Abstract
Primary kidney neuroendocrine tumors (NETs) are rare renal malignancies. However, detecting and monitoring neuroendocrine neoplasms remains challenging because of their nonspecific nature. We herein present a case involving a 53-year-old woman who experienced episodes of intermittent abdominal pain, dizziness, and nausea for a period of 5 days. Computed tomography urography revealed a small (approximately 19- × 16-mm) nodular shadow in the left kidney. The nodular shadow exhibited slightly lower density than the surrounding tissue as well as enhancement, with a portion protruding into the renal sinus region. Histological and immunohistochemical analyses of the biopsy specimen from the mass indicated a well-differentiated NET. After analysis of this case, we performed a literature review and herein discuss various techniques for imaging and pathological diagnosis of renal NETs. Additionally, we provide insights into the treatment options and prognosis for affected patients. By combining this case study with the existing published literature, we aim to offer a valuable reference for clinicians treatment patients diagnosed with renal NETs.
Keywords
Introduction
Neuroendocrine neoplasm (NEN) is a type of epithelial tumor primarily characterized by its neuroendocrine differentiation. Because of the widespread distribution of neuroendocrine cells throughout the body, these tumors can develop in multiple organs. Approximately 70% of NENs are found in the gastroenteropancreatic system, 25% in the respiratory system, and the remaining 5% in various other organs. 1 Neuroendocrine tumors (NETs) originating from the kidney are considered rare within the spectrum of renal malignant tumors, accounting for less than 1% of renal epithelial malignant tumors. 2 In August 2022, a case of NET was diagnosed in our department. We herein discuss the key clinical features, imaging characteristics, pathological findings, treatment options, and prognosis of this disease based on a comprehensive literature review.
Case report
The reporting of this study adheres to the CARE guidelines. 3 In accordance with local and national guidelines, ethics approval was not required for this study. The patient provided written informed consent for both her treatment and the publication of her medical case details, including any accompanying images.
A 53-year-old woman presented with a 5-day history of intermittent abdominal pain, dizziness, and nausea. Physical examination revealed no remarkable abnormalities, and gross hematuria was not present. Computed tomography urography (CTU) revealed a small (approximately 19- × 16-mm) nodular shadow in the left kidney. The nodular shadow exhibited slightly lower density than the surrounding tissue as well as enhancement, with a portion protruding into the renal sinus region (Figure 1). Renal magnetic resonance imaging revealed round lesions exhibiting long T1 and short T2 signals in the left kidney (Figure 2). These lesions appeared hyperintense on diffusion-weighted imaging, showed an indistinct border, and measured approximately 20 mm in width (left–right), 18 mm in length (anterior–posterior), and 20 mm in height (superior–inferior). Based on the preoperative assessment, the diagnosis of left renal tumor was established, and laparoscopic partial nephrectomy was performed on 2 August 2022. The surgical procedure was conducted using retroperitoneal access. Intraoperatively, the perirenal fascia was incised, and the perirenal fat was isolated to expose the left kidney. Utilizing intraoperative laparoscopic ultrasound, a mass of approximately 2 cm in diameter located in the central region of the left kidney was identified. The tumor was surgically excised along with a portion of the kidney tissue. Upon gross examination, the specimen displayed a nodular consistency with dimensions of approximately 3.0 × 2.5 × 1.8 cm. The cut surface of the specimen exhibited vibrant colors and a soft texture. The pathological diagnosis of the left renal tumor was a well-differentiated NET. The tumor exhibited mitotic figures at a rate of 12 per 10 high-power fields, along with areas of necrosis. Notably, there was no involvement of the surgical stump. Immunohistochemistry analysis (Figure 3) demonstrated the following: pan cytokeratin (+), CD56 (+), synaptophysin (+), cytokeratin 7 (−), CD10 (−), vimentin (−), Wilms’ tumor protein 1 (−), p63 (−), Pax-8 (−), Ki-67 (+, about 5%), and somatostatin receptor 2 (3+).
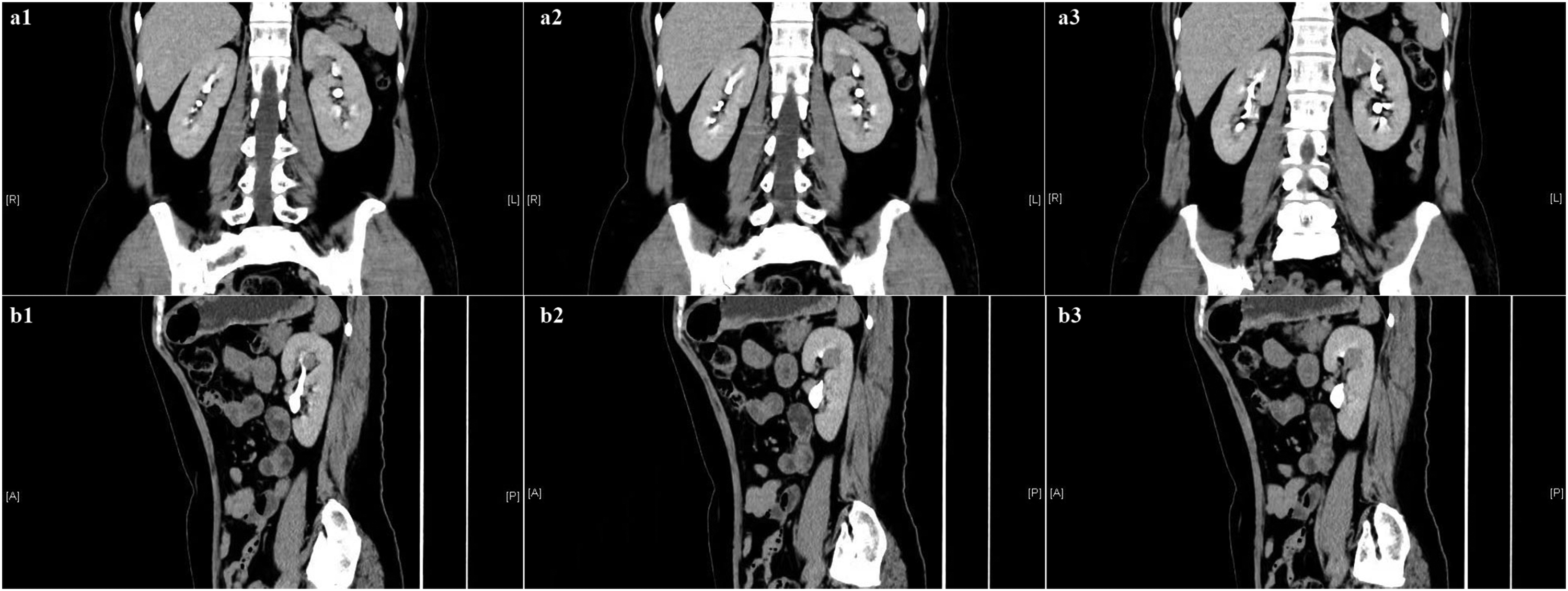
Preoperative computed tomography urography images. The contrast-enhanced scans showed heterogeneous enhancement of the tumor in the (a1–3) coronal and (b1–3) sagittal images; no enhancement was observed in the central necrotic area.

Preoperative computed tomography urography and magnetic resonance imaging. The contrast-enhanced scans showed heterogeneous enhancement of the tumor in the (a1–3) arterial and (b1–3) venous phases; no enhancement was observed in the central necrotic area. (c1–3) Renal magnetic resonance imaging also showed the tumor in the left kidney.

Tumor tissues were subjected to histologic examination using hematoxylin and eosin staining. (a) 20× and (b) 40×. The tumors primarily consisted of cells that exhibited uniformity with round to oval nuclei. The cytoplasm exhibited a pale eosinophilic to amphophilic appearance.
Postoperative positron emission tomography/computed tomography (PET/CT) revealed nodular lesions with tracer uptake enhancement in the left outer lobe and right anterior lobe of the liver, exhibiting a maximum standardized uptake value of approximately 13.3. However, corresponding CT imaging showed no foci with abnormal density in these areas. Because liver metastases were suspected, the patient began postoperative endocrine therapy involving intramuscular injections of octreotide acetate microspheres for injection at a dosage of 30 mg every month. At the 4-month follow-up, the patient’s condition was stable. The kidney that underwent surgical treatment showed good postoperative recovery, and there were no indications of metastatic lesions in the contralateral kidney (Figure 4).
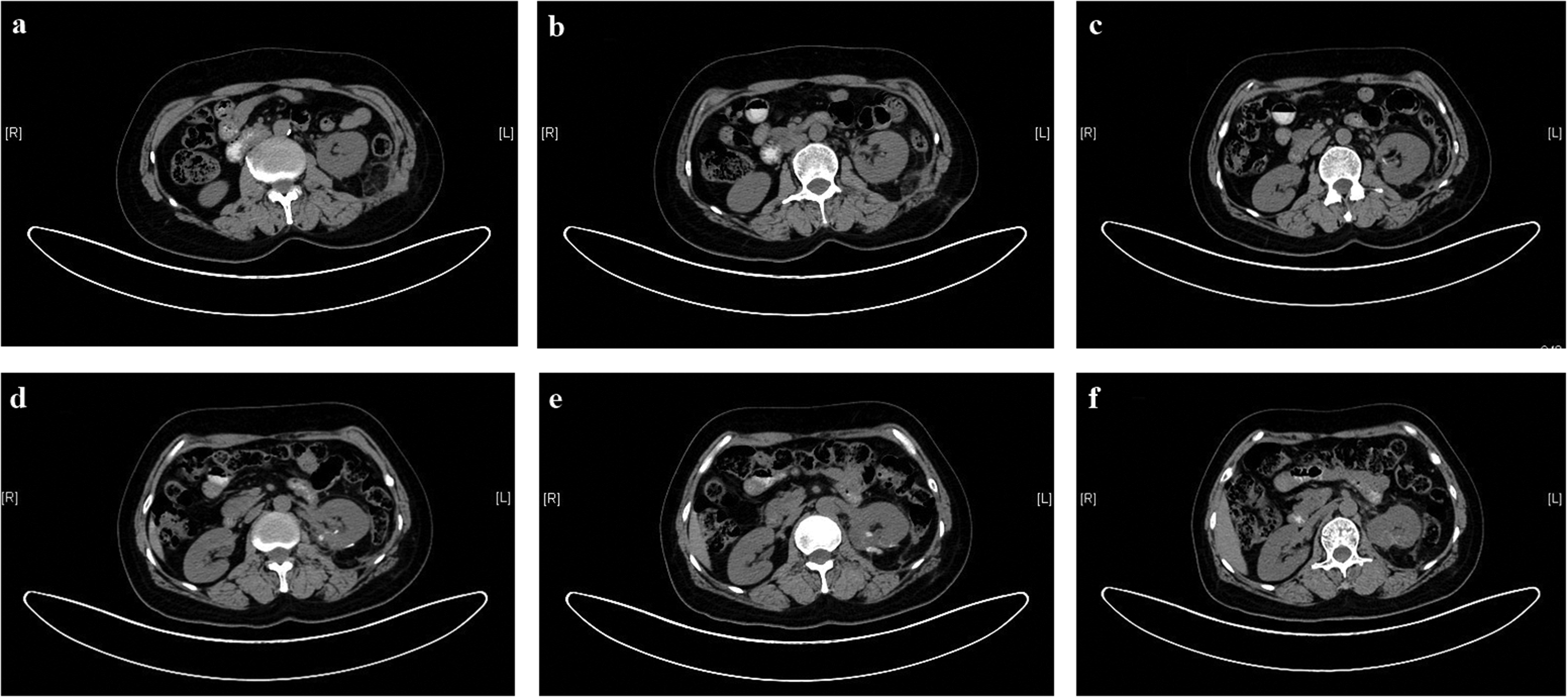
Postoperative computed tomography images. (a)–(f) The images show good recovery of the left kidney and no metastatic lesion in the opposite kidney.
Discussion
NETs can be found in nearly every tissue, including both endocrine and non-endocrine organs and tissues. NETs primarily occur in the digestive tract and lungs; they are relatively rare in the urinary system.1,2 The pathogenesis of renal NETs remains unclear. 4 The World Health Organization has classified NENs based on their histological characteristics, which include small cell neuroendocrine carcinoma, large cell neuroendocrine carcinoma, well-differentiated NET, and paraganglioma. The inclusion of paraganglioma in the classification aims to enhance the overall classification system for NETs.5–7
Clinical presentation
According to previous reports, renal NETs predominantly affect individuals aged 40 to 60 years, with no notable sex predilection.8–10 Approximately 25% to 30% of cases are diagnosed incidentally. 11 NETs of the urinary system primarily manifest in the kidneys and bladder. Typical symptoms include abdominal or flank pain, the presence of an abdominal mass, unintended weight loss, and hematuria. Symptoms resulting from hormone production or carcinoid syndromes are infrequent and occur in less than 10% of cases. 12
Imaging features
Detecting and monitoring NETs and evaluating their response to therapy can be challenging because of the lack of specificity of these tumors. CT imaging commonly reveals single renal masses in that tend to be large in size and fairly well-circumscribed. Contrast-enhanced CT frequently exhibits poorly vascularized or hypovascular tumors with limited enhancement. 10 We ruled out renal cyst and renal angiomyolipoma by CTU before the operation. Renal cysts appear as areas of low density on CTU and do not exhibit enhancement, whereas renal tumors demonstrate enhancement on CTU. Additionally, renal angiomyolipoma typically displays a negative CT value due to the presence of adipose tissues, whereas renal cancer exhibits a positive CT value. 13 Historically, the visualization of NENs depended on less effective structural imaging techniques such as CT and magnetic resonance imaging. However, the primary approach for visualizing NENs now revolves around molecular imaging. Radiolabeled octreotide is a somatostatin analog with high affinity for somatostatin receptors. Primary NETs and metastases exhibit high-affinity receptors for somatostatin in 87% of cases. Advancements in PET have led to the recognition and widespread application of somatostatin receptor imaging combined with PET/CT technology. This combined approach has gained international acceptance and is actively utilized in the field of NET imaging. The use of PET/CT improves test sensitivity and serves multiple purposes in the management of NETs. It is valuable for diagnosing NETs, staging the disease, and monitoring post-treatment recurrence or metastasis.4,14,15
Pathologic features
Well-differentiated NENs typically exhibit distinct, well-defined nodules with minimal necrosis. Under microscopic examination, the tumor cells are organized in nests, trabeculae, adenoid patterns, or structures resembling inulin-like masses. Neuroendocrine carcinomas, particularly small cell carcinomas, are characterized by high invasiveness, poorly circumscribed boundaries, and the frequent presence of necrosis. When examined microscopically, these tumors display a lack of typical organoid architecture. Instead, they consist of poorly differentiated small round or spindle-shaped cells arranged in glandular, trabecular, or nest-like patterns. The nuclei of these cells are hyperchromatic, and there is a high frequency of mitotic figures.8,10 Immunohistochemical markers play a crucial role in diagnosing renal NETs. The most common epithelial and neuroendocrine cell markers expressed in NETs include neuron-specific enolase, chromogranin A, and synaptophysin. The second most common immunohistochemical markers include vimentin, CD56, cytokeratin, and CD99. However, CD10, cytokeratin 7, and kidney-specific cadherin are negative markers. The specificity of synaptophysin in the diagnosis of NENs may reach 100%. Among these markers, CD56 has high sensitivity but low specificity and can be used as a screening indicator.10,16–19
Treatment and prognosis
NETs are characterized by their low level of malignancy and slow rate of growth. In terms of treatment, surgical intervention is typically the initial and preferred approach. The recommended surgical method is radical resection, which involves removing the tumor along with the surrounding lymph nodes. Romero et al. 10 found that 47% of patients with well-differentiated NETs who underwent radical nephrectomy remained free of tumors during an average follow-up period of 43 months. Patients diagnosed with well-differentiated NETs are often found to have regional lymph node metastasis during nephrectomy. Although these tumors can metastasize to distant viscera, patients typically experience extended survival. 7 Therefore, long-term follow-up is necessary.
Because of the high likelihood of metastasis and the challenges in early detection, small cell renal cell carcinoma and large cell renal cell carcinoma often cannot be treated surgically. As a consequence, patients diagnosed with these types of carcinomas typically have an average survival time of less than 1 year. For high-grade neuroendocrine carcinoma, radical renal resection on the affected side is recommended because this type of carcinoma tends to exhibit local invasion or metastasis. Additionally, retroperitoneal lymph node dissection may be performed alongside the surgery. Furthermore, cisplatin-based chemotherapy has shown significant improvement in survival rates for patients diagnosed with high-grade neuroendocrine carcinoma of the kidney. 20 Research has shown that patients who receive chemotherapy as the sole treatment may have a more favorable prognosis than patients who undergo surgery alone or in combination with chemotherapy. 21
Poor prognostic factors include older age, male sex, larger tumor size, and tumors not confined to the renal parenchyma. In such cases, complete surgical removal of all visible tumor tissues is considered the preferred and most effective treatment approach. 22
Conclusion
Primary kidney NETs are rare malignant tumors. They pose a diagnostic challenge in medical imaging because of their similarity with other renal tumors. However, pathological examination and immunohistochemical analysis are considered crucial methods for accurate clinical diagnosis. Conservative treatment approaches are generally ineffective in managing primary kidney NETs. Therefore, surgical intervention, often in the form of partial resection, is commonly selected as the preferred treatment option.
Footnotes
Author contributions
Changxun Liu conceived the study, analyzed the data, and wrote the manuscript. Xiaokun Yang selected the clinical case and supervised the study. Yixin Qi and Youzhi Zhang analyzed the data and generated the figures. All authors read and approved the final manuscript.
Data availability statement
All data in this study are included within the article. Further inquiries may be directed to the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
