Abstract
Background:
Dual human epidermal growth factor receptor 2 (HER2) blockade with trastuzumab and pertuzumab (TP) is a standard therapy of metastatic and localized HER2-positive breast cancer (BC), but its activity in breast cancer brain metastases (BCBM) is unknown.
Methods:
Patients with HER2-positive BCBM were identified from the Vienna Brain Metastasis Registry and clinical data including patient characteristics, therapies and overall survival (OS) were obtained. Patients were grouped into ‘TP’, ‘other-HER2-targeted therapy’ and ‘no-HER2-targeted therapy’ according to received first-line systemic therapy after diagnosis of BCBM. Radiological re-assessment of intracranial lesions was performed in patients treated with TP as systemic first-line therapy according to RANO response criteria for brain metastases (BM).
Results:
A total of 252 HER2-positive BC patients with BM were available for this analysis. Patients treated with TP as systemic first-line therapy after diagnosis of BM had a significantly longer OS compared with treatment with other-HER2-targeted therapy and no-HER2-targeted therapy (44 versus 17 versus 3 months, p < 0.001; log-rank test). Among radiologically re-assessed patients treated with TP as systemic first-line therapy after diagnosis of BM, 5/14 patients (35.7%) had complete intracranial remission (CR), 8/14 patients (57.1%) partial intracranial remission (PR), 1/14 patients (7.1%) stable intracranial disease (SD) and 0/14 patients (0.0%) progressive intracranial disease (PD) as best response resulting in an intracranial objective response rate (iORR) of 92.9% and an intracranial clinical benefit rate (iCBR) of 100.0%.
Conclusion:
First-line therapy with dual HER2-inhibition of TP after BM diagnosis was associated with the longest median OS times in patients with BCBM.
Introduction
Survival prognosis of breast cancer (BC) patients has been markedly improved over the last two decades. This was most pronounced in human epidermal growth factor receptor 2 (HER2)-positive disease due to the implementation of different HER2-targeted therapies.1,2 With increasing survival rates due to effective systemic disease control, brain metastases (BM) are being observed in 30% to 55% of metastatic HER2-positive BC patients.3–5 Local treatment still represents the standard of care in symptomatic BM patients; however, systemic therapy approaches have been increasingly incorporated into the multimodal management of BM patients. 6 Small molecules like the HER2-targeted tyrosine kinase inhibitors (TKI) lapatinib, neratinib and tucatinib did achieve meaningful intracranial responses, especially when applied in combination with chemotherapy.7–10 Of note, even large molecules such as the HER2-directed antibodies trastuzumab and pertuzumab accumulate in breast cancer brain metastases (BCBM) as was shown in positron emission tomography-computed tomography (PET-CT) studies using radioactively tagged antibodies.11,12 In line with these data, the antibody-drug-conjugate trastuzumab-emtansine (TDM1) was associated with favourable intracranial response rates when applied in symptomatic as well as asymptomatic BM patients. 13
Dual HER2-inhibition by trastuzumab (targeting extracellular domain 4) and pertuzumab (directed against extracellular domain 2) (TP) is highly efficacious as evidenced by a clinically relevant prolongation of overall survival (OS) when administered in conjunction with chemotherapy as first-line therapy in the metastatic setting. In addition, the combination has been approved as neoadjuvant treatment in HER2-positive early-stage BC and adjuvant therapy in patients at high recurrence risk.14–16 TP prolonged BM-free survival (BMFS) over trastuzumab alone in a post hoc analysis from the pivotal CLEOPATRA study, whereas little is known regarding the intracranial efficacy of dual antibody treatment on established BCBM. 17 Therefore, we investigated the outcome of BCBM patients treated with dual HER2 blockade of TP in comparison with patients treated with other HER2-targeting drugs and patients treated with no HER2-directed therapies.
Material and methods
Patients
Overall, 267 patients treated between 1990 and 2019 for HER2-positive BCBM at the Medical University of Vienna were identified from the Vienna Brain Metastasis Registry. Of these 267 patients, 13 had to be excluded due to incomplete information regarding therapy and 2 due to diagnosis of a second primary tumour. Therefore, 252 patients were available for this retrospective analysis (Supplemental Figure S1). For 61 patients (24.2%) who were diagnosed before the year 2000, immunohistochemical staining for HER2 was not available at initial diagnosis of BC but was performed at some point during their course of disease either from the primary tumour or a metastatic lesion. If leptomeningeal carcinomatosis (LC) was present concomitantly to diagnosis of parenchymal BM, patients were also eligible for inclusion. Information relating to patient demographics, case history and survival were collected by retrospective chart review. This study was conducted in accordance with the Declaration of Helsinki and approval by the institutional review board (IRB) was obtained (1167/2019). According to the IRB, no written consent from the subjects was necessary due to the retrospective study design.
All patients were managed by a dedicated team of BCBM specialists at an academic breast centre; treatment decisions were taken in an interdisciplinary tumour conference. Treatment was performed according to best clinical evidence and according to current standard of care.
Immunohistochemical analysis
Receptor status was obtained by chart review as the vast majority had been diagnosed in-house at the certified Department of Pathology, Medical University of Vienna according to international guidelines. In brief, oestrogen receptor (ER) and progesterone-receptor (PgR) immunohistochemical stainings were performed according to prevailing American Society of Clinical Oncology/United States and Canadian Academy of Pathology (ASCO/USCAP) recommendations [CONFIRM SP1 clone for ER and 1E2 clone for PgR, respectively (Ventana, Tucson, AZ, USA) or clones 1D5 and 636 for ER and PgR, respectively (Dako, Glostrup, Denmark)]; hormone receptor expression was estimated as the percentage of positively stained tumour cells, using the Reiner Score, applying a 10% cutoff for positivity.18–20 HER2-status was assessed by immunohistochemistry [HER2 clone 4B5 (Ventana) or HercepTest® (Dako)] and dual colour fluorescent in situ hybridization [FISH; PathVision® HER2 DNA probe kit (Vysis Inc., Downers Grove, IL, USA); CISH; INFORM HER2 dual ISH (Ventana)]. Tumours were classified as HER2-positive if they had a staining intensity of 3+ on the Herceptest®; tumours with staining intensity of 2+ were tested by FISH for HER2 DNA amplification. 21
Radiological assessment
Intracranial radiological re-assessment of patients treated with TP as systemic first-line therapy after diagnosis of BM was performed by an experienced neuroradiologist. The longest diameter of each target lesion (up to a maximum of five BM per patient) was measured at baseline and subsequently at every available restaging magnetic resonance imaging (MRI). Best responses had been classified (using the sum of the diameter of all target lesions) according to RANO response criteria for BM. 22
Complete response (CR) was defined as disappearance of all measurable lesions. Partial response (PR) was defined as ⩾30% reduction of the greatest diameters of measurable lesions, no increase in lesion size and no new lesions. Stable disease (SD) was defined as ⩽30% decrease and ⩽20% increase without the appearance of new lesions. Progressive disease (PD) was defined as ⩾20% increase in tumour size, appearance of new lesions or an increase of applied corticosteroids. Intracranial clinical benefit rate (iCBR) was defined as percentage of patients with intracranial CR, PR and SD. Intracranial objective response rate (iORR) was defined as percentage of patients with intracranial CR and PR. In 12 patients, just MRI reports, but no images were available. Therefore, this subgroup analysis contains 14/26 patients (53.8%) treated with TP.
Statistical analysis
For comparisons, patients were grouped in three groups based on the received first-line systemic treatment after diagnosis of BM: TP, other-HER2-targeted therapy and no-HER2-targeted therapy. OS was defined as interval from first diagnosis of BM until death or last date of follow up and estimated with the Kaplan–Meier product limit method. To test for differences between two parameters, the chi-square test was used for binary variables and the Kruskal-Wallis test for differences in mean ranks between more than two variables. To test for differences between OS curves, the log-rank test was used. BMFS was defined as the interval from diagnosis of metastatic breast cancer until diagnosis of BM. Two-tailed p values < 0.05 were considered to indicate statistical significance. The association of systemic first-line therapies after diagnosis of BM with OS and the differential effect of different anti-HER2 substances when used as first-line treatment were the main points of interest of the present study.
The diagnosis specific graded prognostic assessment score (DS-GPA) including BC subtype (luminal A, luminal B/Her2-positive, triple-negative, HER2-positive), age (<60, >60 years) and Karnofsky performance status (<50, 60, 70–80, 90–100) is the best established prognosticator of outcome in BCBM. 23 To investigate the independent association of systemic first-line therapy with OS, we predefined to perform four different multivariate analyses: within the first three analyses, the association of DS-GPA, time period of initial BC diagnosis (<2000, 2000–2010, >2010) as well as different first-line local therapies for BM [stereotactic radiosurgery, surgical resection, whole brain radiotherapy (WBRT)] with systemic first-line therapy after diagnosis of BM was tested. Within a fourth multivariate analysis, DS-GPA and type of first-line HER2-targeted therapy after BM diagnosis were entered. Here, HER2-targeted therapy was entered as a categorical variable with no-HER2-targeted therapy as the reference variable. A multivariate analysis was performed using the Cox Regression model. Due to the exploratory and hypothesis-generating design of the present study, no adjustment for multiple testing was applied and no formal sample-size calculation was conducted. 24
All statistics were calculated using statistical package for the social sciences (SPSS®) 26.0 software (SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
A total of 252 patients with HER2-positive BCBM were available for analysis. Median age at initial diagnosis of BC was 49.5 years (range 27–84) and at diagnosis of BCBM 53.5 years (range 27–85). All 252 patients (100.0%) were female; 61/252 patients (24.2%) were initially diagnosed before the year 2000, 138/252 patients (54.8%) between 2000 and 2010 and 53/252 patients (21.0%) after the year 2010. Of the 252 patients, 154 (61.1%) received HER2-targeted therapy after diagnosis of BCBM whereas 98/252 patients (38.9%) did not. Regarding therapy, 26/252 patients (10.3%) received TP, 128/252 patients (50.8%) other-HER2-targeted therapy and 98/252 patients (38.9%) no-HER2-targeted therapy as systemic first-line treatment after diagnosis of BM. Patients characteristics according to these subgroups are listed in Table 1 and details of applied systemic first-line HER2-targeted therapies are listed in Table 2.
Patients characteristics according to systemic first-line therapy after diagnosis of BM.

p values < 0.05 were considered to indicate statistical significance.
BC = breast cancer; BM = brain metastases; ER = oestrogen receptor; HER2 = human epidermal growth factor receptor 2; OS = overall survival; TP = combination of trastuzumab and pertuzumab.
Applied therapies after diagnosis of BM.
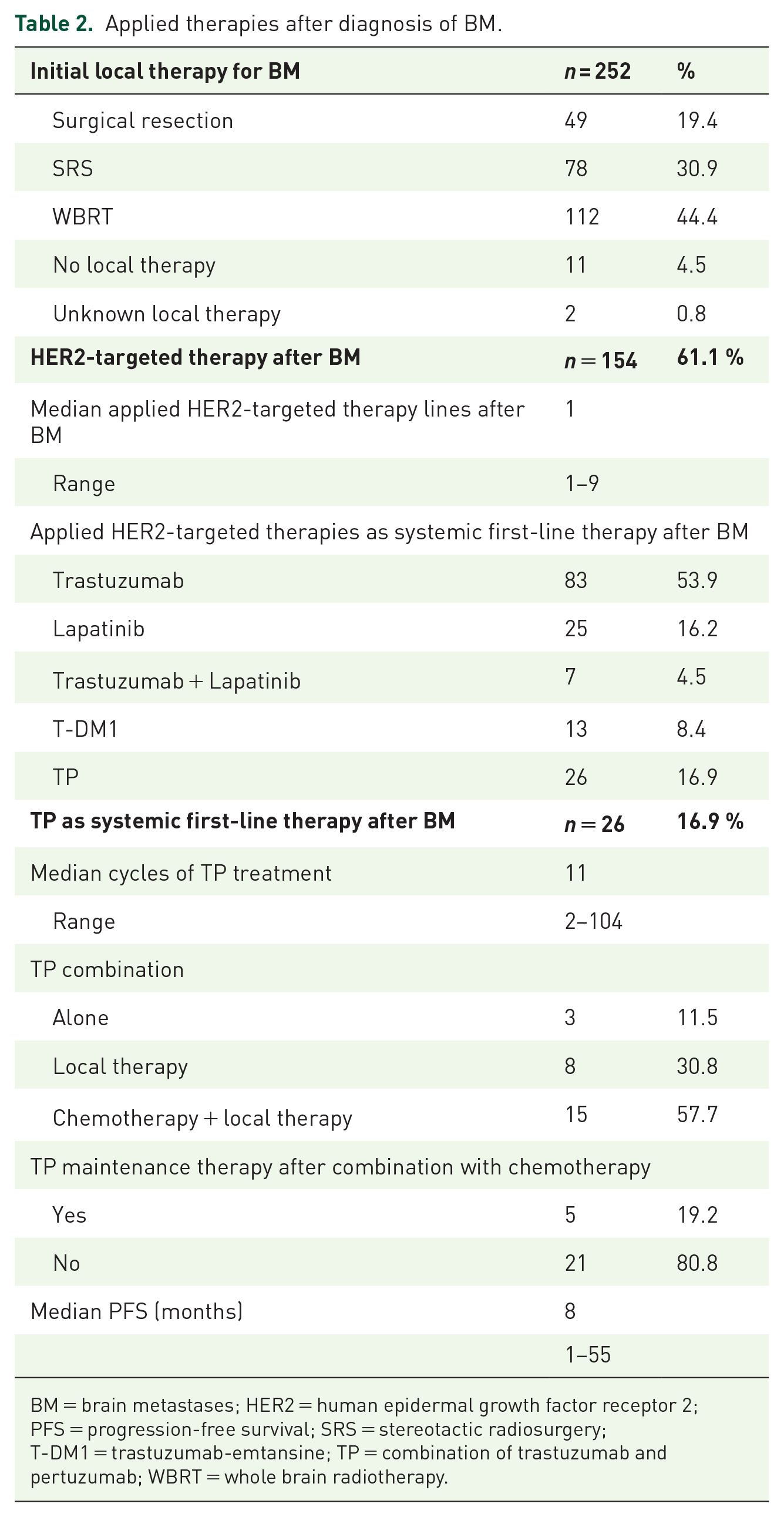
BM = brain metastases; HER2 = human epidermal growth factor receptor 2; PFS = progression-free survival; SRS = stereotactic radiosurgery; T-DM1 = trastuzumab-emtansine; TP = combination of trastuzumab and pertuzumab; WBRT = whole brain radiotherapy.
Dual blockade of TP as systemic first-line therapy after diagnosis of BCBM
Of the total of 252 patients, 26 (10.3%) received TP as systemic first-line therapy after diagnosis of BCBM. Within these patients, median progression-free survival (PFS) was 8 months (range 1–55) and median number of applied cycles was 11 (range 2–104); 3/26 patients (11.5%) received TP as monotherapy, 8/26 patients (30.8%) concomitantly with, or directly after local BM treatment and 15/26 patients (57.7%) in combination with chemotherapy regimens as well as local therapy. In 5/26 patients (19.2%), the initial combination of TP with chemotherapy regimens was consequently deescalated to TP maintenance therapy.
In 14/26 patients (53.8%) treated with TP after diagnosis of BM, MRI images to reclassify response according to RANO BM response criteria were available. The median diameter of all target lesions at the beginning of TP was 19.5 mm (range 10–28 mm) and 7.5 mm (range 0–26 mm) at best response. The median absolute change of diameter was a decrease of 10.5 mm (range −28 to +10 mm) and the median relative change of diameter a decrease of 64.1% (range −100.0 to 4.0%) at best response. Of the 14 patients treated with TP, 5 (35.7%) had CR, 8 (57.1%) PR, 1 (7.1%) SD and 0 (0.0%) PD as best response, resulting in an iORR of 92.9% and an iCBR of 100.0%. Radiological re-assessed intracranial response rates according to TP combinations are summarized within Table 3 and visualized in Figure 1.
Summary of radiologically re-assessed iORR and iCBR according to different applications of dual blockade with TP as systemic first-line treatment after diagnosis of BM.

BM = brain metastases; CHT = chemotherapy; iCBR = intracranial clinical benefit rate; iORR = intracranial objective response rate; LT = local therapy; ptx = patients; TP = combination of trastuzumab and pertuzumab.

Waterfall-plot of radiologically re-assessed intracranial response rates of patients treated with TP as systemic first-line therapy after diagnosis of BM: x-axis: patients according to TP treatment type. y-axis: relative change of target lesion’s diameter from baseline to best response.
OS outcome analyses
Patients diagnosed with BC after the year 2010 had a significantly longer OS compared with patients diagnosed before the year 2000 and patients diagnosed between 2000 and 2010 (22 versus 12 versus 11 months, p = 0.002; log-rank test) (Figure 2a). Patients treated with TP as systemic first-line therapy after diagnosis of BM had a significantly longer OS compared with treatment with other-HER2-targeted therapy and no-HER2-targeted therapy (44 versus 17 versus 3 months, p < 0.001; log-rank test) (Figure 2b).

Kaplan–Meier estimates for OS. (a) Median OS according to time period of BC diagnosis (p = 0.002; log-rank test). (b) Median OS according to treatment with TP, other-HER2-targeted therapy and no-HER2-targeted therapy as systemic first-line treatment after diagnosis of BM.
Time to intracranial progression after diagnosis of BM was only numerically associated with systemic first-line therapy (10 months with TP versus 9 months with other-HER2-targeted therapy versus 7 month with no-HER2-targeted therapy, p > 0.05; log-rank test). Time to extracranial progression after diagnosis of BM was not significantly associated with systemic first-line therapy (4 months with TP versus 5 months with other- HER2-targeted therapy versus 4 months with no- HER2-targeted therapy, p > 0.05; log-rank test).
To evaluate the independent impact of systemic first-line treatment after BM diagnosis, we performed four predefined multivariate analyses. Within the first analysis, including DS-GPA and type of systemic first-line therapy after diagnosis of BCBM, DS-GPA [hazard ratio 1.77 (95% CI: 1.36–2.32; p < 0.001, Cox proportional hazards model)] as well as type of systemic first-line therapy after diagnosis of BCBM [hazard ratio (HR) 2.15; 95% confidence interval (CI): 1.70–2.71; p < 0.001, Cox proportional hazards model] were shown to be independent prognosticators of OS. Within a second analysis including the time period of initial BC diagnosis (<2000, 2000–2010, >2010) as well as type of systemic first-line therapy after diagnosis of BCBM, only the latter remained significant [HR 0.93 (95% CI: 0.75–1.15; not significant) versus HR 2.23 (95% CI: 1.76–2.84; p < 0.001, Cox proportional hazards model)]. Within a third analysis including different first-line local therapies (stereotactic radiosurgery, surgical resection, WBRT, none) as well as type of systemic first-line therapy after diagnosis of BCBM, both factors were shown to be associated independently with OS [HR 1.11 (95% CI: 1.00–1.23; p = 0.043) versus HR 2.29 (95% CI: 1.81–2.89; p < 0.001, Cox proportional hazards model)]. To further investigate the differential impact of different HER2-targeted therapies applied as systemic first-line treatment after diagnosis of BM, both DS-GPA and type of systemic first-line therapies after BM were entered into a fourth multivariate analysis. Within this analysis, DS-GPA (HR 1.94 [95% CI: 1.47–2.57; p < 0.001, Cox proportional hazards model)] and all HER2-targeted therapies except for the combination of trastuzumab and lapatinib remained significantly correlated with OS with TP presenting the lowest HR [TP: HR 0.15 (95% CI: 0.08–0.30; p < 0.001); TDM-1: HR 0.31 (95% CI: 0.15–0.62; p < 0.001); lapatinib: HR 0.39 (95% CI: 0.24–0.64; p < 0.001); trastuzumab: HR 0.61 (95% CI: 0.44–0.83; p = 0.002); trastuzumab and lapatinib: HR 0.53 (95% CI: 0.23–1.21; not significant); Cox proportional hazards model]. Results of this multivariate analysis are visualized within Figure 3.

Forest plot of a multivariate analysis with DS-GPA and different systemic first-line therapies applied after diagnosis of BCBM.
Within a separately analysed subgroup of 21 patients (8.3%) with concomitant LC at diagnosis of solid BM, no significant association of median OS with systemic first-line therapies could be observed (4 months with TP versus 3 months with other-HER2-targeted therapy versus 1 month with no-HER2-targeted therapy, p > 0.05; log-rank test).
Discussion
Systemic therapy has become an important treatment option in patients with BCBM. The HER2-positive and luminal subtypes have a favourable OS prognosis in comparison with triple negative BC even upon the diagnosis of BM with a median survival of up to 24 months. 23 In consequence, treatments such as WBRT with the ensuing risk for neurocognitive decline have to be considered with caution.25,26 This has led to a surge of interest in the intracerebral effect of systemic drugs. As BC patients with BM were frequently excluded from registration trials, only limited knowledge regarding a direct effect of systemic treatment on BM is available. Despite all limitations, retrospective outcome analyses in BCBM are therefore of interest.
In our patient cohort, the application of HER2-targeted therapy as systemic first-line therapy after diagnosis of BM was significantly associated with prolonged OS compared with no HER2-targeted treatment. Of note, the effect was independent of the DS-GPA and time period of diagnosis. Importantly, 40% of the included patients did not receive HER2-targeted treatment as they were diagnosed mainly before continued administration of HER2-directed therapy after local treatment for BM was applied routinely. Moreover, over the last decade the treatment pattern, especially in BM treatment, changed and an increasing fraction of patients is treated routinely with systemic therapy. 27 However, the survival benefit of systemic first-line therapy in our study appeared to be most pronounced with TP, where an OS of 44 months was observed, which supports the ongoing application of systemic therapies in BCBM patients. Previous case series have already suggested a survival benefit with continuation of systemic treatment after BM diagnosis. OS in these studies, however, was usually not correlated with DS-GPA. In addition, it is still unresolved if the main OS effect of systemic treatment is due rather to systemic disease control as opposed to a direct effect of BM. In fact, approximately half of BC patients with BM die with progressing BM while the extracranial disease is stable. 28 To overcome these issues, the DS-GPA was included into a multivariate analysis to correct for the best-established prognostic factors. In addition, we evaluated the response rates with TP applied as systemic first-line therapy in patients with and without local treatment by radiological re-assessment of central nervous system (CNS) response rates from brain MRI scans wherever available. Therefore, survival data in our study are believed to be linked to a direct effect of systemic treatment of BCBM.
A direct effect of TKIs in BM is meanwhile well established: in a prospective single-arm phase II study, Lin et al. 7 reported an intracranial response rate of 6% with single-agent lapatinib in progressive BCBM; an additional 21% of patients had some minor degree of disease shrinkage. In an extension cohort evaluating the combination of lapatinib and capecitabine, a CNS response rate of 20% was observed. With a 66% response rate observed in the LANDSCAPE trial, this combination was later established as a potential standard as upfront therapy of asymptomatic to oligosymptomatic patients with multiple HER2-positive BCBM. 8 More recently, neratinib, a second-generation TKI of epithelial growth factor receptor (EGFR) and HER2, in combination with capecitabine yielded an intracranial response rate of 33% in patients with and 49% in patients without prior lapatinib treatment. 9 In line with the assumption that TKIs are most likely to reach clinically relevant concentrations within BM tissue due to their molecular size and permeability characteristics, addition of the highly selective HER2 TKI tucatinib to trastuzumab and capecitabine resulted in an improved progression-free survival (PFS) even in the subgroup of BC patients with active BM in a recently published phase II trial. 29 However, also TDM-1 – although significantly larger in size and therefore earlier considered of not being able to penetrate into BM tissue – did achieve meaningful intracranial responses. 13 In contrast, no prospective trial has evaluated the activity of TP in BCBM patients so far. However, within the PHEREXA phase III trial of BC patients receiving pertuzumab in addition to trastuzumab and capecitabine during or after progression upon trastuzumab, a non-significant OS benefit was observed, which appeared to be more pronounced in the subgroup of patients with stable BM at baseline. 30 Therefore, we decided to focus on a potential direct effect of TP in BCBM. In our study, systemic first-line treatment with TP after BM diagnosis yielded the best OS results as compared with other types of HER2-directed therapy or no further HER2-directed treatment (44 versus 17 versus 3 months). Addressing the question of a direct effect of TP on BM, iORR and iCBR among radiologically re-assessed patients were 92.9% and 100.0%, respectively.
While clinically interesting, our results need to be interpreted with caution due to the retrospective design of our study. Unfortunately, no information with regard to quality of life of our patients under TP treatment was available, which should be addressed within prospective studies. In addition, trastuzumab and pertuzumab are today commonly used in the neoadjuvant, adjuvant and/or first-line metastatic setting, limiting the use of dual antibody treatment in later lines. However, patients receiving TP as systemic first-line therapy in our study were initially significantly more often diagnosed with stage IV disease, pre-treated with Her2-targeted therapies and generally in worse performance status and still benefited the most.
In summary, this is the first analysis investigating differences in outcome between the currently available, modern HER2-targeted therapies in BCBM patients. We were able to show that prognosis of these patients improved over the last decades due to new treatment approaches. We observed a significant difference in terms of outcome between different HER2-targeted therapies applied as systemic first-line treatment with the longest OS associated with TP. In addition, TP yielded clinically relevant intracranial response rates. Prospective clinical trials on HER2-targeted therapies in BCBM patients are warranted to validate the present results.
Supplemental Material
sj-tiff-1-tam-10.1177_17588359211009002 – Supplemental material for Favourable outcome of patients with breast cancer brain metastases treated with dual HER2 blockade of trastuzumab and pertuzumab
Supplemental material, sj-tiff-1-tam-10.1177_17588359211009002 for Favourable outcome of patients with breast cancer brain metastases treated with dual HER2 blockade of trastuzumab and pertuzumab by Elisabeth Sophie Bergen, Amelie Binter, Angelika Martina Starzer, Gerwin Heller, Barbara Kiesel, Kristina Tendl-Schulz, Zsuzsanna Bago-Horvath, Julia Furtner, Johannes Leitner, Ruth Exner, Florian Fitzal, Karin Dieckmann, Georg Widhalm, Matthias Preusser, Anna Sophie Berghoff and Rupert Bartsch in Therapeutic Advances in Medical Oncology
Footnotes
Conflict of interest statement
AMS has received travel support from PharmaMar. KT-S has received honoraria for lectures or advisory board participation from Roche as well as travel support from Roche, Novartis and Merck Sharp & Dohme GmbH (MSD). ZB-H has received personal fees and honoraria for participation on advisory boards from Novartis, Biomedica and Roche and travel support from Roche. FF has received travel and scientific support from Comesa, Novartis, Roche, Astra Zeneca, Pfizer, Myriad, Nanostring, Bondimed (Polytech, Integra) and Lilly, honoraria for advisory board participation from Pfizer, Astra Zeneca, Lilly and Roche, serves as editor for Springer and as founder of the Breast Analyzing Tool. GW received restricted travel grants from NX Development Corp. MP has received honoraria for lectures, consultation or advisory board participation from the following for-profit companies: Bayer, Bristol-Myers Squibb, Novartis, Gerson Lehrman Group (GLG), CMC Contrast, GlaxoSmithKline, Mundipharma, Roche, Astra Zeneca, AbbVie, Lilly, Medahead, Daiichi Sankyo, Sanofi, Merck Sharp & Dome. The following for-profit companies have supported clinical trials and contracted research conducted by MP with payments made to his institution: Böhringer-Ingelheim, Bristol-Myers Squibb, Roche, Daiichi Sankyo, Merck Sharp & Dome, Novocure, GlaxoSmithKline, AbbVie. ASB has received research support from Daiichi Sankyo (⩽10000€), Roche (>10000€) and honoraria for lectures, consultation or advisory board participation from Roche Bristol-Meyers Squibb, Merck, Daiichi Sankyo (all <5000€) as well as travel support from Roche, Amgen and AbbVie. RB has received research support from Daiichi, Novartis, Roche and honoraria for lectures from Accord, Astra-Zeneca, BMS, Celgene, Eli-Lilly, Novartis, Pfizer, Pierre-Fabre, Roche, Sandoz as well as for advisory board participation from Astra-Zeneca, Celgene, Daichi, Eisai, Eli-Lilly, MSD, Novartis, Pfizer, Pierre-Fabre, Puma, Roche, Samsung. The other authors declare that they have no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research budget of the Medical University of Vienna.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
