Abstract
Pulmonary sarcomatoid carcinoma (PSC), a rare subtype of non-small cell lung cancer, is highly malignant and has a poor prognosis. Treatments for PSC are presently limited. Traditional treatments provide fewer benefits to PSC patients and are associated with early recurrence and metastasis. Surgical intervention is the preferred option for early-stage PSC patients, whereas chemotherapy, radiotherapy, immunotherapy, and other targeted therapies are recommended for advanced PSC patients. Targeted therapy is only effective in a small number of PSC patients. The initial efficacy of immune checkpoint inhibitors has been acceptable in patients with advanced PSC; therefore, much attention on related biomarkers has been sought. This article aimed to review the research progress of PSC immunotherapy and related diagnostic and prognostic biomarkers in recent years.
Plain language summary
Pulmonary sarcomatoid carcinoma (PSC), a rare subtype of non-small cell lung cancer (NSCLC), is highly malignant and has a poor prognosis. Traditional treatments provide fewer benefits to PSC patients and are associated with early recurrence and metastasis. Targeted therapy is only effective in a small number of PSC patients. The initial efficacy of immune checkpoint inhibitors (ICIs) has been acceptable in patients with advanced PSC, therefore, much attention on related biomarkers has been sought. This article aimed to review the research progress of PSC immunotherapy and related diagnostic and prognostic biomarkers in recent years.
Keywords
Introduction
Pulmonary sarcomatoid carcinoma (PSC), a rare histological subtype of non-small cell lung cancer (NSCLC), is characterized by its extreme aggressiveness and high mortality, with a 5-year survival rate of advanced PSC of less than 5%. 1 PSC accounts for 0.1–0.5% of all primary lung cancers.2,3 In the 2021 World Health Organization (WHO) Classification of Lung Tumors, 4 PSC is primarily classified into pleomorphic carcinoma, carcinosarcoma, and pulmonary blastoma, and pleomorphic carcinoma is further subcategorized into pleomorphic carcinoma, giant cell carcinoma, and spindle cell carcinoma. Presently, the NSCLC treatment model is used for the management of PSC. Surgery is considered the optimal treatment option for resectable PSC, 5 but approximately 70% of patients are diagnosed with locally advanced or metastatic disease at the time of initial diagnosis where curative surgery is not an available option. 6 Currently, the standard treatment strategy for advanced PSC is still under debate, with poor efficacy of conventional chemotherapy and radiotherapy. Therefore, we should focus on targeted and immunotherapies.7–9 Nevertheless, only a minority, around 30%, of PSC patients harboring driver gene mutations have access to targeted therapies. 10 The clinical management of PSC remains challenging, particularly for patients with advanced disease who are in critical need of innovative and efficacious treatment to prolong their survival. Immune checkpoint inhibitors (ICIs) have emerged as the standard of care for managing various solid tumors, including NSCLC, with a substantial improvement in overall survival (OS) in patients with advanced solid tumors. 11 But the effectiveness and safety of ICIs in treating PSC remain uncertain, with only a subset of PSC patients eliciting immune response and showing sustained clinical benefit. Therefore, there is a growing interest in exploring immunotherapeutic targets to better optimize immunotherapy and enhance the therapeutic efficacy of ICIs in PSC. This comprehensive review highlights recent advancements in immunotherapy and related biomarker targets for PSC for optimizing immunotherapeutic strategies in the future.
Immunotherapy for PSC
Single-agent immunotherapy
Back-line treatment
A retrospective study involving 37 patients with stage III/IV PSC treated with ICI monotherapy as second line and beyond therapies showed favorable responses, with an overall objective response rate (ORR) of 40.5%, a disease control rate (DCR) of 64.8%, and a median OS of 12.7 months [95% confidence interval (CI): 0.3–45.7]. Further, six patients (16.2%) reported treatment-related adverse events (TRAEs), such as diarrhea and interstitial pneumonia. 12 Another similar study investigated 175 patients with PSC, of whom 49 patients were enrolled in the efficacy analysis cohort, 40 patients received pembrolizumab, 7 patients received nivolumab, and 2 patients received atezolizumab. The study found an ORR of 49.0%, median progression-free survival (PFS) of 7.2 months (95% CI: 4.9–9.5), and median OS of 22.2 months (95% CI: 7.0–37.3). TRAEs were seen in 13 patients (26.5%), including TRAEs of grade 4 or 5, such as immune-associated pneumonia in three patients (6.1%). 13 These retrospective studies found single-agent immunotherapy as a promising choice as the back-line treatment. Toyozawa et al. 14 conducted a multicenter, single-arm phase II trial (NCCH1603), evaluated nivolumab monotherapy as the second-line treatment in 13 patients with relapsed PSC, and determined an ORR of 30.8% and a DCR of 53.8% with grade ⩾3 TRAEs in 15.4% of patients.
The above studies have shown that immunotherapy is more clinically effective and generally well tolerated as a treatment option for patients with advanced PSC than chemotherapy, even as a backline treatment option, making single-agent immunotherapy a promising treatment option for patients with PSC.
First-line treatment
Prior investigations produced preliminary evidence supporting the efficacy of ICI monotherapy as a first-line treatment option for PSC. However, the number of studies that supported single-agent immunotherapy as an effective first-line therapy for PSC remains limited. The NCCH1703 study, a multicenter, single-arm phase II trial evaluated programmed death-1 (PD-1) monoclonal antibody pembrolizumab, in previously untreated PSC patients with high expression of programmed death ligand-1 (PD-L1) [tumor proportion score (TPS) of ⩾50%]. The study analysis determined an ORR of 68.2% and a DCR of 81.8%, and the incidence of grade ⩾3 TRAEs was 15.4%. 14 Further, retrospective analyses and case reports demonstrated the effectiveness of single-agent immunotherapy as first-line treatment in PSC. In a previous study involving 21 PSC patients, three patients received immune monotherapy and two patients were treated with pembrolizumab. Among the two, one had an OS of 12.6 months, while the other had an OS exceeding 25.4 months. Additionally, patients who received sintilimab as a first-line treatment showed a PFS of over 5 months and did not reach the OS endpoint. Besides, no serious TRAEs (grade ⩾3) were noticed. 15 Matsumoto et al. 16 reported a case with advanced PSC who received pembrolizumab monotherapy as first-line treatment and achieved a partial response (PR) after three treatment cycles with an acceptable safety profile.
Collectively, single-agent immunotherapy demonstrates better efficacy and tolerable adverse effects as a first-line treatment for PSC when compared to conventional chemotherapy, but it still needs further verification using large-scale prospective clinical trials with longer follow-up periods.
ICI as combination therapy
ICIs when combined with antiangiogenic drugs
Vascular invasion is present in 90% of PSC patients, which is associated with poor prognosis, 17 and early administration of antiangiogenic drugs may improve the patient’s response to immunotherapy. 18 An open-label, multicenter, single-arm phase II clinical trial (ChiCTR2000032649) investigated 16 patients with stage IIIB–IV PSC in two groups (camrelizumab + apatinib, n = 3; camrelizumab, n = 13). In the camrelizumab plus apatinib group, the ORR was 66.7%, the DCR was 66.7%, and the median duration of response (DoR) was 2.53 months (95% CI: 1.4–3.1); in the camrelizumab monotherapy group, the ORR was 54.5%, the DCR was 90.9%, and a median DoR of 3.19 months (95% CI: 1.1–6.9). TRAEs of grade 3 or 4 were elevated AST/ALT (6.25%) and pituitary inflammation (6.25%), and no treatment-related deaths were reported. 19 This combination therapy of camrelizumab with apatinib has a relatively high ORR, and we need more prospective studies to demonstrate the feasibility of this therapy. In a study conducted by Qian et al., in 21 patients diagnosed with stage III/IV PSC analyzed, 14 patients received a combination of ICIs with anlotinib, and 3 patients received single-agent immunotherapy. The results demonstrated that among the seven patients treated with tislelizumab in combination, two patients achieved PR and four patients achieved stable disease. All four patients treated with camrelizumab plus anlotinib achieved PR and survived beyond 14 months. Additionally, a patient treated with pembrolizumab plus anlotinib achieved PR, PFS of 10.5 months, and OS of 14.2 months. The median PFS from single-agent immunotherapy and ICIs combined with anlotinib were 8.0 and 9.4 months, respectively, while the median OS were 19.0 and 22.8 months, respectively. Importantly, both regimens were generally well tolerated. 15 Furthermore, Li et al. 20 reported a case of advanced PSC switched to tislelizumab plus anlotinib therapy after failure of first-line conventional chemotherapy, and after 13 cycles of the combined treatment, the patient achieved complete response (CR) and was still on the drug.
The aforementioned studies and the case report provided evidence supporting the potential of ICIs in combination with antiangiogenic drugs for achieving effective antitumor activity in patients with PSC. However, further validation through prospective studies is warranted to substantiate these findings. Professor Li’s team from Sichuan Cancer Hospital is currently conducting an open-label, multicenter, single-group, phase II clinical trial (NCT04725448) to evaluate the therapeutic efficacy of toripalimab in combination with bevacizumab- and platinum-based chemotherapy in patients diagnosed with primary advanced PSC.
ICIs when combined with chemotherapy
The National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines (2019, V7) recommend chemotherapy in combination with ICIs (class 1) for advanced non-squamous NSCLC with high PD-L1 expression (PD-L1 of TPS ⩾ 50%) and no expression of epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK). 21 Even in populations with PD-L1 TPS of <50%, such treatment modalities can still provide a survival benefit. 22 The effectiveness of this combination therapy was supported by preliminary data from preclinical models, finding a synergistic effect between ICIs and chemotherapeutics, resulting in improved clinical outcomes in patients with advanced NSCLC.23–25
Presently, prospective clinical trials on the effectiveness of ICIs plus chemotherapeutics in PSC patients are in progress. In a recent retrospective analysis that included 21 patients with localized or locally advanced PSC, four patients received platinum-based chemotherapy plus ICI and three patients received single-agent immunotherapy. The results showed that all patients who received chemotherapy plus pembrolizumab achieved PR, with one patient in continued remission for 11.7 months and the other had a PFS of 10 months and an OS of up to 30.6 months; the patient who received chemotherapy plus sintilimab had a PFS of 9.6 months and did not achieve an OS; the patient who received the combination of chemotherapy with durvalumab showed rapid progression. Moreover, the median PFS from single-agent immunotherapy and chemotherapy plus immunotherapy were 8.0 and 9.6 months, and the median OS were 19.0 and 30.6 months, respectively. TRAEs of grade ⩾3 were not found. 15 Okauchi et al. presented a case report of a 60-year-old male with advanced primary PSC characterized by high PD-L1 expression (TPS >75%). This patient was treated with pembrolizumab in combination with platinum-based chemotherapy, and after six cycles of the combined therapy, the patient achieved a significant general remission with a PR by the efficacy assessment and experienced no significant TRAEs. 26 Likewise, Kong et al. reported about a female patient with advanced PSC who was treated with camrelizumab plus doxorubicin and cisplatin as first-line therapy. Due to the reduction in leukopenia grade (grade IV), the patient was switched from combination therapy to camrelizumab monotherapy and achieved PR after six cycles of combination therapy, with the sustained response for more than 20 months. 27
The combination of ICIs with chemotherapeutics has shown promising clinical efficacy with manageable toxicity in advanced PSC patients. However, further evaluation through prospective clinical studies is warranted to confirm these findings of its effectiveness and to regularly use in clinical practice.
Dual immunotherapy
Dual immunotherapy, a promising approach to enhance the antitumor immune response, involves simultaneous targeting of two different immune checkpoints. The two drugs used in combination have distinct mechanisms of action and potential synergy, complementing each other for their mechanism of action. Spatially, the PD-1 pathway primarily regulates T-cell activity at the periphery, while the cytotoxic T lymphocyte antigen 4 (CTLA-4) pathway acts mainly in the lymph nodes. Temporally, the CTLA-4 antibody acts early in T-cell development, preceding the action of PD-1 antibody/PD-L1 antibody, which occurs at the effector stage.28,29
Given the significant benefit of dual immunotherapy observed in patients with NSCLC, 30 Kim et al. conducted a non-randomized, open-label, multicenter, single-arm phase II clinical trial (NCT03022500) to evaluate the efficacy and safety of durvalumab and tremelimumab in the treatment of PSC patients with a total of 18 patients with treatment relapse or metastatic PSC. At the data cutoff point, the ORR was 26.7%, and median PFS and OS were 5.9 months (95% CI: 1.1–11.9) and 15.4 months (95% CI: 11.1–not reached), respectively. AEs occurred in 16 patients (88.9%), with two cases who discontinued the treatment due to TRAEs of grade ⩾3. Importantly, no deaths related to treatment-related adverse events (TRAEs) were reported during the study period. 31
Several prospective clinical trials with dual immunotherapeutics for PSC are underway, including NCT02834013, a non-randomized, open-label, multicenter phase II clinical trial designed to evaluate the clinical efficacy of nivolumab and ipilimumab in patients with advanced PSC. We are eagerly awaiting the results of these trials. The current ongoing and registered clinical studies of ICIs for the treatment of PSC are presented in Table 1.
Trials with ICIs in PSC patients.
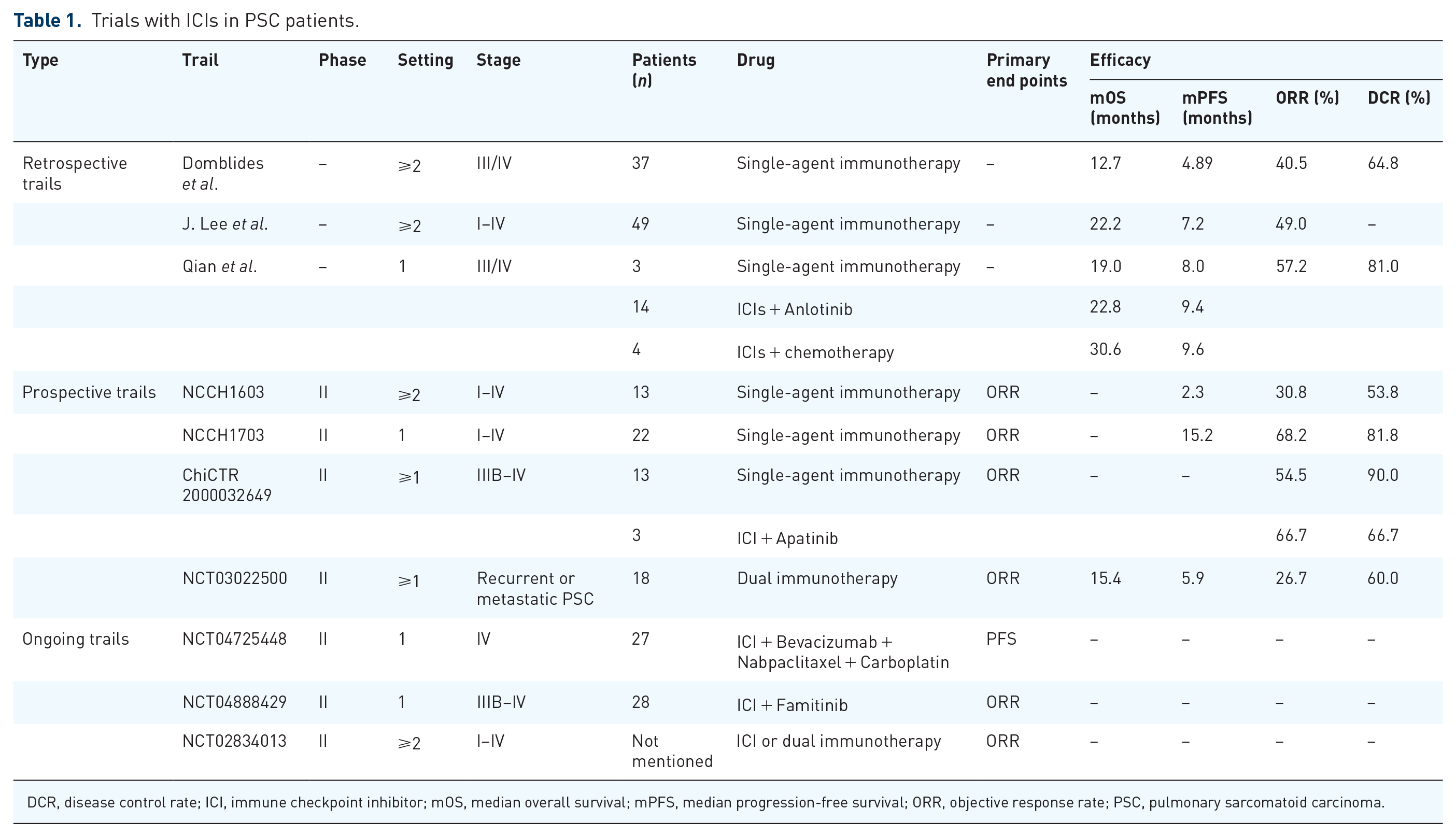
DCR, disease control rate; ICI, immune checkpoint inhibitor; mOS, median overall survival; mPFS, median progression-free survival; ORR, objective response rate; PSC, pulmonary sarcomatoid carcinoma.
Immunotherapy for PSC with driver gene mutations
PSC is a type of NSCLC with a high frequency of gene mutations, which are carried by 69–80% of PSC patients.32,33 For example, about one-third of PSC patients have MET exon 14 skipping mutation (MET ex14), which is a factor of poor prognosis in advanced PSC patients. 34 A multicenter, single-arm, open-label phase II clinical trial enrolled 70 patients with METex14-positive NSCLC, including 25 patients with PSC. Savolitinib showed promising antitumor activity in PSC patients with an ORR of 40.0% and an mPFS of 5.5 months. 35 Unfortunately, there is a lack of studies of immunotherapy in METex14-positive PSC.
Kirsten rat sarcoma viral oncogene homolog (KRAS) mutation is one of the common mutations in PSC, with KRAS G12C being the most common type of mutation. 33 Furthermore, in PSC patients, KRAS mutations were positively correlated with tumor mutational burden (TMB) and PD-L1 expression, 36 and they had a higher response rate to pembrolizumab. 37 AMG510 has promising efficacy in NSCLC with KRAS G12C mutations, 38 but its utility in PSC patients is unknown.
The frequency of EGFR mutations in PSC patients generally ranges from 10% to 19%.39,40 The efficacy of EGFR tyrosine kinase inhibitors (TKIs) and ICIs in PSC patients has rarely been reported. Tamura et al. 41 reported that a patient with EGFR exon 19 deletion in PSC achieved CR that lasted 35 months after receiving gefitinib as first-line therapy. A patient in a retrospective study with an EGFR exon 18 mutation, a Tumor protein p53 (TP53) mutation, and low TMB (4 mutations per Megabase, mut/Mb) developed early progression after immunotherapy. 12 Likewise, a case report of a patient who also carried an EGFR exon 18 mutation, combined with TP53 mutation and low TMB (6.15 mut/Mb), had a similar outcome to the above patients, 42 which may imply that patients with PSC who have an EGFR mutation may benefit less from immunotherapy.
In PSC patients, the incidence of ALK fusion mutations was 2.3% 43 and the incidence of ALK rearrangements was 3.5%. 44 In a case report, patients carrying ALK fusion-positive PSC, who had disease progression during treatment with pembrolizumab even though high PD-L1 expression was detected, experienced effective tumor remission after switching to targeted therapy with an ALK inhibitor. 45 Therefore, in PSC patients initially diagnosed as ALK-positive with high PD-L1 expression, upfront targeted therapy may be the best therapeutic option for patients compared with immunotherapy. Lin et al. 46 reported that a patient with advanced PSC harboring an ALK rearrangement achieved PR after 7 months of treatment with crizotinib.
The results of the latest study showed that 5.3% of PSC are SMARCA4-deficient. 47 Thoracic SMARCA4-deficient undifferentiated tumors (SD-UTT) are a unique entity in the fifth edition of the WHO classification of thoracic tumors, 4 which is a rare tumor with a high degree of malignancy and low efficacy of conventional chemotherapy,48–50 and studies have found that immunotherapy and Enhancer of zeste homolog 2 inhibitor-targeted therapy have achieved better results.51–53 However, based on histopathological and immunohistochemical features, SMARCA4-deficient PSC could not be classified as SD-UTT. 54 For SMARCA4-deficient PSC patients, we need to validate the effectiveness of targeted and immunotherapy in future clinical trials.
At present, targeted therapy and immunotherapy are the mainstream direction in the field of PSC treatment. It is necessary to conduct genetic testing on patients before treatment, and the results of genetic testing can not only guide the selection of targeted therapy, but also have a certain suggestive effect on the efficacy of immunotherapy.
Immunotherapy biomarker targets
Immunotherapeutics, specifically ICIs, have shown promising results in terms of sustained responses and improved survival outcomes in most patients with PSC. But a subset of patients does not have optimal benefits from immunotherapy, emphasizing the need for identifying reliable biomarkers in that subpopulation that may respond favorably to immunotherapy.
Programmed death ligand-1
PD-L1 has emerged as a widely studied biomarker and its expression in tumor tissues has been used for predicting the efficacy of immunotherapy. PD-L1 protein is expressed on both tumor cells and antigen-presenting cells and serves as an immune checkpoint molecule. It binds to PD-1 on the surface of T cells, inhibits T cell activation, and causes subsequent evasion of immune surveillance and destruction by tumor cells, thereby promoting tumor progression (Figure 1).55,56
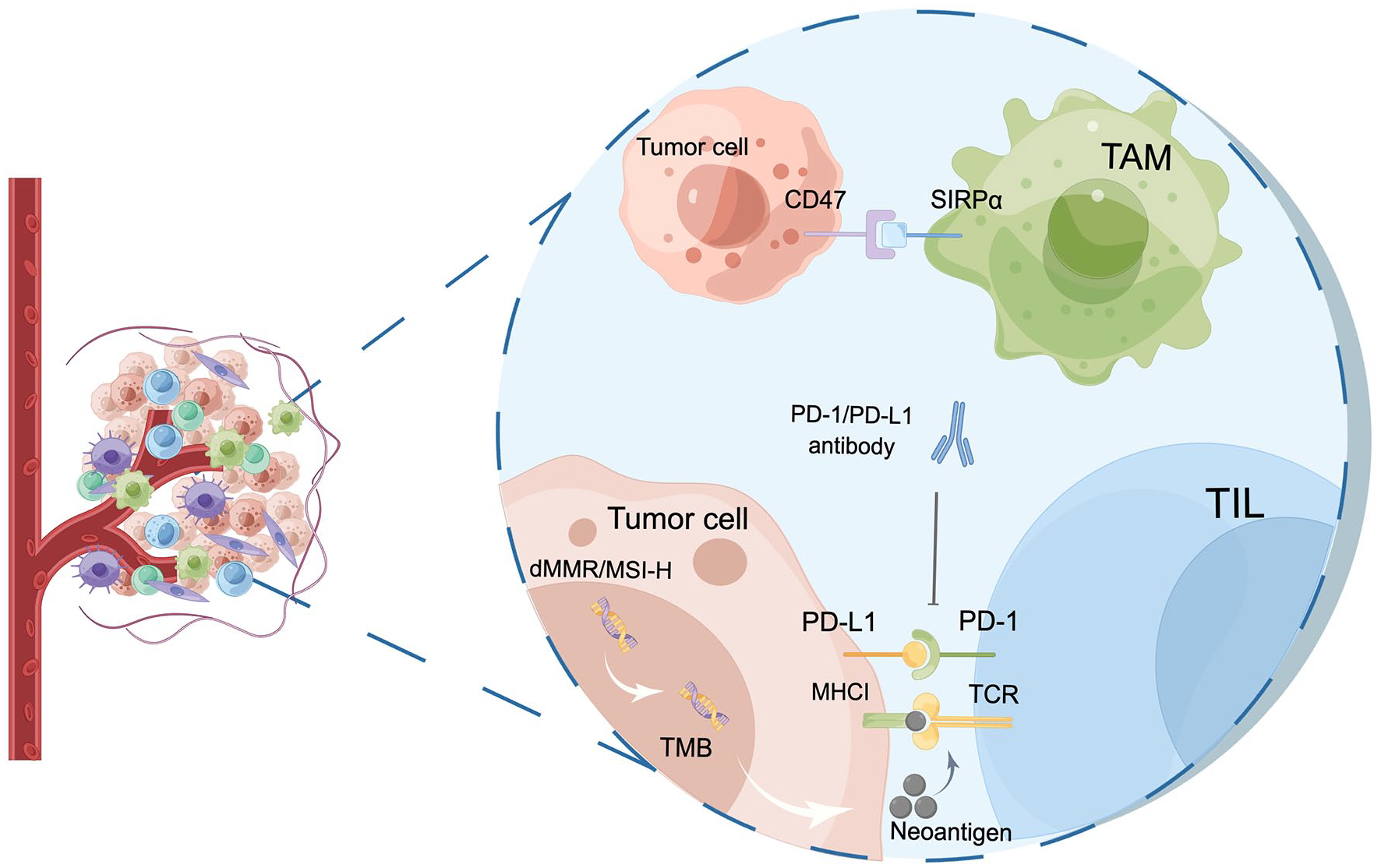
Main biomarkers for predicting treatment efficacy of ICIs. First, the binding of PD-1 to its ligand PD-L1 on tumor or immune cells can potently inhibit the cytotoxic T-cell response, resulting in tumor cells evading immune surveillance. Furthermore, dMMR and MSI-H exhibit a robust correlation with elevated TMB. The heightened TMB levels are associated with an increased tumor neoantigen load, which augments immune recognition and fosters the development of an antitumor immune response. Third, TILs recognize endogenous antigenic peptides presented by MHC class I molecules, which play a pivotal role in orchestrating the immune response to eliminate tumor cells. Lastly, CD47, by binding to SIRPα expressed on the surface of macrophages, propagates immunosuppressive signals downstream, thus contributing to immune evasion by tumor cells.
In PSC, high PD-L1 expression has been found as a distinctive characteristic, with significantly elevated levels compared to conventional NSCLC, as demonstrated in a comparative gene expression study. 57 Lee et al. conducted an extensive study involving 175 PSC patients, of which 125 patients were included in the cohort for the PD-L1 expression analysis. The overall PD-L1 positive rate was 89.6% (112/125). Further efficacy analysis revealed that ORR was observed exclusively in the high PD-L1 expression group, with a notable ORR of 58.5%. In contrast, no objective response events were reported in the low/negative/unknown PD-L1 expression group. 13 Another retrospective analysis of 37 patients with stage III/IV PSC demonstrated a median PD-L1 expression of 70% (ranging from 0% to 100%), with a trend toward patients with higher PD-L1 expression in responding to immunotherapy. The ORR for PD-L1-positive patients was 58.8%, compared with 0% for PD-L1-negative patients. 12 Similarly, within a pooled cohort of 90 patients diagnosed with PSC who were treated with immunotherapy, a substantial 73.3% of patients showed positive PD-L1 expression. Additionally, patients with a PD-L1 of ⩾1% had a greater survival benefit compared to PSC patients with a PD-L1 of <1%, with a median PFS of 14.4 months (95% CI: 4.6–24.2) and 2.7 months (95% CI: 1.4–4.1), respectively, and the percentage of patients achieving DCR increased with rising levels of PD-L1 expression. 58
Despite accumulating evidence indicating a correlation between PD-L1 expression levels and response to immunotherapy, the use of PD-L1 as a biomarker remains controversial, with several limitations hindering its widespread use. One major challenge is the lack of standardization of PD-L1 detection methods, with numerous platforms and antibodies available for PD-L1 assessment. Moreover, different platforms utilize diverse scoring systems to determine PD-L1 expression levels, and there are no universally accepted criteria to define the negative versus positive PD-L1 expression. 59
Tumor mutational burden
In 2020, the U.S. Food and Drug Administration approved the TMB parameter as another immunotherapy biomarker. TMB is defined as the total count of somatic mutations in the genome of a tumor cell. As somatic mutations can result in the generation of neo-antigens, an increased TMB is associated with a higher probability of neo-antigens being presented by major histocompatibility complex proteins, which in turn may enhance T cell recognition (Figure 1).60,61
High TMB, defined as ⩾10 mut/Mb, has been frequently observed in patients with PSC, as evidenced by a retrospective study examining tissue specimens from 15,867 patients with NSCLC, including 125 cases of PSC. The study revealed a significantly higher proportion of PSC cases with high TMB (⩾20 mut/Mb) compared to non-PSC NSCLC (20% versus 14%). 10 In a retrospective analysis conducted by Domblides et al., 37 patients with stage III/IV PSC were assessed for the treatment efficacy of ICIs. The findings demonstrated a median TMB of 18 mut/Mb, with 87.5% of cases exhibiting high TMB (⩾10 mut/Mb), and a notable difference in median OS between the high-TMB population (18 months) and the low-TMB population (1.84 months). 12
The potential use of high TMB as a predictive tool for evaluating immune system efficacy in patients with PSC holds promise. Despite its predictive value in immunotherapy treatment outcomes, the clinical application of high TMB levels is constrained by several limitations. First, the assessment of TMB expression necessitates a substantial quantity of tumor tissue, and obtaining a larger tissue may be challenging in advanced PSC cases. Moreover, there is considerable variability in the calculation methods employed for determining TMB, lacking a uniform standard across the field.
Tumor microenvironment-related biomarkers
The dynamic interplay between the tumor parenchyma and tumor interstitium within the TME has been recognized as a pivotal determinant of cancer progression. 62 The immune cellular components constituting the TME encompass a diverse array of cell types, including T and B lymphocytes, tumor-associated macrophages (TAMs), dendritic cells, natural killer cells, neutrophils, and bone marrow-derived myeloid-derived suppressor cells (Figure 1). 63
The majority of PSC patients showed immune-inflamed type and type I (PD-L1+/CD8+, adaptive immune resistance) resistance, underlying the notable responsiveness of PSC patients to immunotherapy. 64 A comprehensive analysis of surgical specimens from 75 PSC patients and 54 NSCLC cases demonstrated that PSC patients exhibited higher immune cell infiltration, including CD3+ tumor-infiltrating lymphocytes (TILs) and CD163+ TAMs, compared to NSCLC patients. Moreover, increased infiltration of CD3+ and CD4+ TILs in PSC patients was associated with longer OS. Interestingly, tumors positive for PD-L1 demonstrated significantly higher infiltration of CD8+ TILs and CD163+ TAMs compared to PD-L1-negative tumors (10% versus 8%; 27% versus 20%). 65 CD8+ cytotoxic T lymphocytes upregulate immune checkpoint receptors, such as PD-L1 and CTLA-4 on tumor cells, rendering them more susceptible to immunotherapy. 66 Therefore, CD8+ TIL density may serve as a sensitive predictor for immunotherapy efficacy. In a study by Chen et al., a high density of CD8+ TILs was observed in tissues from 49% of 100 PSC patients, with mean OS and PFS of 92.3 and 71.6 months, respectively, compared to 31.2 and 20.2 months in patients with low CD8+ TIL density, respectively. 67
The aforementioned studies highlighted the significant correlation between TILs, TAMs, and PD-L1 positive expression, which can serve as potential predictors for immunotherapy response in a certain context. However, it should be noted that despite their potential as biomarkers, tumor microenvironment-related biomarkers have not yet reached sufficient adequacy in predicting response to immunotherapy in PSC patients, primarily due to the limited availability of prospective trials in this regard. Therefore, well-designed prospective studies are required to validate the prognostic and predictive value of these biomarker targets in PSC patients undergoing immunotherapy.
Cluster of differentiation 47
The cluster of differentiation 47 (CD47), a widely expressed membrane glycoprotein, plays a crucial role in enabling tumor cells to evade immune surveillance by interacting with macrophage-expressed signaling regulatory protein alpha, resulting in a ‘don’t eat me’ signal (Figure 1). 68
CD47 checkpoint is an emerging molecule that holds promise to be used as a therapeutic target and immunotherapy biomarker, but it warrants further investigation for its potential clinical applications. A retrospective analysis was conducted on 148 patients with PSC and revealed that 54 (36.5%) patients were positive for PD-L1 expression, 78 (52.7%) patients were positive for CD47 expression, and 36 (24.3%) patients had co-expression of PD-L1 and CD47. Notably, there was a significant correlation observed between PD-L1/CD47 co-expression and a higher density of CD8+ TILs and CD68+ TAMs. Furthermore, patients exhibiting positive CD47 expression and PD-L1/CD47 co-expression showed a poorer prognosis. 69 Consequently, PD-L1/CD47 has the potential to serve as a novel predictive marker for dual-targeted therapy in PSC patients, thereby highlighting its prospects for the clinical setting.
In conclusion, PSC is characterized by elevated PD-L1 expression and TMB, accompanied by notable immune infiltration, implying the potential benefits of ICIs therapy in PSC patients. Nevertheless, the limitations of individual biomarkers in specific populations that may benefit from immunotherapy vary. Hence, integrating multiple biomarkers to construct robust and effective comprehensive prediction models could facilitate the identification of more effective treatment modalities, thereby optimizing clinical outcomes and advancing precision immunotherapy.
Conclusion and perspectives
Immunotherapy has promised better clinical outcomes and a manageable safety profile in patients with PSC, providing renewed hope for those who had failures with conventional therapies. However, the efficacy of single-agent immunotherapy in PSC is limited, and combination immunotherapy approaches are emerging as a potential therapeutic option in current clinical practices. Advances in targeted therapies have provided new ideas for the treatment of PSC patients. In particular, for PSC patients with MET ex14 mutations, MET-TKI-targeted therapy offers hope for long-term survival. In conclusion, the treatment approach for PSC should continue to follow the model of NSCLC, wherein TKIs or ICIs are chosen based on the patient’s driver mutation status. Because there are few prospective clinical trials on targeted therapies, a combination of immunotherapy-based treatments is more important for PSC patients. To further evaluate the efficacy and risk, it is imperative to identify ideal biomarkers and develop personalized treatment strategies for PSC patients. Nonetheless, the high heterogeneity of PSC poses challenges in relying solely on single biomarkers for accuracy, and a comprehensive model needs to be explored in the future combining PD-L1, TMB, TILs, and other indicators to screen out the most favorable population who can achieve the maximum effectiveness from immunotherapy. Currently, targeted therapies and immunotherapies are increasingly used in the field of PSC, which heralds the beginning of the precision therapy model for treating PSC; however, larger prospective studies are warranted to further validate these findings in PSC patients.
