Abstract
Mixed neuroendocrine non-neuroendocrine neoplasms of the gastrointestinal tract are rare, with only a few cases documented in the literature. In particular, there are no reports of a rectal mixed neuroendocrine non-neuroendocrine neoplasm with regional lymph node metastasis of the neuroendocrine tumor component. Here, we present the case of a man in his early 70s with an incidental finding of multiple colonic polyps during a routine colonoscopy, including a 0.9-cm sessile polyp in the rectum. Repeat colonoscopy was performed for endoscopic mucosal resection. Histopathological examination revealed a biphasic tumor comprising a well-differentiated adenocarcinoma and a grade 1 neuroendocrine tumor, leading to a diagnosis of rectal mixed neuroendocrine non-neuroendocrine neoplasm. Because the adenocarcinoma component had invaded the submucosa and involved the resection margin, an ultralow anterior resection was performed. Notably, postoperative pathological analysis revealed metastasis of the neuroendocrine tumor component to a regional lymph node, an unusual finding not previously reported in the literature. Despite this, the patient remained recurrence-free for 2 years without adjuvant therapy. To the best of our knowledge, this is the first such case to be reported in the literature. Given the limited understanding of mixed neuroendocrine non-neuroendocrine neoplasms, particularly cases with low-grade neuroendocrine components, further case accumulation is necessary to develop standardized treatment guidelines.
Introduction
Mixed neuroendocrine non-neuroendocrine neoplasms (MiNENs) of the gastrointestinal tract are rare, with a limited number of case reports and small case series available in the literature. The World Health Organization (WHO) first introduced the term “mixed adenoneuroendocrine carcinoma” (MANEC) in 2010 to describe gastrointestinal tumors composed of both neuroendocrine and exocrine components, each constituting at least 30% of the tumor. In the 2019 WHO classification, this term was replaced with “MiNEN” to encompass a broader spectrum of tumors, including non-adenocarcinomatous components and neuroendocrine tumors (NETs) other than neuroendocrine carcinoma (NEC). Both entities are morphologically and immunohistochemically recognizable as a discrete component comprising 30% of the neoplasm. 1 The non-neuroendocrine component is most commonly adenocarcinoma, but other tumor types may also be present. The neuroendocrine component can be an NEC or NET, depending on the mitotic rate and the Ki-67 proliferation index. MiNENs are an uncommon tumor category, with an estimated annual incidence of 0.01 per 100,000 individuals. 2 Because of their low incidence, only a few studies have investigated their epidemiological and clinical characteristics. 3 Here, we report a case of rectal MiNEN with regional lymph node (LN) metastasis of the grade 1 NET component. To the best of our knowledge, this is the first such case to be described in the literature.
Case report
A man in his early 70s was referred to Chungbuk National University Hospital in August 2023 for endoscopic mucosal resection (EMR) after multiple colonic polyps were detected during a routine colonoscopy. He had no specific symptoms, and his medical history was unremarkable. Physical examination revealed no abnormalities. A repeat colonoscopy was then performed for EMR of the colonic polyps. A total of eight colonic polyps were detected, with the largest one, a sessile polyp in the rectum, measuring 0.9 cm at its greatest dimension (Supplementary Figure 1).
Microscopically, the tumor comprised two distinct components: well-differentiated atypical glands, consistent with colonic adenocarcinoma (60%), and sheets of epithelioid oval-to-polygonal neoplastic cells arranged in nests, trabeculae, and possible tubules, consistent with an NET (40%); therefore, a diagnosis of rectal MiNEN was made (Figure 1(a) to (d)). Immunohistochemical staining revealed that the adenocarcinoma component was positive for CK20, while the neuroendocrine component was positive for CD56 and synaptophysin. No mitotic activity was observed in the neuroendocrine component, and the Ki-67 proliferation index was 1.4%, classifying it as grade 1 (Figure 2(a) to (d)). Based on these findings, the tumor was diagnosed as a rectal MiNEN. Among the remaining seven polyps, five were identified as tubular adenomas with low-grade dysplasia, and two were hyperplastic polyps.

The tumor comprised two distinct components: well-differentiated atypical glands indicative of colonic adenocarcinoma (red square), and sheets of epithelioid neoplastic cells arranged in nests, trabeculae, and possible tubules, which are characteristic of neuroendocrine tumors (yellow square). The entire tumor excised by endoscopic mucosal resection was bisected, and two slides were prepared from each half. These whole tumor sections correspond to panels a and b (50× magnification). Higher-magnification views of the red and yellow squares highlight the adenocarcinoma component in panel c and the neuroendocrine tumor component in panel d, respectively (200× magnification).

(a) Immunohistochemical staining of the MiNEN. The glandular portion was diagnosed as adenocarcinoma, which was CK20-positive (a). The neuroendocrine component was positive for CD56 (b) and synaptophysin (c). The Ki-67 proliferation index of the neuroendocrine component was 1.4% (d) (magnification, all 100×). MiNEN: mixed neuroendocrine non-neuroendocrine neoplasm.
Because the adenocarcinoma component had invaded the submucosa and involved the resection margin, an ultralow anterior resection was performed. The resected portion of colon contained no residual tumor (Supplementary Figure 2); however, 1 of the 16 regional LNs removed contained a metastasis from an NET. Because of the rarity of this condition, immunohistochemical staining of the LN metastasis was performed, revealing metastatic tumor cells positive for CD56 and synaptophysin (Figure 3(a) to (d)). The final diagnosis was MiNEN, classified as pT1N1aM0, corresponding to pathological stage IIIA according to the eighth edition of the TNM Classification of Malignant Tumors. Adjuvant therapy was not considered, and the patient remained alive with no evidence of recurrence over an approximately 20-month follow-up period. The patient provided informed consent for treatment. Written consent for publication was also obtained. The reporting of this study conforms to the Case Report (CARE) guidelines. 4
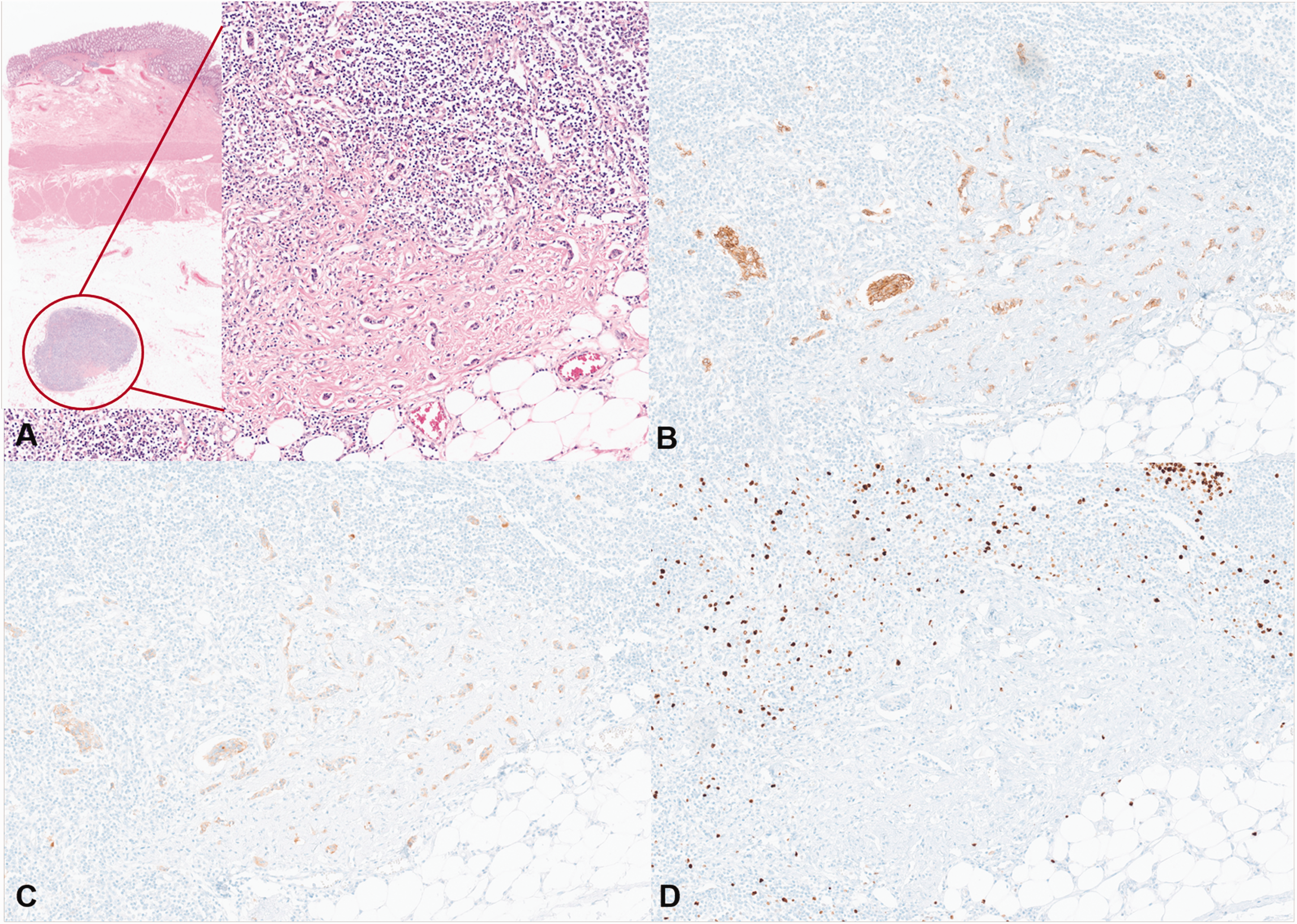
Regional lymph node metastasis of a well-differentiated neuroendocrine tumor component. (a) Low-power view of the resected colon specimen showing a lymph node with metastatic involvement (red circle, inset). The boxed area highlights the metastatic lesion, which is shown at higher magnification in the main panel (hematoxylin and eosin). Immunohistochemical staining of the metastatic tumor cells demonstrated positivity for CD56 (b) and synaptophysin (c), with a Ki-67 proliferation index of <1% (d) (magnification, all 200×).
Discussion
Mixed tumors exhibiting both exocrine and neuroendocrine morphological features are often encountered in pathological practice and can originate in various organs; however, because of their nonspecific colonoscopic and macroscopic presentation, histopathological evaluation and immunohistochemistry are essential for accurate diagnosis. Endoscopic findings often reveal semicircular tumors with deep ulceration or a mass protruding into the lumen. 5 Despite their identification, the rarity of this diagnosis, limited published data, and inconsistent use of terminology pose challenges to understanding the epidemiology, prognosis, and optimal management strategies for MiNENs. Nevertheless, these tumors are typically associated with aggressive behavior and poor prognosis. 3
In general, MiNENs are characterized by aggressive biological behavior, often driven by a poorly differentiated neuroendocrine component. In contrast, in almost all cases, the non-neuroendocrine component displays adenocarcinoma histology.6,7 The prognosis of colorectal MiNENs is believed to be worse than that of pure adenocarcinoma and is comparable to that of poorly differentiated NEC, especially at the metastatic stages.6–8 The metastatic risk appears to be closely linked to the grade of the neuroendocrine component.6,9
In a systematic review of 71 studies comprising 752 patients with MANECs of the lower gastrointestinal tract, Grossi et al. 10 reported the distribution of primary tumor sites as follows: appendix (60.3%), colon (29.3%), rectum (9.2%), and anal canal (1.2%). The mean age at diagnosis was 64.2 ± 13.6 years, with a male-to-female ratio of 1.25. Over 80% of the tumors had a high-grade (grade 3) neuroendocrine component. Among 152 patients with follow-up data, the median overall survival was 12.3 months. Independent prognostic factors included tumor location, grade III histology, tumor size >2 cm, T3–T4 stage, LN involvement, and distant metastasis. 10 Zhao et al. 11 reported a 3-year overall survival rate of 48.3% in patients with colorectal MANEC, with an LN metastasis rate of 58.8% (107 of the 182 cases).
However, evidence specifically addressing LN metastasis in MiNENs with a low-grade NET component—such as in our case—is extremely limited. We conducted a targeted literature review using the terms “colon MiNEN,” “rectal MiNEN,” and “rectum MiNEN” across PubMed and Google Scholar databases. Cases labeled as MANEC were excluded due to the obligatory inclusion of NEC in that definition. Additionally, cases lacking clear information on tumor location or histological subtypes were excluded. This search identified 17 case reports encompassing 20 colorectal MiNEN cases (Table 1).12–28 Of these, 18 involved NEC, one was classified as grade 3 NET, and only one case involved a grade 1 NET component.
Clinicopathological features of previously reported cases of colorectal MiNEN.

F: female; LN: lymph node; M: male; NA: not available; ND: not described; NEC: neuroendocrine carcinoma; NET: neuroendocrine tumor; MiNEN: mixed neuroendocrine non-neuroendocrine neoplasm.
Fourteen cases demonstrated LN metastasis based on either histopathological or radiological evaluation, four had no LN metastasis, and in two cases, LN status could not be determined. Of the 14 cases with confirmed LN metastasis, 8 had documented metastasis from the neuroendocrine component. Among these eight cases, two also showed concurrent adenocarcinoma metastasis. In the remaining six cases, the histological type of LN metastasis was not specified. Notably, the sole MiNEN case with a grade 1 NET component showed no LN metastasis and remained recurrence-free during follow-up. 13 In contrast, our case uniquely demonstrated regional LN metastasis derived from a grade 1 NET component.
To better contextualize our observation, we reviewed large-scale studies reporting LN metastasis rates in early-stage rectal NETs and colorectal adenocarcinomas. In a retrospective study involving 601 patients with rectal NETs of grade 1 or 2, the LN metastasis rate was 5.2% among the 515 patients with grade 1 NETs. 29 In comparison, a population-based analysis of 27,649 patients with pT1-stage colorectal adenocarcinoma reported an overall LN metastasis rate of 13%. 30 These data suggest that, overall, LN involvement is more common in early-stage colorectal adenocarcinoma than in low-grade rectal NETs. Given this context, the occurrence of LN metastasis from the grade 1 NET component—rather than the adenocarcinoma—in our case is unusual and warrants further biological consideration.
The distinct metastatic behavior may reflect fundamental biological differences between the tumor components. Although the pathogenesis of MiNENs remains incompletely understood, several theories have been proposed to explain their biphasic morphology. The collision theory suggests the coexistence of two independent neoplastic clones. The common precursor theory posits divergent differentiation from a single progenitor cell. Molecular studies increasingly support a monoclonal origin with shared clonal genetic alterations observed in both components. Furthermore, when the neuroendocrine component is high-grade, it frequently exhibits more extensive chromosomal and genetic abnormalities than the non-neuroendocrine component. 12 This suggests that a stepwise progression toward neuroendocrine differentiation may occur, potentially conferring metastatic potential even in morphologically low-grade NETs. Accordingly, the selective metastasis of the grade 1 NET component in our case may reflect early acquisition of molecular changes that favored lymphatic dissemination.
The recommended treatment approach for MiNENs depends on the tumor stage and whether the neoplasm is primary or recurrent. 7 In cases in which the tumor is localized and resectable, surgical excision remains the preferred curative option. Treatment decisions should be guided by the most aggressive histological component of the tumor. In our case, an ultralow anterior resection was performed due to invasion of the adenocarcinoma into the submucosa and involvement of the resection margin. However, subsequent pathological analysis of the resected specimen unexpectedly revealed regional LN metastasis of the NET component, specifically a grade 1 NET—an occurrence not previously reported. This case underscores the importance of recognizing the potential for LN metastasis even in grade 1 NET components within rectal MiNENs, particularly in the absence of known risk factors. Clinicians should be aware of this rare but significant possibility. These findings further highlight the need for further case accumulation and research to establish standardized treatment protocols, particularly for rectal MiNENs with a low-grade neuroendocrine component.
The role of neoadjuvant chemotherapy for MiNENs remains uncertain, with limited supporting evidence. Although neoadjuvant chemotherapy is believed to target the adenocarcinoma component, it is recommended more commonly for tumors in which the adenocarcinoma component predominates; however, current data do not support its use for preventing or delaying recurrence of the neuroendocrine component. When both components are present and equally aggressive, treatment targeting the neuroendocrine component should be prioritized, as it is the primary driver of metastatic behavior. Current literature suggests that adjuvant chemotherapy following surgical resection may help reduce the risk of recurrence in most colorectal MiNEN cases. 20
To the best of our knowledge, this is the first reported case of a rectal MiNEN with regional LN metastasis of the grade 1 NET component. Pathologists and physicians must be vigilant for this condition. Managing MiNENs remains challenging, with surgery, and in some cases, neoadjuvant therapy serving as the cornerstones of treatment. As more cases are reported, our understanding of the epidemiology and prognosis of this rare entity will improve, ultimately aiding the development of standardized management strategies.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251345907 - Supplemental material for Lymph node metastasis of grade 1 neuroendocrine tumor from rectal mixed neuroendocrine non-neuroendocrine neoplasm: A case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605251345907 for Lymph node metastasis of grade 1 neuroendocrine tumor from rectal mixed neuroendocrine non-neuroendocrine neoplasm: A case report by Taek-Gu Lee, Ki Bae Kim, Jun Su Lee, Chang Gok Woo and Seung-Myoung Son in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251345907 - Supplemental material for Lymph node metastasis of grade 1 neuroendocrine tumor from rectal mixed neuroendocrine non-neuroendocrine neoplasm: A case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605251345907 for Lymph node metastasis of grade 1 neuroendocrine tumor from rectal mixed neuroendocrine non-neuroendocrine neoplasm: A case report by Taek-Gu Lee, Ki Bae Kim, Jun Su Lee, Chang Gok Woo and Seung-Myoung Son in Journal of International Medical Research
Footnotes
Acknowledgements
Artificial intelligence tools (ChatGPT by OpenAI) were used to improve the language and clarity of the manuscript. All intellectual content and interpretation are solely those of the author. The author would like to thank Dr. Ok-Jun Lee for their assistance in histological data collection.
Author contributions
Taek-Gu Lee and Seung-Myoung Son designed the study; Ki Bae Kim and Jun Su Lee acquired clinical data; Taek-Gu Lee and Chang Gok Woo wrote the manuscript; Seung-Myoung Son revised the manuscript. All authors issued final approval for the version to be submitted.
Data availability statement
Not applicable.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
Not applicable.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
