Abstract
Advanced breast cancer (ABC) that is positive for hormone receptors (HRs) and human epidermal growth factor receptor 2 (HER2) is a cancer subtype with distinctive characteristics. The primary treatment guidelines suggest that a combination therapy comprising anti-HER2 therapy and chemotherapy should be administered as the initial treatment for HR-positive/ HER2-positive (HR+/HER2+) ABC. However, crosstalk between the HR and HER2 pathways can partially account for the resistance of HR+/HER2+ disease to HER2-targeted therapy. This, in turn, provides a rationale for the concomitant administration of HER2-targeted therapy and endocrine therapy (ET). Many clinical studies have confirmed that the combination of HER2-targeted therapy and ET as a first-line treatment is not inferior to the combination of HER2-targeted therapy and chemotherapy, and support its use as a first-line treatment choice for HR+/HER2+ ABC. Other drugs, such as antibody–drug conjugates, cyclin-dependent kinase 4/6 inhibitors, phosphatidylinositol 3-kinase–protein kinase B (AKT)–mammalian target of rapamycin inhibitors, and programmed cell death protein 1 or programmed cell death ligand 1 inhibitors, may also improve the prognosis of patients with breast cancer by blocking signaling pathways associated with tumor proliferation and break new ground for the treatment of HR+/HER2+ ABC.
Keywords
Introduction
Since 2020, breast cancer has become the most prevalent cancer worldwide.1,2 Among breast cancers, 20% overexpress human epidermal growth factor receptor 2 (HER2) and nearly 50% overexpress hormone receptors (HRs), including estrogen receptor (ER) and/or progesterone receptor (PR). 3 Compared to HR−/HER2+ breast cancer, HR+/HER2+ disease is more aggressive, leading to a lower first 5-year recurrence risk and poorer prognosis. 4 However, the recurrence rate markedly increases after the 5th year, resulting in similar long-term outcomes for patients with HR+/HER2+ and HR−/HER2+ breast cancer. 5 HR+/HER2+ breast cancer cells that highly express ER have similar biological characteristics and behaviors to those of HR−/HER2+ cells. 6 Furthermore, luminal A or B subtypes – which are hormone dependent with low epidermal growth factor receptor (EGFR)/HER2 pathway activity but a high mutation rate of the phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha – are less sensitive to HER2-targeted therapy but have a favorable prognosis. 7 However, 30% of patients present with marked HER2 overexpression, strong HER2/EGFR pathway activity, and a high tumor cell proliferation rate. These features contribute to the therapy responsiveness and prognostic differences observed among HR+/HER2+ patients. Most guidelines have established anti-HER2 therapy plus chemotherapy as the standard first-line treatment for patients with HR+/HER2+ advanced breast cancer (ABC). 8 We aimed to investigate the optimal treatment regimen for patients with HR+/HER2+ breast cancer while also summarizing some innovative therapeutic approaches tailored to this patient population, to provide better guidance for clinical practice.
Molecular biological characteristics of HER2+/HR+ breast cancer cells
HER2 regulates the development and growth of tumor cells
HER2, a member of the HER family, consists of three main components – an intracellular protein tyrosine kinase domain, an extracellular ligand-binding domain, and a single-chain transmembrane domain – which are responsible for receiving extracellular signals and activating downstream signaling pathways. 9 In the HER family, HER1, HER3, and HER4 all have high-affinity specific ligands, making HER2 the only receptor in the family without a known high-affinity specific ligand. 10 HER2 primarily undergoes heterodimerization with other members of the HER family and binds to ligands to form complexes. The formation of these complexes triggers the phosphorylation of the tyrosine kinase domain in the cytoplasm, activating the kinase and downstream pathways. 11 These pathways mainly include the Ras–Raf–mitogen-activated protein kinase (MAPK) pathway and the phosphatidylinositol 3-kinase (PI3K)–AKT–mammalian targets of rapamycin (mTOR) pathway (Figure 1), which are closely related to the promotion of tumor cell growth and proliferation, and the inhibition of apoptosis.
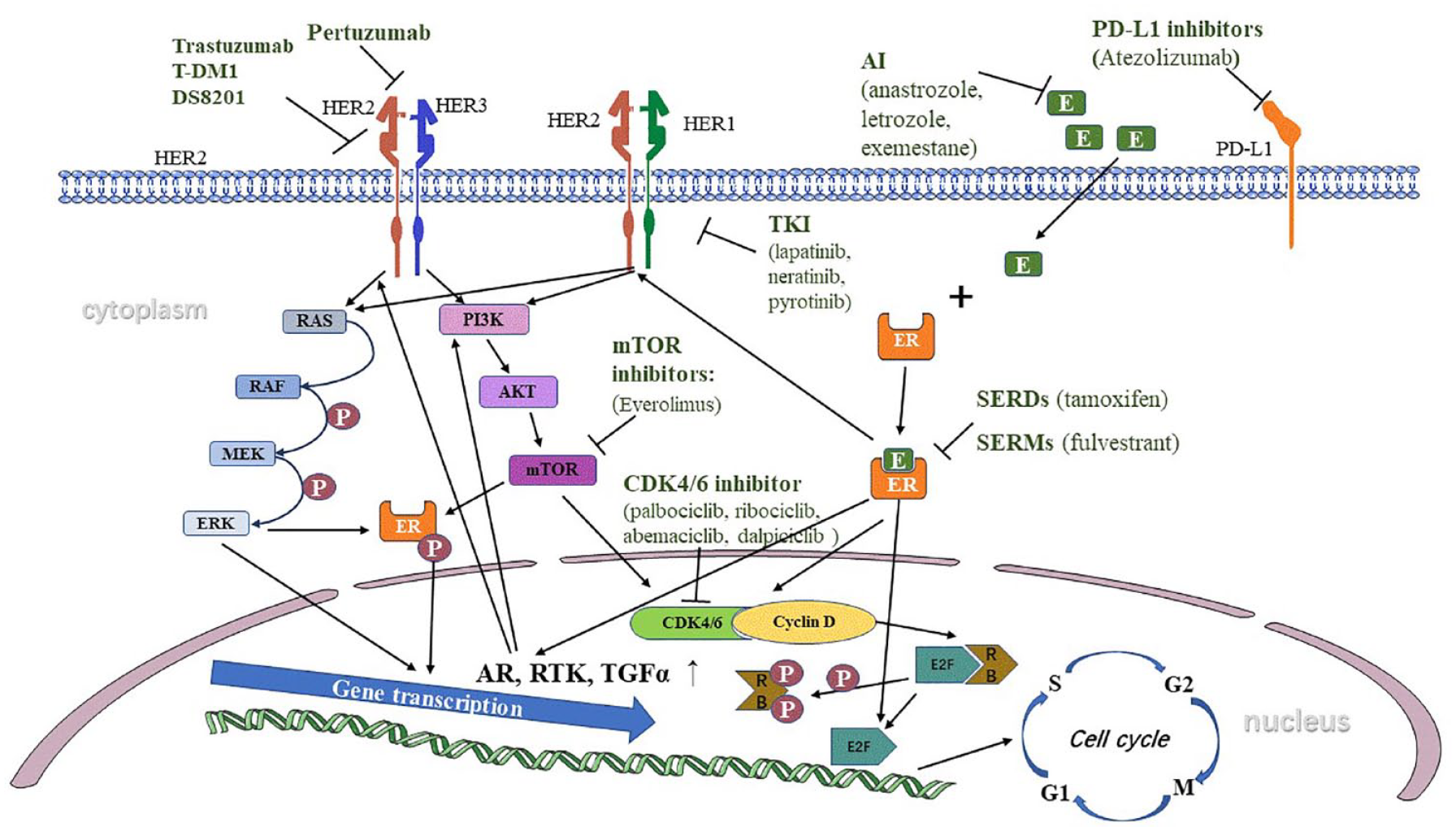
Crosstalk between HER2 signaling pathway and ER signaling pathway and drugs for HR+/HER2+ advanced breast cancer patients.
ER regulates the development and growth of tumor cells
The growth of breast cancer cells is largely regulated by the activation of ER by the steroid hormone estrogen. The activation of the ERα and ERβ nuclear receptors promotes the proliferation and survival of both normal and tumor tissues through genomic regulation, by promoting gene transcription, and non-genomic regulation, via activation of related signaling pathways. 12 Estrogen binds the ER and regulates the cell cycle by inducing the expression of the MYC gene and the G1/S-specific cyclin D1. 13 Upon binding of estrogen to the nuclear receptor portion of ER, the signal is transmitted to interact with transcription factors (such as Fos and Jun) or the estrogen response elements of target genes. This interaction leads to the activation of activating protein 1 or specificity protein 1 binding sites in the promoter regions of target genes, followed by the initiation of the transcription and translation of relevant genes. 14 ER stimulation by estrogen also upregulates the transcription of several growth factors that are crucial for breast development, including insulin-like growth factor-1 biphasic proteins and epithelial growth factor.15,16 The estrogen–ER axis that contributes to normal breast development also plays a role in breast hyperplasia and tumorigenesis. Due to the strong dependence of breast tumor development on the estrogen–ER axis, endocrine therapy (ET) has become an important treatment modality for premenopausal and postmenopausal patients with ER-positive breast cancer.
Interaction between the HER2 and ER signaling pathways
Activation of the HER signaling pathway promotes endocrine resistance in tumor cells
Studies have confirmed that HER2 and its downstream signaling pathways promote endocrine resistance in HR+/HER2+ breast cancer cells at the cellular level. HER2 amplification activates selective survival pathways, such as the PI3K–AKT and MAPK pathways, reducing the sensitivity of breast cancer cells to ET. 17 Laboratory studies revealed that the application of HER2 inhibitors to HER2-amplified tamoxifen-resistant MCF-7 cells could reverse tamoxifen resistance, indicating that HER2 and its downstream signaling pathways are involved in endocrine resistance in breast cancer cells. 17 In the tumor microenvironment, where estrogen levels are extremely low, both heterodimers and homodimers can activate downstream signaling pathways, including PI3K–AKT and Ras–Raf–Mek–MAPK, through HER1 and HER2. This increases the sensitivity of breast cancer cells to estrogen, promoting proliferation and potentially leading to endocrine resistance. 18 In the absence of estrogen, HER2 can still regulate estrogen-related signaling pathways in two distinct ways. First, the signaling pathway mediated by HER2 can decrease the expression level of ER while increasing ER phosphorylation, thereby activating the downstream signaling pathway initiated by ER (Figure 1). 19 Second, the activation of the HER1 and HER2 signaling pathways in low-estrogen conditions can recruit ER co-activators and disrupt the interaction between ER co-repressors and ER. This leads to the stimulation of ER-mediated signaling pathways. 20
Activation of ER-related signaling pathways promotes tumor cell resistance to HER2-targeted therapy
HER2 overexpression is involved in the development of endocrine resistance, and ER and associated downstream pathways can induce resistance to anti-HER2 therapy in breast cancer cells by interacting with HER2 signaling pathways. ERα activation is mainly responsible for breast malignancies. 21 Following ERα activation, growth factor receptors – such as the androgen receptor, tumor growth factor α, and receptor tyrosine kinases associated with its downstream pathways – are also highly expressed. The elevated expression of these growth factor receptors simultaneously augments the activity of pathways downstream of HER2, leading to resistance to HER2 therapy in HER2+ breast cancer.22 –24 ER activation can activate EGFR, HER2, other growth factor receptors, and HER2-targeted therapy resistance-related kinase cascades, such as PI3K–AKT, leading to cell migration and upregulation of the chemokine receptor CXCR4. 25 A study demonstrated that administration of HER2-targeted therapy can upregulate the expression of ERα and ER-related genes and fail to inhibit the phosphorylation of AKT and ERK, leading to tumor proliferation. 26 Meanwhile, AIB1, an important ERα coactivator, is upregulated by HER2-targeted therapy and causes the activation of ER signaling and continuous growth of HER2-targeted therapy-resistant breast cell lines. 26 In addition to promoting the activity of pathways downstream of HER2, ERα can directly activate HER2 on the cell membrane and prompt the proliferation of tumor cells.27,28
In vitro studies found that breast cancer cell lines resistant to lapatinib developed differential upregulation of ER-related genes compared with lines that were not resistant, which indicates the involvement of ER signaling in acquired HER2-targeted therapy resistance. 29 Adding anti-estrogen therapy to lapatinib stopped ER+/HER2+ breast cancer cells from developing acquired lapatinib resistance. 26 In mice bearing xenografted HR−/HER2+ tumors, the tumors were converted to ER+ after 2 weeks of lapatinib neoadjuvant therapy, providing support for the use of combination anti-HER2 and ET. 25 Another study found that patients with HER2+ metastatic breast cancer who developed primary resistance to trastuzumab-derivative of maytansine (T-DM1) were negative for HER2 gene overexpression and positive for ER and/or PR by immunohistochemistry, highlighting that the ER pathway may contribute to the development of resistance to HER2-targeted therapy. 30
Anti-HER2 therapy
Anti-HER2 medications have emerged as crucial elements of the therapeutic approach for patients with HR+/HER2+ ABC. Several anti-HER2 drugs are commonly employed in clinical practice, as discussed below.
Monoclonal antibodies
Trastuzumab is the first humanized monoclonal antibody targeting HER2; its main mechanisms of action are binding to extracellular domain IV of HER2 and suppressing the expression of downstream signals. 31 Moreover, trastuzumab is capable of binding to HER2 expressed on the surface of breast cancer cells, thereby stimulating nonspecific immune cells to elicit cytotoxic effects through antibody-dependent cellular cytotoxicity. 32 Since its approval by the US Food and Drug Administration in 1998, it has been extensively and irreplaceably used in treating patients with HER2+ breast cancer. However, for patients with HER2+ ABC who received prior trastuzumab-based therapy, harboring a high HER2 gene copy number may be associated with a worse prognosis. 33 Pertuzumab binds to extracellular domain II of HER2 and hinders the heterodimerization of HER2 and HER3 to obstruct the downstream signaling pathways, ultimately impeding tumor growth. 34 Without affecting dimerization, pertuzumab also demonstrated a complementary mechanism to trastuzumab. 35
Tyrosine kinase inhibitors
Tyrosine kinase inhibitors (TKIs), such as lapatinib, pazopanib, and lenvatinib, are extensively utilized in clinical practice. Lapatinib, a small-molecule TKI, binds reversibly to the HER1 and HER2 intracellular tyrosine kinase domain that contains an ATP binding site. This suppresses phosphorylation and blocks downstream signaling, which inhibits tumor growth and promotes tumor cell apoptosis. 36 Approximately 25% of patients with HER2+ breast cancer treated with trastuzumab experienced rapid recurrence. 37 Lapatinib-based treatment is effective in trastuzumab-pretreated patients with HER2+ ABC. 38 Pyrotinib is an irreversible pan-ErbB TKI that has been confirmed to suppress the proliferation of HER2-overexpressing breast cancer cells both in vivo and in vitro. 39 In contrast to lapatinib, pyrotinib can broadly inhibit the downstream signaling pathways of HER1, HER2, and HER4. 40 Neratinib, an irreversible pan-ErbB TKI, produces the same pharmacological effects as pyrotinib. 41
Antibody–drug conjugates
In addition to the widespread usage of monoclonal antibodies and small-molecule TKIs, there has been a gradual emergence of antibody–drug conjugates (ADCs) in recent years. T-DM1 and trastuzumab deruxtecan (DS-8201) are notable among these ADCs. T-DM1 is a novel anti-HER2 ADC in which trastuzumab is bound to the microtubule inhibitor maytansine. 42 The combination mediates synergistic effects by ensuring selective binding of the cytotoxic agent to malignant breast cells, thereby increasing its tumor cell cytotoxicity while reducing the occurrence of adverse events (AEs). 43 DS-8201 belongs to a novel class of anti-HER2 ADC that employs an innovative conjugation strategy to link the anti-HER2 monoclonal antibody and the topoisomerase I inhibitor DXd (an ixabepilone derivative). The anti-HER2 antibody component of DS-8201 is a humanized monoclonal IgG1 with an amino acid sequence that is identical to trastuzumab. 44 In laboratory investigations, DS-8201 has demonstrated the ability to hinder the proliferation of tumor cells with elevated or diminished HER2 expression, as well as that of cells that have developed T-DM1 resistance. 45
ET drugs
Hormone therapy plays an important role in treatment regimens for patients with HR+/HER2+ ABC. Selective estrogen receptor modulators (SERMs), selective estrogen receptor downregulators (SERDs), and aromatase inhibitors (AIs) have gained approval for adjuvant therapy in breast cancer patients with ER-positive tumors. 46 In premenopausal women, aromatase, predominantly produced by the ovaries, is converted into estrogen. In postmenopausal women, however, estrogen is mainly synthesized in non-glandular tissues (such as subcutaneous fat), and its production markedly declines. 47 This leads to completely distinct hormonal therapy strategies for premenopausal and postmenopausal women diagnosed with ABC.
Aromatase inhibitors
AIs are typically categorized into non-steroidal and steroidal types. Anastrozole and letrozole are common examples of non-steroidal drugs, whereas exemestane represents the most frequently utilized steroidal variant. 48 AIs exert their effects in postmenopausal women by inhibiting the conversion of androgens to estrogens, leading to reduced estrogen levels throughout the body. This ultimately impedes the proliferation of estrogen-dependent tumor cells and contributes to therapeutic efficacy.
Selective estrogen receptor modulators
SERMs, such as tamoxifen, competitively bind to ERs and exhibit a dual agonist/antagonist capacity. By blocking the binding of ligands to receptors, SERMs effectively inhibit ER downstream pathways. These drugs are primarily employed in premenopausal patients. 49
Selective estrogen receptor downregulators
SERDs, such as fulvestrant, primarily function by inducing the degradation of the ERα protein or blocking ER transcriptional activity.50 –52 A recent study suggested that fulvestrant and analogous ER antagonists exert their inhibitory effects predominantly via reducing nuclear ER activity. 53 Currently, some orally administered SERDs with pharmacological properties that may be superior to fulvestrant are also under development. 54
Comparison of different treatment regimens for patients with HR+/HER2+ ABC
Targeted therapy for HER2 used in conjunction with chemotherapy has become an established first-line treatment for patients with HER2+ ABC. The phase III clinical trial for H0648g verified the remarkable efficacy of combining HER2-targeted therapy with chemotherapy, demonstrating that patients receiving chemotherapy and HER2-targeted therapy achieved longer median progression-free survival (mPFS) than patients receiving single-agent chemotherapy (7.4 versus 4.6 months, respectively; p < 0.001). 55 In addition, the NCIC CTG MA.31 study compared the efficacy of trastuzumab and the small-molecule TKI lapatinib, both in combination with paclitaxel. The trastuzumab group had a significantly longer mPFS compared with the lapatinib group (11.3 versus 9.0 months, respectively; p = 0.001). Patients with higher continuous ER expression values achieved longer PFS, indicating that the subset of patients with HR+/HER2+ cancer had a better prognosis than those with HR−/HER2+ cancer in the study. In terms of overall survival (OS), the lapatinib group did not show an advantage over the trastuzumab group (p = 0.03). 56 For patients with HR+/HER2+ ABC, combining a targeted HER2 monoclonal antibody with chemotherapy has yet to be established as the superior choice for first-line treatment. The advent of dual-targeted first-line treatments was heralded by the CLEOPATRA trial, which revealed a difference of 6.3 months (18.7 months for pertuzumab + trastuzumab + docetaxel group versus 12.4 months for trastuzumab + docetaxel group) in mPFS between patients receiving dual HER2-targeted therapy versus trastuzumab plus chemotherapy, and a difference of 15.7 months in median OS (56.5 versus 40.8 months), for whose difference is larger than the difference in mPFS. 57 According to subgroup analysis, the combination of dual-targeted anti-HER2 therapy with chemotherapy has clinical efficacy for patients bearing HR+/HER2+ ABC [hazard ratio = 0.74; 95% confidence interval (CI), 0.58–0.96]. 57 The PUFFIN (YO29296) study in Chinese patients revealed that, compared with trastuzumab plus chemotherapy, dual-targeted anti-HER2 therapy plus chemotherapy prolonged the mPFS of patients with HER2+ ABC for 4 months and that of patients with HR+/HER2+ ABC for 5.4 months, which is similar to the findings of CLEOPATRA.57 –59 Median OS was immature58,59 (Table 1).
Comparison of different treatment options from published articles.

ET has been a crucial aspect of the treatment protocol for patients with HR+ tumors. In this section, we aim to elucidate the pivotal role of ET in the therapeutic approach for patients with breast cancer and present the trial results in Table 1.
Trastuzumab plus ET versus trastuzumab
The TAnDEM study compared the effectiveness of trastuzumab in conjunction with anastrozole versus trastuzumab monotherapy as the first-line treatment in patients with HR+/HER2+ breast cancer. 60 It illustrated that the combination of ET and anti-HER2 therapy provided greater clinical benefit than monotherapy with trastuzumab (mPFS: 4.8 versus 2.4 months, respectively; p = 0.0016). There was no significant difference detected in the OS of the trastuzumab group and ET plus trastuzumab group (18.5 versus 23.9 months, respectively; p = 0.325), but a prolongation of 4.6 months was achieved by trastuzumab plus ET. 60 Patients receiving anastrozole alone were allowed to switch to another treatment arm after disease progression, which may have contributed to the reduction in OS benefit with trastuzumab plus anastrozole, by prolonging OS in the anastrozole alone group. 60 Furthermore, 70% of the patients in the trastuzumab monotherapy group experienced disease progression but still obtained clinical benefit after receiving a combination therapy of trastuzumab with anastrozole. 60 These findings further confirmed that the combination use of anti-HER2 therapy and ET can successfully reverse resistance to single-agent anti-HER2 therapy. However, the incidence of grade 3 or higher AEs in the trastuzumab combined with anastrozole group was 28% (involving mainly diarrhea, fatigue, and vomiting), which was higher than that in the trastuzumab monotherapy group (16%). Taking into account both the side effects and clinical survival benefits, the combination of anti-HER2 therapy and ET would still be the preferred first-line treatment for this subset of patients (Table 1).
TKIs plus ET versus ET
To determine whether the first-line treatment choice for patients with HR+/HER2+ ABC can eradicate the need for anti-HER2 therapy, the phase III clinical trial EGF30008 (NCT00073528) compared the survival benefits of letrozole plus lapatinib and letrozole with placebo for the first-line treatment of patients with HR+/HER2+ ABC. 61 The letrozole combined with the lapatinib group attained a longer mPFS (8.2 versus 3.0 months, compared with letrozole with placebo; p = 0.008). Furthermore, analysis of the clinical benefit rates (28% versus 15%, combination versus placebo; p = 0.021) and objective response rates (48% versus 29%, combination versus placebo; p = 0.003) revealed that the patients in the combination therapy group achieved better clinical benefits than those in the letrozole with the placebo group. However, consistent with the results of TAnDEM, the OS for patients was not significantly improved by adding lapatinib to letrozole, indicating that the efficacy of lapatinib plus letrozole needs further validation.60,61 Analysis of AEs revealed that the incidence of grade 3 or higher AEs was higher in the letrozole monotherapy group. Thus, based on either the survival benefit or incidence of AEs, letrozole combined with lapatinib may be the superior first-line therapy choice for patients with HR+/HER2+ ABC (Table 1). For patients in the eLEcTRA study, adding trastuzumab to letrozole improved mPFS by 10.8 months. The OS data were not provided, but no significant difference was found between the OS of the letrozole alone and trastuzumab plus letrozole groups. 62 Due to the relatively small sample size of the study, the results of eLEcTRA should be interpreted with caution.
Anti-HER2 therapy plus ET versus anti-HER2 therapy plus chemotherapy
A real-world study examined data from a database encompassing 6234 patients diagnosed with ABC of the HR+/HER2+ subtype (Supplemental Table 1). 63 The patients had undergone monotherapy chemotherapy, monotherapy ET, combination anti-HER2 treatment with chemotherapy, or combination anti-HER2 treatment with ET. Multivariate analysis revealed that receiving ET was an independent factor for improved patient prognosis. The cohort that received combination ET and anti-HER2 treatment exhibited the highest 5-year OS rate (47.5% versus 39.8% for patients receiving chemotherapy + anti-HER2 therapy) (Supplemental Table 1). Moreover, in the multivariate subgroup analysis that controlled for other prognostic factors, patients who received ET plus anti-HER2 treatment had a lower risk of mortality than those who received chemotherapy and anti-HER2 treatment (hazard ratio: 0.74; 95% CI, 0.61–0.91; p = 0.004). 63
In a Chinese patient population, the SYSUC-002 (NCT01950182) trial compared the clinical advantages of trastuzumab in combination with ET (group) and trastuzumab in combination with chemotherapy therapy (CT group) as a first-line treatment for individuals with HR+/HER2+ ABC. 64 All patients with HR+ disease were defined as having >10 ER+ and/or PR+ cells. After a median follow-up period of 30.2 months, the mPFS was 19.2 months (95% CI, 16.7–21.7) in the single-targeted anti-HER2 therapy with ET group and 14.8 months (95% CI, 12.8–16.8) in the single-targeted HER2 therapy combined with chemotherapy group, resulting in a difference of 4.4 months (hazard ratio = 0.88; 95% CI, 0.71–1.09; p < 0.0001) (Table 1). In addition, single-agent anti-HER2 therapy combined with ET has a lower rate of AEs. The majority of AEs in the ET group were grades 1–2. Joint pain (16.8%), muscle pain (16.3%), and fatigue (15.8%) were the most commonly reported AEs. By contrast, alopecia (63.8%), leucopenia (50.0%), and nausea (47.5%) were the most frequently reported AEs in the CT group. Patients in the ET group had a significantly lower prevalence of grade 3–4 AEs compared to those in the CT group [6 (3.1%) versus 100 (51.0%); p < 0.01], confirming that ET plus HER2-targeted therapy can improve the quality of life of the patients.
Dual anti-HER2 therapy plus ET versus single-agent trastuzumab plus ET
The CLEOPATRA trial demonstrated, via subgroup analysis, a survival advantage when utilizing dual-targeted combination chemotherapy in patients with HR+/HER2+ ABC. That finding prompted the PERTAIN (NCT01491737) study to compare the efficacy of trastuzumab plus pertuzumab and AI with trastuzumab plus AI to investigate whether dual-targeted anti-HER2 therapy could provide more distinct clinical benefit than single-targeted anti-HER2 therapy in this patient population.65,66 Chemotherapy was added at the discretion of the clinician. The results of the study revealed that dual-targeted anti-HER2 therapy coupled with ET had greater survival advantages than single-targeted therapy combined with ET, with a mPFS of 18.89 months compared to 15.8 months, respectively (p = 0.0070). No deaths due to severe side effects were reported in either of the two experimental groups. However, the rates of grade 3 or higher side effects were 50.8% and 38%, respectively. The primary side effects of dual-targeted anti-HER2 therapy were nausea, diarrhea, and alopecia. Based on the findings of this study, dual-targeted therapy with pertuzumab and trastuzumab would be the preferred first-line treatment for patients in the HR+/HER2+ group in combination with ET.
Dual anti-HER2 therapy plus ET and chemotherapy versus dual anti-HER2 therapy plus chemotherapy
RegistHER was a real-world clinical trial that tested the efficacy of various treatment strategies, including trastuzumab plus chemotherapy with and without ET (T + CT/T + CT + HT) for HR+/HER2+ ABC. 67 The mPFS was 20.4 versus 9.5 months in the groups with ET and without ET, respectively. Adding ET to trastuzumab + chemotherapy prolonged patients’ mPFS for 10.9 months, which is longer than 6.3 months in the CLEOPATRA trial. The median OS for patients in the T + CT group was 36.7 months, while not reached for patients in the T + CT + HT group, p < 0.001. The administration of ET improved the outcomes of patients with HR+/HER2+ ABC when combined with trastuzumab-based therapy, although further validation is needed.
Possible new options for patients with HR+/HER2+ ABC
Numerous novel therapeutic drugs or treatment strategies are currently being developed with the aim of improved efficacy in patients with HR+/HER2+ tumors. The following summarizes published clinical trials with available data and more details are presented in Table 2. In addition, Table 3 outlines information regarding ongoing clinical trials to better illustrate cutting-edge research directions.
New option for HR+/HER2+ ABC patients.

ABC, advanced breast cancer; CDK4/6, cyclin-dependent kinase 4/6; ER, estrogen receptor; HER2, human epidermal growth factor 2; HR, hormone receptor; mOS, median overall survival; mPFS, median progression-free survival; mTOR, mammalian target of rapamycin; NR, not reached; PD-L1, programmed cell death ligand 1; PI3K, phosphatidylinositol 3-kinase; T-DM1, trastuzumab-derivative of maytansine.
Ongoing clinical trials on treatment for HR+/HER2+ ABC patients.

ABC, advanced breast cancer; AI, aromatase inhibitors; CDK4/6, cyclin-dependent kinase 4/6; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; OFS, overian function suppression; T-DXd, trastuzumab deruxtecan.
DS-8201
DS-8201, a type of ADC, is composed of an anti-HER2 antibody, a cleavable tetrapeptide-based linker, and a cytotoxic topoisomerase I inhibitor. 44 DESTINY-Breast01 evaluated the efficacy of DS-8201 in patients with HER2+ ABC who had previously received T-DM1 treatment. 68 With a median of six previous treatments, the patients achieved a mPFS of 16.4 months, notably longer than the 8.8 months for the heavily pretreated patients with ABC receiving neratinib plus capecitabine and 9.6 months for the patients who previously received trastuzumab plus a taxane and received T-DM1 as a second-line therapy.69,70 The impressive efficacy of DS-8201 in patients with HER2+ cancer has brought it into the limelight. Compared with the standard treatment strategy of anti-HER2 therapy plus chemotherapy, DS-8201 extended the mPFS of patients with HER2+ ABC by almost 11 months in DESTINY-Breast02. 71 In addition, in DESTINY-Breast03, the DS-8201 group achieved a mPFS of 28.8 months, whereas the T-DM1 group achieved only 6.8 months. 72 In the HR+/HER2+ subgroup, the mPFS was 26.2 months for DS-8201-treated patients and 6.9 months for T-DM1-treated patients, demonstrating the potent therapeutic effect of DS-8201 in the HR+/HER2+ cohort. The incidence of grade 3 or higher treatment-related AEs remained similar between the DS-8201 and T-DM1 groups. A greater incidence of drug-related interstitial lung disease or pneumonia was observed in the DS-8201 group. Neither group experienced any AEs of grade 4 or 5, suggesting that DS-8201 is a safe option for patients with ABC. Breast cancer cells pathologically documented as HER2+ or HER2++ with a negative result by fluorescence in situ hybridization (HER2-low tumor) did not show significant responses to traditional anti-HER2 therapy. DESTINY-Breast04 recruited HER2-low patients to compare the efficacy of DS-8201 and chemotherapy in this patient population. Patients treated with DS-8201 achieved a longer mPFS than those treated with chemotherapy (10.1 versus 5.4 months, respectively; p < 0.001), indicating that DS-8201 may be a new option for the treatment of HR+/HER2-low patients. 73 DESTINY-Breast09 is comparing T-Dxd with or without pertuzumab with the standard of care (taxane, trastuzumab, and pertuzumab) as a potential first-line treatment for patients with HER2+ ABC, which may change the standard first-line treatment for HER2+ breast cancer (Table 3).
Cyclin-dependent kinase 4/6 inhibitors
In addition to the interaction between the HR and HER2 pathways, factors that regulate the cell cycle also contribute to resistance to HER2-targeted therapy and ET for HR+/HER2+ breast cancer. 74 In physiological conditions, cyclin-dependent kinases (CDKs) and cyclins interact to ensure the proper progression of the cell cycle. CDKs 4/6 bind with cyclin D, resulting in the formation of the cyclin D–CDK4/6 complex. The complex phosphorylates retinoblastoma protein, which releases the E2F transcription factor and promotes the transition of the cell cycle from phase G1 to S. 75 In ER+ breast cancer cells, overactivation of cyclin D–CDK4/6 often leads to a loss of cell cycle control, resulting in the limitless proliferation of tumor cells (Figure 1). 76 Currently, CDK4/6 inhibitors are being employed as a standard first-line treatment for patients with HR+ ABC.77,78 For HR+/HER2+ breast cancer, a preclinical study demonstrated that overexpression of cyclin D contributed to resistance to HER2-targeted therapy. Cells that survived anti-HER2 therapy showed nuclear overexpression of cyclin D1 and CDK4, suggesting hyperactivity of the cyclin D1–CDK4/6–phosphorylated retinoblastoma protein axis in anti-HER2 therapy-treated cell lines. 79 HER2 ligand interaction activates the PI3K–AKT pathway and cyclin D1, leading to resistance to trastuzumab and other kinds of anti-HER2 therapy. 78 Palbociclib effectively inhibited the growth of ER+/HER2+ and ER−/HER2+ breast cancer cell lines as a single agent in a dose-dependent manner. 80 These preclinical results provided biological evidence for the combination use of a CDK4/6 inhibitor plus HER2-targeted therapy.
A phase Ib clinical trial enrolled patients with advanced/metastatic HER2+ breast cancer who had previously been treated with trastuzumab and a taxane in any setting to analyze the safety and efficacy of ribociclib conjugated with T-DM1. The mPFS for patients with HR+/HER2+ was 10.8 months (95% CI, 1.3–19.3 months). Ribociclib in combination with T-DM1 was well tolerated in patients with HR+/HER2+. 81 Designed as a randomized study of a CDK4/6 inhibitor in combination with ET and anti-HER2 therapy compared with standard chemotherapy in patients pretreated with at least two HER2-targeted treatments, MonarcHER first reported the positive result of an improved mPFS in group A (abemaciclib, trastuzumab, and fulvestrant) compared with group C (chemotherapy and trastuzumab). 82 The adverse effects reported in group A were more pronounced (nausea, vomiting, and diarrhea) than in group C, but the adverse effects were typically transient and controllable. PATINA validated the value of adding palbociclib to trastuzumab and ET for maintenance after induction therapy in the first-line setting in patients with HER2+ ABC with two to four prior lines of anti-HER2 therapy. 83 For the luminal B and luminal A subgroups, the mPFS figures were 10.6 and 8.3 months, respectively. The AEs were all clinically manageable. Adding CDK4/6 inhibitors and ET to anti-HER2 drug-based treatments could be established as a novel chemotherapy-free option with tolerable side effects for HR+/HER2+ ABC after further validation.
PI3K–AKT–mTOR inhibitors
As described above, both the ER and HER2 pathways interact with the PI3K–AKT–mTOR pathway. To explore the possibility of reversing resistance to HER2-targeted therapy, investigators conducted the BOLERO-3 study to examine the effectiveness of the combination of trastuzumab, vinorelbine, and everolimus or placebo in patients with HER2+ ABC. 84 The overall population reported a mPFS of 7.00 months for the everolimus group and 5.78 months for the placebo group (p = 0.0067). However, the efficacy of everolimus for the HR+ cohort was not as pronounced as for the HR− cohort. Serious AEs occurred in 117 (42%) patients in the everolimus group and 55 (20%) patients in the placebo group. For patients with HR+ disease, physicians should consider the potential benefits and risks of everolimus on an individual basis and closely monitor patients for any side effects during everolimus administration. In the first-line setting, BOLERO-1 tested the efficacy of adding everolimus to trastuzumab and paclitaxel for patients with HER2+ ABC. 85 The overall population achieved a mPFS of 14.95 months in the everolimus group and 14.49 months in the placebo group (p = 0.1166). In the subgroup analysis, a mPFS of 20.27 was achieved in the everolimus group versus 13.08 months in the placebo group (p = 0.0049, significance threshold = 0.0044). Based on the results of BOLERO-1, a subgroup analysis for the HR+ population was not formally conducted. The safety profile was generally consistent with the results previously reported in BOLERO-3. 85 Although studies suggested that patients with HR+ breast cancer may not benefit from treatment containing everolimus, given the heterogeneity of the tumors, more biomarkers should be explored and more trials should be designed to identify patients who may benefit from everolimus-containing treatments.
Programmed cell death protein 1/programmed cell death ligand 1 inhibitors
Programmed cell death protein 1 (PD-1)/programmed cell death ligand 1 (PD-L1) inhibitors can obstruct the interaction between PD-1 and PD-L1 molecules, thereby reversing T-cell exhaustion and augmenting the cytotoxic anti-tumor effects of CD8+ T cells. 86 Research on the therapeutic effectiveness of PD-1 and PD-L1 inhibitors has mainly been conducted in patients with triple-negative breast cancer. 87 Nevertheless, attempts are currently being made to explore the efficacy of PD-1 and PD-L1 inhibitors in patients with HR+/HER2+ ABC. The combination of T-DM1 and atezolizumab has the potential to enhance the toxicity of anti-HER2 drugs and simultaneously strengthen anti-tumor immunity. Based on this premise, the KATE2 study explored the efficacy of combining T-DM1 and atezolizumab for the treatment of patients with HER+ ABC who had previously been treated with trastuzumab and a taxane and experienced disease progression. 88 The atezolizumab group had a mPFS of 8.2 months, as compared with 6.3 months in the placebo group (p = 0.33). However, in the subgroup analysis, patients receiving T-DM1 plus atezolizumab had a mPFS of only 6.8 months, which did not exceed the 8.5 months in the placebo group. Although the T-DM1 and atezolizumab combination therapy did not yield positive results in the study, it did uncover a novel treatment regimen for patients with HR+/HER2+ ABC. The efficacy of combining a HER2-targeted therapy with PD-1 or PD-L1 inhibitors requires further validation.
Summary
Together, the trial results indicate that, for patients with HR+/HER2+ ABC, adding ET to a dual anti-HER2 therapy-based treatment regimen may be a favorable option. By reducing the development of resistance to HER2-targeted and endocrine therapies, and delaying the use of chemotherapy, the combination of anti-HER2 and ET not only provides clinical benefit but also improves the quality of life of patients with ABC. The combination of CDK4/6 inhibitors, ET, and HER2-targeted therapy has also achieved remarkable efficacy in patients with HR+/HER2+ ABC, and numerous clinical studies are underway to further confirm its clinical effectiveness. Considering that no positive results have been achieved in studies on adding PI3K–AKT–mTOR inhibitors and PD-1 or PD-L1 inhibitors to the regimens, it will be necessary to explore new combination therapies and further validate treatment options to fully unleash the potential of the drugs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231220501 – Supplemental material for Endocrine therapy plus HER2-targeted therapy, another favorable option for HR+/HER2+ advanced breast cancer patients
Supplemental material, sj-docx-1-tam-10.1177_17588359231220501 for Endocrine therapy plus HER2-targeted therapy, another favorable option for HR+/HER2+ advanced breast cancer patients by Yuehua Liang, Xiaoran Liu, Zehui Yun, Kun Li and Huiping Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
