Abstract
Biliary tract cancers (BTCs), consisting of intrahepatic and extrahepatic cholangiocarcinoma and gallbladder cancer, are an aggressive, heterogeneous malignancy. They are most often diagnosed in the locally advanced or metastatic setting, at which point treatment consists of systemic therapy or best supportive care. Our understanding of the tumor microenvironment and the molecular classification has led to the identification of targetable mutations, such as isocitrate dehydrogenase 1 and fibroblast growth factor receptor 2. Despite the identification of these genomic alterations, until recently, little advancement had been made in the first-line setting for advanced BTC. While immunotherapy (IO) has revolutionized the treatment of many malignancies, the use of IO in BTC had yielded limited results prior to TOPAZ-1. In this review, we discuss the systemic therapeutic advances for BTC over the past decade, the rationale for immunotherapy in BTC, prior trials utilizing IO in BTC, and current and emerging immune-based therapeutic options. We further analyze the culmination of these advances, which resulted in the approval of durvalumab with gemcitabine and cisplatin for the first-line treatment of BTC per TOPAZ-1. We also discuss the results of KEYNOTE-966, which similarly reported improved clinical outcomes with the use of pembrolizumab in combination with gemcitabine and cisplatin.
Introduction
Biliary tract cancers (BTCs) are a diverse group of malignancies classified by their primary anatomical site of origin and consist of intrahepatic cholangiocarcinoma (iCCA), extrahepatic cholangiocarcinoma (eCCA), and gallbladder cancer (GBC). 1 While they represent only 15% of primary liver cancer, their incidence has been steadily increasing over the past three decades, coinciding with a rising mortality burden.2,3 BTC are aggressive malignancies with a 5-year overall survival (OS) rate of less than 10%. 4 Most patients are diagnosed with metastatic or locally unresectable disease, correlating with a poorer prognosis and limited therapeutic options, including systemic therapy, clinical trials, or best supportive care. 5
In this narrative review, we describe the decade of progress in therapy for advanced BTC patients, culminating in the recent approval of durvalumab in combination with gemcitabine and cisplatin for first-line therapy due to the TOPAZ-1 trial.
Pre-TOPAZ-1 first-line treatment for BTC
Despite the rising incidence and mortality rate, until recently, little advancement had been made in the treatment of advanced BTC. Over a decade ago in 2010, the landmark phase III trial ABC-02 established cisplatin and gemcitabine as the standard of care for first-line treatment of advanced or metastatic BTC.
6
This study showed an improved progression-free survival (PFS) of 8.0 months for patients treated with cisplatin and gemcitabine compared to 5.0 months for patients treated with gemcitabine monotherapy, with an accompanying improvement in median OS of 11.7
Several other clinical trials have evaluated multi-agent chemotherapy compared to doublet chemotherapy with disparate results.8–12 The addition of nanoparticle albumin-bound (nab)-paclitaxel to cisplatin and gemcitabine in a phase II trial led to a remarkable median OS of 19.2 months (95% CI, 13.2 months to not estimable) and a median PFS of 11.8 months (95% CI, 6.0–15.6). 13 Unfortunately, the phase III randomized clinical trial, SWOG 1815 (NCT03768414), failed to show a statistically significant improvement in median OS between gemcitabine and cisplatin compared to gemcitabine, cisplatin, and nab-paclitaxel. 14 Subset analyses showed that patients with locally advanced disease did significantly better with the triplet regimen and could be an option in this subset, though this treatment option is not currently included in international guidelines.
There has been interest in examining the utility of fluoropyrimidines in BTC, as well. In Japan, a phase III study compared gemcitabine and cisplatin to gemcitabine and S-1 for the treatment of chemotherapy naïve recurrent or advanced BTC.
15
The combination of gemcitabine/S-1 was found to be non-inferior to gemcitabine/cisplatin. The median OS was 13.4 months with gemcitabine/cisplatin and 15.1 months with gemcitabine/S-1 (HR, 0.945; 90% CI, 0.78–1.15;
Second-line treatment for BTC
Across all these studies, patients inevitably progress on first-line therapy. Treatment for BTC in the second-line setting has yielded limited results. The ABC-06 trial assessed 5-fluorouracil (5-FU) and oxaliplatin (FOLFOX) and active symptom control (ASC) to ASC alone.
18
While the study met its primary endpoint of improved OS, the benefit was minimal with approximately 1 month of improved median OS [6.2
Molecularly targeted therapy
The identification of genomic alterations through the use of next-generation sequencing has broadened the treatment landscape for BTC for those with actionable alterations, such as isocitrate dehydrogenase 1 (IDH1) (10–20%), fibroblast growth factor receptor 2 (FGFR2) fusion/rearrangement (15%), v-raf murine sarcoma viral oncogene homolog B1 (BRAF) (5%), human epidermal growth factor receptor 2 (HER2) (15%), though alterations are known to vary between anatomic site of origin.22–24
The most significant advancement in biomarker-guided therapy has been the subset of patients with FGFR2 fusions/rearrangements. FGFR2 inhibitors pemigatinib, infigratinib, and futibatinib have received accelerated approval for the treatment of BTC with FGFR2 fusions/rearrangements, with response rates of 23–42%.25–27 Multiple novel agents targeting FGFR2 are presently in clinical trials and showing promising results. In addition, for patients with IDH1 mutations, the phase III ClaIDHy study established ivosidenib as the second-line treatment after demonstrating a 1.3-month improvement in PFS, and a 4.8-month improvement in OS when adjusted for crossover. 28 Novel IDH1/2 inhibitors are also being investigated in ongoing clinical trials. Combined BRAF and MEK inhibition therapy has also shown promising results for those with BRAF V600E mutations, as have the combination of anti-Her2 therapy with pertuzumab and trastuzumab.29,30 Both are now included in the NCCN guidelines. 21 Less common but actionable alterations include microsatellite instability (MSI)/mismatch repair deficient (dMMR) (~1–2%), neurotrophic tyrosine receptor kinase (NTRK) (<1%), and rearranged during transfection (RET) (<1%).31–33
Rationale for immunotherapy responsiveness in BTC
While this expanding landscape offers additional treatment options for a subset of patients, the presence of a targetable mutation, which occurs in a minority of patients, does not guarantee durable clinical benefit. New treatment strategies applicable to the majority of patients with BTC are urgently needed. While immunotherapy has revolutionized the treatment of many malignancies, the benefit of immunotherapy in BTC is more recently being recognized. Treatment with immunotherapy has largely centered around the use of immune checkpoint inhibitors (ICIs), namely monoclonal antibodies to programmed cell death 1, (PD-1), programmed cell death ligand 1 (PD-L1), and cytotoxic T-lymphocyte-associate protein 4 (CTLA-4). PD-1, a transmembrane protein expressed in T cells, B cells, and myeloid cells, regulates T-cell activation by binding to its ligands, programmed death ligands 1 and 2 (PD-L1, PD-L2).34,35 PD-1 binding inhibits T-cell proliferation, production of interleukin-2, interferon-γ (IFN-γ), tumor necrosis factor-α, and reduces T-cell survival. The binding of CTLA-4 to its ligands B7-1 and B7-2 leads to T-cell anergy and apoptosis while preventing CD28 co-stimulation.36,37 As the BTC research community works to optimize immune approaches, a detailed understanding of the immunobiology and tumor microenvironment (TME) of BTC is necessary.
The use of integrated whole-genome and epigenomic analyses has led to a better understanding of radically different molecular and biological landscapes.
38
In one unique molecular cluster, immune cell infiltration within cholangiocarcinoma was noted to have upregulation of
While the subset analyses have suggested those BTCs likely to respond to IO, these hypotheses need to be validated in prospective studies with biospecimen interrogation. Further investigations have deepened our comprehension of the role of chemotherapy in enhancing the efficacy of IO, a strategy inclusive of a broader patient population.42–44 Certain chemotherapy agents, including gemcitabine and cisplatin, have been shown to induce immunogenic cell death and immunomodulatory effects through diverse pathways.45–48 Gemcitabine, for example, has been shown to induce tumor cell apoptosis, enhance the cross-priming of CD8+ T cells, and modulate dendritic cells to enhance tumor-specific T-cell responses.49,50 Several other studies have shown that conventional chemotherapy can eliminate myeloid-derived suppressor cells (MDSCs), deplete regulatory T cells, and disrupt tumor stroma.51–55 Combination therapy in preclinical models has also been shown to enhance T-cell responses. Effects include increasing dendritic cell function by augmenting T-cell response and upregulation of HLA1 expression. These mechanisms thereby restore the immune visibility of tumor cells to T cells. 56 This preclinical evidence led to clinical trials assessing combinatorial therapies, and, with improved clinical outcomes, set the stage for exploration in BTC.57,58
Immunotherapeutic approaches for BTC
Several studies have investigated the use of IO as a single agent and as a combination therapy in BTC (Table 1).
Selected published clinical trials of IO in BTC.

A, atezolizumab; BTC, biliary tract carcinoma; C, cobimetinib; D, durvalumab; DCR, disease control rate; DGC, durvalumab + gemcitabine + cisplatin; GC, gemcitabine + cisplatin; GCA, gemicitabine + cisplatin + atezolizumab; GCAB, GCA + bevacizumab; IO, immunotherapy; N, nivolumab; ORR, objective response rate; OS, overall survival; P, pembrolizumab; PFS, progression-free survival; PGC, pembrolizumab + GC; T, tremelimumab; TDGC, tremelimumab + DGC.
Single-agent immunotherapy
The investigation of single-agent immunotherapy in the treatment of microsatellite stable BTC has shown limited benefit, often in small and/or non-randomized studies. In an early phase Ib study, KEYNOTE-028, patients with advanced BTC who had received ⩾1 line of prior therapy received pembrolizumab, a PD-1 inhibitor.59,71 PD-L1 positivity was an eligibility criterion and was defined as ⩾1% PD-L1 expression in tumor and associated inflammatory cells or positive staining in stroma. Among the 23 evaluable patients, the objective response rate (ORR) was 13.0% (3/23), and the median PFS was 1.8 months (95% CI, 1.4–3.1). The median OS was 5.7 months (95% CI, 3.1–9.8). Grade 3–5 treatment-related adverse events (AEs) occurred in 16.7% of patients (no grade 5), and 20.8% of patients experienced an immune-mediated AE. While this study showed modest activity, durable responses were appreciated in the three responders (21.5, 51.4, and 53.2 months). KEYNOTE-158, a phase II non-randomized trial, examined the activity of pembrolizumab in a larger cohort of advanced BTC, though it enrolled patients regardless of PD-L1 status.
72
The objective response rate (ORR) fell to only 5.8% (6/104, all partial response (PR); 95% CI, 2.1–12.1%). The median PFS was 2.0 months (95% CI, 1.9–2.1), and the median OS was 7.4 months (95% CI, 5.5–9.6). Of the patients who were evaluated for PD-L1 expression, ORR was numerically higher but not statistically significant for those with PD-L1 expression (6.6%
Several studies have also evaluated anti-PD1 nivolumab as monotherapy in patients with advanced BTC who progressed on prior lines of chemotherapy with varying results. In the study by Kim
A novel first-in-class bifunctional fusion protein, bintrafusp alfa (M7824), was evaluated as monotherapy as a treatment for refractory BTC. Bintrafusp alfa is composed of a human anti-PD-L1 IgG1 monoclonal antibody fused with two extracellular domains of the transforming growth factor β receptor II. 63 The ORR was 20% (6/30 patients). Given the clinical activity and durable responses for a subset of patients, a phase II/III clinical trial evaluating bintrafusp alfa in combination with gemcitabine and cisplatin was opened (NCT04066491). However, the study was discontinued after an independent committee determined that the primary endpoint of improved OS was unlikely to be met.
Throughout the clinical trials assessing monotherapy, multiple themes emerge. First, response and clinical benefit are limited with ORR ranging between 3.3% and 20%, with the largest series suggesting the RR is 6%. However, in the subset of patients who do respond, the durable response is long-lasting. This raises the question of how to identify patients who will have prolonged benefit from IO and emphasizes the need for further prospective trials with correlative analysis to delineate this inquiry.
Combinatorial IO strategies
The use of combinatorial immunotherapy with PD-1 and CTLA4 blockade has proven clinical benefit in several different malignancies and has been explored within BTC.74–76 In the CA209-538 prospective multicenter phase II trial for rare cancer, 39 patients with BTC received nivolumab 3 mg/kg and ipilimumab 1 mg/kg every 3 weeks for four doses followed by nivolumab monotherapy 3 mg/kg every 2 weeks. 64 There was an ORR of 23% (9/39) with a disease control rate of 44%. Consistent with prior monotherapy IO trials, durable responses were appreciated in a subset of patients with a median DOR NR (range, 2.5 to ⩾23 months). Of note, six patients were treatment-naïve, none of whom responded to combination treatment. In a larger study assessing the combination of durvalumab (20 mg/kg every 4 weeks) and tremlimumab (1 mg/kg every 4 weeks), ORR for patients with BTC was only 10.8% (7/65) with a median DOR of 8.4 months. 62 The median PFS was only 1.6 months (95% CI, 1.4–2.8) with a median OS of 10.1 months (95% CI, 6.5–11.6). Toxicities were consistent with prior studies, though treatment-related serious AEs were reported in nine (13.8%) patients with BTC. The IMMUNOBIL GERCOR D18-1 PRODIGE-57 study assessing the combination of durvalumab (1500 mg Q4 weeks) and tremelimumab (75 mg Q4 weeks X4 cycles) for recurrent and advanced BTC reported a similar ORR of 9.7%. 77 Median PFS and median OS were 2.5 months (95% Cl, 2.0–3.2) and 8.0 months (95% Cl, 5.7–11.7), respectively. Thus, the combination of IO agents did not yield as profound of benefit noted in other malignancies.
Combination immunotherapy
Novel combinations to enhance the efficacy of IO have been explored. Preclinical models have shown tumor effect when treated with MEK inhibitors and PD-(L)1 inhibitors, and combination treatment may result in enhanced PD-L1 expression, CD8+ T-cell infiltration, and major histocompatibility complex class I expression.78–81 In a randomized phase II clinical trial, patients with unresectable or metastatic BTC were randomized to receive atezolizumab (anti-PD-L1) monotherapy or a combination of atezolizumab and cobimetinib (MEK inhibitor). 65 The study met its PFS endpoint of doubling PFS but with limited responses (<5% in both arms), though the durability of response was noted with the combination therapy response ongoing for >2 years. An ongoing randomized trial is testing the combination with a CD27 agonist to build on these results, based on preclinical data suggesting that the MEK inhibition was contributing to decreased T-cell cytotoxic function that could be rescued with a T-cell agonist (NCT04941287).
Epigenetic agents have also been utilized in combination with immunotherapy for the treatment of BTC (Figure 1). In a phase IB trial of guadecitabine, a DNA methyltransferase inhibitor, and durvalumab, a PD-L1 inhibitor, 23 patients with pre-treated advanced BTC received combination therapy (NCT03257761). 82 One patient had a partial response (5%) lasting 12 months, and five patients had stable disease (22%). The median PFS was 1.9 months, while the median OS was 8.6 months. Grade 3/4 AEs included only myelosuppression. While the clinical efficacy was limited, a subset of patients experienced a durable response. Furthermore, results of the combination of entinostat, a histone deacetylase inhibitor, with nivolumab for the treatment of metastatic cholangiocarcinoma are eagerly anticipated (NCT03250273). 83
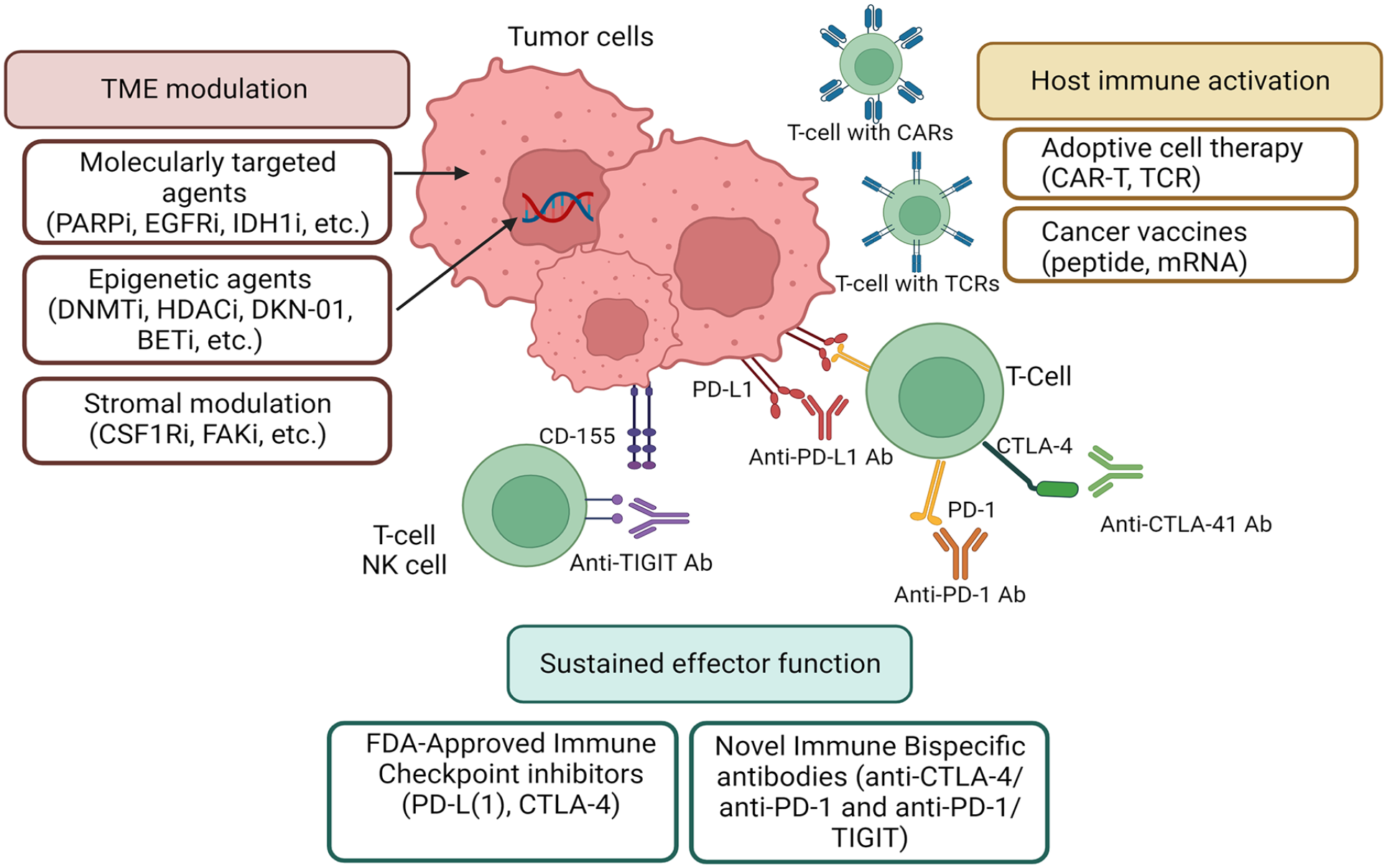
Novel approaches for the treatment of BTC. Currently, multiple novel approaches are being explored for the treatment of BTC. Novel immune bispecific antibodies (anti-CTLA-4/anti-PD-1 and anti-PD-1/anti-TIGIT), as well as FDA-approved immune checkpoint inhibitors in combination with cytotoxic chemotherapy, are being studied. The host immune system is underdoing activation by adoptive cell therapy and cancer vaccines. Molecularly targeted, epigenetic, and stromal modulation therapies that modulate the TME are also under investigation.
In addition, the efficacy of vascular endothelial growth factor (VEGF) inhibitors in combination with IO has been investigated both preclinically and clinically. Preclinical studies have shown that the immunosuppressive effects of VEGF lead to impaired CD8+ T-cell infiltration, inhibition of dendritic cell maturation and antigen presentation, upregulation inhibitory immune checkpoints, and accumulation of immunosuppressive cells, including regulatory T cells, MDSCs, and tumor-associated macrophages.
70
In addition to reverting the immunosuppressive effects of VEGF, the antiangiogenic VEGF inhibitors can augment the activity of PD-1 and PD-L1 inhibitors. This provides a rationale for the combination of PD-(L)1 blockade and VEGF inhibitors and has been an efficacious strategy in the treatment of multiple malignancies, including hepatocellular carcinoma, renal cell carcinoma, and endometrial carcinoma.84–87 In the LEAP-005 study, 31 patients with advanced BTC were treated with lenvatinib, a multiple tyrosine kinase inhibitor to VEGF, and pembrolizumab in the second-line setting.
66
Initial results of this ongoing study showed an ORR of 9.7% (95% CI, 2.0–25.8%). The disease control rate was compelling at 68% (
Rationale for anti-PD-1 (PD-L1) in combination with cytotoxic therapy
Since the investigation of IO monotherapy, several studies have investigated the efficacy of combination chemotherapy and IO, as preclinical evidence has shown synergy between cytotoxic chemotherapy and IO, as discussed above.42–44,48,56 IMBrave 151, a phase II randomized multicenter clinical trial, is assessing the combination of PD-L1 and VEGF blockade in addition to the backbone of gemcitabine and cisplatin (NCT04677504).
69
In this study, patients with untreated advanced BTC are randomized to either atezolizumab and bevacizumab or atezolizumab and placebo in combination with a backbone of gemcitabine and cisplatin. The median PFS for the atezolizumab and bevacizumab cohort was 8.4 months, while the median PFS in the atezolizumab and placebo cohort was 7.9 months and not statistically different. DOR ⩾6 months was 89% for atezolizumab and bevacizumab
Meanwhile, in phase I clinical trial, treatment-naïve patients with unresectable or metastatic BTC were eligible to receive nivolumab (240 mg every 2 weeks) in addition to standard dosing gemcitabine and cisplatin. 61 Of the 30 patients enrolled, 11 (37%) patients had a partial response by central assessment (37% ORR), and a median OS was 15.4 months. Treatment with combination therapy was well-tolerated with improved OS compared to IO alone, setting the stage for larger randomized control trials assessing combination chemotherapy and IO.
A pivotal phase II nonrandomized, single-center study assessed the combination of gemcitabine, cisplatin, and durvalumab with or without tremelimumab for treatment-naïve BTC.
67
Disease control rate was an unparalleled 100% (
Results of TOPAZ-1: a new first-line regimen
In the TOPAZ-1 study, patients with treatment naïve locally advanced, unresectable, or metastatic BTC were randomized 1:1 to receive either durvalumab or placebo in combination with gemcitabine and cisplatin.
68
Gemcitabine (1000 mg/m2) and cisplatin (25 mg/m2) were administered on days 1 and 8 of each 21-day cycle for up to eight cycles and durvalumab (1500 mg) or placebo was administered on day 1 of each cycle. Following eight cycles of treatment, either durvalumab (1500 mg) or placebo was continued once every 28 days until disease progression, unacceptable toxicity, or withdrawal from the study. This multi-center study enrolled 341 patients in the durvalumab group and 344 patients in the placebo group. Patients were stratified by disease status (unresectable
While not a primary objective, population subsets were further assessed. HRs most notably favored locally advanced (0.49; CI, 0.26–0.88) compared to metastatic disease (0.83; CI, 0.68–1.02). 89 Treatment of recurrent disease (HR, 0.56; CI, 0.32–0.96) had more favorable outcomes compared to initially unresectable disease (HR, 0.84; CI, 0.69–1.03). When analyzing OS based on primary tumor location, iCCA and eCCA maintained an OS benefit. However, the OS benefit for all patients with GBC favored the placebo cohort, and the OS was 10.7 months in the durvalumab group and 11.0 months in the placebo group (HR, 0.94; CI, 0.65–1.37). Importantly, these subset analyses based on anatomical subsite did not reach statistical significance. Per the authors, investigations are ongoing to understand these outcomes. The Asia subgroup trended toward improved OS with an HR of 0.72 (0.56–0.94) compared to the non-Asia subgroup with an HR of 0.89 (0.66–1.19). PFS also favored the Asia subgroup (HR, 0.67; CI, 0.53–0.83) compared to the rest of the world (HR, 0.87; CI, 0.68–1.12). Authors attribute this trend to longer follow-up in the Asia subgroup and imbalance in baseline characteristics, such as recurrent disease and Eastern Cooperative Oncology Group (ECOG). That said, it must be noted that the overall results of TOPAZ-1, while positive, did not reach the magnitude of benefit in the randomized phase II trial that served as the rationale for the study; the randomized phase II study was a fully Asian population, and there may well be immunobiological and/or pharmacogenomic differences that account for the increased benefit observed in Asian patients.
One of the exploratory outcomes of TOPAZ-1 was to assess efficacy outcomes by tumor mutations. The majority of patients, 64% (
The safety analysis included 680 patients, 338 in the durvalumab group and 342 in the placebo group. 68 Any grade AEs occurred in nearly all patients (99.4% in the durvalumab group and 98.8% in the placebo group) with grade 3 or 4 events occurring in 75.7% and 77.8% of patients, respectively. The most common AEs were similar between treatment groups and consisted of anemia, nausea, and neutropenia. The rate of immune-related AEs was 12.7% in the durvalumab group compared to 4.7% in the placebo group. Quality of life (QOL) was similar for both arms, as well. 91 Results of the safety analysis were consistent with prior studies and similar between treatment groups, and give comfort to treating with the IO and chemotherapy TOPAZ-1 regimen in terms of tolerability.
TOPAZ-1 was practice changing, as the only advancement to the treatment of BTC in the first-line setting since ABC-02 over a decade before.6,68 Yet, the OS benefit, while statistically significant, was only 1.3 months greater with durvalumab compared to placebo and highlights the need for optimization of treatment paradigms for BTC. This trial also raises a number of questions. First, while the addition of durvalumab led to improved OS and PFS, it is uncertain whether durvalumab during the first 6 months of chemotherapy provides clinical benefit. The Kaplan–Meier curves for OS and PFS are relatively uniform in the first 6 months with separation of the curves occurring after induction chemotherapy. While further investigation is warranted, the benefit of durvalumab may be most appreciable following induction chemotherapy and may limit immune-related AEs. Furthermore, a subset of patients, those represented by the tail of the Kaplan–Meier OS curves, experienced lasting benefits from durvalumab and are arguably the most clinically meaningful results from TOPAZ-1. The skew toward Asian patients and those with iCCA are important considerations; further analysis is needed to determine why there was continued benefit and how to determine which patients will experience prolonged OS. Despite these outstanding questions, TOPAZ-1 has advanced the treatment paradigm and led to the U. S. Food and Drug Administration approval of gemcitabine, cisplatin, and durvalumab in the first-line setting for BTC in September 2022.
KEYNOTE-966: a confirmatory study
KEYNOTE-966 (NCT04003636), a randomized, double-blind phase III trial, investigated the efficacy and safety of gemcitabine and cisplatin with or without pembrolizumab, a PD-1 inhibitor, in patients with treatment-naïve metastatic or unresectable BTC.92,93 Enrolling internationally, patients were randomized 1:1 to either gemcitabine, cisplatin and pembrolizumab or gemcitabine, cisplatin, and placebo. Given the cumulative toxicities, cisplatin (1000 mg/m2) was administered up to eight cycles, whereas gemcitabine (25 mg/m2) had no maximum number of cycles. Pembrolizumab (200 mg IV Q3 weeks) could be administered for up to 35 cycles, 2 years in total. Patients were stratified by site of origin, disease stage, and geographic region (Asia
The results of KEYNOTE-966 were remarkably similar in many respects to TOPAZ-1. The median OS at final analysis was 12.7 months in the pembrolizumab cohort compared to 10.9 months in the placebo cohort (HR, 0.83; CI, 0.72–0.95,
Population subsets were also analyzed. There was a lower degree of benefit in eCCA (HR, 0.99; CI, 0.73–1.35) and GBC (HR, 0.96; CI, 0.73–1.26) compared to iCCA (HR, 0.76; CI, 0.64–0.91). The authors hypothesize that this is likely because of increased comorbidities in this patient population. Patients enrolled outside of Asia (HR, 0.8; CI, 0.67–0.96) had a slightly higher degree of benefit compared to those enrolled within Asia (HR, 0.88; CI, 0.72–1.08). Those with CPS < and ⩾1 had similar outcomes (HR, 0.84; CI, 0.62–1.14
AEs were similar between the two cohorts with 99% of patients experiencing any AE. Treatment-related AEs of grade 3–4 were noted in 70% of the pembrolizumab cohort and 69% of the placebo cohort, and treatment-related death was noted in 2% of the pembrolizumab cohort and 1% of the placebo cohort. Similar to TOPAZ-1, safety analysis was comparable to prior studies with similar rates of AEs between the cohorts. No new safety concerns were raised, again providing reassurance.
KEYNOTE-966 validates the findings of TOPAZ-1 and establishes gemcitabine, cisplatin, and pembrolizumab as another first-line option for the treatment of advanced/metastatic BTC (Table 2). Though we cannot directly compare between clinical trials, the median OS was almost 13 months in both studies and the median PFS was 1–1.5 months longer than control arms. Both studies have advanced the treatment paradigm for BTC. Intrahepatic cholangiocarcinoma patients made up the largest subset of both trials and trended toward increased benefit.
First-line treatment options for metastatic biliary tract cancer.
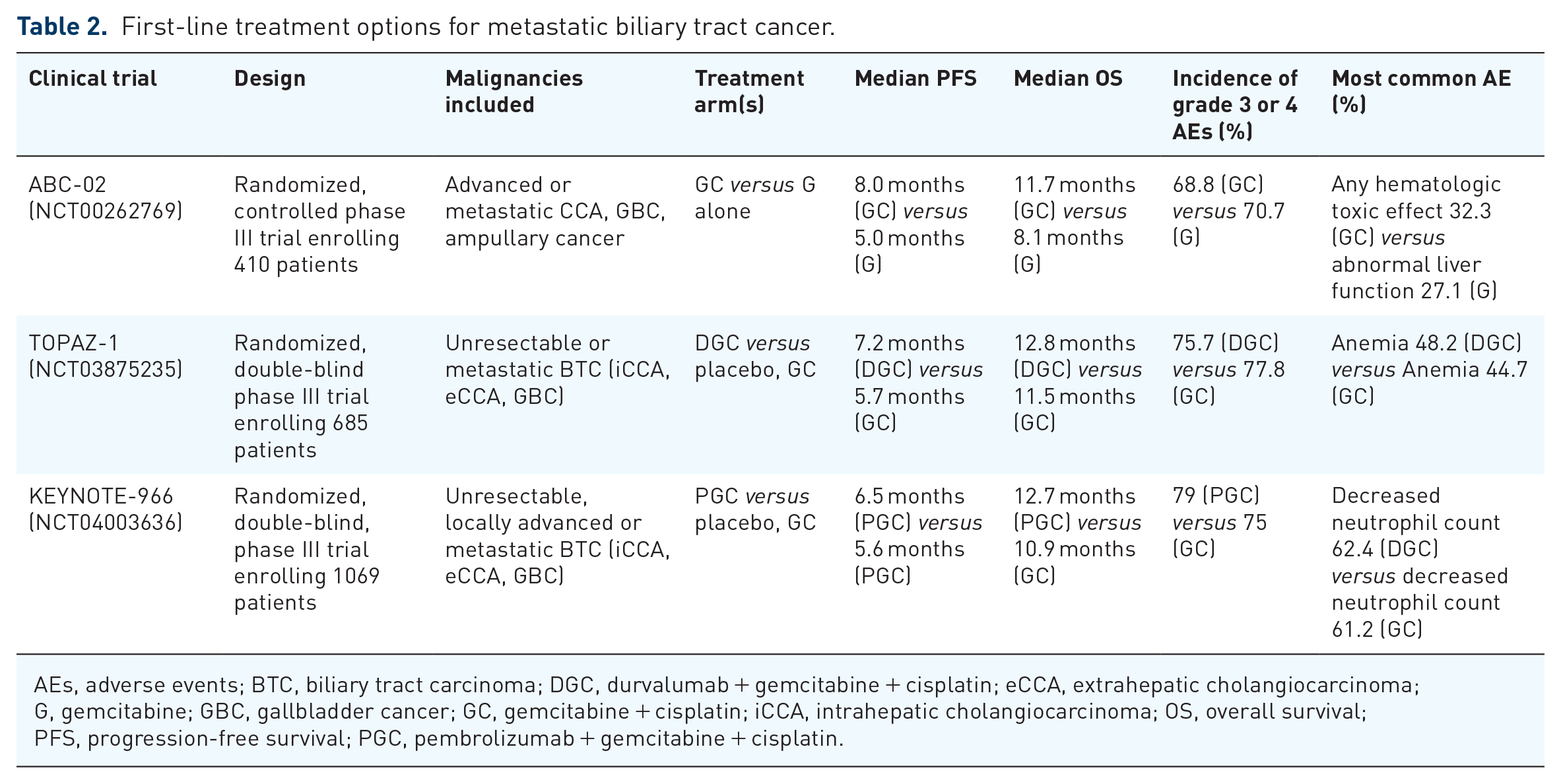
AEs, adverse events; BTC, biliary tract carcinoma; DGC, durvalumab + gemcitabine + cisplatin; eCCA, extrahepatic cholangiocarcinoma; G, gemcitabine; GBC, gallbladder cancer; GC, gemcitabine + cisplatin; iCCA, intrahepatic cholangiocarcinoma; OS, overall survival; PFS, progression-free survival; PGC, pembrolizumab + gemcitabine + cisplatin.
Yet, there are key differences between KEYNOTE-966 and TOPAZ-1. At 24 months, the median OS was 18% in the placebo group in KEYNOTE-966 and only 10.4% in the placebo group in TOPAZ-1. The continuation of gemcitabine as maintenance therapy in KEYNOTE-966 could account for this difference and support the use of gemcitabine beyond the initial 6 months of induction chemotherapy, though patient drug exposure shows that patients received gemcitabine for a median of one more month than cisplatin. KEYNOTE-966 enrolled a predominantly Western
While there has been progress in the treatment of BTC recently for the first time in over a decade, there is still progress to be made with median OS of advanced/metastatic BTC of only slightly more than 1 year. At this time, we await further analysis based on tumor mutations and circulating tumor DNA, as well as a final analysis of DOR, to deepen our understanding of this patient population.
Unanswered questions following TOPAZ-1 and KEYNOTE-966
The many questions as to the investigation of immunotherapy and timing of treatment modalities have arisen. TOPAZ-1 and KEYNOTE-966 have established the use of immunotherapy in the metastatic setting for BTC, paving the way for investigation of the use of immunotherapy in the adjuvant setting. The phase II DEBATE trial is investigating neoadjuvant gemcitabine and cisplatin with or without durvalumab followed by postoperative durvalumab for patients with localized, resectable cholangiocarcinoma and gallbladder carcinoma (NCT04308174). This trial will shed light on the use of perioperative immunotherapy for BTC. Furthermore, the timing of targeted therapy for those with a targeted molecular alteration has yet to be clearly defined. As previously mentioned, patients with a molecular target benefited from first-line durvalumab, gemcitabine, and cisplatin in the TOPAZ-1 trial. 90 Yet, the question of when to introduce targeted therapy remains. The SAFIR-ABC10 trial (NCT05615818) aims to investigate whether the introduction of a targeted therapy after four cycles of current first-line standard-of-care treatment for advanced BTC is superior to continuing standard treatment. This trial, which aims to begin in January 2024, will provide insight into this therapeutic strategy.
Novel approaches for the treatment of BTC
Currently, ongoing clinical trials are investigating novel combinatorial strategies to advance the treatment of BTC (Figure 1). Vaccine therapy in BTC is an evolving area of research. While multiple peptide vaccines specifically targeting Wilms Tumor-1 (WT-1), Mucin-1 (MUC1), or with use of multiple peptides have been investigated, clinical benefit has been limited.94–98 Recently, the addition of IO to personalized neoantigen vaccination resulted in robust neoantigen-specific T-cell reactivity and led to direct vaccine-induced tumor cell killing.
99
This human pilot study highlights the therapeutic synergy of this personalized combination that may be utilized in other tumor types moving forward. Moreover, mRNA-based vaccines are increasingly becoming regarded as a hotspot for cancer immunotherapy and have demonstrated immunogenicity in other tumor types.100–102 Huang
Furthermore, adoptive cell therapy is an immunotherapeutic strategy to augment the number, specificity, and reactivity of T cells against tumors and involves genetic modification of T cells to express chimeric antigen receptors (CAR) or tumor antigen-specific T-cell receptors. 105 This strategy has been explored in BTC using therapeutic targets of epidermal growth factor receptor (EGFR) and HER-2.106,107 In a phase I study, 19 patients received EGFR-specific CAR T cells after conditioning with nab-paclitaxel and cyclophosphamide. The DCR was 65% with a median PFS of 4 months, and the CAR T-cell infusion was well-tolerated. 106 In the phase I clinical trial evaluating HER2-specific CAR T treatment, the median PFS was 4.8 months. Of the 11 enrolled patients, 1 had a PR, and 5 achieved stable disease (SD). 107 Similar to efforts to increase immunogenicity and efficacy of vaccination, the addition of immunotherapy with anti-PD-1 blockade warrants further investigation to improve the clinical benefit of adoptive cell therapy in solid tumors. 108
Further preclinical investigation has identified potential novel strategies for improving treatment efficacy. In mouse models with mutant IDH1 cholangiocarcinoma, inhibition of IDH1 was shown to stimulate CD8+ T-cell recruitment and IFN-γ expression and led to TET2-dependent induction of IFN-γ response genes in tumor cells. Treatment resistance to IDH1 inhibition was demonstrated when CD8+ T cells were depleted or when TET2 or IFN-γ receptor 1 was ablated. CTLA4 blockade was able to overcome this immunosuppression, providing evidence that the combination of IDH1 inhibition and CTLA4 blockade is an effective strategy for this subset of cholangiocarcinoma and that both immune function and the IFN-γ–TET2 axis are necessary for response.
109
Similarly, Wu
Additional clinical trials are investigating the use of IO with epigenetic therapy (NCT03250273), poly (ADP-ribose) polymerase inhibitors (PARPi) (NCT03639935), and DKN-01, an anti-DKK1 monoclonal antibody (NCT04057365) to improve the efficacy of immunotherapeutic strategies in BTC (Table 2). Phase I clinical trials are also assessing the use of bromodomain and extra-terminal inhibitors (NCT05252390), and preclinical investigation has highlighted the therapeutic potential of an enhancer of Zeste homolog 2 inhibitor for the treatment of CCA. 111 Ongoing efforts are also in process to assess the TME of molecular subsets of patients, such as those with FGFR alternations or IDH1 mutations, as well as the utility of molecularly targeted agents to synergize with IO approaches. Stromal modulation is another area of active investigation with preclinical studies showing the therapeutic potential of focal adhesion kinase inhibitors, 112 while an early-phase clinical trial is assessing the combination of colony-stimulating factor-1 receptor and anti-PD-1 antibody (NCT04301778) in cholangiocarcinoma.
Finally, the phase II GEMINI-Hepatobiliary study is investigating the use of bispecific antibodies for treatment-naïve, advanced BTC (NCT05775159). One arm is studying volrustomig (MEDI5752), a CTLA-4/anti-PD-1 bispecific antibody, in combination with gemcitabine and cisplatin, while a second arm is investigating AZD2936, an anti-PD-1 and TIGIT bispecific antibody in combination with gemcitabine and cisplatin.
Future directions and conclusion
BTCs are a heterogeneous group of malignancies that pose a therapeutic challenge given their diversity and late stage at diagnosis. Over the past decade, the therapeutic potential of immunotherapy has become increasingly recognized within BTC with investigational approaches utilizing monotherapy, combinatorial, and synergistic approaches with chemotherapy and IO. For the first time in over a decade, advancement for the treatment of advanced or metastatic BTC in the first-line setting has been proven with the results of TOPAZ-1 leading to a median OS of 12.8 months with the combination of gemcitabine, cisplatin, and durvalumab and with the results of KEYNOTE-966 leading to a median OS of 12.7 months with combination of gemcitabine, cisplatin, and pembrolizumab. While these represent new first-line treatments, further optimization of the treatment paradigm for BTC is needed.
Given the heterogeneity of BTC and the complexity of immunotherapy-based approaches, a one-size-fits-all approach is unlikely to be successful, and future optimal treatment must prioritize individual approaches. The design of robust and translationally focused clinical trials, as well as early referral for clinical trials, is paramount to the success of the treatment of this disease. A better understanding of the TME of BTC is imperative for the development of novel and effective therapeutics. Further immune classification may help to identify which patients are likely to have robust and durable responses to IO, and a deepened understanding of the mechanisms of resistance to IO, as well as molecularly targeted agents, may lead to novel therapeutic strategies. Ultimately, further characterization and understanding of the TME, as well as novel combinatorial strategies, may improve the clinical outcomes of this devastating malignancy.
