition, variability in the interpretation of HER2 IHC scores (0 vs 1+) by pathologists21,22 may contribute to the inconsistent HER2 status before and after NAC. Our study found that transitions from HER2-zero to low expression were not associated with specific clinicopathological features, whereas transitions from HER2-low to zero were related to the HR status. This finding aligns with those of Kang et al., who noted that ER-positive patients were most prevalent in the constant HER2-low subgroup and least prevalent in the constant HER2-zero subgroup. 6
Currently, the prognostic implications of HER2-low status remain controversial. In a large-scale study involving nearly 1 million patients with breast cancer, those with HER2-low status showed better OS than those with HER2-zero status. 23 Similarly, another extensive study focusing on triple-negative breast cancer (TNBC) found that HER2-low tumors were associated with improved survival relative to HER2-zero tumors. 11 A multicenter study also reported that the recurrence-free interval (RFI) was better for patients with a HER2-low status than those with a HER2-zero status in a non-pCR population. 7 Furthermore, long-term follow-up research indicated that the HER2-low status was associated with increased OS and DFS in high-risk patients identified through genetic testing. 24 By contrast, some studies reported no significant prognostic differences between HER2-low and HER2-zero expressions.12,13,25–28 These discrepancies may be attributed to the shorter follow-up periods and the smaller sample sizes, particularly among patients with TNBC. In view of the association between ER-positive and HER2-low expression, 12 our previous survival analysis of TNBC indicated that patients with a HER2-low status had better breast cancer-specific survival than those with HER2-zero status; this difference persisted in patients with non-pCR or higher stages. 29 Denkert et al. also reported that HER2-low tumors showed improved DFS and OS when compared with HER2-zero tumors in HR-negative and non-pCR populations. 10 These findings imply that the prognostic differences between HER2-zero and HER2-low tumors were more pronounced in high-risk patients. No difference was noted between the HER2-low and HER2-zero groups in terms of DFS and OS comparison. However, the results of retrospective studies may be influenced by various confounding factors, which is why future prospective studies with large sample sizes are needed to explore the prognostic impact of HER2-low expression.
HER2 changes after NAC may impact the accuracy of research findings regarding the prognostic implications of HER2-low status. Our study analyzed the effect of transitions between HER2-zero and HER2-low on prognosis, revealing that patients whose HER2 status transitioned from zero to low had better OS than those with constant HER2-zero status in an HR-negative population. Similarly, a study on metastatic breast cancer found that the HER2-zero to HER2-low transition subgroup demonstrated superior OS than the constant HER2-zero subgroup. 30 In addition, Kang et al. reported that the HER2-low subgroup transitioning to HER2-zero exhibited a better prognosis than the constant HER2-zero subgroup. 6 These findings seem to suggest that HER2 low is associated with a better prognosis. However, a multicenter study found that the HER2-zero to HER2-low subgroup had the worst RFI among ER-positive patients, whereas the constant HER2-low subgroup had the best RFI. The study did not detect any prognostic significance in HER2 transitions within the overall or ER-negative populations. 7 Our multivariate analysis supports HR, with T and N staging as the main prognostic factors. HER2 changes do not affect DFS, but they affect OS, probably due to the confounding factors, warranting further research. These controversial research findings may be attributed to the selection bias inherent in retrospective studies. Different treatment plans for patients after surgery or recurrence may also lead to bias in research results. Thus, further researches, especially prospective clinical trials, are needed to clarify the prognostic significance of HER2 transitions.
The prognosis for patients with TNBC remains poor, 31 especially those with non-pCR after NAC. Our findings suggest that constant HER2-zero expression is associated with worse OS only in HR-negative populations. Therefore, HER2 retest in the residual tumor after NAC for TNBC could be necessary. By categorizing non-pCR TNBC based on dynamic HER2 transitions, we may be able to provide more tailored treatment opportunities. For patients whose HER2 status changes from zero to low after NAC, novel ADCs targeting HER2 could be beneficial. For those with persistently HER2-zero expression, additional effective treatments, beyond capecitabine, olaparib, or pembrolizumab, need to be explored to enhance treatment efficacy.
Nonetheless, our study has certain limitations that should be considered. The retrospective nature of this study introduces the potential for bias, which is a significant limitation. The HER2 status was assessed via IHC by pathologists from Henan Cancer Hospital and extracted from pathological reports, without re-evaluation by a central laboratory. This approach could contribute to inconsistencies in the HER2 status determination. In addition, due to a notable lack of data on histological grade, it was not included in our analysis, which also represents a limitation of the study. The results of this study lack validation from external independent cohorts; this is also a limitation. Trastuzumab deruxtecan showed a therapeutic effect on breast cancer with HER2 ultra-low expression. This ultra-low expression of HER2 probably exhibited biological characteristics similar to those of HER2-low expression. The lack of analysis on HER2 ultra-low expression is also a limitation of this study.
Conclusion
HER2 transitions between low and zero expression were frequently observed after NAC, which exhibited significant heterogeneity of the HER2 expression. Except for the worst OS in the constant HER2-zero subgroup, no significant differences in DFS and OS were observed with respect to the changes of HER2-zero and HER2-low. However, the clinical outcomes of the constant HER2-zero subgroup were only validated in the HR-negative population, which may have been influenced by confounding factors. The prognostic significance of HER2-zero and HER2-low changes after NAC requires further exploration through prospective studies.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251392070 – Supplemental material for Prognostic significance of HER2-low and HER2-zero status before and after neoadjuvant chemotherapy in patients with HER2-negative breast cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251392070 for Prognostic significance of HER2-low and HER2-zero status before and after neoadjuvant chemotherapy in patients with HER2-negative breast cancer by Youzhao Ma, Jingyang Zhang, Lina Wang, Dechuang Jiao, Xiuchun Chen and Zhenzhen Liu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
