Abstract
Background:
Sex disparities are known modifiers of health and disease. In neuroendocrine neoplasms (NENs), sex-based differences have been observed in the epidemiology and treatment-related side effects.
Objectives:
To examine sex differences in demographics, diagnoses present during hospital admission, comorbidities, and outcomes of hospital course among hospitalized patients with NENs.
Design:
Retrospective analysis.
Methods:
A descriptive analysis of sex differences was performed on patients with NENs discharged from U.S. community hospitals in 2019 from the National Inpatient Sample (NIS), Healthcare Cost and Utilization Project, and Agency for Healthcare Research and Quality.
Results:
A total of 7334 patients with NENs were identified; 4284 patients had primary NENs, and 3050 patients had metastatic NENs. In total, 48.7% were males and 51.3% were females. Distributions of race and ethnicity, and payer types differed by sex (p < 0.001 and p = 0.027, respectively). For race and ethnicity, there were more females in White, Black, and Native American races, and Hispanic ethnicity. For payer types, female predominance was seen with Medicare, Medicaid, private insurance, and self-pay groups. Sex differences were seen in diagnosis made during hospital stay. In all NENs, oral (p = 0.036) and neurologic (p < 0.001) diagnoses were more common in females; ascites (p = 0.002), dysphagia (p = 0.002), biliary ductal obstruction (p = 0.014), and jaundice (p = 0.048) were more common in males. In primary NENs, ascites (p < 0.001) was male predominant. In metastatic NENs, dysphagia (p = 0.003) and jaundice (p = 0.034) were male predominant, whereas females had more headaches (p < 0.001). Nausea and vomiting were female predominant in all NENs (p < 0.001), primary (p = 0.044), and metastatic (p < 0.001) NENs. For comorbidities, arthropathies (p < 0.001), depression (p < 0.001), hypothyroidism (p < 0.001), other thyroid disorders (p < 0.001), chronic pulmonary disease (p = 0.002), and obesity (p < 0.001) were female predominant.
Conclusion:
There were sex differences in the race and ethnicity, payer types, diagnoses present during hospital admission, and comorbidities among the 2019 NIS hospital discharge sample of patients with NENs.
Plain language summary
This study explored sex differences in neuroendocrine neoplasms (NENs) using 2019 hospital discharge data. There were variations in demographics, diagnoses, and comorbidities between males and females. Female patients had higher rates of oral and neurologic issues, while males had more ascites, dysphagia, and jaundice. Certain comorbidities like arthritis, depression, hypothyroidism, and obesity were more common in females. Hospital stays and mortality rates did not differ significantly based on sex. In summary, this research highlights distinct sex-related patterns in NENs, shedding light on potential areas for tailored interventions or further investigation.
Keywords
Introduction
Neuroendocrine neoplasms (NENs) are relatively rare tumors that possess both neural and endocrine properties that most commonly arise in the gastroenteropancreatic (GEP) system and lungs.1,2 Generally, NENs are classified based on the clinical behavior, histology, and proliferation rate into low-grade indolent neuroendocrine tumors (NETs) or high-grade aggressive neuroendocrine carcinomas (NECs) although histological grades do not always correlate with clinical behavior. 3 In these discordant circumstances, clinical judgment precedes histology which may result in the treatment of low-grade tumors with chemotherapy. 3 Although most NENs usually have indolent tumor biology and clinical course, approximately 10%–20% of NENs are NECs which are highly proliferative and characterized by rapid disease progression which warrants treatment.4,5
The incidence and prevalence of NENs are increasing steadily in the United States (U.S.) and globally owing to the increase in early detection. The annual age-adjusted incidence of NENs in the U.S. was 1.09 per 100,000 persons in 1973 and increased to 6.98 per 100,000 persons by 2012 across all sites, stages, and grades.5,6 Since many patients with NENs usually have an indolent disease course and long survivorship, being able to predict and mitigate acute and chronic treatment-associated tissue injury or systemic side effects is important. 7 Based on prior large epidemiologic and retrospective studies, it appears that there are sex-based differences in the epidemiology of NENs and treatment-related side effects.8 –11 Sex differences are also known as an important modulator of symptoms, patient behavior, access to therapy, and treatment-related toxicities in medicine and pharmacology but remain inadequately explored in oncological research. 12 Hence, studying sex differences in NENs can potentially aid in optimizing treatment selection for patients to better predict outcomes and mitigate toxicities. 8
Sex and gender disparities are widely known modifiers of health and disease, impacting many aspects of medicine ranging from biology to social behaviors. 13 The term “sex” refers to the biological differences between males and females determined by chromosomes whereas “gender” refers to the sociocultural construct which includes the roles, norms, and behaviors expected of males and females. 12 The complex interaction of biological sex and gender expression influences not only response to treatment but also access to healthcare due to social or economic inequalities. In recent years, sex differences have been increasingly recognized in oncology but remain largely underexplored. 14 In our retrospective study, hospitalized patients with the diagnoses of NEN were of interest because this specific population may reflect the impact of sex differences on acute and chronic treatment-associated tissue injury or systemic side effects and outcomes of patients with NENs.
Given that sex differences may influence treatment-related side effects, patient outcomes, and cost of healthcare, the aim of this study is to determine whether sex differences are present among patients with the diagnosis of NENs during their hospital stay in the U.S. Several preexisting studies on sex-based differences focus on epidemiology and survival of NENs in a general population and treatment toxicities in patients enrolled in clinical trials.6,8,9 However, to the best of our knowledge, there are no dedicated studies evaluating sex-based differences specifically among hospitalized patients with the diagnoses of NENs to explore both acute and chronic treatment-associated tissue injury or systemic side effects and patient outcomes. Using a large publicly available inpatient dataset, we examined the impact of sex differences on the demographics, diagnoses present during hospital stays, comorbidities, and outcomes of hospitalized patients with the diagnosis of NENs during their hospital course.
Materials and methods
Using a retrospective approach, a descriptive analysis was performed on the patient data from the National Inpatient Sample (NIS), Healthcare Cost and Utilization Project, and Agency for Healthcare Research and Quality (AHRQ). 15 These patient data approximates a 20% stratified sample of all discharges from U.S. community hospitals, excluding rehabilitation and long-term acute care hospitals. 15 This database was used to define an unweighted sample of patients with NENs who were discharged from U.S. community hospitals in 2019. Unweighted data was used because the aim of the study was to explore sex differences for this sample of hospitalized patients diagnosed with NENs, and not to calculate nationally representative estimates. We selected patients from 2019 because the sample size from 2020 was smaller than anticipated presumably due to the impact of COVID-19 on the number of admissions. Patients with primary and metastatic NENs were identified using the Clinical Classifications Software Refined for International Classification of Disease, Tenth Revision, clinical modification (ICD-10-CM) principal diagnosis of “malignant neuroendocrine tumors” (NEO066), excluding “secondary Merkel cell carcinoma” (ICD-10-CM code C7B1). 16 Primary NEN is defined as a localized disease, whereas metastatic NEN refers to the involvement of secondary organs. Refer to the Supplemental Table for a list of ICD-10 codes for primary and metastatic NEN.
Our primary comparison variable for patient sex was defined according to the indicator provided within the NIS Inpatient Core File. Data elements from the Core file used to define demographics included patient age at admission, race and ethnicity, median household income in zip code, primary expected payer, and urban/rural location of residence. The Core file also contained up to 40 ICD-10-CM diagnoses present during hospital admission, which we grouped according to organ system into oral, gastrointestinal, hematologic, constitutional, dermatologic, neurologic, and other categories (see Tables 2–4 for subcategory detail). Diagnoses present during hospital admission refer to conditions that were diagnosed during the hospital course which are distinct from comorbidities. Using the NIS Diagnosis and Procedure Groups File, comorbidities were defined according to 15 distinct categories identified by the AHRQ Elixhauser Comorbidity Software Refined for ICD-10-CM diagnosis codes (see Table 5 for the list of comorbidities). 17 Length of hospital stay and inpatient mortality were defined from the Core File.
Chi-square, Fisher’s Exact, and Independent t tests were used, as appropriate, to determine the statistical significance of differences between males and females in terms of demographics, diagnoses present during hospital admission, comorbidities, length of hospital stay, and mortality. All analyses were conducted using Stata/SE 17.0. 18 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement through the Enhancing the Quality and Transparency Of Health Research network 19 (Supplemental File 1).
Results
Sex differences in demographics of patients with NENs during hospital admission
A total of 7334 patients with NENs were identified in the 2019 NIS database. A total of 4284 patients had primary NEN, and 3050 patients had metastatic NEN. Overall, 48.7% were males and 51.3% were females. In primary NEN, 47.8% were males and 52.2% were females. In metastatic NEN, 49.9% were males and 50.2% were females (Figure 1). The mean age in years was 64.8 (standard deviation 12.8) in males and 63.6 (standard deviation 13.8) in females (p < 0.001). Statistical differences were seen when comparing race and ethnicity by sex (p < 0.001). There were more females than males in Black, White, and Native American races, and Hispanic ethnicity (Figure 2). There was a higher number of males in Asian Pacific Islanders as compared to females. Statistical differences were also seen when comparing primary payer types by sex (p = 0.027). Female predominance was seen in Medicare, Medicaid, private insurance, and self-pay groups while there was a male predominance in the no-charge group although both self-pay and no-charge groups had lower numbers (Figure 3). Patient sex did not differ significantly with respect to median household income or urban/rural location of residence. This is summarized in Table 1.

Sex differences in primary, metastatic, and all NENs.
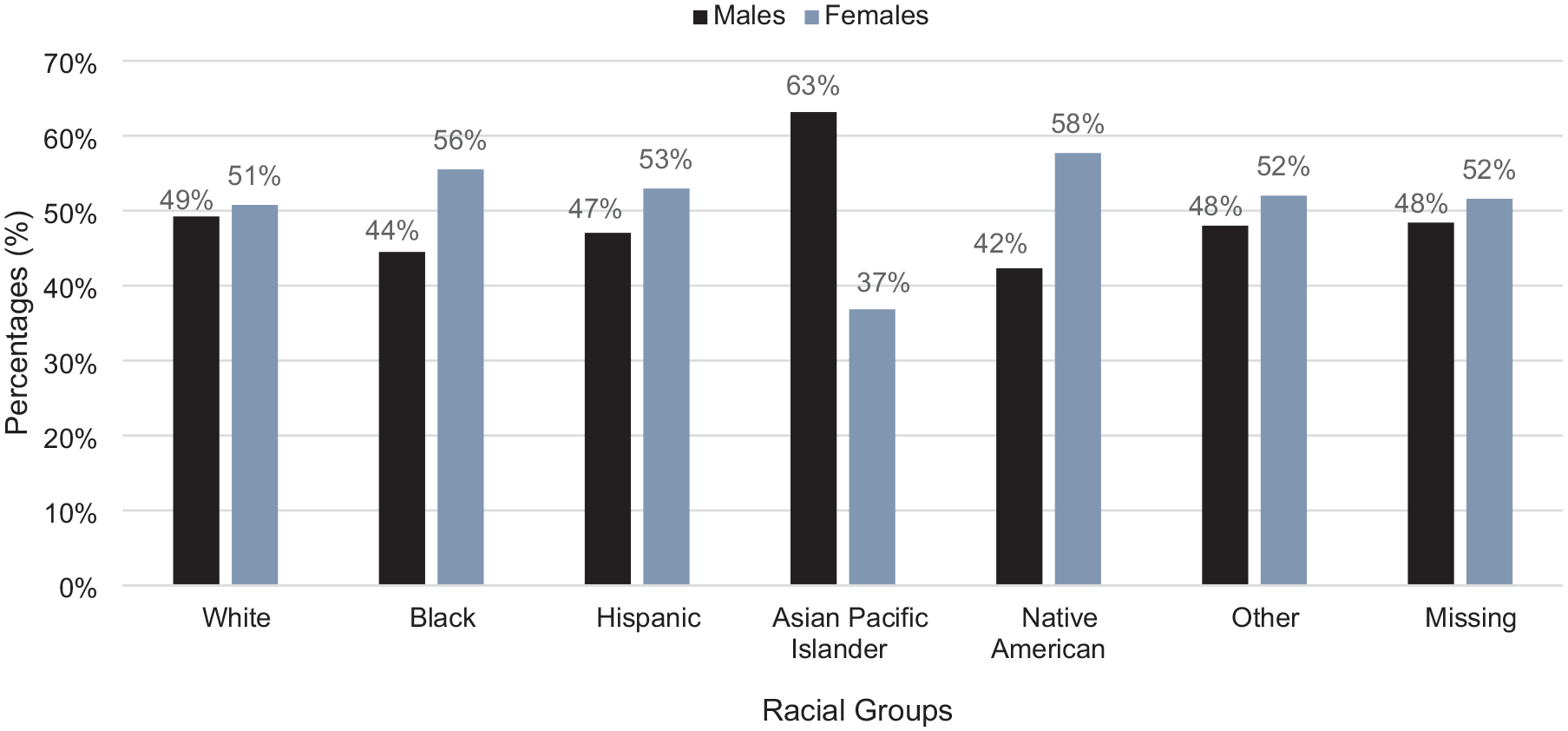
Percentages of males and females in racial and ethnic groups in all NENs.

Percentages of males and females in different primary payer types in all NENs.
Demographics of male and female hospitalized patients with NENs from 2019 public NIS database.

Ind. t-test, independent t-test; NCHS, National Centre for Health Statistics; NENs, neuroendocrine neoplasms; NIS, National Inpatient Sample.
Sex differences in diagnoses present during hospital admission in patients with NENs
All NENs
For diagnoses present during hospital admission in patients with all NENs, female predominance was seen in oral (males 1.9% vs females 2.6%, p = 0.036) and neurologic (males 1.1% vs females 3.8%, p < 0.001) categories. Diagnoses during hospital stay in the oral category were driven by mucositis and stomatitis whereas the neurologic category consists of headaches. Although sex was not significantly associated with the overall gastrointestinal category, there were significant differences seen when considering specific gastrointestinal diagnoses by sex. Ascites (males 8.4% vs females 6.6%, p = 0.002), dysphagia (males 5.0% vs females 3.5%, p = 0.002), biliary ductal obstruction (males 2.9% vs females 2.1%, p = 0.014), and jaundice (males 0.6% vs females 0.3%, p = 0.048) were more common in males whereas nausea and vomiting (males 3.9% vs females 6.0%, p < 0.001) were predominant in females (Figure 4). Mucositis and stomatitis occurred more commonly in females but there were no sex differences in taste alteration. There were no sex differences in cachexia and weight loss. This is summarized in Table 2.

Gastrointestinal diagnoses among patients with all NENs during hospital stay.
Diagnoses present during hospital stay among patients with all NENs.

p-Values are calculated based on Chi-square test unless the counts are less than 5. If counts are less than 5, Fisher’s exact test is used to calculate p-values.
NEN, neuroendocrine neoplasms.
Primary NENs
In patients with primary NENs, sex differences were seen in the oral, gastrointestinal, constitutional, and neurologic categories. There was a male predominance in gastrointestinal (males 23.2% vs females 20.5%, p = 0.033) and constitutional (males 10.7% vs females 8.4%, p = 0.011) categories whereas there was a female predominance in neurologic (males 1.0% vs females 3.9%, p < 0.001) and oral (males 1.6% vs females 2.5%, p = 0.05) categories. Among the diagnoses within the gastrointestinal category, ascites (males 7.9% vs females 5.3%, p < 0.001) was significantly more common in males, whereas nausea and vomiting (males 3.7% vs females 5.0%, p = 0.044) were female predominant (Figure 5). Males experienced more cachexia (males 4.1% vs females 2.9%, p = 0.033), whereas females experienced more headaches (males 1.0% vs females 3.9%, p < 0.001). There was an inclination toward more fever in males and mucositis, stomatitis, and taste alteration in females, although the differences were only marginally statistically significant. This is summarized in Table 3.

Gastrointestinal diagnoses among patients with primary NEN during hospital stay.
Specific diagnoses present during hospital stay among patients with primary NENs.

p-Values are calculated based on Chi-square test unless the counts are less than 5. If counts are less than 5, Fisher’s exact test is used to calculate p-values.
NEN, neuroendocrine neoplasms.
Metastatic NENs
In metastatic NENs, sex differences were observed in neurologic as well as specific gastrointestinal diagnoses. Sex differences were seen in specific gastrointestinal diagnoses. Dysphagia (males 5.3% vs females 3.1%, p = 0.003) and jaundice (males 0.7% vs females 0.2%, p = 0.034) were more common in males whereas nausea and vomiting (males 4.2% vs females 7.5%, p < 0.001) were common in females (Figure 6). Females experience headaches (males 1.3% vs females 3.7%, p < 0.001) more commonly as compared to males. There were no sex differences in weight loss or cachexia. This is summarized in Table 4.
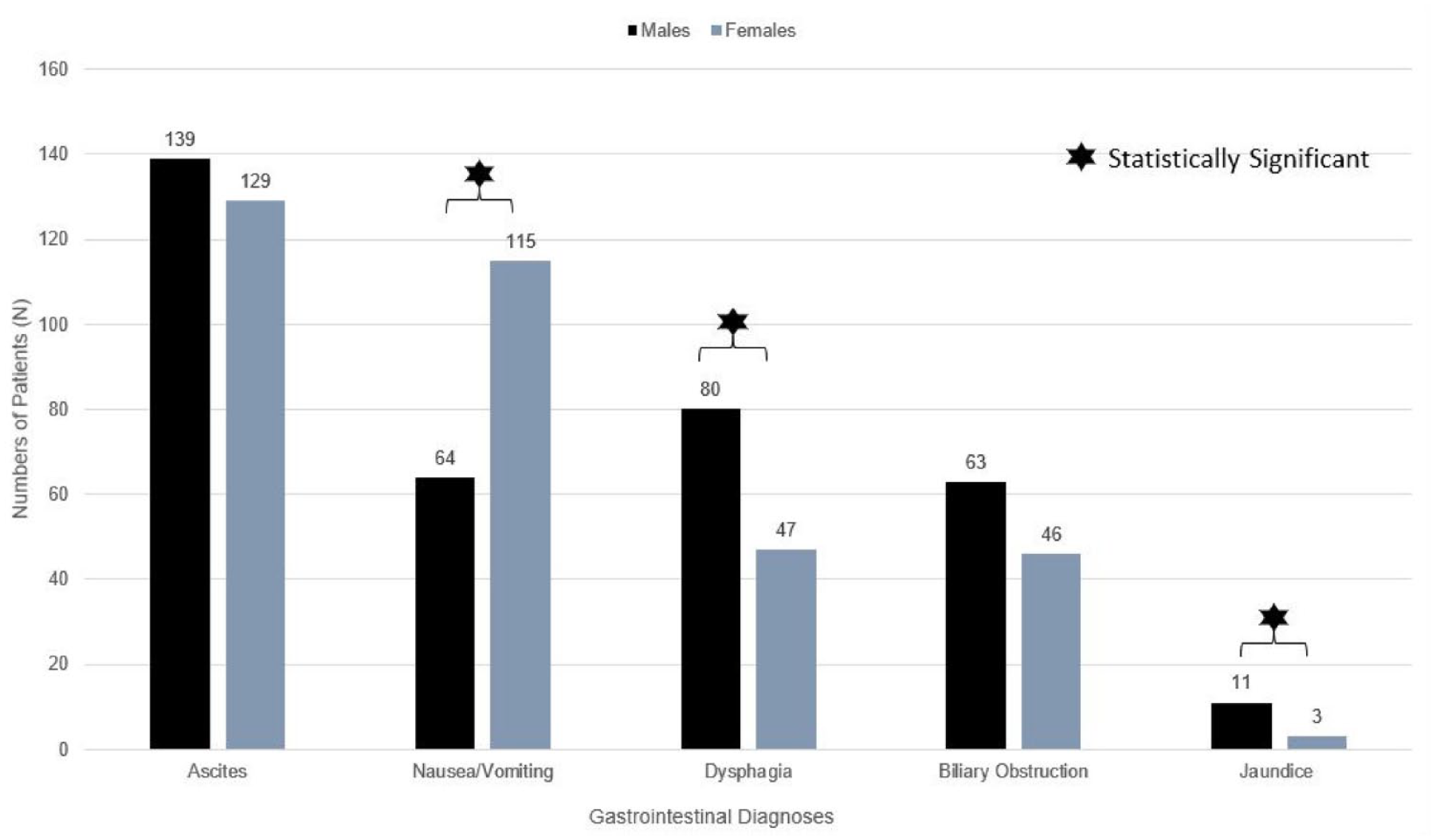
Gastrointestinal diagnoses among patients with metastatic NEN during hospital stay.
Diagnoses present during hospital stay among patients with metastatic NENs.

p-Values are calculated based on Chi-square test unless the counts are less than 5. If counts are less than 5, Fisher’s exact test is used to calculate p-values.
NEN, neuroendocrine neoplasms.
Sex differences in comorbidities and outcomes of hospital admission in patients with NENs
We found statistically significant sex differences in medical comorbidities and outcomes. In all NENs, arthropathies (males 1.7% vs females 4.0%, p < 0.001), depression (males 9.7% vs females 15.2%, p < 0.001), hypothyroidism (males 8.4% vs females 20.4%, p < 0.001), and other thyroid disorders (males 1.1% vs females 2.6%, p < 0.001) were statistically more common in females as compared to males. On the other hand, alcohol use disorder (males 3.2%–3.6% vs females 0.5%–0.9%, p < 0.001) and complicated hypertension (males 22.3% vs females 15.8%–18.1%, p ⩽ 0.001) were statistically more common in males compared to females in all NENs, primary NENs, and metastatic NENs groups. We noted a male predominance in acquired immune deficiency syndromes (males 0.6% vs females 0.2%, p = 0.013) in the all NENs group, similar to the metastatic NENs group (males 0.5% vs females 0.0%, p = 0.04). In diabetes with and without complications, statistically a male predominance was observed in all NENs groups (males 16.9% vs females 14.8%, p = 0.017 and males 13.2% vs females 11.4%, p = 0.022 respectively) but not when primary NENs and metastatic NENs groups were analyzed separately. Although no major sex differences were seen in the metastatic NEN group for chronic pulmonary disease and obesity, females in the primary NENs group had significantly more concomitant chronic pulmonary disease (males 23.2% vs females 27.0%, p = 0.004) and obesity (males 14.5% vs females 19.3%, p < 0.001). No major sex differences were seen in peripheral vascular disease in all NENs groups, but male predominance was seen specifically in the metastatic NENs group (males 7.8% vs females 5.7%, p = 0.023). This is summarized in Table 5. There was no statistical difference with respect to in-hospital mortality between males and females. Although there is statistical significance in the mean length of hospital stay for males and females, it is not clinically significant. However, elective admissions were statistically female predominant as compared to males (males 26.4% vs females 30.9%, p < 0.001). This is summarized in Table 6.
Comorbidities of hospitalized patients with NENs in all, primary, and metastatic NENs.

p-Values are calculated based on Chi-square test unless the counts are less than 5. If counts are less than 5, Fisher’s exact test is used to calculate p-values.
NEN, neuroendocrine neoplasms.
Outcomes of patients with NENs during hospital stay.

Ind. t-test, independent t-test; NEN, neuroendocrine neoplasms.
Discussion
In our analysis of hospitalized patients with NENs, there were more females than males in all stages of NENs. This finding is consistent with a retrospective, population-based study using nationally representative data from the Surveillance, Epidemiology, and End Results (SEER) program which evaluated 64,971 patients with NETs from 1973 to 2012 and showed that 34,233 (52.7%) were females. 6 Although our data did not show statistically significant differences in survival, probably due to the inpatient population sample, several large epidemiologic studies have shown that female sex is associated with better overall survival. Since we observed more hospitalized females with NEN as compared to males, this raises the possibility of an association between hospitalization and improved survival. It is unclear if the overall survival seen in the female population from other studies is a result of healthcare accessibility, biology, or behavior. Alternatively, improved survival in females could also be attributed to inherent biological characteristics which favor survival, and hence contribute to increased hospitalizations that may or may not be related to the diagnosis of NENs.
Although our study is unable to specify the primary site and specific stages of NENs, it is important to note that sex differences may vary depending on these variables.10,20 For example, based on data from England’s National Cancer Registry and Analysis Service that examined 14,834 cases from 2012 to 2018, appendix and lung sites were predominantly diagnosed in females but stomach, pancreas, small intestine, colon, and rectal NENs were most frequently diagnosed in males. 10 The U.S. SEER Registry also showed that females were more likely to have a primary NEN in the lungs, stomach, appendix, or cecum, while males were more likely to have a primary tumor located in the thymus, duodenum, pancreas, small intestine, or rectum. 21
In our study, there was a trend toward more females hospitalized with the diagnosis of NENs in White, Black, and Native American races, and Hispanic ethnicity whereas there was a male predominance in Asian Pacific Islanders. The observed higher proportion of females in the White race in our study was reflective of the published estimates of the prevalence of NENs in the U.S. 22 Notably, our study has limitations inherent to the NIS database, including limited data on location of disease, specific disease stages, and treatment regimens which restricts extrapolation and further comparison with data from other countries. However, we found interesting observations that may provide unique insights when looking into published data on patients with NENs from South America, China, and Japan. In our analysis, hospitalized Asian Pacific Islanders with NENs were predominantly males. Asian Pacific Islanders refer to persons with origins in the Far East (i.e., East and Southeast Asia), Indian subcontinent, or the Pacific Islands. Interestingly, large local epidemiological studies in China, Taiwan, and Japan also showed similar predominance in males despite the variation in environment and healthcare system as compared to the U.S.23 –25 This finding proposes a possible component of biologic difference between males and females in Asian Pacific Islanders. In addition, local epidemiological studies from Chile and Ecuador showed male predominance, whereas there were more females with NENs in Argentina and Panama.26 –29 These findings postulate that other than possible biological variations, healthcare system accessibility and patient behavior may also be important factors influencing the observed sex differences among patients with NENs. Further studies in this area are warranted.
There was a statistically significant difference in the distribution of payer type by sex. In the U.S., health insurance and affordability of healthcare are major challenges.30,31 Payer types are generally categorized into commercial insurance, government insurance, and uninsured or self-pay. Individuals who are considered low income or very low income are eligible for Medicaid, a type of government insurance, although requirements vary slightly based on different states and other factors. 32 Among hospitalized patients with NENs in our study, the percentage of females in the Medicaid and self-pay groups was distinctively higher compared to males. Our findings suggest that there may be a higher percentage of hospitalized females with NENs from lower socioeconomic groups as compared to males. Since we also observed an overall female predominance in hospitalized patients with NENs, this may further postulate that regardless of socioeconomic status and primary payer types, females have a higher tendency to seek health care services. One study evaluated 509 new adult patients randomly assigned to primary care clinics and monitored their use of health care services for 1 year. This study found that females had significantly lower self-reported health status, lower education, and had significantly higher mean number of visits to their primary care clinic and diagnostic services than compared to males. 33 Several studies also showed higher expenditure and utility of healthcare services in females as compared to males.34,35 With regards to cancer care, females are known to experience more adverse events (AEs) from chemotherapy than males which may lead to increased utilization of healthcare services and hospitalizations.36,37
Sex differences in patterns of illness and treatment responses are well recognized but the pathophysiology remains to be further elucidated. Although the primary site and stage of NEN affect the predominant symptoms presented regardless of sex or gender, studies have shown a biological basis that differs between males and females.38,39 Sex differences occur in genetics, energy storage, and metabolism, as well as hormonal factors.39,40 Males and females also differ in terms of pharmacodynamics and pharmacokinetics which determine drug absorption, distribution, metabolism, and excretion. These factors affect drug efficacy resulting in distinct outcomes when exposed to the same compound, rendering one more susceptible to serious AEs and requiring various diagnostic and therapeutic interventions. 39 Our analysis revealed differences in the pattern of diagnoses (symptoms and comorbidities) between hospitalized males and females with NENs. Gastrointestinal diagnoses were common in both males and females. However, males experienced more specific localized symptoms of dysphagia, dyspepsia, ascites, biliary ductal obstruction, and jaundice whereas females predominantly experienced non-specific symptoms such as nausea and vomiting, headaches, mucositis, and stomatitis. Some studies have shown that females have slower gastric emptying and colon transit and these are affected by phases of menstruation. 38 Sex hormones also affect pain processing and females were shown to be more affected by visceral pain which is usually difficult to localize as compared to somatic pain.38,40 Biopsychosocial models may also explain gastrointestinal symptoms. Psychosocial stressors include environmental factors and psychological distress that influence the brain and the gut are all strongly associated with abdominal symptoms. 38
In terms of comorbidities, females hospitalized with NENs more commonly had obesity and chronic pulmonary disease. Obesity is a common risk factor for many diseases and overall poor health outcomes. It is unclear how obesity impacts the health outcomes of females with NENs. Some studies have supported that obesity is both a risk factor for developing NENs as well as a good prognostic factor with a positive correlation between survival outcomes and increasing BMI.41,42 Females also had more arthropathies and thyroid disorders which are diseases that have an inflammatory or immunological basis. Females have been shown to elicit a more vigorous adaptive immune response as compared to males due to the upregulation of cellular effectors of human adaptive immunity by estrogenic hormones which may be influenced by treatment types. 39 On the other hand, males had more hypertension, diabetes, and peripheral vascular disease which were more cardiovascular in nature. Biologically, the means by which sex modulates cardiovascular cells are complex. 43 However, increasing evidence suggests that sex-related differences in the cardiovascular system already exist at birth and are due to purely biological mechanisms, that is, genes and sex hormones. 43 It has also been shown that renin-angiotensin aldosterone system that underlies hypertension is influenced significantly by estrogen status which fluctuates throughout the menstrual cycle. 44 Taken together, sex hormones including pre- or post-menopausal status play a crucial role in symptoms and comorbidities which may influence the observed sex differences. Females also had more depression whereas males had more alcohol use disorder. These findings may be due to sex differences in behavioral responses and coping mechanisms. Despite these differences, the length of hospital stays and mortality during hospitalization were similar in both males and females.
Although these findings supported the presence of sex differences in hospitalized patients with NENs, there were several significant limitations to our study due to the information available from the data and caution should be taken for generalizability. We were only able to analyze our data by primary and metastatic NENs and not by clinical subtypes such as grade or degree of differentiation. The other major limitation of our study is the inability to specify the primary site, specific disease stages, or the treatment regimen of the patients. Since our data captures all patients admitted with the diagnosis of NENs regardless of the primary reason for admission, it is not possible to determine the specific NEN disease timeline for each patient whether they were at the onset or progression of the disease. We are also not able to correlate our findings directly with cancer-related treatments and timing, and hence not able to differentiate between symptoms related to disease or treatment. This study utilized the NIS database, but our sample is unweighted and thus caution must be taken in generalizing results to the population of the U.S. Instead, this unweighted inpatient sample provides a descriptive means to develop further dedicated studies to explore sex differences in a systematic manner for the improvement of patient management.
Although prior studies have shown sex differences in epidemiological and treatment-related side effects among patients with NENs, our study provided further insights into the modulators and characteristics that influence these differences which are primarily hypothesis-generating in approach. We also identified sex differences in specific signs, symptoms, and comorbidities which could be factors to consider when selecting treatment options. Our findings have shown convincing evidence to recommend further investigation into sex differences in patients undergoing treatment and participating in clinical trials for NENs to provide better guidance in treatment choices and clinical decision-making. Traditionally, clinical trials do not focus on sex differences when considering treatment type, treatment toxicities, comorbidities, and quality of life. Our study highlights the importance of considering sex differences when designing future clinical trials and comparing different treatment regimens and adverse effects. Based on our findings regarding diagnoses present during hospital admissions, it is worth further exploring the impact of sex differences on the overall burden of these diagnoses beyond hospital stay, since it may implore the actual impact of treatment-associated side effects and quality of life.
Conclusion
This study showed statistically significant sex differences in the demographics, diagnoses present during hospital admission, and comorbidities among the 2019 NIS hospital discharge sample of patients with NENs in the U.S. There were no sex-based differences for in-hospital mortality. Given that the incidence and prevalence of NENs are increasing with a commensurate increase in survivorship, there is an even greater need to understand the impact of sex and gender on patient outcomes such as survival and side effects, quality of life, and costs to the patient and the health care system. We advocate that cancer researchers examine sex and gender as independent variables in clinical and translational research.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241292271 – Supplemental material for Impact of sex differences on patients with neuroendocrine neoplasms during hospital admission
Supplemental material, sj-docx-1-tam-10.1177_17588359241292271 for Impact of sex differences on patients with neuroendocrine neoplasms during hospital admission by Wan Ying Tan, Laura D. Cramer, Namrata Vijayvergia, Maryam Lustberg and Pamela L. Kunz in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241292271 – Supplemental material for Impact of sex differences on patients with neuroendocrine neoplasms during hospital admission
Supplemental material, sj-docx-2-tam-10.1177_17588359241292271 for Impact of sex differences on patients with neuroendocrine neoplasms during hospital admission by Wan Ying Tan, Laura D. Cramer, Namrata Vijayvergia, Maryam Lustberg and Pamela L. Kunz in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
