Abstract
Breast cancer remains a leading cause of brain metastases (BM), which carry a poor prognosis. The current approach to managing BMs in breast cancer patients involves a combination of local therapies (surgery, radiotherapy) and systemic treatments. Developing newer antibody–drug conjugates (ADCs) has sparked a revolution in metastatic breast cancer (MBC) care. ADCs such as ado-trastuzumab emtansine, trastuzumab deruxtecan, and sacituzumab govitecan have demonstrated significant improvement in patient outcomes and are standard of care in the treatment of MBC. Most of the ADC registration studies included patients with stable BMs but excluded individuals with active BM, making intracranial (IC) response assessment a challenge. Promising data has recently emerged, suggesting relevant IC activity for certain ADCs and ongoing studies in patients with active BM that will expand our knowledge. This review aims to summarize the effectiveness of approved ADCs as well as promising new ADCs in development for breast cancer with BM.
Introduction
In recent years, there have been significant strides toward developing effective therapeutic options for patients with metastatic breast cancer (MBC). Up to 30% of patients with MBC develop brain metastases (BM) during their disease, with higher rates of BM of 25%–46% seen in triple-negative breast cancer (TNBC) and 30%–50% in human epidermal growth factor receptor 2 (HER2)-positive MBC. 1 Historical overall survival (OS) rates for breast cancer with brain metastases (BCBM) remain inferior to patients without BCBM (overall 7.5 months; HER2-positive 13.2 months; TNBC 4.5 months). 1 There has been improvement in OS in patients with HER2-positive BCBM. Nevertheless, there remains an unmet need for effective systemic therapies for the treatment of patients with BCBM across all breast cancer subtypes.
Initial treatment options for BCBM typically include local therapy with surgery for select solitary lesions, radiosurgery, or whole-brain radiation. Systemic therapies following local therapy improve OS for luminal and HER2-positive BCBM.2,3 To date, no systemic therapy has demonstrated improved survival for patients with triple-negative BCBM. Systemic therapies often include chemotherapy, antibody–drug conjugates (ADCs), and tyrosine kinase inhibitors (TKIs). Surgery is considered for patients with a solitary brain lesion or symptomatic space-occupying lesion. For those who are not surgical candidates and who have a lower volume of metastatic lesions, stereotactic radiosurgery (SRS) is the preferred approach over whole brain radiotherapy (WBRT), given the concern for neurological toxicities associated with WBRT. WBRT is reserved for those with high volume, symptomatic, or progressive BM, not amenable to SRS. If WBRT is recommended, phase III trials have demonstrated that sparing the hippocampus during radiotherapy and administering memantine better preserves cognitive function and patient-reported symptoms, with no difference in intracranial (IC) progression-free survival (PFS) and OS. 4
The systemic treatment of patients with BCBM remains a challenge as many, though not all, historical cytotoxic chemotherapies did not effectively cross the blood–brain barrier (BBB). Another challenge is that BCBM often occur late in the disease course, when treatment resistance has already occurred. The BBB is composed of non-fenestrated endothelial cells connected by adherens junctions, gap junctions, scaffolding proteins, and tight junctions that function together as a barrier to pathogens but concurrently restrict drug penetration to lipophilic, lower molecular weight molecules of under 400–500 daltons (Da) 5 (Figure 1). Another significant contributor to drug resistance in treating BCBM is the efflux transporter systems of the BBB. These transporters, including P-glycoprotein, breast cancer-resistance protein, and multidrug resistance-associated proteins, actively transport drugs back out of the central nervous system (CNS), thereby hindering their penetration, which may explain the poor historical response rates to single-agent chemotherapy in BCBM. 6

Translocation of ADCs across the blood–brain barrier.
Small-molecule TKIs, neratinib (557 Da), tucatinib (480 Da), and lapatinib (581 Da) have historically shown efficacy in the treatment of BCBM with penetration into BM due to their low molecular mass. 7 Of interest, lapatinib has minimal penetration across an intact BBB, but nevertheless, the more permeable blood–tumor barrier (BTB) allows for therapeutic levels and CNS activity in BM compared to adjacent normal brain. 8 In the phase II LANDSCAPE trial testing lapatinib and capecitabine as initial treatment of BM (i.e., in lieu of radiotherapy) from HER2-positive breast cancer, two-thirds of patients had objective CNS responses. 9 Regarding neratinib, a subgroup analysis of the NALA trial, evaluated patients with HER2-positive BCBM after two or more lines of treatment. The neratinib + capecitabine cohort had improved PFS and CNS outcomes compared with the lapatinib + capecitabine cohort, with an intracranial overall response rate (IC-ORR) of 26.3% and 15.4%, respectively. 10 Similarly, in TBCRC 022, which was a phase II study evaluating neratinib and capecitabine for progressive HER2-positive BCBM, the neratinib + capecitabine combination demonstrated activity against refractory BCBM in both the lapatinib naïve and lapatinib-treated cohorts, with an IC-ORR of 49% (95% confidence interval (CI), 32%–66%) versus 33% (95% CI, 10%–65%), respectively. 11 Similar results confirming IC efficacy with IC-ORR of ~30% were seen in the T-DM1 + neratinib cohort that was recently published. 12
Specifically, tucatinib has more efficient BBB penetration and demonstrated evidence of IC activity in preclinical models of HER2-positive BCBM. 13 HER2CLIMB was the landmark clinical trial of tucatinib, trastuzumab, capecitabine, which included nearly half of patients with HER2-positive BCBM, including active BM (untreated lesions or lesions that had progressed after prior radiation) and allowing for prospective evaluation of IC response and CNS-PFS. The addition of tucatinib to trastuzumab and capecitabine doubled the IC-ORR, reduced the risk of IC progression or death by 68%, and reduced the risk of death by 42%, with a median OS of 18.1 months in the tucatinib arm versus 12.0 months in the control arm. 14 In the exploratory subgroup analysis of patients with BM, median OS was 9.1 months longer in the tucatinib-combination group (21.6 months; 95% CI, 18.1–28.5) versus the placebo-combination group (12.5 months; 95% CI, 11.2–16.9). The tucatinib-combination group also showed a reduced risk of developing new brain lesions by 45.1% versus the placebo-combination group (hazard ratio, 0.55 (95% CI, 0.36–0.85)). 15 This landmark trial set the bar for future trials of HER2-directed TKIs.
There is growing evidence that neovascularization and tumor progression within the CNS leads to disruption of the BBB, leading to what is known as the BTB. 16 The BTB is more permeable and heterogeneous than the BBB due to disruption of the junctional proteins, and studies have shown that similar concentrations of trastuzumab can be detected in both BM and extracranial bone metastasis of HER2-positive MBC patients17,18 (Figure 1). 18 This disrupted BTB may also allow for larger molecules including ADCs to penetrate. ADCs are composed of a monoclonal antibody connected to a cytotoxic payload via a chemical linker, with the goal to provide targeted chemotherapy and to reduce systemic toxicity. Newer generation ADCs have novel properties such as cleavable linkers allowing for the bystander effect and potent drug-to-antibody ratios (DAR), which likely contribute to their enhanced efficacy in the brain. 19 In 2013, ado-trastuzumab emtansine (T-DM1) became the first ADC approved for the treatment of HER2-positive breast cancer showing improved PFS and OS compared to lapatinib and capecitabine with a median OS of 30.9 versus 25.1 months, respectively. 20 Since then, several next-generation ADCs targeting HER2 and anti-trophoblast cell-surface antigen 2 (TROP2), including trastuzumab deruxtecan (T-DXd), sacituzumab govitecan (SG), and datopotamab deruxtecan (Dato-DXd) have improved outcomes for patients living with MBC. T-DXd was FDA-approved in 2019 for use in previously treated metastatic HER2-positive breast cancer with two or more anti-HER2 therapies based on the results of the DESTINY-Breast01 study. 21 T-DXd has also been FDA-approved for use in HER2-low expressing MBC based on the results of the DESTINY-Breast04 trial. 22
SG was the first TROP2 ADC that was FDA-approved in 2021 for patients with unresectable locally advanced or metastatic TNBC who have received two or more prior systemic therapies based on the ASCENT trial, where SG showed improved PFS and OS compared to chemotherapy. 23 Since then, SG has also garnered FDA approval in 2023 for unresectable locally advanced or metastatic hormone receptor (HR)-positive, HER2-negative breast cancer who were previously treated with endocrine-based therapy and at least two additional systemic therapies based on the results of the TROPICS-02 study. 24
The pivotal trials that led to the approval of the use of ADCs in MBC excluded patients with active BCBM. Small retrospective and prospective analyses have helped us understand the promise of ADCs in BCBM but data remains limited. Therefore, the IC efficacy of ADCs in untreated or active/progressing BM remains unclear and is being investigated in multiple ongoing trials. In this narrative review, we will focus on the existing and growing evidence of the IC efficacy of different ADCs in the treatment of BCBM.
Approved agents
Ado-trastuzumab emtansine
The first FDA-approved ADC for HER2-positive breast cancer, T-DM1, is made up of the monoclonal antibody trastuzumab, which binds the HER2 receptor, coupled to the cytotoxic agent emtansine, which inhibits microtubule polymerization. This ADC has a drug to antibody (DAR) ratio of 3.5:1. T-DM1 is approved for use in metastatic HER2-positive breast cancer with progression on prior taxane and trastuzumab based on the results of the EMILIA trial, which showed significantly improved PFS and OS compared with lapatinib and capecitabine in patients with previously treated HER2-positive locally advanced or MBC. Patients with CNS-only disease, and those with untreated, recently treated, or symptomatic CNS metastases were excluded. In the retrospective exploratory analysis of the EMILIA trial assessing the outcomes of patients with CNS metastases, aproximatelly 10% of patients in both cohorts had previously treated and stable CNS metastases at baseline. The rate of CNS progression in patients was not statistically different between T-DM1 versus capecitabine and lapatinib, at 22.2% and 16%, respectively. The PFS for T-DM1 versus capecitabine and lapatinib was also similar (5.9 vs 5.7 months, respectively). For patients with asymptomatic BM at baseline, median OS significantly improved with T-DM1 when compared to capecitabine and lapatinib, with 26.8 versus 12.9 months, respectively. However, this retrospective analysis was limited since patients with progressive CNS metastases were excluded and could not evaluate the efficacy of T-DM1 in treating CNS disease. 25
The prospective KAMILLA trial, which evaluated T-DM1 in patients with previously treated HER2-positive MBC, included 398 patients (19.9%) with baseline CNS metastases. A total of 269 patients experienced new brain lesions during T-DM1 treatment, including 115/398 of patients with baseline BM (28.9%) and 154/1604 of patients without baseline BM (9.6%), in which patients were allowed to continue T-DM1 in those with controlled systemic disease. In the 126 patients who had measurable BM, the best ORR (complete response (CR) + partial response (PR)) was 21.4%, with a clinical benefit rate (CBR) (CR + PR + stable disease lasting ⩾6 months) of 42.9%. A ⩾30% reduction in the major diameter of Response Evaluation Criteria in Solid Tumors (RECIST)-evaluated BM was seen in 42.9% of these patients. In the 67 patients without prior radiotherapy before study entry, almost half showed a ⩾30% reduction in the major diameters of BM, suggesting a direct therapeutic effect of T-DM1 in BM that is not confounded by prior treatment with radiotherapy. Overall, the findings support that T-DM1 is active and well tolerated in HER2-positive BCBM, and brain target lesion responses were observed in both patients with and without prior radiotherapy. 26 In the TH3RESA study, T-DM1 also showed significantly longer PFS and OS when compared to the treatment of physician’s choice in previously treated HER2-positive advanced breast cancer. In the subgroup analysis of patients with stable brain lesions, longer median OS of 17.3 months was seen with T-DM1 compared to 12.6 months with the treatment of physician’s choice, further supporting the efficacy of T-DM1 in patients with BM. 27
The HER2CLIMB-02 trial looked at the efficacy of tucatinib when combined with T-DM1 in previously treated HER2-positive locally advanced and MBC. Like HER2CLIMB, it was among the first registration trials to include patients with stable, progressing, or untreated BM. About 44% of the study population had either active or stable BM at enrollment. Results showed that the addition of tucatinib to T-DM1 significantly improved median PFS with 9.5 versus 7.4 months for the tucatinib and control arms, respectively, in patients with previously treated HER2-positive locally advanced/MBC, including those with active BM. The hazard ratio for PFS in the cohort with BM at baseline was 0.639, which was consistent with the hazard ratio of the overall population of 0.759 in the tucatinib + T-DM1 arm. The OS data is still immature. 28
Notably, the data about T-DM1 preventing BCBM has been limited. In the KATHERINE trial, which compared adjuvant T-DM1 or trastuzumab in patients with HER2-positive early breast cancer who had residual invasive disease at surgery after receiving neoadjuvant therapy containing a taxane (with or without anthracycline) and trastuzumab, the T-DM1 arm showed improved invasive disease free survival outcomes. Despite the improved outcomes observed with T-DM1, approximately 5% of patients in both treatment groups experienced their first relapse in the CNS, with no decrease in the T-DM1 arm. 29
Trastuzumab deruxtecan
T-DXd is composed of a HER2-targeted monoclonal antibody linked to the cytotoxic payload deruxtecan, a topoisomerase I inhibitor. It has a higher DAR ratio of 8.1 compared to T-DM1, delivering a higher concentration of cytotoxic deruxtecan to both target cells expressing the HER2 antigen. T-DXd has an effect not only in HER2-positive cells but also in surrounding cells (cancer and immune), which is known as the bystander effect. The bystander effect is mainly mediated by pharmacologic determinants of the ADC, including a cleavable linker (either chemically labile or enzyme-labile), which allows for the release of the payload in the surrounding tumor microenvironment, as well as the permeability of the payload, which allows diffusion to adjacent cells following internalization. 30 In the subgroup analysis of the phase I DESTINY-Breast01 trial that included 24 patients with treated BM, T-DXd showed similar efficacy in patients with a history of CNS metastases with an extracranial ORR of 58.3% and PFS of 18.1 months compared to the overall population of those without BM where ORR was 61.3% and PFS was 16.4 months. At 11.1 months follow-up, 8 of the 24 patients had a partial response (PR) or CR, with only 4 patients having CNS progression (including 2 with baseline CNS metastases). Of the 17 patients with measurable brain lesions, 7/17 patients (41.2%) had a PR (defined as ⩾30% decrease in size) in the brain, and 7/17 patients (41.2%) had stable disease. 31
The DESTINY-Breast03 trial, which included those with clinically inactive or previously treated BM, showed the superiority of T-DXd compared to T-DM1 in metastatic HER2-positive breast cancer previously treated with trastuzumab and a taxane, with 2-year OS rate of 77% versus 69.9%, respectively. About 15% of patients in both T-DXd and T-DM1 cohorts had BM at baseline. For patients with BM, PFS was 15.0 months for T-DXd versus 3.0 months for T-DM1. For patients without BM, median PFS was not reached for T-DXd versus 7.1 months for T-DM1. IC-ORR was 65.7% versus 34.3% with T-DXd versus T-DM1, respectively. T-DXd was moved to second-line treatment for HER2-positive MBC based on these results. 32 In both subgroup analyses of the DESTINY-Breast01 and 03 trials, T-DXd demonstrated activity against stable BM.
Although the DESTINY-Breast trials have primarily included those with stable/pretreated BM, some trials have allowed patients with active BM. For example, in those with baseline BM in the DESTINY-Breast03 analysis, around half of the patients in both the T-DXd arm (20/43 patients) and in the T-DM1 arm (19/39 patients) had not received prior local BM treatment, which technically is classified as active BM per the FDA, further supporting the antitumor activity of T-DXd in active BM. 33 In a pooled exploratory IC analyses for the DESTINY-Breast01, -02, and -03 trials, 104/148 patients (70.3%) had treated BM and 44/148 patients (29.7%) had untreated BM. IC-ORR for those treated with T-DXd in the treated versus untreated BM cohorts were similar at 45.2% and 45.5%, respectively. The results showed that T-DXd led to higher rates of CNS responses in both treated and untreated BM compared to the comparator arms. 34 Since then, there have been several prospective phase II studies looking into the efficacy of T-DXd in patients with active BM. The DEBBRAH trial is a phase II study evaluating T-DXd in patients with HER2-positive and HER2-low advanced breast cancer, which included patients with stable, untreated, or progressing BM. T-DXd showed IC activity with overall ORR-IC in those with active BM of 46.2% with a disease control rate of 88.9%, suggesting both IC and extracranial efficacy of T-DXd. 35 The other prospective phase II TUXEDO-1 trial enrolled 15 patients with metastatic HER2-positive breast cancer with newly diagnosed/untreated or progressing BM and were treated with T-DXd. At 12 months follow-up, CNS-ORR was 73.3%, with PFS of 14 months. For the six patients with newly diagnosed, untreated BM, response rate was 100%, suggesting that CNS responses were due to T-DXd, not prior radiotherapy, and that T-DXd was effective in metastatic HER2-positive breast cancer patients with active BM. 36
One of the limitations of these prospective phase II studies is their relatively small sample size. In ROSET-BM, a larger retrospective study including 104 patients, T-DXd showed efficacy in IC-ORR, IC-PFS, PFS, and OS in those with HER2-positive BCBM (including 90 patients with active BM) and sustained systemic and CNS disease control in leptomeningeal carcinomatosis. In another systematic review and meta-analysis of 10 studies looking at BCBM that were treated with T-DXd, results also support the IC activity of T-DXd in patients with stable and active BM for HER2-positive BCBM. There was an IC-ORR of 61% and an IC-CBR of 70%, with ORR of 68% in those stable BM versus 60% in those with active BM, and PFS of 15 months. 37 Data from the ongoing DESTINY-Breast07 trial, which looked at T-DXd and pertuzumab compared to T-DXd as first-line treatment in HER2-positive MBC (including those with stable BM), was presented at ASCO 2024. Results showed an ORR of 77.3% (80% CI: 70.0–83.6) with T-DXd versus 82.0% (80% CI: 73.1–88.8) with T-DXd and pertuzumab. At 12 months, the PFS was 77.3% (80% CI: 69.0–83.7) versus 89.4% (80% CI: 81.9–93.9) with T-DXd alone and T-DXd plus pertuzumab, respectively. 38 Based on these promising results, there are many ongoing trials, including DESTINY-Breast12 and HER2CLIMB04, which are evaluating T-DXd either as monotherapy or in combination with other agents in those with HER2 expressing breast cancer with active BM (Tables 1–3). In addition to showing efficacy in the treatment of patients with HER2-positive breast cancer, T-DXd has been shown to be an effective treatment for patients with HER2-low-expressing tumors, irrespective of the HR status. 39 DESTINY-Breast04 was a phase III trial that revealed that T-DXd significantly improved PFS and OS in patients with HER2-low expressing MBC treated with T-DXd when compared to physician’s choice chemotherapy (PCT). In the subgroup analysis of those with baseline BM, IC responses favored T-DXd over PCT in patients with clinically stable and treated baseline BM with IC-ORR of 25% for T-DXd compared to 0% for PCT.22,40
Active clinical trials of ADCs for breast cancer patients with ACTIVE brain metastases.
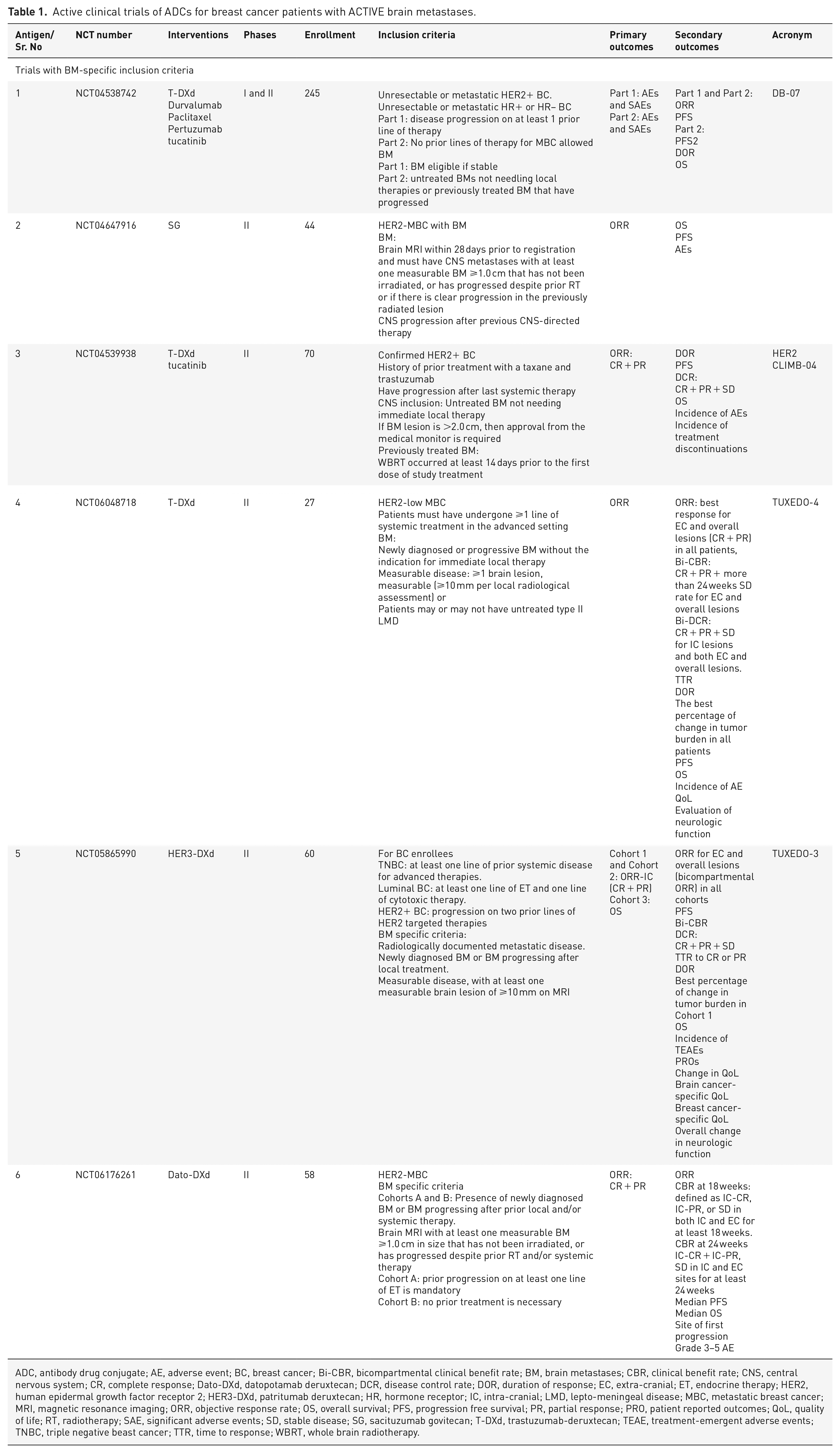
ADC, antibody drug conjugate; AE, adverse event; BC, breast cancer; Bi-CBR, bicompartmental clinical benefit rate; BM, brain metastases; CBR, clinical benefit rate; CNS, central nervous system; CR, complete response; Dato-DXd, datopotamab deruxtecan; DCR, disease control rate; DOR, duration of response; EC, extra-cranial; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; HER3-DXd, patritumab deruxtecan; HR, hormone receptor; IC, intra-cranial; LMD, lepto-meningeal disease; MBC, metastatic breast cancer; MRI, magnetic resonance imaging; ORR, objective response rate; OS, overall survival; PFS, progression free survival; PR, partial response; PRO, patient reported outcomes; QoL, quality of life; RT, radiotherapy; SAE, significant adverse events; SD, stable disease; SG, sacituzumab govitecan; T-DXd, trastuzumab-deruxtecan; TEAE, treatment-emergent adverse events; TNBC, triple negative beast cancer; TTR, time to response; WBRT, whole brain radiotherapy.
Active clinical trials of T-DXd for breast cancer patients with brain metastases.
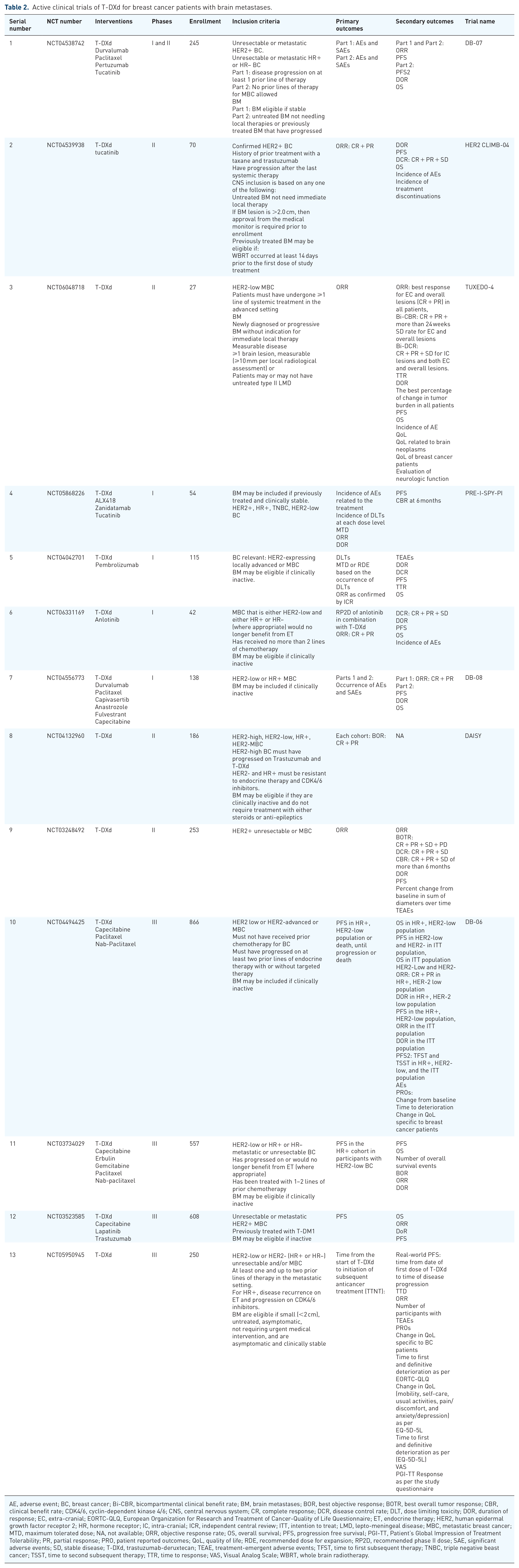
AE, adverse event; BC, breast cancer; Bi-CBR, bicompartmental clinical benefit rate; BM, brain metastases; BOR, best objective response; BOTR, best overall tumor response; CBR, clinical benefit rate; CDK4/6, cyclin-dependent kinase 4/6; CNS, central nervous system; CR, complete response; DCR, disease control rate; DLT, dose limiting toxicity; DOR, duration of response; EC, extra-cranial; EORTC-QLQ, European Organization for Research and Treatment of Cancer-Quality of Life Questionnaire; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; IC, intra-cranial; ICR, independent central review; ITT, intention to treat; LMD, lepto-meningeal disease; MBC, metastatic breast cancer; MTD, maximum tolerated dose; NA, not available; ORR, objective response rate; OS, overall survival; PFS, progression free survival; PGI-TT, Patient’s Global Impression of Treatment Tolerability; PR, partial response; PRO, patient reported outcomes; QoL, quality of life; RDE, recommended dose for expansion; RP2D, recommended phase II dose; SAE, significant adverse events; SD, stable disease; T-DXd, trastuzumab-deruxtecan; TEAE, treatment-emergent adverse events; TFST, time to first subsequent therapy; TNBC, triple negative beast cancer; TSST, time to second subsequent therapy; TTR, time to response; VAS, Visual Analog Scale; WBRT, whole brain radiotherapy.
The DESTINY-Breast06 trial looked at T-DXd versus chemotherapy in HER2-low or HER2-ultralow (immunohistochemistry 0 with membrane staining), HR-positive MBC population who progressed on endocrine therapy and who have not received prior chemotherapy for MBC. Results presented at ASCO 2024 showed that T-DXd significantly improved PFS with 13.2 versus 8.1 months compared to chemotherapy in the HER2-low cohort (hazard ratio, 0.62 (95% CI: 0.51–0.74), p < 0.0001). Similar results were seen in the HER2-ultralow cohort as well. However, data regarding BCBM in this trial was not included and further studies would need to address that gap in a prospective fashion, particularly in patients with active BM where the evidence to date is based on only very small numbers of patients. 41
Sacituzumab govitecan
SG is another ADC made up of an anti-TROP2 monoclonal antibody linked to SN-38, a topoisomerase I inhibitor with a high DAR of 7.6. 42 In a single center, prospective, window of opportunity trial studying concentrations of SG, SN-38, and SN-38G in 20 patients undergoing craniotomy for BCBM, therapeutic levels of the SN-38 payload were reached in resected BM: the mean total concentration of SN-38 was 626 nM via mass spectrometry after receiving SG 10 mg/kg IV the day prior to craniotomy. Preliminary data reported from the study included two out of the four BCBM patients who were treated and underwent mass spectrometry analysis who demonstrated subsequent PR in unresected lesions (ORR of 50% at 12 weeks). 43 SG became the first ADC approved for treatment of previously treated metastatic TNBC based on the phase III ASCENT trial which showed improved PFS (5.6 vs 1.7 months, with hazard ratio of 0.41; 95% CI, 0.32–0.52; p < 0.001) and OS (12.1 vs 6.7 months, with hazard ratio for death, 0.48; 95% CI, 0.38–0.59; p < 0.001) with SG compared to PCT, respectively. The study included those who had stable BM for at least 4 weeks before treatment, but they were excluded from the primary end-point analysis. 23 In the post hoc analysis of the ASCENT study of 61 patients with stable or treated BM, SG demonstrated numerically better outcomes than PCT with PFS (2.8 vs 1.6 months) and tumor responses (3% vs 0%). However, SG showed a similar OS to PCT of 7.0 versus 7.5 months, respectively. 44 These studies have been limited in sample size and ongoing studies are needed to define the efficacy of SG across the BBB in those with active BM (Table 4). In a retrospective multicenter real-world analysis of the efficacy of SG in 12 patients with stable (n = 7) and active BM (n = 5), the IC disease control rate was 42%, with no significant difference in median IC PFS or OS in the groups with active or stable BM. 45 This trial highlights the poor outcomes of patients with triple-negative BCBMs and the ongoing unmet need.
Active clinical trials of multi-ADCs for breast cancer patients with brain metastases.

ADC, antibody–drug conjugate; AE, adverse event; BC, breast cancer; BM, brain metastases; CBR, clinical benefit rate; CCND1, cyclin D1; CDK4/6, cyclin-dependent kinase 4/6; CR, complete response; DCR, disease control rate; DFS4, disease free survival at 4 years; DLT, dose limiting toxicity; DOR, duration of response; HCQ, hydroxychloroquine; HER2, human epidermal growth factor receptor 2; MBC, metastatic breast cancer; MRD, minimal residual disease; ORR, objective response rate; OS, overall survival; PFS, progression free survival; PR, partial response; QoL, quality of life; SD, stable disease; T-DXd, trastuzumab-deruxtecan; TEAE, treatment-emergent adverse events; TNBC, triple negative breast cancer; TTR, time to response.
Active clinical trials of SG for breast cancer patients with brain metastases.

AE, adverse event; BM, brain metastases; CBR, clinical benefit rate; CDK4/6, cyclin-dependent kinase 4/6; CNS, central nervous system; CR, complete response; DLT, dose limiting toxicity; DOR, duration of response; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; HT, hormonal therapy; MBC, metastatic breast cancer; MRI, magnetic resonance imaging; ORR, objective response rate; OS, overall survival; PD1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1; PFS, progression free survival; PR, partial response; QoL, quality of life; RP2D, recommended phase II dose; RT, radiotherapy; SAE, significant adverse events; SD, stable disease; SG, sacituzumab govitecan; SRS, stereotactic radiosurgery; T-DXd, trastuzumab-deruxtecan; TNBC, triple negative beast cancer; TTR, time to response; WBRT, whole brain radiotherapy.
SG is also FDA-approved for the treatment of patients with previously treated unresectable locally advanced or metastatic HR-positive, HER2-negative breast cancer based on the results of the TROPICS-02 study. The study included those with stable BM and showed that SG had statistically significant improvement in PFS compared with chemotherapy, and a 3.2-month median improvement of OS. 24 SWOG S2007 (NCT04647916) is a prospective, phase II trial that will evaluate the IC-ORR of SG for HER2-negative breast cancer patients with BM and includes those with active BM.
Agents under investigation
Datopotamab deruxtecan
Dato-DXd is another anti-TROP2-directed monoclonal antibody linked to derivative of exatecan, a topoisomerase inhibitor, as the cytotoxic payload. The TROPION-PanTumor01 is a phase I study evaluating Dato-DXd in patients with previously treated solid tumors, which included a HR-positive/HER2-negative cohort of 41 patients as well as a TNBC cohort of 44 patients. Those with clinically inactive BM or previously treated BM over 2 weeks ago were eligible for enrollment in the study. Those with active or untreated BM were excluded. Results showed an ORR of 26.8% and 31.8% for the HR-positive/HER2-negative breast cancer and TNBC cohort, respectively. PFS in the HR-positive/HER2-negative breast cancer and TNBC cohort was 8.3 and 4.4 months, respectively. Overall, Dato-DXd showed promising clinical activity in pretreated advanced HR-positive/HER2-negative breast cancer and TNBC and there are ongoing phase III studies to confirm these results 46 (Table 5).
Active clinical trials of Dato-DXd for breast cancer patients with brain metastases.

ADC, antibody–drug conjugate; AE, adverse event; AESI, adverse event of special interest; BC, breast cancer; BM, brain metastases; BOR, best objective response; CBR, clinical benefit rate; CR, complete response; DCR, disease control rate; DLT, dose limiting toxicity; DOR, duration of response; Dato-DXd, datopotamab deruxtecan; EC, extra-cranial; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; IC, intra-cranial; MBC, metastatic breast cancer; MRI, magnetic resonance imaging; NA, not available; ORR, objective response rate; OS, overall survival; PD1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1; PFS, progression free survival; PR, partial response; PRO, patient reported outcomes; QoL, quality of life; RT, radiotherapy; SD, stable disease; TEAE, treatment-emergent adverse events; TFST, time to first subsequent therapy; TNBC, triple negative beast cancer; TROP2, trophoblast cell surface antigen 2; TSST, time to second subsequent therapy; TTD, time to deterioration; TTR, time to response; WBRT, whole brain radiotherapy.
TROPION Breast01 was a phase III trial comparing Dato-DXd versus chemotherapy in patients with previously-treated inoperable or metastatic HR-positive, HER2-negative breast cancer. The study included those with clinically inactive BM or previously treated BM if they had completed therapy over 2 weeks ago prior to trial enrollment. Dato-DXd demonstrated ORR of 36.4%, along with statistically significant and improved PFS (6.9 months) compared with PCT (4.9 months), with a manageable safety profile. Final OS data is still pending. 47
There is also the ongoing phase III TROPION-Breast05 trial, evaluating Dato-DXd alone and in combination with durvalumab compared with PCT plus pembrolizumab in patients with programmed cell death ligand 1 (PD-L1) positive locally recurrent inoperable or metastatic TNBC. 48 To address the BCBM population who have active or untreated BM, another ongoing phase II trial, DATO-BASE, is looking at the effectiveness of Dato-DXd in metastatic TNBC and HR-positive/HER2- negative breast cancer with active BM. 49
The TUXEDO-2 is a phase II study assessing Dato-DXd in patients with TNBC with active BM. Out of the eight patients accrued, six (75%) had newly diagnosed BM while two (25%) had progressive BM. At the time of data cut off, three objective IC responses were observed, equivalent to an ORR of 37.5%. 50
Patritumab deruxtecan
Patritumab deruxtecan (HER3-DXd) is a novel ADC made up of an anti-HER3 monoclonal antibody linked to an exatecan-derived topoisomerase I inhibitor. HER3 overexpression has been associated with a poor prognosis. In a phase I/II trial looking at heavily pretreated advanced breast cancer, HER3-DXd had durable efficacy in patients across multiple clinical subtypes of HER2-positive, TNBC, and HR breast cancer, including 11.5% of patients with stable BM. The ORR rate was 30.1% for HR-positive/HER2-negative BC, 22.6% for TNBC, and 42.9% for HER2-positive BC. Across all breast cancer subgroups, the ORR was 28.6% for both the group with baseline BM as well as no baseline BM. 51 In the HERTHENA-Lung01 trial evaluating HER3-DXd in treatment of EGFR-mutated NSCLC after progression on EGFR inhibitor and platinum-based chemotherapy, HER3-DXd showed clinically meaningful efficacy with durable responses, including in CNS metastases, with a CNS ORR of 33.3% in 30 patients with nonirradiated BM. 52 There are ongoing trials evaluating HER3-DXd, including phase II TUXEDO 3 trial evaluating the efficacy of HER3-DXd for MBC with active BM who had received at least one line of prior systemic therapy for advanced disease 53 (Table 6).
Active clinical trials of miscellaneous ADCs for breast cancer patients with brain metastases.

ADC, antibody–drug conjugate; AE, adverse event; AESI, adverse event of special interest; BC, breast cancer; Bi-CBR, bicompartmental clinical benefit rate; BM, brain metastases; BOR, best objective response; CBR, clinical benefit rate; CEACAM5, CEA cell adhesion molecule 5; CDK, cyclin-dependent kinase; CNS, central nervous system; CR, complete response; CRR, complete response rate; ctDNA, circulating tumor DNA; DCR, disease control rate; DLT, dose limiting toxicity; DOR, duration of response; ECOG-PS, Eastern Cooperative Oncology Group-Performance Status; EDL, expansion dose level; EOT, end of treatment; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; LMD, lepto-meningeal disease; MBC, metastatic breast cancer; MRI, magnetic resonance imaging; OD, optimal dose; OR, objective response; ORR, objective response rate; OS, overall survival; PD-1 Ab, programmed cell death protein 1 antibody; PD-L1, programmed cell death ligand 1; PFS, progression free survival; PR, partial response; QoL, quality of life; RD, recommended dose; RDE, recommended dose for expansion; ROR2, Receptor Tyrosine Kinase Orphan Factor 2; RP2D, recommended phase II dose; SAE, significant adverse events; SD, stable disease; SOC, standard of care; SRS, stereotactic radiosurgery; TEAE, treatment-emergent adverse events; TNBC, triple negative beast cancer; T-DXd, trastuzumab-deruxtecan; TROP2, trophoblast cell surface antigen 2; TTP, time to progression; TTR, time to response; WBRT, whole brain radiotherapy.
Trastuzumab duocarmazine
Trastuzumab duocarmazine is another novel ADC with the anti-HER2 monoclonal antibody trastuzumab linked to the cytotoxic payload duocarmazine. It has a DAR of 2.4–2.8. The TULIP trial was a phase III study looking at trastuzumab duocarmazine versus PCT (lapatinib and capecitabine, trastuzumab combined with either vinorelbine, capecitabine, or eribulin) in previously treated HER2-positive MBC who progressed on two or more lines of HER2-directed therapy. The study included those with stable BM and showed superior PFS of 7.0 versus 4.9 months for trastuzumab duocarmazine compared to PCT, respectively. OS was longer in the trastuzumab duocarmazine group, but not statistically significant, with 21.0 months in the trastuzumab duocarmazine cohort versus 19.5 months in the PCT cohort. 54
Enfortumab vedotin
Enfortumab vedotin (EV) is another ADC composed of a monoclonal antibody targeting Nectin-4, which is a cell adhesion molecule highly expressed in epithelial tumors, linked with the cytotoxic payload, monomethyl auristatin E, which is a microtubule disruptor. In in vitro and in vivo preclinical studies, the antitumor activity of EV has been demonstrated for both HR-positive and TNBC. 55 The ongoing EV 202 phase II trial is looking at the efficacy and tolerability of EV in previously treated advanced solid tumors, with a HR-positive/HER2-negative as well as a TNBC cohort. The results presented at ASCO 2024 showed an ORR of 19% in the TNBC cohort of 42 patients and 15.6% in the HR-positive/HER2-negative cohort of 45 patients. OS was 12.9 months in the TNBC cohort versus 19.8 months in the HR-positive/HER2-negative group. 56 Activity in patients with active BM is unknown yet and will be evaluated in the future (Table 6).
Discussion
Many ADCs, including T-DM1, T-DXd, and SG have already made their way into and revolutionized the treatment of MBC. Although there have been major advances in ADCs for HER2-positive BCBM with the approvals of T-DM1 and T-DXd, therapeutic options after T-DXd still need further investigation. Despite these therapeutic advances made in recent years, BCBM remains difficult to treat with modest activity to date for ADCs, especially with HER2-negative BCBM. There is a continued need for novel agents and more inclusion in clinical trials. Many of the trials leading to the approval of these ADCs only included patients with treated or asymptomatic BM. Although some post hoc and subgroup analyses of these trials have demonstrated that ADCs show IC activity, a better understanding of IC efficacy of ADCs and the inclusion of patients with active BM in clinical trials, such as the TUXEDO-1 and DEBBRAH trials, is needed to optimize treatment of BCBM.
Many questions remain about the optimal sequence of using ADCs. The MARIANNE trial suggested T-DM1 was a reasonable first-line therapeutic option for metastatic HER2-positive BC, as it was non-inferior but not superior to the combination of trastuzumab and taxane. However, with the improved PFS after the addition of pertuzumab to trastuzumab and taxane in the CLEOPATRA trial, that combination has remained the standard of care (SOC) for first-line metastatic HER2-positive breast cancer. 45 There are multiple other ongoing studies looking at whether first-line ADCs would be superior to the current SOC.
More research is also necessary to look at whether certain biomarkers may predict both IC and extracranial response to ADCs. The DAISY phase II trial looked at the efficacy of T-DXd in MBC patients that were divided into three cohorts based on their HER2 status of either HER2-positive, HER2-low, or HER2-0. In the subgroup of patients with BM for the DAISY study, PFS was 13 and 4.1 months in the HER2-positive and HER2-low patients, respectively. 57 Results suggest that HER2 expression is a determinant of T-DXd efficacy, with ORR of 70.6% in the HER2-positive cohort, ORR of 37.5% in the HER2-low cohort, and ORR of 29.7% in the HER2-0 cohort. 58 Another study looking at the dynamic HER2 expression in BCBM showed that 40% of HER2-0 primary or extracranial tumors gain HER2 expression in the brain, which may have significant therapeutic implications given improved OS seen with HER2-directed ADCs and emerging evidence of their CNS activity. Noninvasive diagnostics to assess HER2 expression in BCBM are strongly needed. 59 TROP2, HER3, and other biomarker expression and association with treatment efficacy are still unknown and future studies are warranted.
Since the SOC for symptomatic BM is still local therapy with surgery or targeted radiation, another area of investigation is how to best select between local or radiotherapy approaches versus systemic therapy, and how to safely sequence modalities if both are needed. Several retrospective studies have suggested that concurrent ADCs (T-DM1 or T-DXd) and radiotherapy were associated with a higher risk of symptomatic radiation necrosis in HER2-positive breast cancer. 60 This also becomes especially clinically relevant in cases where patients are at risk for increased CNS toxicity if they need salvage radiotherapy after progression on systemic treatment with ADCs. Further investigation into the safety of these combined modality treatments and with different ADCs is needed.
Many other trials underway are also assessing the combination of ADCs with systemic treatments, such as chemotherapy, immune checkpoint inhibitors, or targeted therapies (such as poly ADP ribose polymerase inhibitors). DESTINY-Breast07 is an ongoing phase I/II trial evaluating T-DXd monotherapy or in combination with other agents (including durvalumab, pertuzumab, paclitaxel, durvalumab + paclitaxel, or tucatinib) in previously untreated HER2-positive MBC. The study included patients with active or stable, untreated, or previously treated and progressive BM. Preliminary results at the European Society of Medical Oncology (ESMO) Breast 2024 showed promising efficacy in both the T-DXd and the T-DXd + pertuzumab cohort with ORR in the 80% for both groups. 38 The ongoing DESTINY-Breast09 also includes patients with inactive or treated and asymptomatic BM and is a phase III trial looking at T-DXd with or without pertuzumab compared with SOC taxane, trastuzumab, and pertuzumab as first-line treatment in patients with HER2-positive MBC. 61
Immune checkpoint inhibitors have also been tested in combination with ADCs, with some studies showing promising results in BCBM. In the KATE2 study, the combination of atezolizumab and T-DM1 did not show improved PFS in advanced HER2-positive breast cancer. 51 BEGONIA is a phase I/II trial with a combination of Dato-DXd + durvalumab as first-line treatment in metastatic TNBC. Updated results showed ORR was 79% with a PFS of 13.8 months, suggesting that the Dato-DXd and durvalumab combination has durable response rates in first-line treatment of metastatic TNBC. 62 As above, TROPION-Breast05 is evaluating Dato-DXd alone and in combination with durvalumab in patients with advanced or metastatic TNBC whose tumors are PD-L1 positive. 48 There are ongoing phase II trials with SG and pembrolizumab in first-line treatment of metastatic TNBC (ASCENT 04). 63 The Morpheus-panBC trial is an umbrella study evaluating multiple treatment combinations in patients with locally advanced/MBC. Interim results from the atezolizumab and SG arm in the Morpheus-panBC trial for first-line treatment for inoperable locally advanced and metastatic TNBC patients were presented as ESMO Breast 2024. ORR was 76.7% with combination atezolizumab and SG versus 66.7% with atezolizumab plus nab-paclitaxel. PFS data was immature but did trend toward benefit with atezolizumab plus SG with 12.2 versus 5.9 months with the control arm, showing promising activity with first-line atezolizumab plus SG treatment. 64 However, a major limitation of these studies is the exclusion of active BM patients and thus the lack of data about CNS activity of immunotherapy combinations, which warrants further study.
Based on the known IC efficacy of TKIs, with tucatinib per the HER2CLIMB trial, there may be a synergistic effect in the CNS when combined with ADCs. This is supported by the results of the HER2CLIMB-02 looking at the combination of tucatinib with T-DM1 as discussed above. 28 The ongoing HER2CLIMB-04 phase II trial is evaluating the combination of tucatinib plus T-DXd in patients with HER2-positive advanced/MBC and includes those with active BM. 65
Conclusion
In conclusion, ADCs represent a significant advancement in the treatment of BCBM, offering a targeted approach to a previously challenging clinical issue. Clinical trials and emerging data have already demonstrated that ADCs can effectively reduce tumor burden and improve PFS and OS, leading to FDA approvals for T-DM1, T-DXd, and SG in the treatment of MBC. However, there remains limited but promising data showing the activity of ADCs in patients with active BM. Given the high mortality and morbidity associated with BM, further studies including patients with active BM are needed to address this unmet need. Subgroup analyses have already been critical in discovering novel therapeutic agents that have IC efficacy. This is best exemplified by the subgroup analyses such as that of the NALA trial which demonstrated IC efficacy of the neratinib + capecitabine combination; those of the KAMILLA trial and TH3RESA trial which uncovered the IC efficacy of TDM-1; and lastly, that of DESTINY-Breast01 which illuminated the IC efficacy of T-DXd. Thus, retrospective subgroup analyses of early-stage clinical trials of novel ADCs will be crucially important in obtaining evidence of IC efficacy. This, in turn, will help to identify promising ADCs that would be more likely to yield a positive read-out in clinical trials that are designed to assess IC efficacy against active BCBM lesions. We have provided a comprehensive list of novel ADCs in early-stage clinical trials in which patients with stable, not active, BCBM lesions are eligible to participate (Tables 2–6). Future studies should also focus on optimizing drug delivery mechanisms, minimizing resistance, and exploring combinations with other modalities to further enhance efficacy. Many ongoing clinical trials will provide further insights into the efficacy and safety of ADCs and combination therapy, potentially offering a new therapeutic option for this challenging patient population with previously limited options.
