Abstract
Introduction:
Immune checkpoint inhibitors (ICIs) have demonstrated promising efficacy as monotherapy in patients with pulmonary sarcomatoid carcinoma (PSC). We performed the current multi-institutional, real-world study to assess the efficacy of ICIs plus chemotherapy in patients with PSC.
Methods:
All consecutive patients with locally advanced or metastatic PSC from three centers treated with ICIs between January 2018 and July 2021 were enrolled. Programmed death ligand 1 (PD-L1) expression was stained and evaluated using immunohistochemical with 22C3. Single-cell RNA sequencing (scRNA-seq) was performed in two patients with PSC and two patients with adenocarcinoma to understand the cell-type-specific transcriptome landscape of cancer cells and tumor microenvironment (TME) of PSC.
Results:
A cohort of 42 PSC patients was identified. In the overall population, the objective response rate (ORR) was 73.8%, median progression-free survival (mPFS) was 10.3 months and median overall survival was not reached and 2-year survival rate was 51.2%. For 34 treatment-naïve patients who received first-line ICIs plus chemotherapy, the ORR was 70.6%, mPFS was 10.3 months and 2-year survival rate was 57.8%. In patients with PD-L1 tumor proportion score (TPS) < 1%, 1–49%, and ⩾50%, the ORR was 33.3%, 72.7%, and 85.7% and mPFS was 6.0, 6.7, and 10.3 months, respectively. Notably, two patients with transformed PSC from lung adenocarcinoma after epidermal growth factor receptor-tyrosine kinase inhibitor treatment also responded well to ICIs plus chemotherapy. scRNA-seq revealed immune-cell-inflamed TME, lower intratumoral heterogeneity, and activated immune response pathway in PSC.
Conclusions:
Our study demonstrated remarkable efficacy of ICIs plus chemotherapy as first-line therapy for patient with locally advanced or metastatic PSC.
Keywords
Introduction
Pulmonary sarcomatoid carcinoma (PSC) is a rare subset of non-small-cell lung cancer (NSCLC), characterized by poorly differentiated tumors with a component of sarcoma or sarcoma-like elements 1 and highly aggressive biological behaviors.2–5 PSC has been reported to be associated with poor prognosis and low responsiveness to first-line chemotherapy, with an objective response rate (ORR) of 16.5% and overall survival (OS) of only 6.3 months. 6 Therefore, developing novel therapeutic strategies to improve the dismal prognosis of patients with this malignance is in high demand.
Recent multi-omics analyses have revealed high programmed death ligand 1 (PD-L1) expression, high tumor mutational burden (TMB), and a T-cell-inflamed tumor microenvironment (TME) in PSC.7–9 All these features define this group most likely to respond to immune checkpoint inhibitors (ICIs). Intriguingly, a recent, multi-institutional retrospective study found that the ORR was 40.5%, progression-free survival (PFS) was 4.89 months, and OS was 12.7 months in 37 French patients with PSC who received ICI monotherapy as second- or later lines therapy. 10 Furthermore, another study conducted in Korea also elucidated the outstanding efficacy of ICIs as monotherapy in PSC, with an ORR of 49%, median PFS of 7.2 months, and median OS of 22.2 months. 11 All these findings support ICIs as a promising choice in patients with PSC.
To date, ICIs in combination with platinum-based chemotherapy have been standard of care first-line treatment in patients with metastatic NSCLC without actionable oncogenic drivers, regardless of PD-L1 expression.12,13 Although prospective studies that directly compare ICI monotherapy and ICIs plus chemotherapy in first-line setting for patients with metastatic NSCLC are not available, several network meta-analyses reported that combination therapy confers an improved ORR and PFS compared with ICI monotherapy in patients with PD-L1 tumor proportion score (TPS) ⩾ 50% tumors.14,15 Due to the impressive efficacy of ICIs plus chemotherapy in metastatic NSCLC and most of the patients with PSC have high PD-L1 expression, 11 to evaluate the efficacy of ICIs in combination with chemotherapy in PSC is of high clinical interest. Therefore, we performed the current multi-institutional real-world study to assess the efficacy of ICIs plus chemotherapy in patients with locally advanced or metastatic PSC.
Material and methods
Patients and data collection
Electronic databases of three participating cancer centers (Shanghai Pulmonary Hospital, Hunan Cancer Hospital, and Anhui Provincial Hospital) were systematically searched for patients diagnosed with PSC between January 2018 and July 2021. PSC was diagnosed according to the 2015/2021 World Health Organization classification of lung tumors.1,16 Tumor stage was determined according to the 8th edition of TNM Classification for lung cancer. 17 Only patients with locally advanced or metastatic PSC (stage IIIA/IIIB/IIIC, IV) treated with ICI-based therapy at least more than two cycles were enrolled. The electric medical records were retrospectively reviewed and detailed clinicopathologic characteristics, including gender, age, smoking history, Eastern Corporation Oncology Group performance status, diagnosis method, metastatic sites, oncogenic mutation status, regimens used in any line, and PD-L1 expression were extracted and collected. EGFR, ALK, ROS1, RET, KRAS, NRAS, PIK3CA, BRAF, MET exon 14 (METex14), and HER2 were routinely detected by multiplex real-time PCR (Amoy Diagnostics Co., Ltd, Xiamen, China). Next-generation sequencing was performed in four cases. PD-L1 expression was stained and evaluated using immunohistochemical with 22C3 pharmDx assay (Dako, Carpinteria, CA, USA). The TPS ⩾ 1% was defined as positive PD-L1 expression and TPS ⩾ 50% was defined as high PD-L1 expression.
The study was approved by the institutional review board of each participating centers and was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice.
Evaluation of therapeutic outcomes
Tumor assessments were performed by two experienced medical oncologist (FZ and YZ) per the Response Evaluation Criteria in Solid Tumors criteria 1.1, 18 independently. Any disagreements were resolved by a third senior medical oncologist (CZ). PFS was defined as the time from initiation of ICIs to documentation of disease progression or death, whichever occurred first. Patients without disease progression or death were censored. OS was defined as the time from initiation of ICIs to the date of death from any cause or last follow-up in surviving participants.
Single-cell RNA sequencing
Single-cell RNA sequencing (scRNA-seq) was performed in two patients with PSC and two patients with adenocarcinoma to understand the cell-type-specific transcriptome landscape of cancer cells and TME of PSC. Tissue dissociation and single-cell suspension preparation, scRNA-seq library preparation, and generation of single-cell gene expression matrices were performed as previously described.19,20 To assign one of the 11 major cell types to each cluster, we scored each cluster by the normalized expressions of the following canonical markers: plasma cells (CD79A, JCHAIN, MZB1, IGHG1), stromal cells (DCN, LUM, ACTA2, COL1A1), mast cells (TPSAB1, TPSB2, CPA3), monocytes (CD14, FCN1, FCGR3A, VCAN), macrophages (C1QA, MRC1, CD163), dendritic cells (CD1C, CD1E, FCER1A), B cells (MS4A1, CD79A, CD79), T cells (CD2, CD3D/E/G, TRAC, TRBC1), cancer cells (EPCAM, KRT8, CD24, SOX4), alveolar epithelial cells (SFTPB, SFTPC, ABCA3, LAMP3), and ciliated cells (FOXJ1, TPPP3, PIFO). Gene set enrichment analysis (GSEA) of both cancer cell and T cells was performed with the R package GSVA. Intratumoral heterogeneity (ITH) scores were based on copy number alterations (CNAs) (ITHCNA) and gene expressions (ITHGEX). The calculations of ITH scores were inspired by our previous study. 19 More detailed procedures have been described in the Supplemental Materials.
Statistical analysis
Chi-square test or Fisher’s exact test was used to assess the distribution of the clinicopathological variables. PFS and OS curves were estimated by the Kaplan–Meier method and compared by log-rank test. Cox proportional hazards model was performed, and hazard ratio (HR) and 95% confidence interval (CI) were calculated to determine the survival differences. All of the statistical analyses were performed with Origin (2019b, OriginLab, Northampton, MA, USA), the SPSS statistical software (version 22.0, SPSS Inc., Chicago, IL, USA), and the R package (Version 1.4.1106). Plots were generated by GraphPad Prism (version 7, GraphPad Software Inc., San Diego, CA, USA) and Origin (2019b, OriginLab, Northampton, MA, USA). Two-side p < 0.05 was considered statistically significant.
Results
Patients’ characteristics
A cohort of 42 PSC patients was identified. The baseline characteristics are listed in Table 1. The median age was 64 years (range, 49–78 years). Most patients were males (78.6%, 33/42). The majority of patients were non-smokers (54.8%, 23/42). Most patients were diagnosed with percutaneous lung biopsy samples (69.0%, 29/42) and had stage IV diseases (88.1%, 37/42). Three (7.1%) patients had liver metastases, 9 (21.4%) had brain metastases, 21 (50.0%) had bone metastases, and 11 (26.2%) had adrenal metastases.
Baseline demographics and disease characteristics of the 42 PSC patients who received ICIs.

ECOG, eastern corporation oncology group; ICI, immune checkpoint inhibitors; PD-L1, programmed death ligand 1; PSC, pulmonary sarcomatoid carcinoma.
PD-L1 expression was assessed in 36 (85.7%) patients. Among these 36 patients, PD-L1-positive tumors (TPS ⩾ 1%) were identified in 31 (86.1%) patients and 19 (52.3%) patients had high PD-L1-expressing tumors (TPS ⩾ 50%). Regarding mutational status, three patients had EGFR mutations (2 had EGFR L858R point mutations, 1 had EGFR exon 19 deletion mutation), 14 had KRAS mutations, 3 patients had METex14 skipping mutations, and 2 patients had other molecular alterations (1 had RET fusion and 1 had BRAFV600E mutation).
Six (14.3%) patients had received at least one previous systemic therapy, and 36 (85.7%) patients were treatment-naïve. In the first-line setting, two patients received ICI monotherapy, and 34 patients received ICIs in combination with chemotherapy. The detailed therapeutic regimens were listed in Supplemental Table 1.
ICI efficacy and clinical outcomes in overall cohort
The median follow-up was 16.6 months (interquartile range: 7.8–21.9 months). For the entire cohort, one patient had a complete response and 30 had a partial response. The ORR was 73.8%. Most patients (39/42, 92.9%) had a disease control and had a reduction in tumor size (Figure 1(a)). Responses were observed in both male and female patients, in patients who had brain metastases at baseline, and including in patients who had KRAS mutations, EGFR mutations, or METex14 skipping mutations. Treatment durations for each patient in the overall population are shown in Figure 1(b). The median PFS was 10.3 months (95% CI, 4.3–16.3 months) and median OS was not reached and 2-year survival rate was 51.2% [Figure 2(a) and (b)].

Best percentage change in the tumor sum-of-diameters (a) and treatment durations (b) from baseline for the overall population.
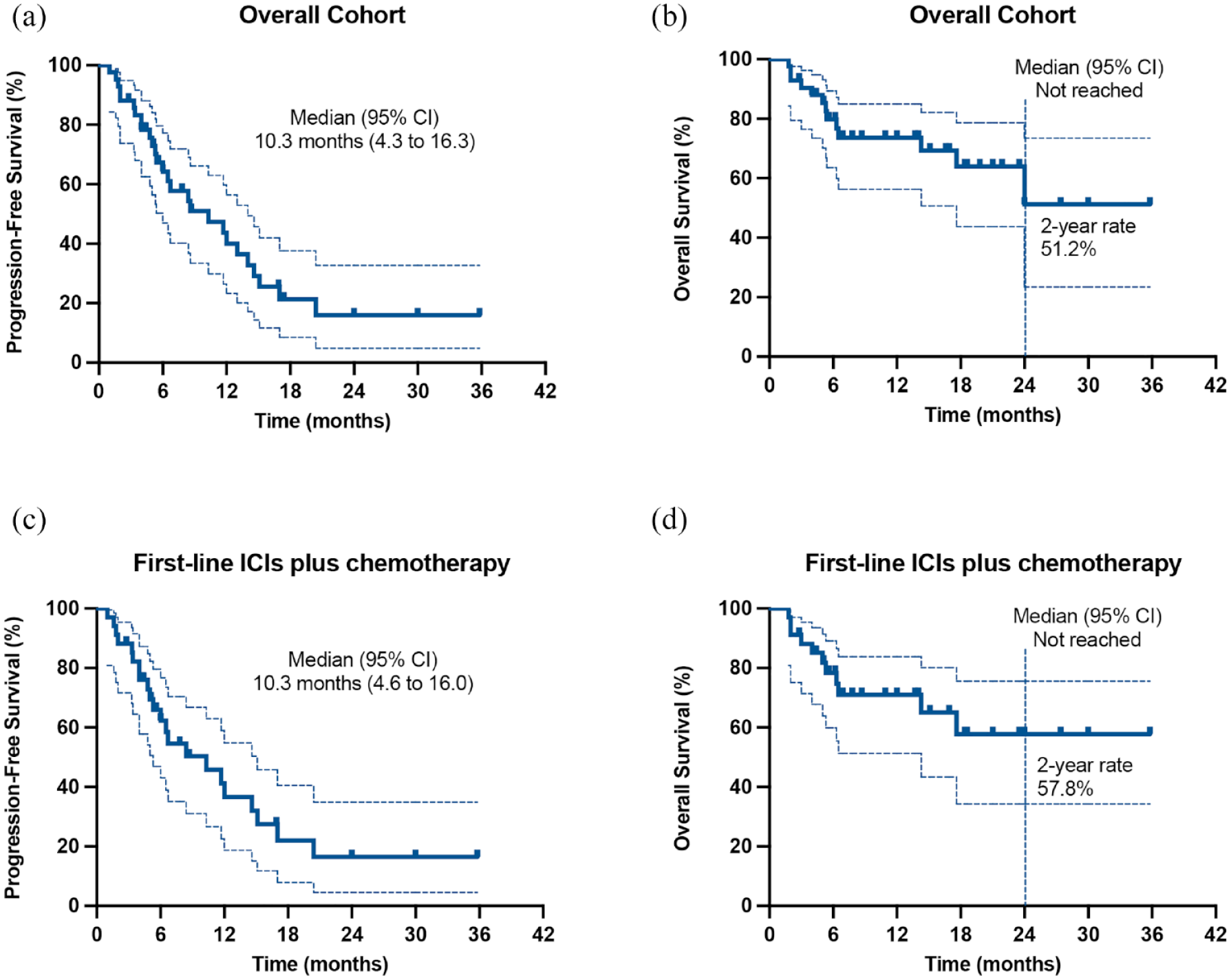
Median PFS and OS in overall population and first-line ICIs plus chemotherapy. (a) median PFS in overall population. (b) Median OS in overall population. (c) Median PFS in first-line ICIs plus Chemotherapy. (d) Median OS in first-line ICIs plus chemotherapy.
The efficacy of ICIs plus chemotherapy as first-line treatment
For 34 treatment-naïve patients who received first-line ICIs plus chemotherapy, one patient had a complete response and 23 had a partial response. Nine patients had stable disease and one patient had progressive disease. The ORR was 70.6%. The median PFS was 10.3 months (95% CI, 4.6–16.0 months) and the median OS was not reached and 2-year survival rate was 57.8% [Figures 2(c) and (d)].
For patients with PD-L1-expressing tumors (⩾1%), the ORR was 80%, median PFS was 10.3 months, and median OS was not reached. Meanwhile, for patients with PD-L1-negative (<1%) or unknown tumors, the ORR was 44.4%, median PFS was 6.0 months (PD-L1-positive versus negative/unknown: HR, 0.72, 95% CI, 0.24–2.16, p = 0.493), and median OS was 14.3 months (PD-L1-positive versus negative/unknown: HR, 0.73, 95% CI, 0.17–3.08, p = 0.633). When using PD-L1 TPS 1% and 49% as cutoffs, the ORR was 33.3%, 72.7%, and 85.7% in patients with PD-L1 TPS <1% population, PD-L1 TPS 1–49% population, and PD-L1 TPS ⩾50% population. While the median PFS was 6.0, 6.7, and 10.3 months (p = 0.861), respectively (Figure 3(a)). The median OS was not reached, not reached, and not reached (p = 0.510), respectively (Figure 3(b)).
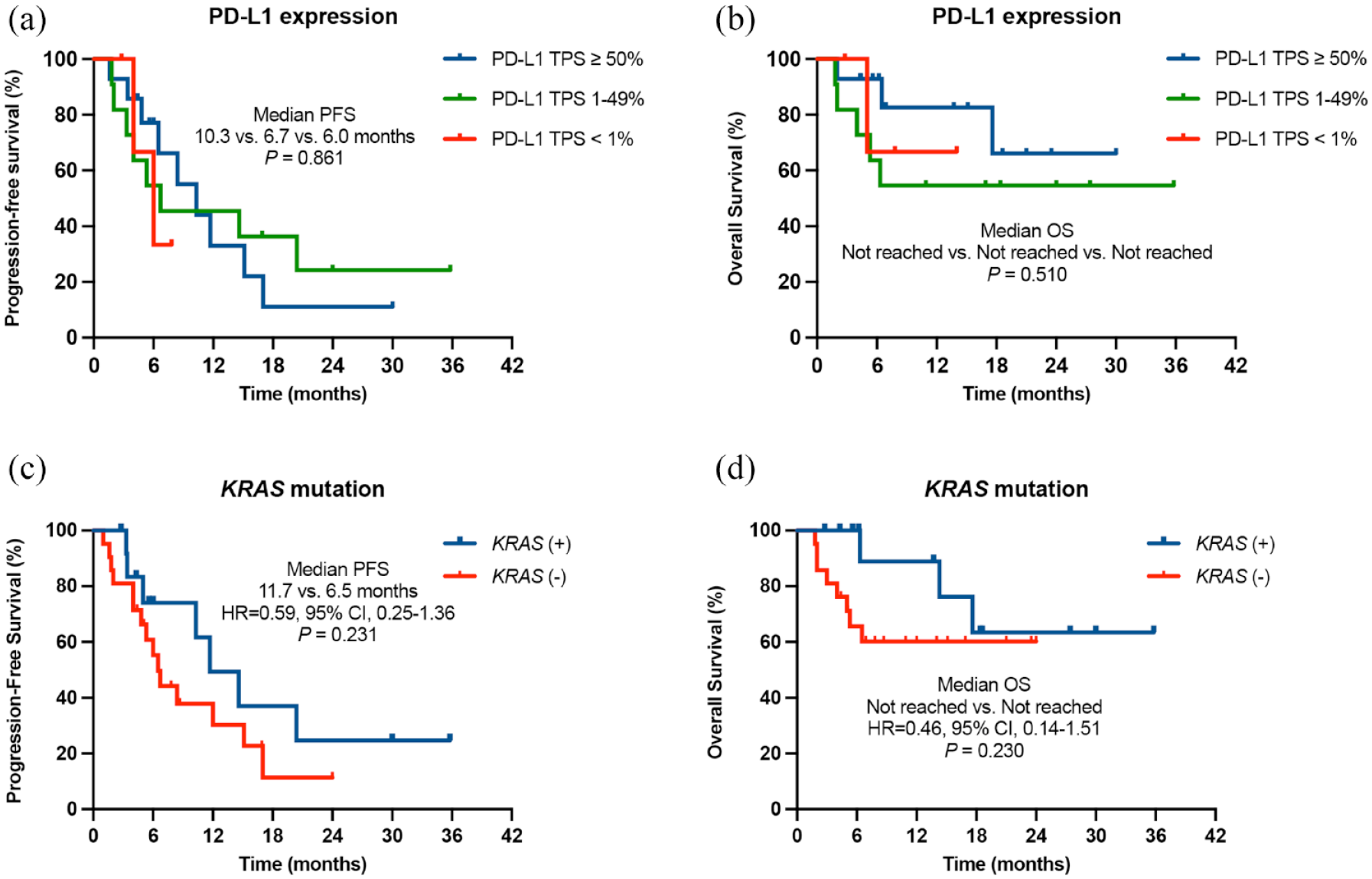
Median PFS and OS in first-line ICIs plus chemotherapy according to PD-L1 expression (a, b) and KRAS mutations (c, d).
In terms of mutational status, the median PFS was 11.7 months versus 6.5 months (HR, 0.59, 95% CI, 0.25–1.36, p = 0.231) in patients with and without KRAS mutations (Figure 3(c)). The median OS was not reached versus not reached (HR, 0.46, 95% CI, 0.14–1.51, p = 0.230) (Figure 3(d)) while the ORR was 76.9% versus 66.7%, respectively. Notably, in three patients harboring METex14 skipping mutations, two patients received pembrolizumab in combination with chemotherapy as first-line treatment (one had PD-L1 TPS 50% tumor and one had PD-L1-unknown tumor) and one patient received nivolumab monotherapy as fourth-line treatment (PD-L1 TPS <1%). Intriguingly, both of these two patients who received combination therapy in first-line therapy had a partial response and the treatment was still going on at the time of this article submission, 4.4 and 8.8 months after the treatment was initiated (Figure 1(b)).
ICIs plus chemotherapy in PSC transformation from lung adenocarcinoma after epidermal growth factor receptor-tyrosine kinase inhibitor treatment
In addition, in our cohort, two patients with EGFR sensitizing mutations (1 had EGFR L858R and 1 had EGFR exon 19 deletion) developed histologic transformation from adenocarcinoma to PSC after failure on previous epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI) treatment in subsequent biopsy (median PFS of previous EGFR-TKI treatment was 10.4 and 10.5 months, respectively) (Figure 4). Both of these two patients had PD-L1-positive tumors (PD-L1 TPS 25% and PD-L1 TPS 85%) after transformation and received ICIs in combination with chemotherapy as second-line therapy (Figure 4). Notably, they all responded well to the combination therapy and had a partial response (Figure 4). Furthermore, the response was durable and the median PFS was 8.6 and 14.0 months, respectively, and both were alive at the last follow-up (the median OS was 16.6 and 21.9 months, respectively).
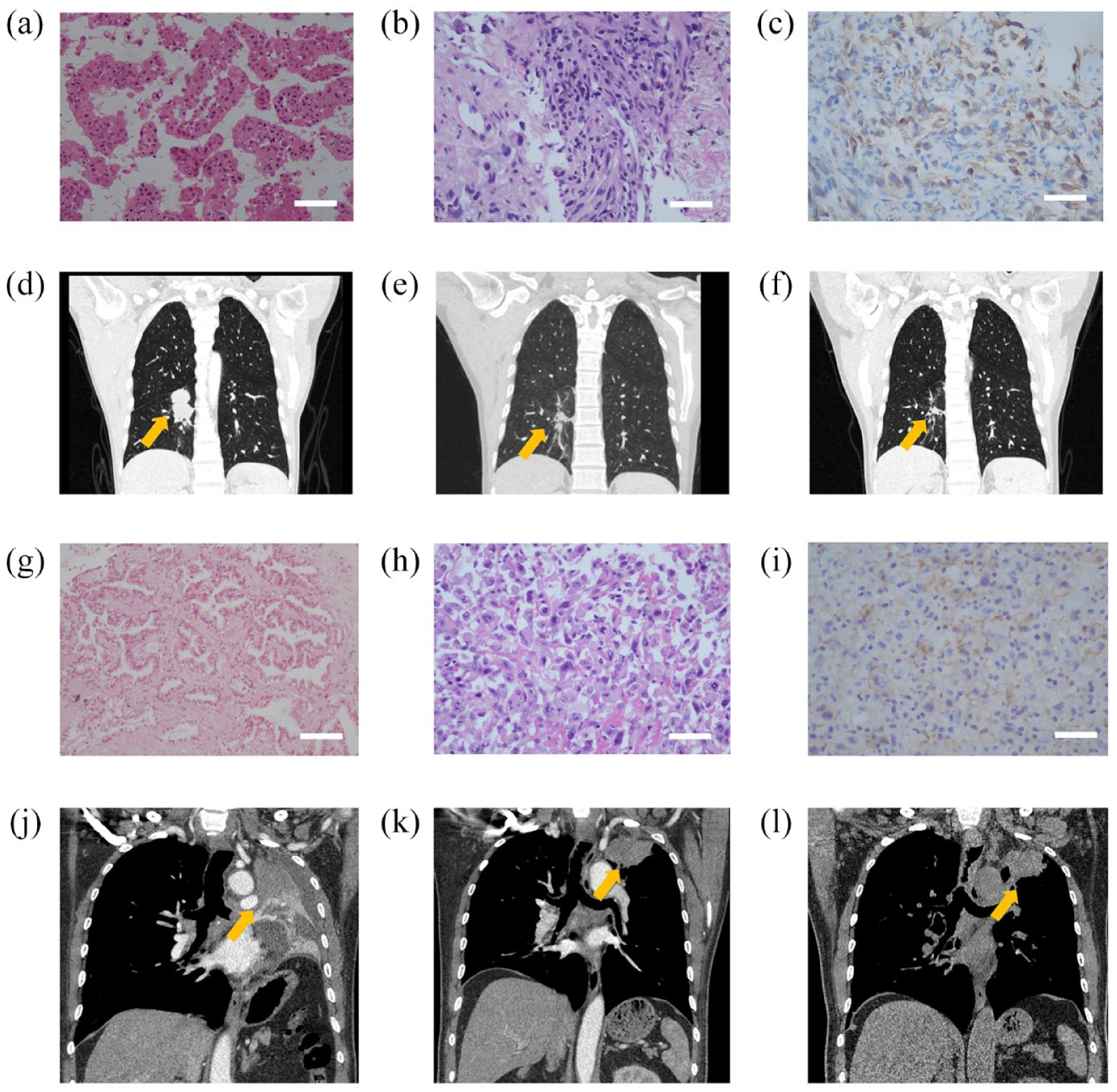
Two cases of ICIs plus chemotherapy in PSC transformation from lung adenocarcinoma after EGFR-TKI treatment. (a, g) HE staining of patients before administration of EGFR-TKI. (b, h) HE staining of patients after failure on EGFR-TKI treatment. (c, i) PD-L1 expression of patients after failure on EGFR-TKI treatment. (d, j) CT images before patients received ICIs plus platinum-based chemotherapy. (e, f) CT images of patient 1 after 6 and 10 months of treatment. (k, l) CT images of patient 2 after 3.6 and 8 months of treatment.
scRNA-seq of PSC
After quality control and filtering steps, a total of 5064 cells were analyzed. In all, 11 major cell types were detected by leveraging canonical cell markers, including plasma cells, stromal cells, mast cells, monocytes, macrophages, dendritic cells, B cells, T cells, cancer cells, alveolar epithelial cells, and ciliated cells. Interestingly, the proportions of cancer cells in PSC were lower than that in adenocarcinoma (Figure 5(a)). On the contrary, immune cells, including macrophages, monocytes, B cells, and dendritic cells, were enriched in PSC (Figure 5(a)). To further classify subgroups of immune cell types, six subtypes were identified according to canonical cell markers, including naïve T cells, CD8+ effector T cells, regulatory T cells, proliferating CD8+ effector T cells, macrophages and neutrophils. Again, although the distribution of immune cells was similar between PSC and adenocarcinoma, PSC had more immune cells in the TME (Figure 5(b)). Regarding ITH, PSC patients had numerically lower ITHCNA and ITHGEX scores compared to adenocarcinoma, although formal statistical analyses cannot be performed due to the sample size (Figure 5(c)). GSEA of cancer cells revealed the top 5 upregulated pathways in PSC (Figure 5(d)). Furthermore, antigen receptor-mediated signaling pathway was upregulated in PSC while basement membrane organization pathway was upregulated in adenocarcinoma (Figure 5(e)). GSEA of T cells, including naïve T cells, CD8+ effector T cells, proliferating CD8+ effector T cells, and regulatory T cells, revealed that antigen processing and presentation pathway and cytokine–cytokine receptor interaction pathway were the most common upregulated pathways in PSC (Figure 5(f)). GO analyses of T cells demonstrated that activation of immune response, adaptive immune response, or antigen receptor-mediated signaling pathway was upregulated in PSC (Figure 5(g)).

scRNA-seq of PSC. (a) UMAP plot of 5064 cells from two patients with PSC and two patients with adenocarcinoma and major cell-type composition of each patient. (b) UMAP plot of immune cells and cell-type composition of each patient. (c) ITH scores based on CNAs (ITHCNA) and gene expressions (ITHGEX). (d) The top 5 enriched pathway of GSEA of cancer cells. (e) Antigen receptor-mediated signaling pathway was upregulated in PSC while basement membrane organization pathway was upregulated in adenocarcinoma. (f). The top 5 enriched pathway of GSEA of T cells, including naïve T cells, CD8+ effector T cells, proliferating CD8+ effector T cells and regulatory T cells and (g). GO analyses of T cells.
Discussion
The present, multi-institutional, real-world study demonstrated a profound efficacy of ICIs in patient with locally advanced or metastatic PSC. The overall ORR was 73.8%, median PFS was 10.3 months, and 2-year survival rate was 51.2%. In contrast to previous studies that evaluated ICI monotherapy in patient who were previously treated with platinum-based chemotherapy, 85.7% (36/42) of patients were treatment-naïve in our study. Intriguingly, in 34 treatment-naïve patients who received first-line ICIs plus chemotherapy, the ORR was 70.6%, median PFS was 10.3 months, and 2-year survival rate was 57.8%. Although the findings were attenuated due to its retrospective nature and small sample, our study, to the best of our knowledge, revealed encouraging efficacy of first-line ICIs in combination with chemotherapy in patients with locally advanced or metastatic PSC.
Due to the rarity of this malignance, prospective study that investigated the efficacy of ICIs in PSC is scarce. Two retrospective studies from French and Korea both demonstrated good efficacy of ICI monotherapy for PSC and revealed a positive correlation between PD-L1 expression and survival outcomes.10,11 Babacan et al. 21 performed a pooled analysis of published literature and five patients at Moffitt Cancer Center to investigate the efficacy of ICIs in patients with PSC. In 90 patients treated with ICI monotherapy, the ORR was 54.5%, and median PFS was 7.0 months. The positive PD-L1 expression rate was 90.4% (59/66). In patients with PD-L1-positive tumors, the ORR was 66.7% and median PFS was 14.4 months while the ORR was 28.6% and median PFS was 2.7 months in patients with PD-L1-negative tumors 21 . KCSG-LU16-07 was phase II study that investigated the efficacy and safety of durvalumab and tremelimumab for recurrent or metastatic PSC. 22 The ORR was 26.7%, median PFS was 5.9 months, and median OS was 15.4 months. 22 Taken together, all these studies elucidated good efficacy of ICI monotherapy or combination therapy for PSC and a positive correlation between PD-L1 expression and tumor response was also observed, despite the overall high PD-L1 expression in PSC.
On the basis of remarkable efficacy of ICI monotherapy in previously treated PSC, the efficacy of first-line ICIs in combination with chemotherapy in treatment-naïve PSC patients in our study may be not beyond expectation. In the all-comer trials of first-line ICIs in combination with chemotherapy in patients with lung adenocarcinoma and squamous cell carcinoma, the ORR ranges from 47.6% to 74.8%12,13,23,24 and median PFS ranges from 5.1 to 11.3 months.12,13,23,24 Overall, the response rate was higher and median PFS was longer in patients with high PD-L1-expressing tumors. Therefore, the high PD-L1 expression in PSC may, at least partially, led to the outstanding efficacy of ICIs plus chemotherapy in our study. Furthermore, immune-cell-inflamed TME, lower ITH scores and activation of immune response pathway in PSC, which have also been observed by scRNA-seq in our study, may also contribute to the good respond to ICI therapy.
Our study also observed promising efficacy of ICIs plus chemotherapy in PSC patient with METex14 skipping mutations. Previous studies have shown limited efficacy of ICI monotherapy in advanced NSCLC patients with METex14 skipping mutations.25,26 Recently, three highly selective MET inhibitors, namely tepotinib, capmatinib, and savolitinib, have demonstrated substantial antitumor activity in advanced NSCLC with METex14 skipping mutations,27–29 particularly in treatment-naïve patients. 28 In the future, as these three MET inhibitors have been approved for patients with METex14 skipping mutations, the role of ICIs plus chemotherapy deserves to be investigated, especially in those who have failed on MET-TKI therapy.
Another intriguing finding was that ICIs plus chemotherapy was effective in patients with transformed PSC from lung adenocarcinoma after EGFR-TKI treatment. Histologic transformation has been identified as an acquired resistance mechanism of EGFR-TKIs and lung adenocarcinoma with PSC transformation after EGFR-TKI treatment has also been observed in clinical cases. 30 The prognosis for patients with PSC transformation was poor after histologic transformation, with a median OS of 2.5 months in a previous study. 30 Notably, in their reported cases, one patient after PSC transformation received chemotherapy plus immunotherapy and the survival time was 16 months. 30 These findings also supported ICIs plus chemotherapy as a potential treatment option for patients with transformed PSC after EGFR-TKI treatment.
Our study has several limitations. First, this was a retrospective and small sample study, owing to the rarity of the disease. Furthermore, varied regimens, including different PD-1 antibodies and chemotherapy drugs, were included in the current study. Therefore, these findings, including scRNA-seq results, should be interpreted with caution and larger sample study is needed to verify these findings. Second, most of the cases were diagnosed with biopsy samples at advanced clinical stage, which could not allow specific PSC subtypes classification. Third, although we tried to adopt scRNA-seq to map the TME of PSC, the sample size was very limited due to the rarity of the disease and difficult diagnosis in pathology. Finally, longer follow-up was needed to confirm the OS benefits of ICIs in combination with chemotherapy in PSC.
In conclusion, the present, multi-institutional, real-world study demonstrated remarkable efficacy of ICIs in combination with chemotherapy as first-line therapy for patient with locally advanced or metastatic PSC. We have previously initiated a prospective, single-arm study to assess the role of camrelizuamb as second-line therapy after platinum-based chemotherapy in patients with locally advanced or metastatic PSC (ChiCTR2000031478). On the basis of these findings, we have amended the trial protocol to prospectively investigate the efficacy of first-line camrelizuamb plus chemotherapy in patients with locally advanced or metastatic PSC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221136759 – Supplemental material for Immune checkpoint inhibitors plus chemotherapy in patients with locally advanced or metastatic pulmonary sarcomatoid carcinoma: a multicentric real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359221136759 for Immune checkpoint inhibitors plus chemotherapy in patients with locally advanced or metastatic pulmonary sarcomatoid carcinoma: a multicentric real-world study by Fei Zhou, Haoyue Guo, Xiaolong Zhou, Huikang Xie, Tian Tian, Wencheng Zhao, Guanghui Gao, Anwen Xiong, Lei Wang, Wei Li, Xiaoxia Chen, Yan Zhang, Jue Fan, Fengying Wu, Yongchang Zhang and Caicun Zhou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359221136759 – Supplemental material for Immune checkpoint inhibitors plus chemotherapy in patients with locally advanced or metastatic pulmonary sarcomatoid carcinoma: a multicentric real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359221136759 for Immune checkpoint inhibitors plus chemotherapy in patients with locally advanced or metastatic pulmonary sarcomatoid carcinoma: a multicentric real-world study by Fei Zhou, Haoyue Guo, Xiaolong Zhou, Huikang Xie, Tian Tian, Wencheng Zhao, Guanghui Gao, Anwen Xiong, Lei Wang, Wei Li, Xiaoxia Chen, Yan Zhang, Jue Fan, Fengying Wu, Yongchang Zhang and Caicun Zhou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
