Abstract
Background:
Although anti-HER2 therapies have been widely used against gastric carcinoma, the prognostic significance of HER2 overexpression remains unclear. Previous studies failed to provide convincible evidence due to inconsistent HER2 evaluation criteria and heterogeneous clinical characteristics.
Objectives:
To figure out the prognostic significance of HER2 expression in gastric cancer, we rigorously designed and conducted this study.
Design:
Meta-analysis.
Data sources and methods:
Record retrieval was performed by searching PubMed, Web of Science, Cochrane Library, Embase, ASCO, and ESMO meeting libraries from inception to November 2022. Cohort studies investigating overall survival comparison between HER2-positive and HER2-negative gastric cancer patients were included. Both resectable and advanced cases were separately collected while HER2 evaluation standards should be consistent across eligible studies. Newcastle–Ottawa Scale was used for quality assessment. Overall survival was the only endpoint and effect size was presented by hazard ratio (HR) with its 95% confidence interval. The pooled calculation was conducted on Review Manager 5.4.
Results:
Thirty studies were eligible, including 9945 patients. Eligible studies were mostly high quality (n = 31). Regarding resectable cases (n = 22), HER2-positive groups had significantly worse prognosis than HER2-negative counterparts (HR 1.56, 95%CI 1.32–1.85, p < 0.00001). For HER2-positive patients with advanced gastric cancer (n = 10), HER2 overexpression was also an unfavorable survival indicator (HR 1.70, 95%CI 1.23–2.35, p = 0.001). Potential heterogeneous studies had been eliminated while outcomes remained stable by sensitivity analysis. Subgroup analysis suggested HER2-positive patients had a poorer prognosis in both East Asian (resectable: HR 1.56; advanced: HR 1.32) and non-East Asian countries (HR 1.58; HR 3.27).
Conclusion:
As a novel survival biomarker in gastric cancer, HER2 overexpression indicates unfavorable prognosis among both resectable and advanced patients, irrespective of East Asian or non-East Asian populations.
Trial registration:
PROSPERO (CRD42020168051).
Introduction
Gastric cancer is currently the fifth most frequent and fourth most lethal malignancy globally. It is estimated that over 1 million gastric cancer cases occurred in 2020, with nearly 770,000 deaths at the same time.1,2 Although therapeutic signs of progress have been achieved in recent years, survival biomarkers for gastric cancer patients remain in scarcity.3,4
Currently, the anti-HER2 monoclonal antibody trastuzumab has been recommended as the first-line option against advanced gastric cancer together with platinum-based chemotherapy.5,6 Nevertheless, unlike breast cancer, the prognostic significance of HER2 overexpression in gastric cancer remains controversial. Several cohort studies hinted that the positivity of HER2 was directly linked to poorer survival,7,8 while others reported that HER2 overexpression was not an independent prognostic indicator among gastric cancer patients.9–11 Meanwhile, inconsistent conclusions could also be observed among previously published meta-analyses in this field (Supplemental Materials). By exploring the methodological designs of previous primary studies and meta-analyses in detail, they evidently had heterogeneous standards regarding HER2 evaluation and incomparable clinical features, which might explain those less harmonious conclusions concerning prognostic values of HER2 expression.
HER2 is one of the most important and commonly examined biomarkers among gastric cancer patients, hence the importance of clarifying its prognostic value. We performed a systematic review and meta-analysis to clarify the survival significance of HER2 overexpression in gastric cancer, with an emphasis on methodology to limit the pooling of data across homogeneous populations.
Methods
Guidelines and registration
Design, calculation, and drafting of our systematic review and meta-analysis were in accordance with standards in PRISMA Checklist 12 (Supplemental Materials) and Cochrane Handbook. Each step was conducted by two individuals in our group (J.C. and M.C.). Any discrepancy was settled by the third investigator (G.W. and K.T.). We registered our systematic review and meta-analysis in PROSPERO (CRD42020168051).
Search strategy
PubMed, Web of Science, Cochrane Library as well as Embase were carefully searched. Meanwhile, ASCO and ESMO Meeting Library were also examined, together with the reference lists of previously published meta-analyses. Our search procedures began on September 3rd until November 12th of 2022, covering records published from January 1966 to November 2022. The title and abstract of each retrieved record were checked first, followed by full-text assessment if necessary. The entire search strategy was listed in Supplemental Materials.
Selection criteria
Studies that met all the following requirements were eligible for inclusion (PICOS framework):
Participant: Patients from eligible studies should be diagnosed with previously untreated resectable or advanced (unresectable, recurrent, or metastatic) gastric cancer (including gastroesophageal junction cancer) from a generalized community without specific selection of pathological, histological, or clinical features. Studies involving patients with synchronous malignancies other than gastric cancer were not permitted. Overall, this was a traditional meta-analysis based on study-level data extraction; therefore, only studies with patients fulfilling the abovementioned criteria were considered.
Intervention: For resectable gastric cancer, all surgeries should be of curative intent. Both perioperative and adjuvant treatments were permitted. For advanced gastric cancer, previous gastrectomy was permitted, irrespective of curative or palliative operations. Either chemotherapy alone or chemotherapy with targeted treatments was qualified.
Comparator: The HER2-positive group was regarded as the experimental group while the HER2-negative counterpart acted as a control. Standards for examining and scoring HER2 expression were strictly in accordance with NCCN guidelines without any exception. 6 Briefly speaking, after immunohistochemical (IHC) grading of surgical or biopsy specimens into 0, 1+, 2+, or 3+, IHC2+ was further examined by in situ hybridization (ISH) methods. Only those with IHC3+ or IHC2+/ISH+ were regarded as HER2 positive and therefore we only included studies with this definition in their methods. Studies using evaluation standards from ToGA trial 5 were therefore also ineligible. In both HER2 positive and negative groups, no comparisons between other targeted markers were allowed.
Outcome: Overall survival data (hazard ratio (HR) or Kaplan–Meier curves) were mandatory. Overall survival data for combined cases from both resectable and advanced gastric cancer without subgroup analysis were not allowed. In addition, to perform a more specific analysis, overall survival data should be separately provided if advanced-stage HER2-positive patients had mixed treatment regimens containing either chemotherapy plus targeted treatments (except for those targeting HER2) or chemotherapy alone. For those studies reported that a mixture of advanced and resectable patients, we only included them if they reported separate survival outcomes.
Study design: Cohort studies reported from January 1966 to November 2022 without language limitations.
Studies were excluded due to the following criteria:
For resectable gastric cancer, the overall follow-up time was less than 3 years.
Quality assessment
The Newcastle–Ottawa Scale was applied to assess the methodological quality of eligible studies. Details of assessment standards of the Newcastle–Ottawa Scale are listed in Supplemental Materials (eTable 1). The full score of each study was 9, while studies with scores equal to or more than 6 were regarded as high-quality studies.
Data extraction and endpoints
Electronic sheets were used to collect original data from the included studies. Baseline clinical features and overall survival data were extracted from the main text or Supplemental Materials. HR results of overall survival from multivariate (in priority) and univariate analysis were both extracted. Meanwhile, Kaplan–Meier curves were applied for survival data extraction if necessary, in accordance with suggestions by Tierney et al. 13 Moreover, for results that were estimated from Kaplan–Meier curves, we also used the p value provided by original studies to test and adjust the outcomes. Overall survival was the primary and only endpoint in our meta-analysis since it was the main survival indicator for gastric cancer patients.
Statistical analysis
Reviewer Manager 5.4 was the statistical platform for our pooled analysis. HR and its 95% confidence interval were used as effect size for pooled analysis of overall survival data. A significant difference was achieved when the pooled confidence interval for HR did not include 1.0, which could also be demonstrated by p < 0.05. According to the Cochrane Handbook, I2 was utilized as an indicator of heterogeneity. A fixed-effects model was suitable for calculations with low heterogeneity (I2 < 50%) while a random-effects model was more reliable for pooled analysis with high heterogeneity. Using STATA 14.0, publication bias was analyzed for pooled analysis with at least 10 studies inside. A symmetrical funnel plot indicated a low risk of publication bias. Several methods were used for performing sensitivity analyses in our meta-analysis, including interchanging between random-effects and fixed-effects models, eliminating low-quality studies (those scored below 6 by the Newcastle–Ottawa Scale) and extra small sample-size studies (<100), since those studies might have less statistical power. Studies based on East Asian and non-East Asian populations were further analyzed by subgroup analysis.
Results
Baseline characteristics
A total of 8408 records were retrieved from electronic databases, among which 32 studies were eligible for our systematic review and meta-analysis with a total population of 9945 patients (Figure 1). Details of search strategies are listed in Supplemental Materials while reasons for each ineligible study by full-text assessments are displayed in eTable 2 (Supplemental Materials). Twenty-two studies were included for resectable gastric cancer analysis, containing an overall 8125 patients (ranging from 75 to 1148 by each study). Most of the included studies were conducted by East Asian countries (n = 18). All studies had a median age of over 60.0 and a male-dominant sex ratio. The majority of included studies shared comparable composition of tumor locations, Lauren classifications, and TNM stages without specific selection. All studies had at least 3 years of follow-up for resectable cases, and most of them reported a 3-year overall survival rate in the HER2-negative group to be at least 60.0% (Table 1). Ten studies were eligible for advanced gastric cancer analysis, with a total population of 1820 patients (ranging from 32 to 321 in each study). The majority of studies originated from East Asian countries (n = 7). The median age of included studies ranged from 53.0 to 67.0, all with a male-dominant sex ratio. Most of the included patients had well performance status (ECOG 0–2) and metastatic lesions. All studies were followed up for at least 12.0 months while the median survival time of the HER2-negative group in most studies had also surpassed 12.0 months (Table 2). None of the included studies reported mixed stages of patients.

Selection flow chart.
Baseline features of included studies (resectable).
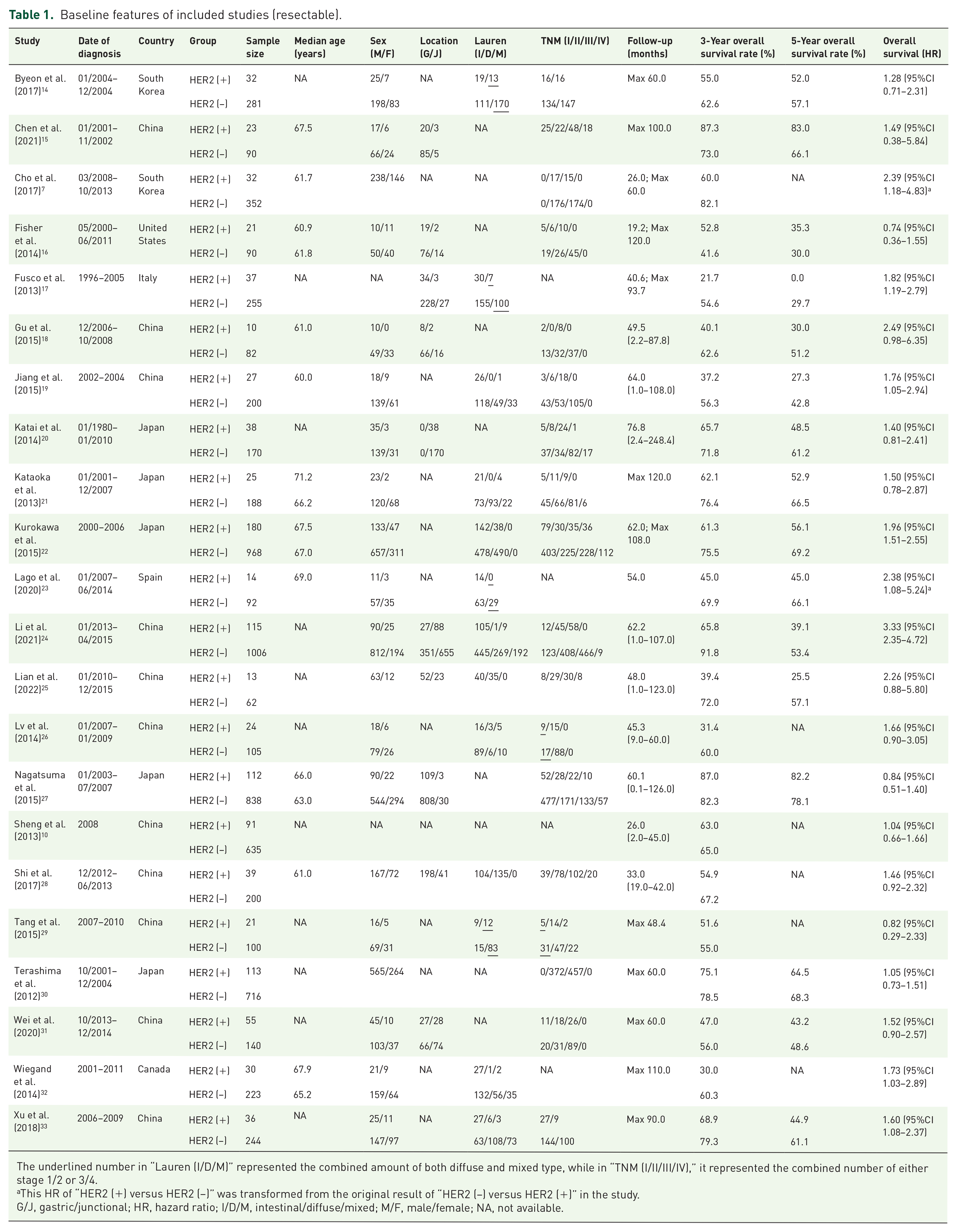
The underlined number in “Lauren (I/D/M)” represented the combined amount of both diffuse and mixed type, while in “TNM (I/II/III/IV),” it represented the combined number of either stage 1/2 or 3/4.
This HR of “HER2 (+) versus HER2 (−)” was transformed from the original result of “HER2 (−) versus HER2 (+)” in the study.
G/J, gastric/junctional; HR, hazard ratio; I/D/M, intestinal/diffuse/mixed; M/F, male/female; NA, not available.
Baseline features of included studies (advanced).
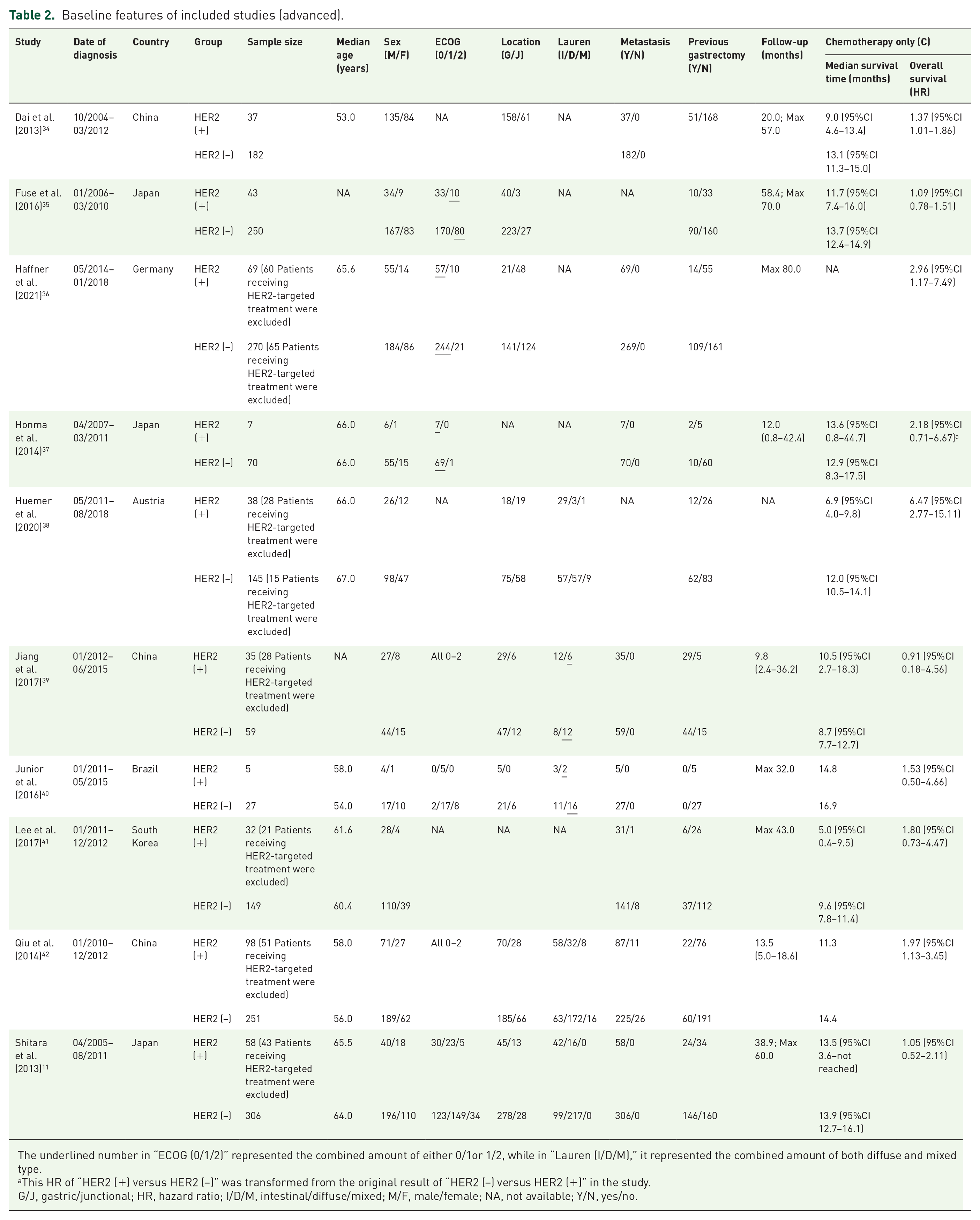
The underlined number in “ECOG (0/1/2)” represented the combined amount of either 0/1or 1/2, while in “Lauren (I/D/M),” it represented the combined amount of both diffuse and mixed type.
This HR of “HER2 (+) versus HER2 (−)” was transformed from the original result of “HER2 (−) versus HER2 (+)” in the study.
G/J, gastric/junctional; HR, hazard ratio; I/D/M, intestinal/diffuse/mixed; M/F, male/female; NA, not available; Y/N, yes/no.
Quality assessment
Concerning resectable cases, all, except one, studies were high quality (at least scored 6) based on the Newcastle–Ottawa Scale, including 11 studies scored 7, 10 studies scored 6, and 1 study scored 5. By further analyzing the results in each category, most studies had full marks in terms of “Outcome,” while some of them obtained relatively low scores concerning “Comparability.” Moreover, since all eligible studies were retrospectively analyzed, none of them reached full scores regarding “Selection” (Figure 2 and eTable 3 in Supplemental Materials).

Quality assessment of eligible studies by the Newcastle–Ottawa Scale.
All studies with advanced-stage patients were high quality based on the Newcastle-Ottawa Scale, including two studies scored 8, five studies scored 7, and three studies scored 6. Via further analyzing the scores in each category, the majority of studies had full marks regarding “Outcome,” while some of them received relatively low scores concerning “Comparability.” Only two prospective studies received full scores concerning “Selection” (Figure 2 and eTable 4 in Supplemental Materials).
Overall survival (resectable)
By pooling 22 studies together, HER2-positive groups had a significantly worse prognosis than HER2-negative counterparts (HR 1.56, 95%CI 1.32–1.85, p < 0.00001). The overall heterogeneity index was I2 = 53% (Figure 3). The risk of publication bias among all included studies was relatively low based on the symmetry of the funnel plot (eFigure 1 in Supplemental Materials).
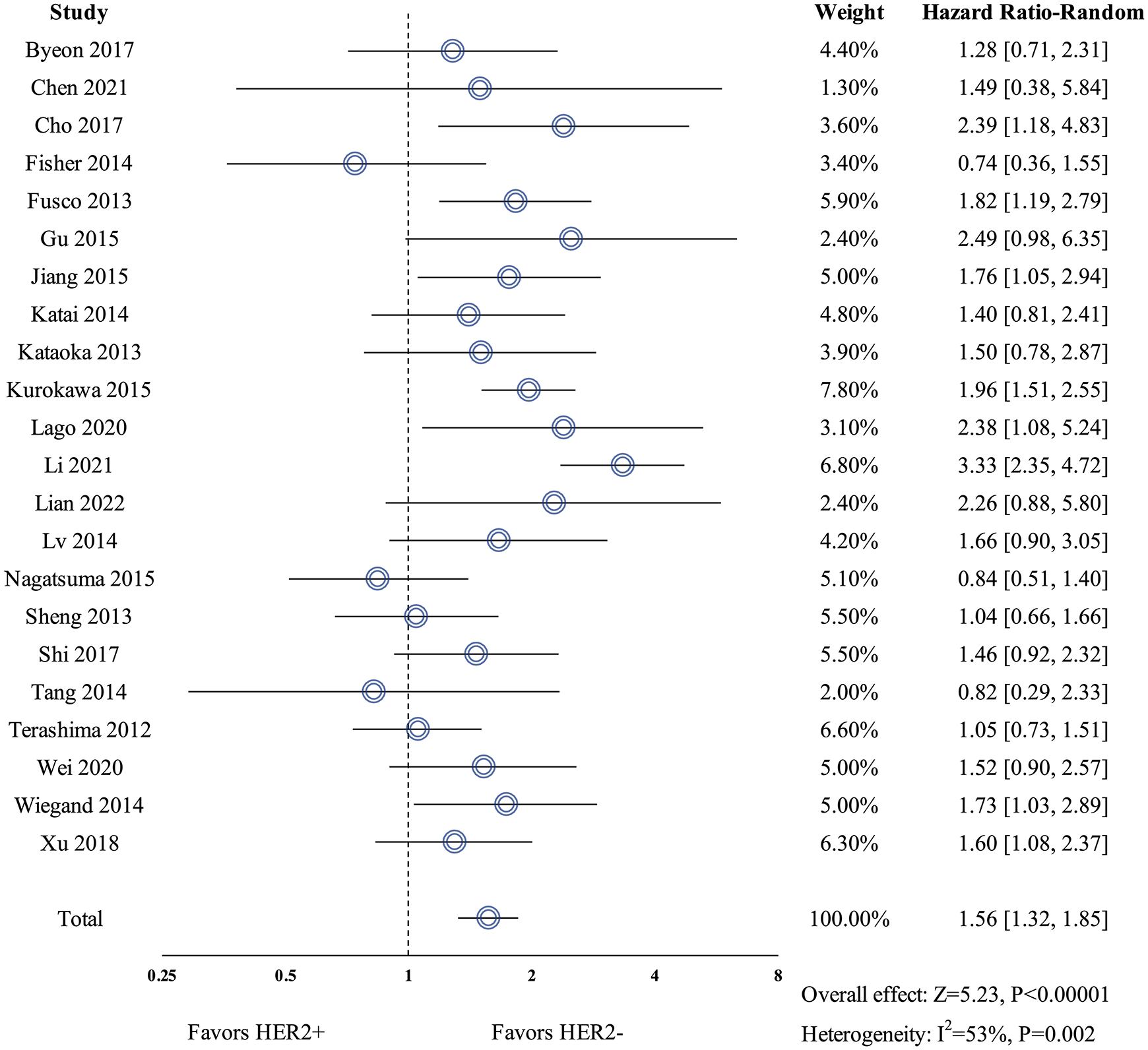
Forest plot of pooled analysis on resectable gastric cancer patients.
Since the heterogeneity level could not be neglected, we conducted comprehensive sensitivity analyses to test the stability of outcomes. Results from all four methods of sensitivity analyses confirmed that the overall pooled outcome was stable (eTable 5 in Supplemental Materials). Moreover, the leave-one-out method confirmed that Li et al. 24 might be the biggest source of heterogeneity since eliminating Li et al. 24 could lower the overall heterogeneity level to I2 = 25% (HR 1.48, 95%CI 1.29–1.70, p < 0.00001) (eTable 5 in Supplemental Materials).
In subgroup analysis based on geographical difference, patients with HER2 positivity also had significantly worse survival than those with HER2-negative expression, irrespective of studies from East Asian (n = 18, HR 1.56, 95%CI 1.29–1.89, p < 0.00001) or non-East Asian countries (n = 4, HR 1.58, 95%CI 1.06–2.36, p = 0.03).
Overall survival (advanced)
For HER2-positive patients receiving chemotherapy only (n = 10), HER2 overexpression was also an unfavorable prognostic indicator (HR 1.70, 95%CI 1.23–2.35, p = 0.001). The overall heterogeneity level was I2 = 56% (Figure 4). The risk of publication bias among eligible studies was not high due to the symmetry of the funnel plot (eFigure 2 in Supplemental Materials).

Forest plot of pooled analysis on advanced gastric cancer patients and sensitivity analysis.
Since the heterogeneity level could not be ignored, we performed comprehensive sensitivity analyses to examine the stability of outcomes. Results from all three methods of sensitivity analyses verified the stability of the overall pooled outcome (eTable 6 in Supplemental Materials). Among all eligible studies, Huemer et al. 38 might be the primary source of heterogeneity by the leave-one-out method since excluding it could help reduce heterogeneity to I2 = 1% (HR 1.37, 95%CI 1.14–1.65, p = 0.0008) (eTable 6 in Supplemental Materials).
Subgroup analysis based on geographical difference suggested that HER2-positive patients had significantly poorer prognosis than HER2-negative counterparts, irrespective of studies from East Asian (n = 7, HR 1.32, 95%CI 1.09–1.60, p = 0.004) or non-East Asian countries (n = 3, HR 3.27, 95%CI 1.46–7.34, p = 0.004).
Discussion
Although HER2 is a vital therapeutic target and routinely detected among gastric cancer patients, its prognostic value remains controversial. Previously published meta-analyses reported inconsistent conclusions on this topic. To be specific, six meta-analyses concluded that HER2 positivity was associated with a poorer prognosis, while two found it irrelevant to survival expectancy (eTable 7 in Supplemental Materials). However, none of these meta-analyses were based on consistent HER2 evaluation standards across included studies, let alone in accordance with the HER2 evaluation system recommended by NCCN guidelines. 6 Moreover, baseline comparability and methodological quality were poorly controlled among most of these meta-analyses, which resulted in high heterogeneity and lowered the credibility of them to become clinically available (eTable 7 in Supplemental Materials).
Learning lessons from previous meta-analyses, we conducted this systematic review and meta-analysis by strictly maintaining homogeneity across included studies and subsequent calculations. All studies must strictly conform with HER2 evaluation standards recommended by NCCN guidelines 6 without any exception. Meanwhile, since resectable and advanced cases were quite different in terms of clinical features and therapeutic reactions, they were separately investigated in our pooled analysis to lower heterogeneity and highlight clinical specificity. Regarding resectable gastric cancer, we confirmed that HER2 positivity was significantly associated with a worse prognosis compared to HER2-negative expression. We then conducted comprehensive sensitivity analyses to test the stability of outcomes as well as find out potential sources of heterogeneity. Regardless of the calculation model, methodological quality, or sample size, pooled results remained stable. Therefore, all our results consistently proved that HER2 was a novel survival biomarker for resectable gastric cancer under current HER2 evaluation standards. From the perspective of each included study, most of them reported either statistical significance or an insignificant tendency of unfavorable survival by HER2 overexpression. And that is why a meta-analysis could lead to clear and significant results here by elevating statistical power. Only three studies reported favorable survival tendency (HR < 1) of HER2 overexpression without statistical significance.14–16 However, they were either with very small sample sizes or unbalanced levels of TNM stages, which were therefore removed by sensitivity analyses without affecting outcome stability. This could also hint that heterogeneous results among other meta-analyses or original studies might be indeed due to their inconsistent standards of HER2 status and incomparable clinical features.
Regarding advanced gastric cancer, since anti-HER2 trastuzumab had been recommended for HER2-positive patients, 6 we only compared survival data of HER2-positive and HER2-negative groups treated by chemotherapy only or chemotherapy plus targeted treatments (except for those targeting HER2), to eliminate impacts from therapeutic interventions. As a result, HER2 overexpression was also a negative indicator of survival among advanced-stage patients. Leave-one-out method of sensitivity analyses indicated that Huemer et al. 38 might be the major source of heterogeneity since the elimination of which significantly lowered the heterogeneity level to I2 = 1%. Moreover, after excluding another two non-East Asian countries-based studies (Haffner et al. 36 and Junior et al. 40 ), the heterogeneity level further reduced to I2 = 0% while the pooled outcome remained stable. This hinted that geographical disparity might have a significant impact on heterogeneity level. Furthermore, like resectable cases, both East Asian and non-East Asian countries subgroups reported similar outcomes that HER2 overexpression was linked to worse survival among advanced-stage patients. This might implicate the potential of global accessibility of our conclusions. All these pooled results seemed consistent with findings of cellular mechanisms, where HER2 overexpression led to activation of multiple downstream proliferative pathways, such as MAPK and PI3K/Akt signaling, 43 making it easier to explain its unfavorable prognostic impacts.
Although our systematic review and meta-analysis was rigorously designed and performed, some limitations were still inevitable. First, the number of included studies and overall population could be more, especially for advanced cases, which could help us to perform more subgroup analyses and sensitivity analyses so that pooled results could be more clinically meaningful and specific. Second, due to lacking original data from the included studies, we could only provide an overall survival analysis. We hoped that more studies could be provided in the future concerning more survival endpoints such as disease-free survival or progression-free survival.
Conclusion
Taken together, based on rigorous approaches and analyses, our study made the first credible pooled evidence suggesting that as a novel survival biomarker in gastric cancer, overexpression of HER2 indicates unfavorable survival outcomes among both resectable and advanced patients, irrespective of East Asian or non-East Asian population. We hypothesized that anti-HER2 therapy may also be a promising option among resectable cases in the future with improved global access.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241271913 – Supplemental material for HER2 becomes a novel survival biomarker for gastric cancer patients: a pooled analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241271913 for HER2 becomes a novel survival biomarker for gastric cancer patients: a pooled analysis by Ji Cheng, Ming Cai, Guobin Wang and Kaixiong Tao in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359241271913 – Supplemental material for HER2 becomes a novel survival biomarker for gastric cancer patients: a pooled analysis
Supplemental material, sj-pdf-2-tam-10.1177_17588359241271913 for HER2 becomes a novel survival biomarker for gastric cancer patients: a pooled analysis by Ji Cheng, Ming Cai, Guobin Wang and Kaixiong Tao in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
