Abstract
Somatostatin analogs (SSA), specifically octreotide and lanreotide, have demonstrated antiproliferative effects in patients with neuroendocrine tumors (NET), a group of rare malignancies of diverse origin and presentation. A prominent feature of NET cells is the expression of G protein-coupled receptors called somatostatin receptors (SSTR). Although these SSTR are not uniformly present in NET, they can be instrumental in the diagnosis and treatment of NET. Apart from their application in nuclear imaging and radionuclide therapy, SSA have proven invaluable in the treatment of hormonal syndromes associated with certain NET (antisecretory effects of SSA), but it took more than two decades to convincingly demonstrate the antiproliferative effects of SSA in metastatic NET with the two pivotal studies PROMID and CLARINET. The current review summarizes three decades of SSA treatment and provides an overview of the clinical trial landscape for SSA monotherapy and combination therapy, including clinical implications and quality of life aspects, as well as ongoing fields of research.
Keywords
Introduction
Neuroendocrine neoplasms (NEN) represent a heterogeneous but well-characterized group of tumors derived from endocrine glands and the diffuse endocrine system (dispersed endocrine cells). 1 Of clinical relevance is their ability to exhibit endocrine activity, with serotonin-driven carcinoid syndrome being a typical example. 1 According to the most recent 2022 WHO Classification of Endocrine and Neuroendocrine Tumors, NEN are classified based on their histomorphology and proliferative activity (i.e. mitotic count and Ki-67 index) into well-differentiated neuroendocrine tumors (NET) grade (G) 1–3 (Ki-67 cutoffs <3%, 3–20%, and >20%, respectively) and into poorly differentiated neuroendocrine carcinomas (NEC, Ki-67 index >20%). 2 This classification is of great prognostic and therapeutic relevance, as NET G1/2 usually show a less aggressive clinical course even in the metastatic stage, whereas NEC are highly aggressive carcinomas resembling small-cell lung cancer (SCLC). 3 Survival is also significantly different depending on the primary tumor location, for example, the median overall survival (OS) of metastatic NET G1/2 of the small intestine is 103 months, of the pancreas 60 months, of the lungs 24 months, and of the colon 14 months. 4
Typical features of NEN are expression of the neuroendocrine vesicle proteins synaptophysin and chromogranin A, and – depending on differentiation – the expression of somatostatin receptors (SSTR), mainly subtypes SSTR2 and SSTR5. 5 SSTR expression is critical for the management of NET, as it not only provides the basis for SSTR-specific functional imaging but also for targeted treatment with somatostatin analogs (SSA) and SSTR-based peptide receptor radionuclide therapy (PRRT).6,7 SSA constitute a standard therapy for the management of metastatic NET and can provide functional control, that is, reduction of hormone production, but also antiproliferative activity under certain conditions.5,8 For example, in slowly growing SSTR-positive metastatic NET of the small intestine and pancreas (Ki-67 index <10%), it is recommended as the first-line option by current guidelines, with PRRT, everolimus, and potentially chemotherapy (in pancreatic NET) as later lines of therapy. 5 Considering that SSA were originally approved in the late 1980s for functional control of hormonally active NET, 9 this represents one of the earliest and most elegant examples of targeted therapy in today’s era of personalized precision oncology. Octreotide (OCT) and lanreotide (LAN) have now been established for the antiproliferative treatment of NET with different areas of application. Several other agents and formulations are being evaluated either as monotherapy or in combination with other compounds. However, despite the broad applicability and the well-known safety profile of these compounds, there still remain open questions regarding the impact of dosing, heterogeneity of SSTR expression, differences in tumor location [e.g. lung versus gastrointestinal (GI)], and the value of combination therapies. This review provides a concise overview of the biological background to the clinical applicability of SSA as antitumor therapy in metastatic NET, covering clinical trial results, state-of-the-art practice, and also patient-reported outcomes.
SSAs – physiology and biological background
Biological background of somatostatin
From a physiological point of view, somatostatin (SST) is a small cyclic peptide, historically known as somatotropin release-inhibiting factor or growth hormone (GH)-inhibiting hormone, with a wide range of inhibitory effects throughout the human organism, including suppressive effects on the GI and endocrine systems as well as modification of neurotransmission and immunomodulatory effects. 10 Relevant effects on the endocrine system include anti-secretory effects on a wide range of hormones, for example, pituitary hormones (GH, prolactin, thyrotropin) and different gastroenteropancreatic (GEP) hormones (insulin, glucagon, pancreatic polypeptide, cholecystokinin, gastric inhibitory peptide, gastrin, secretin, neurotensin, and motilin). 11 In addition, SST also exerts effects on the exocrine system including inhibition of hydrochloric acid, pepsinogen, and intrinsic factor in the upper GI tract, decreased pancreatic enzyme and bicarbonate production, and reduced bile secretion. 11 The complex physiological tasks of this small peptide are reflected in its multiple production sites, including the stomach, intestine, pancreas (delta cells), and the central and enteric nervous system. 12
SST acts by binding to one of five distinct SSTR (SSTR1–5), which belong to the superfamily of G protein-coupled receptors (GPCR). 13 These receptors are physiologically expressed in a variety of organs, including the GI tract, pancreas, hypothalamus, and pituitary gland but also other organs such as the kidneys, adrenal gland, lung, and thyroid. 13 Apart from the physiological expression, SSTR are also found in a wide range of neoplasms, including meningiomas, breast carcinomas, lymphomas, paragangliomas, hepatocellular carcinomas, prostate carcinomas, sarcomas, GH-secreting pituitary adenomas, and NETs. 14 Importantly, the half-life of physiological SST is short at only 1–3 min, underlining the importance of developing more stable analogs for clinical practice. 15 The biological background and, in particular, the signaling pathways associated with SST have been highlighted in several review articles that may be accessed for more detailed information.11,16,17
SSTR expression in NET
SSTR expression represents one of the most important and best-characterized features of NEN, with SSTR2 and SSTR5 being the most commonly expressed and preferentially targeted subtypes.18,19 SSTR expression varies according to the location of the primary tumor with up to 100% presence in well-differentiated midgut NET and up to 90% in pancreatic NET, while being less common in lung NET (e.g. immunohistochemical expression of SSTR2 in 30.8% and SSTR5 in 7.7% of lung NET versus SSTR2 in 81.8% and SSTR5 in 32.7% of GEP-NET, according to one larger study),16,20 and grading (SSTR2A presence in 96–100% of NET G1 and G2 versus 64–71% in NEN G3 specimens, according to another study). 21 Importantly, the detection of SSTR expression can differ using immunohistochemistry (IHC) versus somatostatin-receptor scintigraphy (SRS) or SSTR-positron emission tomography/computed tomography (PET/CT).20,22 These discrepancies in SSTR assessment can pose some pitfalls when prescribing SSA, which was particularly reported for lung NET. Data from Diakatou et al. 20 and other studies show a concordance rate of 64–92.9% for (lung) NEN, comparing IHC and SRS. Even more discordant results were seen in 11/29 pulmonary NET patients (38%) in a study from our center comparing IHC and SSTR-PET/CT. 22 Current guidelines such as the European Society for Medical Oncology (ESMO) and the European Neuroendocrine Tumor Society (ENETS) recommend SSTR2/5 assessment by SSTR-PET/CT/SRS or IHC in the absence of functional imaging as a prerequisite for the antiproliferative use of SSA, also assuming that imaging allows a more comprehensive illustration of SSTR expression in NET patients when compared to IHC.5,23 Strikingly, given the great significance of SSA in NET, only a few disease-related mutations in the SST/SSTR genes are known, hence the reduced SSTR expression observed in SSA-resistant tumors has presumably a different molecular basis. 13 In addition, some patients with symptoms of carcinoid syndrome experience tachyphylaxis, that is, a diminished response to SSA, which might stem from SSTR desensitization/downregulation or other potential mechanisms.24,25 Of note, glucocorticoids have been linked to a suppressed SSTR2 expression, which may explain the low efficacy of SSA in adrenocorticotropic hormone (ACTH)-secreting NET. 26
Antiproliferative effects and development of SSA
The development of synthetic SSA dates back to the early 1980s and octreotide (OCT), an octapeptide (as opposed to the original 14 amino acids in physiological SST), was the first synthetically produced SSA to be launched on the market in 1988 followed by the approval of a long-acting formulation a decade later (i.e. octreotide long-acting release formulation, ‘OCT–LAR’).17,27 While having a comparable mode of action to physiological SSA, OCT has a longer half-life, as it is less sensitive to degradation. 27 In addition to its symptomatic effects in functional GEP-NET and carcinoid syndrome in particular, it has been shown to significantly reduce GH and insulin-like growth factor-1 (IGF1) levels in acromegaly patients, in whom the long-acting release formulation was evaluated in the initial development process. 28 The cyclic octapeptide lanreotide (LAN) is an alternative synthetic SSA with comparable effects that was launched somewhat later in the early 1990s. 17 OCT and LAN both show a similar binding profile, with potent affinity for SSTR2, moderate affinity for SSTR5, and lower affinity for SSTR3. 13
Apart from their approval for the treatment of acromegaly and symptomatic control of hormonal syndromes in GEP-NET, SSA has proven useful in other diseases, including digestive fistulas, acute bleeding from esophageal varices, dumping syndrome, and some pituitary adenomas. 17 Given the pleiotropic inhibitory effects of SSA in vivo and in vitro, it appeared reasonable to assume that the paracrine and autocrine inhibitory effects of SST (either directly on the tumor cell itself through SSTR or by indirect hormonal, antiangiogenic, or immunomodulatory effects) 25 could also have suppressive effects on tumor growth in NET. However, it took more than two decades to confirm this theory with the pivotal studies for OCT and LAN as antitumor agents for NET. The antiproliferative effect of SSA has also been explored in other tumor entities but has not achieved comparable clinical significance despite some early positive data. 29
Interestingly, an analysis of the Surveillance, Epidemiology, and End Results (SEER) database evaluating survival and outcomes of NET patients over time found that OS of patients with disseminated disease increased significantly when comparing patients diagnosed between 1973 and 1987 with patients diagnosed later, demonstrating an improvement in OS from a median of 18–39 months (p < 0.001). 3 This was mainly attributed to the market introduction of OCT, which is further supported by the fact that no increase was noted in the cohort of patients with locoregional disease. 3 This had already been suggested by a 2001 Dutch epidemiological study reporting that patients with metastatic carcinoid tumors diagnosed from 1992 onward had a significantly longer median OS (43 versus 24 months, p = 0.012) than patients diagnosed before 1992 when OCT became available as a symptomatic treatment in the Netherlands. 30
Methods
This literature review applied a comprehensive search strategy for prospective studies involving SSA based on the following search terms on PubMed: [neuroendocrine tumors (MeSH Terms)] AND (octreotide or lanreotide) OR [somatostatin (MeSH Terms)] AND (prospective or phase). In total, 1614 PubMed records were obtained on 10/04/2023, and 80 prospective studies met the search criteria, covering the areas of SSA monotherapy and combination therapy, quality of life (QoL) and symptom control, and various other topics. However, this search method missed some older studies which were not labeled ‘prospective’ or ‘phase’. Hence, search results were supplemented by 13 smaller and older (before 2006) prospective trials analyzed in reviews by Modlin et al. (2010) and Sidéris et al. (2012) who used a broader search strategy.31,32 The following sections and tables provide an overview of prospective studies investigating SSAs in metastatic NET. Retrospective studies that have been implemented for further context are clearly marked as such.
SSA monotherapy in NET
Short-acting SSA for antiproliferative treatment of NET
Following initial results on the growth-inhibiting properties of OCT,33–35 a phase I study published in 1993 investigated OCT (1500–6000 µg) in 14 patients with midgut NET and LAN (2250–9000 µg) in 13 NEN patients of various origin including two SCLC patients. 36 These early data already underlined the excellent toxicity profile of SSA as injection volume and subsequent local reaction were defined as the main dose-limiting events of OCT, while further systemic side effects were negligible. 36 Interestingly, the authors reported not only an overall disease control rate (DCR; i.e. percentage of complete/partial response or stable disease for a certain number of months, varying between studies) of 46% and 38% for OCT and LAN, respectively, but also radiological changes in terms of tumor necrosis in 5/13 (OCT) and 6/13 (LAN) patients evaluated and even a partial remission (PR) in 4 patients for each SSA, including one SCLC patient. 36 Although these objective response rates (ORRs) were not reproducible in large studies and SSA are generally considered to act primarily by disease stabilization, and radiological criteria used at that time do not appear comparable to current standards, these initial data supported further research into the antitumor efficacy of SSA in NET patients. Subsequent phase II trials have used varying doses from 400 to 3000 µg of OCT per day (fractionated to 2–3 daily doses) and have included a wide variety of NET entities, with DCRs of up to 50% reported.37,38
One of the first more detailed results came from the German Sandostatin Multicenter Study, in which a total of 103 patients with different primary tumor locations and endocrine symptoms were treated with 200 µg of OCT three times daily and were monitored for tumor-inhibitory effects over 1 year. 39 In the subgroup of 52 patients with prior confirmed progression, disease control was documented in 36.5% with a median duration of stable disease of 18 months. 39 By contrast, a DCR of 53.8% was reported for the group with prior stable disease. 39 Comparable results were achieved by an Italian multicenter study of 58 patients, with only 27 patients (47%) achieving disease stabilization for at least 6 months, while the majority experienced complete or partial remission only with respect to symptomatic and biochemical control. 37 Several studies have tried high and ‘ultra-high’ doses of short-acting SSA but a reduction in tumor size was seen only in individual patients (in around 5%).40,41
With regard to the combination of short-acting SSA with interferon-alpha, the main previously established NET therapy, Arnold et al. 42 investigated OCT versus OCT plus interferon-alpha in 105 patients in 2005. While there was no difference in the primary endpoint time to treatment failure [either disease-related death, tumor progression, or intolerable adverse events (AEs)], there were significantly more treatment failures due to AEs in the combination arm, and a statistically significant improved global QoL for OCT compared to the combination arm was demonstrated. 42 Comparable results were observed in a smaller randomized trial comparing LAN versus interferon-alpha versus the combination, reporting roughly equal efficacy but more toxicity for the combination. 43 By contrast, a third randomized study suggested a progression-free survival (PFS) benefit when combining OCT and interferon-alpha compared to single-agent OCT, however, in a collective of patients who had all received extended local pretreatment, that is, surgery and hepatic arterial embolization of liver metastases. 44 Given the broad applicability and better-defined antiproliferative effects of long-acting SSA discussed next, this combination is currently limited to application in cases of refractory carcinoid syndrome.
Long-acting SSA for antiproliferative treatment of NET
For both OCT and LAN, more durable formulations were licensed in the 1990s (long-acting release OCT, OCT-LAR; slow release LAN, LAN-SR) and in the following years assessed for antitumor efficacy in NET. 17 For example, in 1999, a larger phase II study on LAN-SR 30 mg every 14 days involving 55 patients with functioning tumors revealed high biochemical/clinical response rates (>50%) and disease stabilization in 25/31 evaluable patients. 45 Regarding the toxicities of this new formulation of SSA, an increased number of (asymptomatic) gallstones was noted (n = 8/30, 27% of assessable patients), leading to an initial warning regarding this symptom. 45 This is presumably due to reduced gall bladder motility and resulted in a recommendation for concurrent cholecystectomy if surgery for a primary tumor was planned.13,45,46 The clinical benefit of long-acting SSA has been demonstrated in several prospective trials evaluating LAN and OCT for antitumor activity (see Table 1).47–54 OCT-LAR is recommended at 30 mg every 4 weeks for antiproliferative purposes, which was also the first regimen to achieve approval based on the PROMID study published in 2009. 55 For LAN, an evolved sustained formulation was chosen for further development, known as LAN Autogel (LAN-AG 120 mg every 4 weeks). 17 Prior to the pivotal LAN trial CLARINET published in 2014, LAN-AG 120 mg every 6 weeks was compared to LAN-microparticles in a phase III study, showing similar efficacy for both, and LAN-AG was also studied in a single-arm phase II study at 120 mg every 28 days.56,57
Prospective studies investigating the antitumor efficacy of SSA monotherapy.

CAAG, chronic autoimmune atrophic gastritis; DCR, disease control rate; GC1, type-1 gastric carcinoids; LAN, lanreotide; LAR, long-acting repeatable; MEN1, multiple endocrine neoplasia type 1; NET, neuroendocrine tumor; No., number; NR, not reached; OCT, octreotide; OLE, open-label extension; OS, overall survival; PBO, placebo; PFS, progression-free survival; SSA, somatostatin analogs; SR, slow release; SSTR, somatostatin receptor; TTP, time to tumor progression.
Comparative data on LAN-AG and OCT-LAR
In general, guidelines make no distinction between OCT and LAN in terms of their antiproliferative efficacy (‘drug class effect’), but study populations in the two pivotal trials differed considerably (e.g. primary tumor location and grading) and resulted also in slightly different approvals by the authorities for each compound.5,7,8 Reliable comparative data on the efficacy of LAN and OCT are lacking, a Spanish R-GETNE registry study (n = 535) showed similar effects of LAN-AG and OCT–LAR on PFS [hazard ratio (HR) of 0.9 for LAN versus OCT, 95% confidence interval (CI): 0.71–1.12]. 81 Furthermore, although not systematically studied, there are anecdotal reports that switching between SSA compounds may result in varying degrees of response.51,82 Pharmacologically, OCT and LAN have a similar affinity for SSTR2 (IC50 0.4 versus 0.75 nmol/L) and SSTR5 (IC50 5.6 versus 5.2 nmol/L), but their long-acting formulations have different pharmacokinetic profiles (OCT–LAR: prolonged plateau phase for about 30 days after an initial increase and a lag phase; LAN-AG: peak concentration on day 1 and then elimination with a half-life of 25.5 days).13,83 The main practical difference with regard to these formulations is the route of administration, as OCT is administered intramuscularly and LAN by deep subcutaneous injection. 5 A recent review highlighted that the injection experience with LAN-AG was preferable to OCT-LAR according to two studies, one of which surveyed nurses and the other patients. 84 Apart from lower rates of treatment discontinuation due to AEs with LAN-AG (3% versus 12% in the respective phase III studies), the safety profile is generally comparable.55,68,81,84 Detailed analyses of the pivotal studies are presented in the following section.
Randomized placebo-controlled trials
Given the rather heterogeneous and often indolent growth behavior of NET, the interpretation of early SSA studies is severely limited by the lack of a placebo group. The first study to conclusively demonstrate that SSA can inhibit tumor progression in NET was the 2009 phase IIIb study PROMID. 55 In this study, treatment-naïve midgut NET patients were randomized to receive OCT-LAR 30 mg or placebo every 4 weeks, with the core study results reported for 85 patients. 55 The primary endpoint time to tumor progression (TTP) was significantly improved at a median of 14.3 months in the OCT group versus 6 months for placebo (HR: 0.34, 95% CI: 0.20–0.59), and 66.7% versus 37.2% experienced disease stabilization at 6 months. 55 Points of discussion include the small number of patients with high liver tumor burden (64/85 patients had less than 10% liver tumor burden) and the predominant G1 collective (>95% in the OCT arm) as well as heterogeneity in terms of functionality (40% with endocrine activity, but there was a similar treatment effect for both groups in the per-protocol subgroup analyses). 55 Particularly, the evidence regarding grading does not reflect the approval status. To date, SSA have failed to demonstrate an OS benefit in prospective studies, most likely related to the high cross-over rates. For the PROMID results, analysis of long-term outcomes also showed no difference in median OS with 84.7 months for OCT and 83.7 months for placebo (HR: 0.83, p = 0.51). 63 Hepatic tumor burden was a strong prognostic factor with 107.6 months median OS in the low tumor burden group and 57.5 months median OS in the high tumor burden group, and the benefit of OCT was mainly seen in patients with low tumor burden (statistically not significant risk reduction of 41%). 63 An interesting post hoc analysis for OCT–LAR was derived from the RADIANT-2 study, which compared everolimus plus OCT with OCT alone. 64 In an analysis of 196 patients treated with OCT alone (41 SSA-naïve), the median PFS for untreated patients was 13.6 months overall and 22.2 months for midgut patients, and 11.1 and 12.0 months, respectively, when pretreated. 64 Median OS was 50.6 and 33.5 months for treatment-naïve and pretreated patients. 64 These data are of interest as tumor progression within 12 months was a prerequisite for inclusion in this study – unlike in the PROMID trial – and although they are only from one study arm, these are very strong data indicating an effect also in patients with progressive NET.
The landmark pivotal study for LAN-AG was the randomized, placebo-controlled phase III CLARINET trial, which remains the largest study on SSA enrolling 204 NET patients. 68 Inclusion criteria were nonfunctioning NET G1/2 with a Ki-67 <10% and a primary located in the pancreas, midgut, hindgut, or with unknown GI primary. 68 Notably, and compared to the PROMID study, most patients (96%) had stable disease in the preceding 3–6 months, and more patients had hepatic tumor volume >25% (33% of patients). 68 The primary endpoint PFS (defined as time to progression/death within 96 weeks) was significantly longer in the LAN group than in the placebo group (median not reached versus 18.0 months; HR: 0.47, 95% CI: 0.30–0.73). 68 In the preplanned subgroup analyses, LAN also achieved improved PFS in patients with NET G2 or patients with higher liver tumor volume (>25%). 68 Subsequently, the CLARINET open-label extension (OLE) study reported a median PFS for LAN of 32.8 months while it also corroborated the favorable long-term safety. 85 The AE data from this study are particularly interesting because only patients with nonfunctioning NET were included, so off-target side effects of SSA, owing to the ubiquitous SSTR expression, especially in the GI tract, are not obscured by the symptoms of carcinoid syndrome. In the core study, treatment-related diarrhea and abdominal pain were common (26% and 14% versus 9% and 2% in the placebo group, respectively), 68 but the OLE study also indicated that AEs generally improved with increasing treatment duration. 85 Notably, while decreased levels of pancreatic enzymes were identified in only 5% of patients on LAN in CLARINET, 68 two prospective studies demonstrated that the incidence of exocrine pancreatic insufficiency can be quite high (20–24%) during long-term SSA therapy.86,87 Furthermore, the extension study provided data on a delayed SSA treatment start, basically for a watch-and-wait approach, as it reported a median PFS of 14.0 months for the 32 NET patients who had PD in the core study and received LAN thereafter. 85 Again, no significant difference in OS was found in CLARINET, but cross-over or other treatments upon disease progression render this comparison challenging. 68
A relevant point of discussion is the limited information on individual tumor growth kinetics in these trials, given the often indolent course of NET with even occasional spontaneous regression reported. For instance, pooled data from 531 NET patients receiving placebo in RCT showed an ORR of 1.52%, a DCR of 52.7%, and a tumor shrinkage rate (any reduction) of 25.2%. 88 On the other hand, a post hoc analysis of the CLARINET trial data looking at the tumor growth rate at baseline found that tumors were growing at a median of 2.1%/month (LAN) and 2.7%/month (placebo), despite being classified as stable disease according to RECIST. 89 That highlights the significance of baseline progression status and growth activity in interpreting ORR and PFS results and it would be valuable to receive this information in future studies.
Further aspects of SSA monotherapy
High-dose SSA
Recently, the phase II CLARINET FORTE trial assessed the PFS of 99 patients with midgut or pancreatic NET G1/2 receiving LAN at a reduced interval (every 14 days) after disease progression on first-line LAN every 28 days. 58 This study showed that an increased dosing frequency could be considered in these patients prior to second-line therapy, reporting an additional 8.3 (midgut NET) and 5.6 months (pancreatic NET) of median PFS and stable disease as a best response in two-thirds of patients, while no new toxicity signals emerged and QoL did not deteriorate. 58 However, patients with a high Ki-67 index (>10%) had a much reduced median PFS (2.8 versus 8.0 months in pancreatic NET and 5.5 versus 8.6 months in the midgut group). 58 Likewise, a recent meta-analysis on high-dose SSA evaluated this study and 10 others, and it also concluded that this approach is only recommended in selected patients. 90 On the other hand, in the case of refractory carcinoid syndrome and stable disease, above-label doses of SSA are common practice and should be considered according to current ESMO and ENETS guidelines.5,8,91
Alternative SSA compounds
The cyclohexapeptide pasireotide is a novel SSA that has a 30- to 40-fold higher binding affinity for SSTR1 and SSTR5 than OCT and LAN. 92 Whether this translates into better symptom and tumor control has been investigated in phase II and phase III trials. Kvols et al. 69 studied symptom relief with pasireotide 150–1200 µg twice daily in NET patients with symptoms refractory or resistant to OCT, observing at least partial symptom control in 27% of patients and barely missing the predefined success threshold of 30%. A phase III trial comparing pasireotide LAR 60 mg versus OCT-LAR 40 mg every 28 days in 110 patients was stopped early because the primary endpoint symptom response was similar in both study arms. 66 Notably, the post hoc analysis of PFS showed a significantly longer median PFS of 11.8 months for pasireotide than for OCT with 6.8 months (HR: 0.46, 95% CI: 0.20–0.98). 66 This is similar to the median PFS of 11 months reported for the 29 treatment-naïve patients with NET included in the phase II study of Cives et al. 67 As discussed in more detail below, adding pasireotide to everolimus failed to elicit any additional PFS benefit. 93 Critically, the incidence of drug-related hyperglycemia was high with pasireotide in all studies, that is, 28.3% (pasireotide) versus 5.3% (OCT) in the phase III trial, 79% (any grade) and 14% (grade 3) reported by Cives et al., and 15.6% with twice-daily pasireotide in the study by Kvols et al., requiring patients to take anti-hyperglycemic medication (45% of all patients in the trial by Cives et al.) and limiting its potential as a first-line option.66,67,69 On the other hand, the hyperglycemic effects of pasireotide can be harnessed in malignant insulinoma, and case reports show promising results, as mentioned in the ENETS 2023 guidelines.94,95
SSA therapy in lung NET
The phase III SPINET trial was planned to provide prospective evidence for the use of SSA in SSTR-positive bronchopulmonary NET but was stopped prematurely due to slow enrollment (also following the incorporation of SSA in the guidelines at that time). 96 Results of the reduced overall cohort of 77 patients (versus 216 initially planned) yielded only a small numerical PFS benefit for LAN, that is, 16.6 versus 13.6 months, with subgroup analysis suggesting no benefit for atypical carcinoids. A subgroup analysis for lung NET in the RADIANT-2 trial is also available, which provides data regarding the use of OCT in these patients. 97 Even though the prospective evidence is more limited than for GEP-NET, current European guidelines suggest SSA as an option in slowly progressing SSTR-positive lung/thymic NET.98,99
SSA in high-proliferative NEN
The evidence for the antiproliferative use of SSA in NET with a Ki-67 >10% particularly in NET G3 is mostly retrospective and limited.84,100 For instance, Merola et al. 101 reported a median PFS for patients with pancreatic NET G2 (Ki-67 >10%) of 12.4 months and for pancreatic NET G3 of 4 months. The ongoing NETTER-2 trial (NCT03972488) may provide insights regarding therapy with SSA in NET G2/3 as it is evaluating the efficacy of 177Lu-Dotatate plus OCT-LAR versus high-dose OCT-LAR alone in patients with GEP-NET of grade 2 or 3 (Ki-67 ⩾10 and ⩽55%). For NEC, small studies tried to combine SSA with standard chemotherapy, with however, unclear add-on value. 102 While the ESMO guidelines only recommend SSA for tumor growth control in NET with a Ki-67 ⩽10%, the National Comprehensive Cancer Network (NCCN) guidelines consider SSA as a potential option in selected patients with NET G3 and favorable biology (low Ki-67 and SSTR positive).5,103 In NEC, however, SSA should not be used for antiproliferative purposes, as stated by the respective ENETS guideline, for instance. 104
Maintenance treatment with SSA
The particularly benign toxicity profile of SSA makes these substances not only interesting for combination therapies but also provides a rationale for their use as maintenance therapy following a (potentially more toxic) induction therapy. The explicit use of LAN maintenance was explored in the recent REMINET study. 105 This trial studied aggressive duodeno-pancreatic NET and compared LAN maintenance with a placebo after pretreatment with various first-line therapies, including chemotherapy (based on temozolomide, dacarbazine, streptozotocin, oxaliplatin) and sunitinib. 105 Results showed an improved PFS at 6 months (73.1% versus 54.2%) and unaffected QoL in both arms so it can be cautiously assumed that this concept could possibly be beneficial. 105 The strategy of a maintenance SSA phase was also pursued in several trials on SSA combination strategies, such as the SONNET study (SSA maintenance after 6 months of temozolomide/LAN in GEP-NET) 106 or even in NEC in the phase II IPO-NEC, which treated GEP-NEC patients with irinotecan/cisplatin followed by monthly OCT. 107
SSA combination therapy in NET
In the last decade, there has been increasing interest in evaluating SSA combination treatments, including combination therapies with interferon, everolimus, PRRT, and chemotherapy due to the low intrinsic toxicity of SSA. However, while no limiting toxicities have been observed, more studies are needed to demonstrate the true additive effect of SSA on other active compounds and to establish whether a sequential treatment approach or combination therapies are preferable. In many centers, it is common clinical practice to continue SSA in NET patients who progress on SSA (if well tolerated), but there is a lack of evidence to strongly support this, especially in nonfunctioning tumors. 84 Furthermore, there is little evidence to currently recommend routine combined use of SSA and other therapies. 84 In the following section, we discuss the available prospective evidence for a selection of combination strategies in the context of advanced or metastatic NET.
Everolimus
Due to the broad use of mammalian target of rapamycin (mTOR) inhibitors in NET, the combination of everolimus and SSA occupies a special position. The pivotal RADIANT-3 and RADIANT-4 studies led to the approval of everolimus in pancreatic and nonfunctional GI/lung NET based on a PFS benefit compared to a placebo.108,109 Interestingly, the phase III RADIANT-2 study investigated the combination everolimus/OCT versus placebo/OCT in progressive functional NET and could not demonstrate a significant PFS (16.4 versus 11.3 months, HR: 0.77, 95% CI: 0.59–1.00) or OS (29.2 versus 35.2 months, HR: 1.17, 95% CI: 0.92–1.49) advantage for everolimus treatment.110,111 However, an additive antitumor effect of everolimus plus OCT could be assumed in terms of tumor shrinkage (75% versus 45% of assessable patients had any decrease, not as defined by RECIST). 110 In 2019, the single-arm EVERLAR study showed similar results for this combination in the setting of progressive nonfunctional GI-NET (12-month PFS rate of 62.3%). 112 Furthermore, the LUNA trial investigated pasireotide/everolimus in progressive lung/thymic NET, reporting a higher proportion of patients without progression at month 9 for the combination than for the two single-agent groups (58.5% versus 39.0% and 33.3%). 113 However, the everolimus-only group had the longest PFS (12.5 months in the everolimus group versus 11.8 months for the combination group and 8.5 months for pasireotide alone). The phase II COOPERATE-2 trial showed no benefit of adding pasireotide to everolimus treatment in terms of PFS in patients with progressive pancreatic NET. 93 Notably, there is another phase II study of OCT co-administered with everolimus for first-line treatment in GI/lung NET, reporting a high ORR (primary endpoint) of 18%,114,115 which is in contrast to other studies of monotherapies.55,108 In summary, while there is a preclinical basis (i.e. IGF pathway) for assuming an additive or synergistic antitumor effect of SSA/everolimus,116,117 the results from these studies are still insufficient to merit a general recommendation for this combination in NET, as stated in the ESMO guidelines, although progressive functional pancreatic NET may be an exception. 5
Chemotherapy
Given the excellent tolerability of SSA, combination with various classical cytostatic drugs is not a limitation. A concept that has been repeatedly investigated in recent years is the combination of SSA with the increasingly applied alkylating agent temozolomide. In the phase II SONNET trial (so far only published as an abstract), this combination resulted in a DCR of 73.5% at 6 months in progressive GEP-NET patients. 118 Following 6 months of LAN maintenance, 71.4% of patients with nonfunctioning NET had stable disease or PR versus 41.7% of patients under observation. 118 In the comparably designed phase II ATLANT trial, concomitant LAN/temozolomide showed a lower benefit in progressive lung/thymus NET patients (DCR at 9 months of 35%), 119 but both trials lacked a control arm. Also, classical GI cancer chemotherapy regimens may safely be combined with SSA and have been evaluated in different phase II studies. For example, monthly OCT-LAR was studied in phase II trials in combination with daily continuous 5-fluorouracil infusion 120 or with daily capecitabine (plus biweekly bevacizumab). 121 The results so far from these two trials (partial responses in 17.8–24.1% and median PFS/TTP of 14.9–22.6 months) indicate good antitumor activity and drug tolerability120,121 and might support a treatment rationale that is based on the antiangiogenic effects of metronomic administration of cytotoxic drugs. 122 In conclusion, cytostatic drugs can be safely combined with SSA but the additive effect remains currently largely unknown.
Bevacizumab
The anti-vascular endothelial growth factor (anti-VEGF) antibody bevacizumab has repeatedly been evaluated in NET, however, with yet mostly unclear clinical value. As for combining bevacizumab with SSA, the SWOG S0518 study randomized 427 NET G1/2 patients to bevacizumab or interferon-α-2b, both in conjunction with OCT-LAR 20 mg every 21 days but could not show a PFS benefit for one drug over the other (16.6 months for the bevacizumab arm versus 15.4 months for the interferon arm, HR: 0.93, 95% CI: 0.73–1.18). 123 The comparator, interferon-α plus OCT, had already been shown to be significantly superior to OCT alone (at 100–200 µg 2–3× daily) in terms of the risk of tumor progression (HR: 0.28, 95% CI: 0.16–0.45) in metastatic midgut NET but this did not translate in longer survival (5-year survival 56.8% versus 36.6%). 44 Thus, bevacizumab plus SSA can be assumed to have an antiproliferative effect. In a phase II study by Bendell et al., 124 pertuzumab showed a high overall response rate of 16% when co-administered with bevacizumab and OCT in progressive NET.
Tyrosine kinase inhibitors
Phan et al. 125 studied pazopanib plus OCT in 52 pancreatic and GI NET patients and achieved an ORR of 21.9% for pancreatic NET, but the observed tumor reduction was not attributed to SSA. Currently available only as an abstract, the results from the phase II/III AXINET study favor OCT plus axitinib compared with OCT plus placebo (ORR 17.5% versus 3.8%, p = 0.0004; median PFS 17.2 versus 12.3 months per investigator assessment, p = 0.169) in the 256 patients with progressive extrapancreatic NET G1/2. 126
Peptide receptor radionuclide therapy
PRRT can currently be considered as a combination therapy in conjunction with SSA because the best available evidence for its efficacy comes from the phase III NETTER-1 trial, in which PRRT plus OCT (co-administered for four PRRT cycles and then as monthly maintenance doses) was superior in terms of PFS to monthly high-dose OCT alone in advanced/metastatic progressive midgut NET, also achieving an ORR of 18% versus 3%.127,128 However, the role of SSA in relation to PRRT, as a combination treatment or as maintenance therapy, is still largely unclear. Although the results of the NETTER-1 study suggest that all cases should receive combination treatment (as specified by the study protocol), the ESMO guidelines recommend combining SSA with PRRT and continuing SSA beyond PRRT in functioning tumors but do not advocate combination treatment for nonfunctioning NET, for which they also point out the absence of evidence regarding maintenance therapy. 5 A retrospective analysis by Yordanova et al. 129 demonstrated that patients with PRRT monotherapy had a significantly shorter median PFS and OS than patients with PRRT plus SSA (combined or as maintenance). Conversely, a recent study by Syguła et al. 130 could not show a PFS or OS benefit for SSA maintenance after disease control achieved by PRRT. In detail, this single-center study randomized 115 patients with nonfunctional NET and with stable disease, partial response, or complete response following PRRT in a 2:1 fashion to SSA or best supportive care. 130 Thus, no definite conclusion can be drawn about the efficacy of this sequential treatment approach in functional NET patients or patients with progressive disease after PRRT.
QoL impact of SSA in NET
Patients with NET have a reduced health-related QoL, particularly when symptoms of carcinoid syndrome (flushing and diarrhea) are present. 131 The symptomatic benefit of SSA in the treatment of malignant carcinoid syndrome was established four decades ago. 132 The observation that palliation of symptoms could also relevantly impact QoL metrics was made as early as 1995 by a placebo-controlled, cross-over study, in which two out of five QoL domains of the Psychosocial Adjustment to Illness Scale (PAIS) were significantly improved in the 11 patients included. 133 While the value of symptomatic control in functional active NET by SSA seems obvious and is not further explored here, it also seems of interest to investigate the impact of SSA on QoL in the antiproliferative therapeutic setting of SSA therapy, and Table 2 provides an overview of observational and interventional studies with QoL assessment in NET.
Prospective SSA trials with HRQoL information.

CGI-S, clinical global impression of severity; CI, confidence interval; EORTC QLQ, European Organization for Research and Treatment of Cancer Quality of Life Questionnaire; FACIT-D, Functional Assessment of Chronic Illness Therapy–Diarrhea; GHQ, General Health Questionnaire; GI, gastrointestinal; HRQoL, health-related quality of life; ISPN, Index de Santé Perceptuel de Nottingham; LAR, long-acting release; No., number; NR, not reached; OCT, octreotide; PAIS, Psychosocial Adjustment to Illness Scale; PEI, pancreatic exocrine insufficiency; PRO, patient-reported outcomes; PROMIS-29, Patient-Reported Outcomes Measurement Information System 29; PRRT, peptide receptor radionuclide therapy; QoL, quality of life; SI-NET, small intestinal neuroendocrine tumor; SSA, somatostatin analogs; TDD, time to definitive deterioration; TTD, time to deterioration.
The most frequent QoL assessment tool in the identified prospective SSA studies was the EORTC QLQ-C30, which was employed in 15/19 studies. The NET-specific instrument EORTC QLQ G.I.NET21 was used in 6/19 trials. In general, the QoL results of these studies are difficult to compare due to differences in the design and duration of the study and QoL assessment as well as in reporting. This heterogeneity was also noted by a systematic review of studies reporting QoL in GEP-NET patients, which equally concluded that these QoL studies were sparse and often methodologically lacking. 142 Furthermore, to assess whether QoL outcomes constitute a sufficient improvement, for example, a statistically and clinically significant improvement in the global health status and not merely in a specific subscale, objective tools such as the ESMO Magnitude of Clinical Benefit Scale (ESMO-MCBS) QoL checklist can be used. 143 For instance, in the two placebo-controlled phase III trials CLARINET and PROMID, no significant differences in the EORTC QLQ-C30 scores between the two groups and compared to the last assessment/after 6 months were reported.55,68 However, a post hoc QoL analysis of the PROMID trial could show that OCT resulted in fewer/later deteriorations (measured by the time to definitive deterioration) with regards to fatigue, pain, and insomnia and that diarrhea was improved in the OCT group and worsened in the placebo arm. 136 Arnold et al. 42 demonstrated in a 2005 study that patients randomized to OCT had improved global QoL, whereas patients randomized to an interferon-alpha/OCT combination had impaired QoL after 3 months of treatment (+11.4 ± 18.6 and −6.4 ± 18.6, respectively, p = 0.003). In comparison, the phase III ELECT study was specifically designed to evaluate the efficacy of LAN versus placebo in terms of carcinoid syndrome treatment, with the primary endpoint being the percentage of days requiring rescue treatment with short-acting OCT. 137 Resulting from the study design, rescue with SSA or various antidiarrheals could have diminished the QoL differences. Nevertheless, apart from a statistically significant difference in the primary outcome, there was a greater improvement from baseline to week 12 in the LAN group in the EORTC-QLQ Global health status/QoL and in the G.I.NET21 GI as well as endocrine symptoms. 137 In addition, compared with more toxic and aggressive therapies that may have a greater adverse impact on QoL, it is noteworthy that SSA do not appear to worsen QoL even when used over a longer period of time (CLARINET), at shortened dosing intervals (CLARINET FORTE study), or as maintenance therapy (REMINET).58,68,105 Finally, the QoL results from SSA studies should also be interpreted in the context of QoL outcomes from other NET treatment options, for instance, listed in a recent systematic review by Gosain et al. 144
Conclusion
This review summarizes over three decades of clinical trials studying the various clinical aspects of applying SSA in patients with metastatic NET of different origins. SSA as antiproliferative compounds have a strong biological rationale and an exceptional safety profile, and their disease-stabilizing (but not tumor-reductive) effects have now been clearly established by phase III trials, making them the cornerstone of (antiproliferative) first-line treatment for indolent functioning as well as nonfunctioning metastatic NET. Apart from their clear-cut role as an antineoplastic first-line therapy for SSTR-positive GEP-NET with Ki-67 <10%, their role in NET of other origins is less clearly defined despite inclusion in various guidelines, even in the absence of solid clinical data. Next to this antiproliferative indication, SSA are also the treatment of choice to ameliorate hormone-related symptoms in NET. Open questions such as their role in combination therapies, the impact of novel diagnostic techniques like liquid biopsies, or the value of SSA use beyond progression are currently being investigated in prospective studies, which are summarized in Table 3.
Clinical trials involving SSA in the treatment of NEN are listed as currently/not yet recruiting on ClinicalTrials.gov.
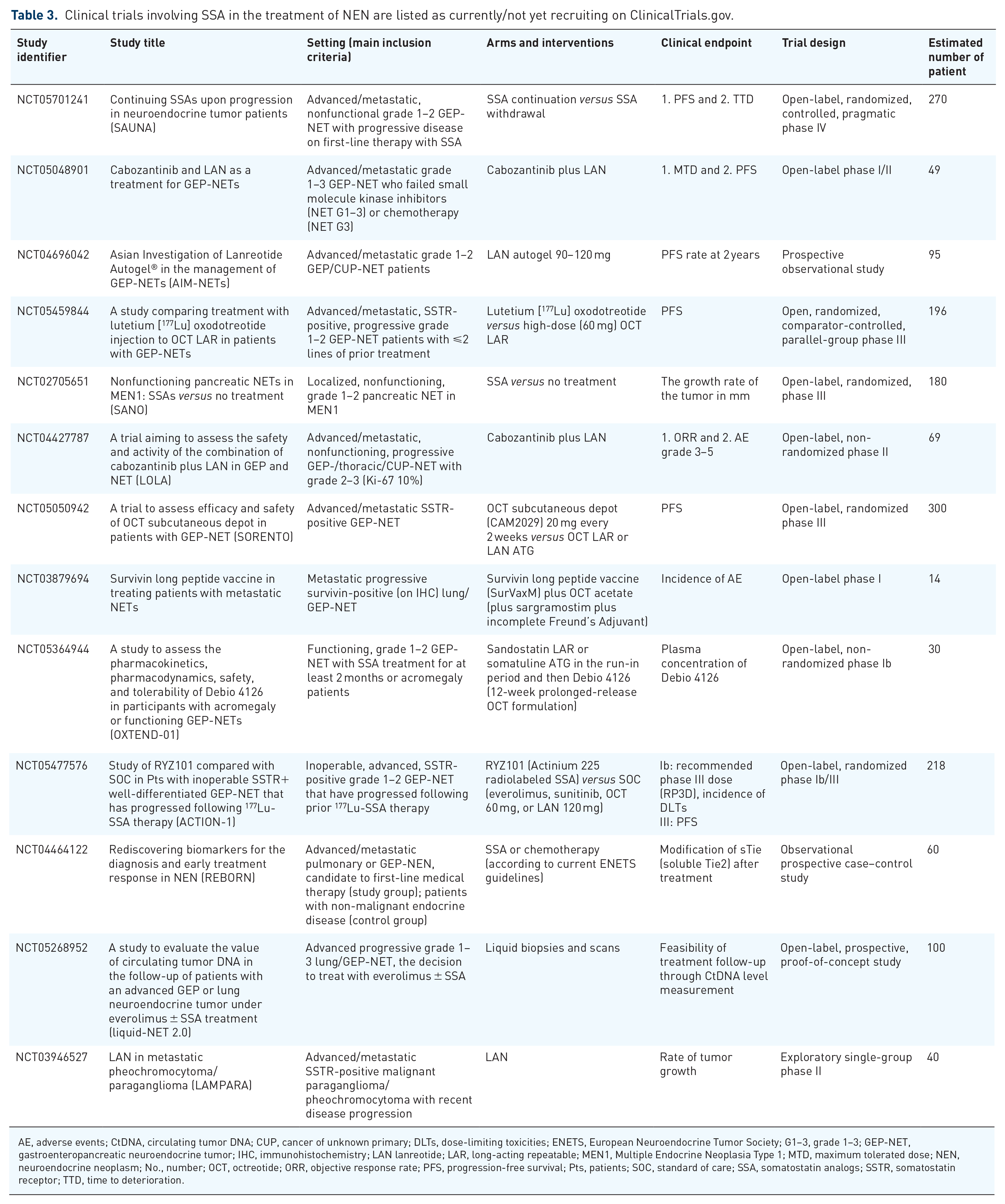
AE, adverse events; CtDNA, circulating tumor DNA; CUP, cancer of unknown primary; DLTs, dose-limiting toxicities; ENETS, European Neuroendocrine Tumor Society; G1–3, grade 1–3; GEP-NET, gastroenteropancreatic neuroendocrine tumor; IHC, immunohistochemistry; LAN lanreotide; LAR, long-acting repeatable; MEN1, Multiple Endocrine Neoplasia Type 1; MTD, maximum tolerated dose; NEN, neuroendocrine neoplasm; No., number; OCT, octreotide; ORR, objective response rate; PFS, progression-free survival; Pts, patients; SOC, standard of care; SSA, somatostatin analogs; SSTR, somatostatin receptor; TTD, time to deterioration.
