Abstract
Background:
Immunotherapy is an emerging antitumor therapy that can improve the survival of patients with advanced non-small-cell lung cancer (NSCLC). However, only about 20% of NSCLC patients can benefit from this treatment. At present, whether patients with driving gene-positive NSCLC can benefit from immunotherapy is one of the hot issues. Therefore, we conducted a meta-analysis to evaluate the efficacy of immunotherapy in patients with oncogene-driven NSCLC and concluded the efficacy of altered subtypes.
Methods:
A literature search was performed using PubMed, Web of Science, and Cochrane databases. The primary endpoints included the objective response rate (ORR), median progression-free survival (mPFS), and median overall survival (mOS) in patients with oncogene-driven NSCLC.
Results:
In all, 86 studies involving 4524 patients with oncogene-driven NSCLC were included in this meta-analysis. The pooled ORRs in clinical trials treated with monoimmunotherapy of EGFR, ALK, and KRAS alteration were 6%, 0%, and 23%, respectively. In retrospective studies, the pooled ORRs of EGFR, ALK, KRAS, BRAF, MET, HER2, RET, and ROS1 alteration were 8%, 3%, 28%, 24%, 23%, 14%, 7%, and 8%, respectively. Among them, the pooled ORRs of KRAS non-G12C mutation, KRAS G12C mutation, BRAF V600E mutation, BRAF non-V600E mutation, MET-exon 14 skipping, and MET-amplification were 33% 40%, 20%, 34%, 17%, and 60%, respectively. In addition, the pooled mPFS rates of EGFR, KRAS, MET, HER2, and RET alteration were 2.77, 3.24, 2.48, 2.31, and 2.68 months, while the pooled mOS rates of EGFR and KRAS alteration were 9.98 and 12.29 months, respectively. In prospective data concerning EGFR mutation, the pooled ORR and mPFS treated with chemo-immunotherapy (IC) reached 38% and 6.20 months, while 58% and 8.48 months with chemo-immunotherapy plus anti-angiogenesis therapy (ICA). Moreover, the pooled mPFS and mOS of monoimmunotherapy was 2.33 months and 12.43 months.
Conclusions:
EGFR-, ALK-, HER2-, RET-, and ROS1-altered NSCLC patients have poor reactivity to monoimmunotherapy but the efficacy of immune-based combined therapy is significantly improved. KRAS G12C mutation, BRAF non-V600E mutation, and MET amplification have better responses to immunotherapy, and more prospective studies are needed for further research.
Plain Language Summary
Immunotherapy is an emerging antitumor therapy that can improve the survival of patients with advanced NSCLC. However, only about 20% of NSCLC patients can benefit from this treatment. At present, whether patients with driving gene positive NSCLC can benefit from immunotherapy is one of the hot issues. Therefore, we conducted a meta-analysis to evaluate the efficacy of immunotherapy in patients with oncogene-driven NSCLC, and concluded the efficacy of altered subtypes. 86 studies involving 4524 patients with oncogene-driven NSCLC were included in this meta-analysis. The pooled ORR in clinical trials treated with monoimmunotherapy was of EGFR, ALK and KRAS alteration was 6%, 0%, and 23%, respectively. While in retrospective studies, the pooled ORR of EGFR, ALK, KRAS, BRAF, MET, HER2, RET and ROS1 alteration was 8%, 3%, 28%, 24%, 23%, 14%, 7% and 8%, respectively. Among them, the pooled ORR of KRAS non-G12C mutation, KRAS G12C mutation, BRAF V600E mutation, BRAF non-V600E mutation, MET-exon 14 skipping and MET-amplification was 33% 40%, 20%, 34%, 17% and 60%, respectively. Additionally, the pooled mPFS of EGFR, KRAS, MET, HER2 and RET alteration was 2.77, 3.24, 2.48, 2.31 and 2.68 months, while the pooled mOS of EGFR and KRAS alteration was 9.98 and 12.29 months. In prospective data concerning EGFR mutation, the pooled ORR and mPFS treated with chemo-immunotherapy (IC) was reached 38% and 6.20 months, while 58% and 8.48 months with chemo-immunotherapy plus anti-angiogenesis therapy (ICA). Moreover, the pooled mPFS and mOS of monoimmunotherapy was 2.33 months and 12.43 months. EGFR, ALK, HER2, RET and ROS1-altered NSCLC patients have poor reactivity to monoimmunotherapy, but the efficacy of immune-based combined therapy is significantly improved. KRAS G12C mutation, BRAF non-V600E mutation and MET amplification have better response to immunotherapy, and more prospective studies are needed for further research.
Introduction
Lung cancer is the leading cause of cancer death and is also the main cause of cancer morbidity and mortality in men. In women, the incidence of lung cancer ranks third, second only to breast cancer and colorectal cancer, and the mortality rate is second only to breast cancer. 1 WHO divides lung cancer into two broad histological subtypes: non-small-cell lung cancer (NSCLC) and small-cell lung cancer. The former is the cause of about 85% of cases, and the latter accounts for the remaining 15%. NSCLC is further subdivided into adenocarcinoma, squamous cell carcinoma, and large-cell carcinoma. 2 In the whole population, the proportion of Kirsten rat sarcoma viral oncogene homolog (KRAS) mutation is about 25–30%, which is also much higher than other types of mutations. The most common type of mutation in KRAS is G12C, accounting for about 13% of non-small-cell lung cancer. 3 Epidermal growth factor receptor (EGFR) mutation and anaplastic lymphoma kinase (ALK) fusion have been found in approximately 20% and 5%, respectively, in advanced NSCLC. Somatic activating v-raf murine sarcoma viral oncogene homolog B1 (BRAF) V600E, human epidermal growth factor receptor 2 (HER2), proto-oncogene receptor tyrosine kinase (ROS1), and rearranged during transfection proto-oncogene (RET) alterations all occurred less than 5% of patients with NSCLC. Mesenchymal to epithelial transition factor (MET) mutations occurred in 2–4% population, and the most common subtype is MET-exon 14 skipping. 4 The development and use of targeted drugs for these specific changes have improved the results of patients with NSCLC harboring oncogene changes. However, the continuous emergence of drug resistance has led to many problems in follow-up treatment, which need to be further solved. Immunotherapy is an emerging antitumor therapy that can improve the survival of patients with advanced NSCLC. However, only about 20% of NSCLC patients can benefit from this treatment. 5 Programmed cell death protein 1 (PD-1)/programmed cell death ligand (PD-L1) drugs such as nivolumab, pembrolizumab, and atezolizumab play a more and more important role in the treatment of NSCLC. Some phase III clinical trials have shown that immune checkpoint inhibitors (ICIs) can significantly prolong the overall survival (OS) of patients with driver-negative advanced NSCLC compared with chemotherapy, either alone or in combination.6–8 However, there are few clinical trials to study whether ICIs are more effective than tyrosine kinase inhibitors (TKIs) or chemotherapy in patients with driving gene-positive NSCLC. The overall therapeutic effect of ICIs is not satisfactory in patients with drug-resistant advanced NSCLC. The data show that immunotherapy can benefit some NSCLC patients with positive driving genes, improve the remission rate, and prolong the survival time of patients.9–11 However, the results of Checkmate 057, OAK, POPLAR, and KEYNOTE-010 studies showed that there was no significant survival benefit in the immunotherapy group compared with the chemotherapy group in patients with EGFR mutant NSCLC.12–15 At present, whether patients with driving gene-positive NSCLC can benefit from immunotherapy is one of the hot issues. Therefore, we analyzed the efficacy of immunotherapy in patients with oncogene-driven NSCLC and concluded the efficacy of altered subtypes. In addition, the efficacy of immuno-combination therapy in different driven genes after using immunotherapy was also deep-dived. This meta-analysis was reported by the PRISMA reporting checklist (Supplemental Tables S1 and S2). 16
Methods
Search strategy and selection criteria
A literature search was performed using PubMed, Web of Science, and Cochrane databases to identify prospective clinical trials and retrospective studies of NSCLC patients treated with ICIs alone or in combination from 2015 to 2022. The following search terms were used: ‘(immune checkpoint inhibitor or immunotherapy or programmed cell death protein 1 or PD-1 or programmed death ligand-1 or PD-L1 or cytotoxic T-lymphocyte-associated protein 4 or CTLA-4 or nivolumab or pembrolizumab or camrelizumab or sintilimab or tislelizumab or toripalimab or cemiplimab or durvalumab or atezolizumab or sugemalimab or avelumab or ipilimumab or tremelimumab or dostarlimab or relatlimab or penpulimab or cadonilimab or serplulimab or sugemalimab or envafolimab or zimberelimab or pucotenlimab or prolgolimab)’ and ‘(non-small cell lung cancer or NSCLC)’ and ‘(EGFR or KRAS or ALK or BRAF or MET or HER2 or ERBB2 or RET or ROS1)’ (Supplemental Table S3). The search was conducted in March 2023.
Study selection
We first defined inclusion and exclusion criteria. The inclusion criteria were as follows: (1) prospective or retrospective observational studies; (2) articles involving patients treated with ICIs alone or in combination; and (3) the study endpoints included objective response rate (ORR), progression-free survival (PFS), or OS. The exclusion criteria were as follows: (1) case reports, reviews, editorials, meta-analyses, and letters; (2) studies that did not focus on any of the abovementioned endpoints; and (3) studies that were not published in English. Two reviewers (J.Chen and W.Lu) evaluated the titles and abstracts of publications identified by the search strategy, and any publication thought to be potentially relevant was retrieved in full. The reviewers then assessed full publications for eligibility. Reviewers were not blinded to study authors or outcomes. The decision to include a study for review was made by consensus between the reviewers (J.Chen and W.Lu). The plan was that disagreements would be resolved by the third author (M.Chen) but none occurred.
Data extraction
Data were extracted by paired reviewers (J.Chen and W.Lu). Disagreements were resolved by consensus. Data extracted included Gene mutation type, ICI class, year of publication, and the presence of at least one measure of activity (ORR, PFS, and OS). Data were extracted from the main text and Supplemental Material.
Study objectives
The main purpose of this study was to investigate the efficacy of immunotherapy in patients with NSCLC with different alterations. Associations between efficacy and gene-altered subtypes were also explored.
Statistical analysis
Descriptive statistics were used to summarize the characteristics data of patients with NSCLC. The main results were summed in a table and a quantitative synthesis was planned for all the reported cases. We performed the random-effects model to pool results to estimate the efficacy of the treatment, and statistical heterogeneity was assessed using the I² test in the random-effects model. When the average ORR of all samples was between 20% and 80%, no transformation was required. When it was less than 20% or over 80%, logit transformation was used. Freeman–Tukey double arcsine transformation was used when there were a large number of values of 0 or 100%. Publication bias was evaluated with the funnel plot asymmetry test. The data analysis was performed using R-Studio (Version 4.2.1, R Studio Team, R Studio Inc. Boston, MA, USA), Stata (Version 17, STATA Corp, College Station, TX, USA), and Microsoft 2016 (Microsoft Corporation, Redmond, Washington, DC, USA).
Results
Results of the systematic search
Our search strategies in ClinicalTrials.gov, PubMed, Web of Science, and Cochrane databases identified a total of 4628 titles. In total, 3378 of them were excluded because of duplication. Therefore, 1250 articles were evaluated by title and abstract, and 918 of them were excluded because of review or meta-analysis. Among the remaining 332 articles, 246 studies were excluded (n = 84 non-relevant subgroup or post-hoc analysis; n = 136 trial design or protocol publication; n = 12 no related outcomes; n = 14 no common treatment). In all, 86 studies were therefore included in the qualitative analysis: 25 contain prospective data from clinical trials, whereas 61 report the outcomes of retrospective studies (Figure 1). There are few prospective clinical studies on immunotherapy of gene mutant-positive NSCLC directly, and most of them are collected from subgroups. Unfortunately, significant heterogeneity was observed in almost all subgroups, which may be attributable to the specificity of the single-arm study. Therefore, the random-effect model was adopted. Concerning retrospective data, the large majority of studies reported outcomes from single-agent anti-PD-1/PD-L1 treatments, while a minority of patients had been exposed to combinations, the ICI evaluated has always been reported.
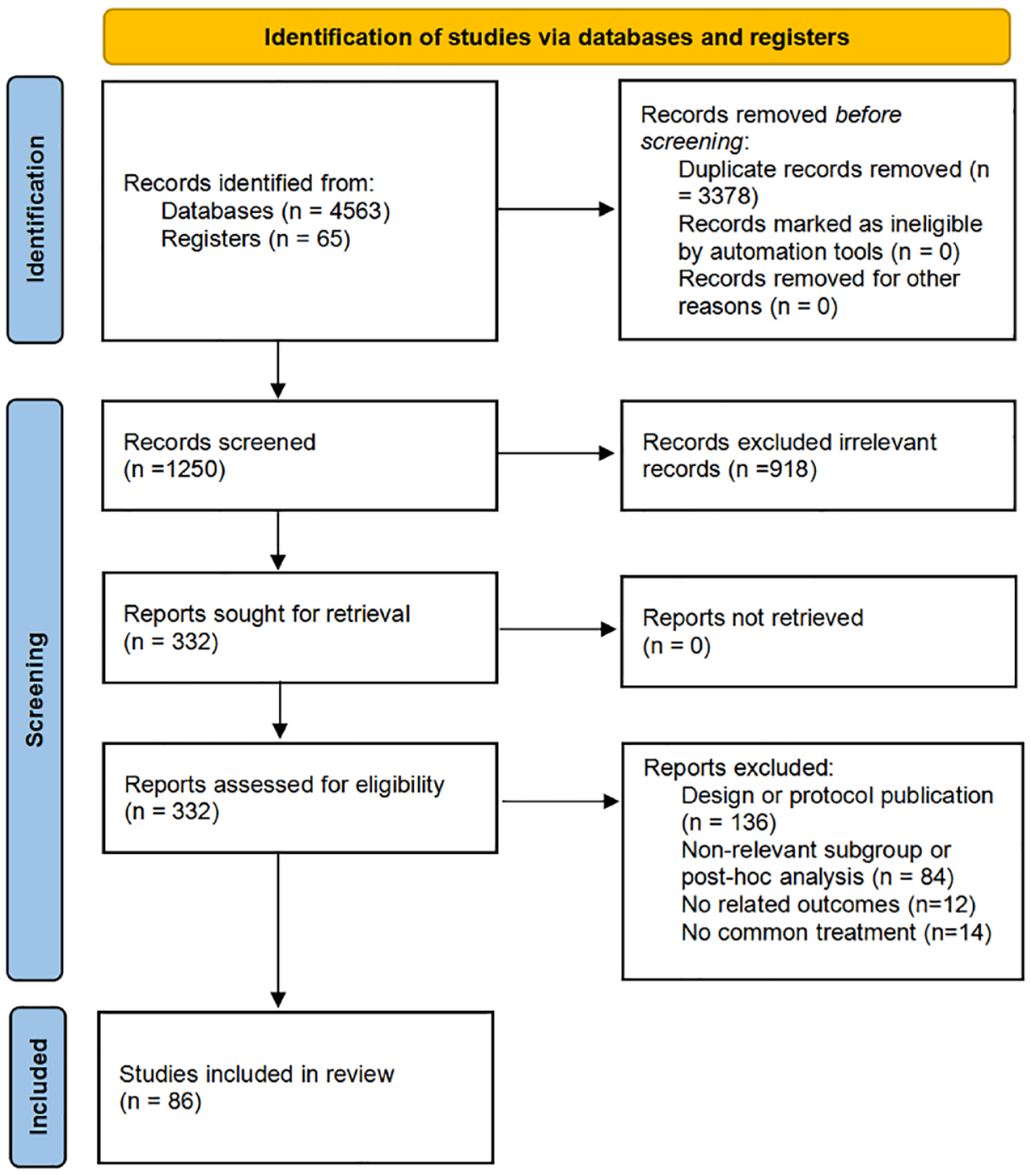
Flowchart diagram of the literature search and study selection.
EGFR-mutant NSCLC
There are 23 clinical trials and 37 retrospective studies in patients with EGFR-mutant-positive NSCLC based on immunotherapy, and most data of them are from subgroups (Tables 1 and 2)9,11,17–74. When pooling activity data in the meta-analysis indeed, the pooled ORR of EGFR-mutant patients treated with immunotherapy in clinical trials was 6% (95% CI: 3–9, I² = 0%), while 8% (95% CI: 6–11, I² = 43%) in retrospective studies (Figure 2). And the pooled ORR was 49% (95% CI: 40–57, I² = 63%) of patients treated with immuno-combination therapy in clinical trials. Among combined therapies, the pooled ORRs of chemoimmunotherapy(IC) and chemoimmunotherapy plus anti-angiogenesis therapy (ICA) were reached 38% (95% CI: 29–48, I² = 48%) and 58% (95% CI: 46–70, I² = 75%), respectively. In addition, the pooled mPFS and mOS of immunotherapy were 2.33 months (95% CI: 1.67–2.98, I² = 72.6%) and 12.43 months (95% CI: 6.43–18.43, I² = 84.3%) in clinical trials, while 2.77 months (95% CI: 1.91–3.62, I² = 95.6%) and 9.98 months (95% CI: 6.58–13.39, I² = 59.7%) in retrospective studies (Figure 3). Moreover, the pooled mPFS and mOS of immuno-combination therapy were 6.99 months (95% CI: 5.89–8.09, I² = 59.3%) and 20.74 months (95% CI: 15.00–26.49, I² = 63.2%), respectively. And among immuno-combination therapy, the pooled mPFS of IC and ICA were 6.20 months (95% CI: 5.32–7.08, I² = 19.4%) and 8.48 months (95% CI: 6.40–10.56, I² = 56.3%), respectively. The data concerning subtypes of EGFR mutation were few, and a retrospective study conducted by Hastings’s team showed that L861Q received the worst efficacy (ORR = 0%, mPFS = 1.3 months) and G719 for the best (ORR = 29%, mPFS = 4.8 months, mOS = 29 months).
Clinical activity of immunotherapy in EGFR-altered NSCLC patients of prospective study.

AE, adverse event; EGFR, Epidermal growth factor receptor; I, monoimmunotherapy; II, double immunotherapy; IC, chemoimmunotherapy; ICA, immunotherapy plus chemotherapy and anti-angiogenesis therapy; mOS, median overall survival; mPFS, median progression-free survival; NA, not available; NR, not reach; ORR, objective response rate.
32 patients were estimated for efficacy and 40 patients were estimated for safety.
Clinical activity of immunotherapy in EGFR-altered NSCLC patients of retrospective study.

EGFR, Epidermal growth factor receptor; I, monoimmunotherapy; IC, chemoimmunotherapy; mOS, median overall survival; mPFS, median progression-free survival; NA, not available; NR, not reach; NSCLC, non-small-cell lung cancer; ORR, objective response rate.

ORR of EGFR-mutant NSCLC patients with monoimmunotherapy or immune-combination therapy. Forest plots of ORR in (a) prospective studies of monoimmunotherapy; (b) retrospective studies of monoimmunotherapy; (c) prospective studies of immuno-combination therapy; (d) prospective studies of chemoimmunotherapy; and (e) prospective studies of chemoimmunotherapy plus anti-angiogenesis therapy.

The mPFS and mOS of EGFR-mutant NSCLC patients with monoimmunotherapy or immuno-combination therapy. Forest plots of mPFS in (a) prospective studies of monoimmunotherapy; (b) retrospective studies of monoimmunotherapy; (c) prospective studies of immuno-combination therapy; (d) prospective studies of chemoimmunotherapy; (e) prospective studies of chemoimmunotherapy plus anti-angiogenesis therapy; (f) Forest plots of mOS in prospective studies of monoimmunotherapy; (g) Forest plots of mOS in retrospective studies of monoimmunotherapy; and (h) Forest plots of mOS in prospective studies of immuno-combination therapy.
KRAS-mutant NSCLC
Because of the gap between different studies, pooling activity data in the collected studies, the outcome suggested that the pooled ORR was 23% (95% CI: 6–39, I² = 87%; Table 3) in the clinical trials while 28% (95% CI: 21–35, I² = 86%) in retrospective studies (Table 4).10,11,14,17,20,21,26,43,48,49,57,58,64,72,75–81 As for these patients, the pooled ORR of KRAS non-G12C mutant NSCLC reached 33% (95% CI: 22–44, I² = 79%), while 40% (95% CI: 25–55, I² = 83%) of KRAS G12C mutant (Figure 4). Besides that, the pooled mPFS and mOS of monoimmunotherapy in retrospective studies were 3.24 months (95% CI: 2.48–4.00, I² = 73.6%) and 12.29 months (95% CI: 10.45–14.13, I² = 19.9%; Figure 5).
Clinical activity of immunotherapy in KRAS-altered NSCLC patients.

I, monoimmunotherapy; IC, chemo-immunotherapy; ICA, immunotherapy plus chemotherapy and anti-angiogenesis therapy; KRAS, Kirsten rat sarcoma viral oncogene homolog; mOS, median overall survival; mPFS, median progression-free survival; NA, not available; NR, not reach; NSCLC, non-small-cell lung cancer; ORR, objective response rate; Pro, prospective study; Retro, retrospective study.
Clinical activity of immunotherapy in ALK-altered NSCLC patients.
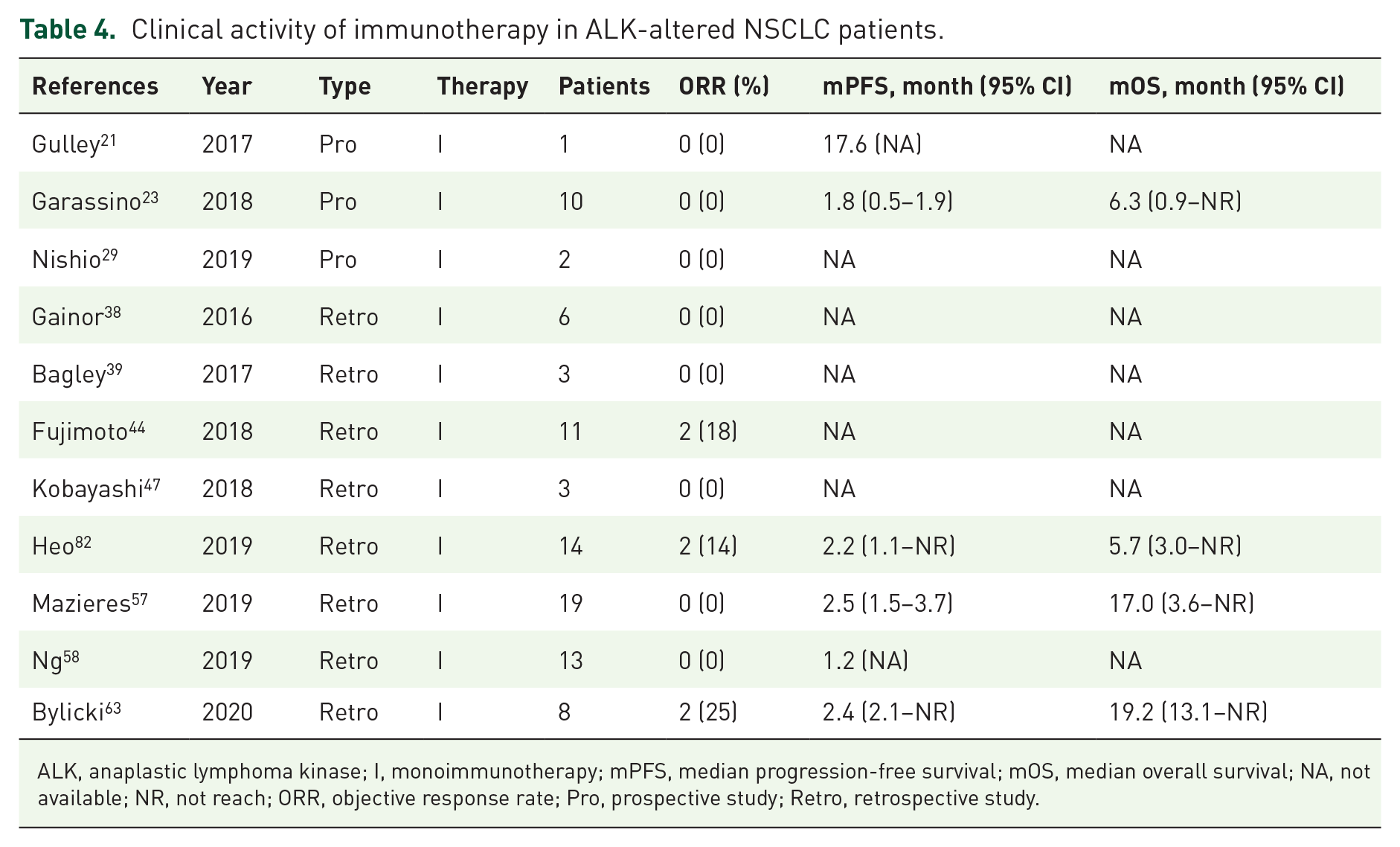
ALK, anaplastic lymphoma kinase; I, monoimmunotherapy; mPFS, median progression-free survival; mOS, median overall survival; NA, not available; NR, not reach; ORR, objective response rate; Pro, prospective study; Retro, retrospective study.
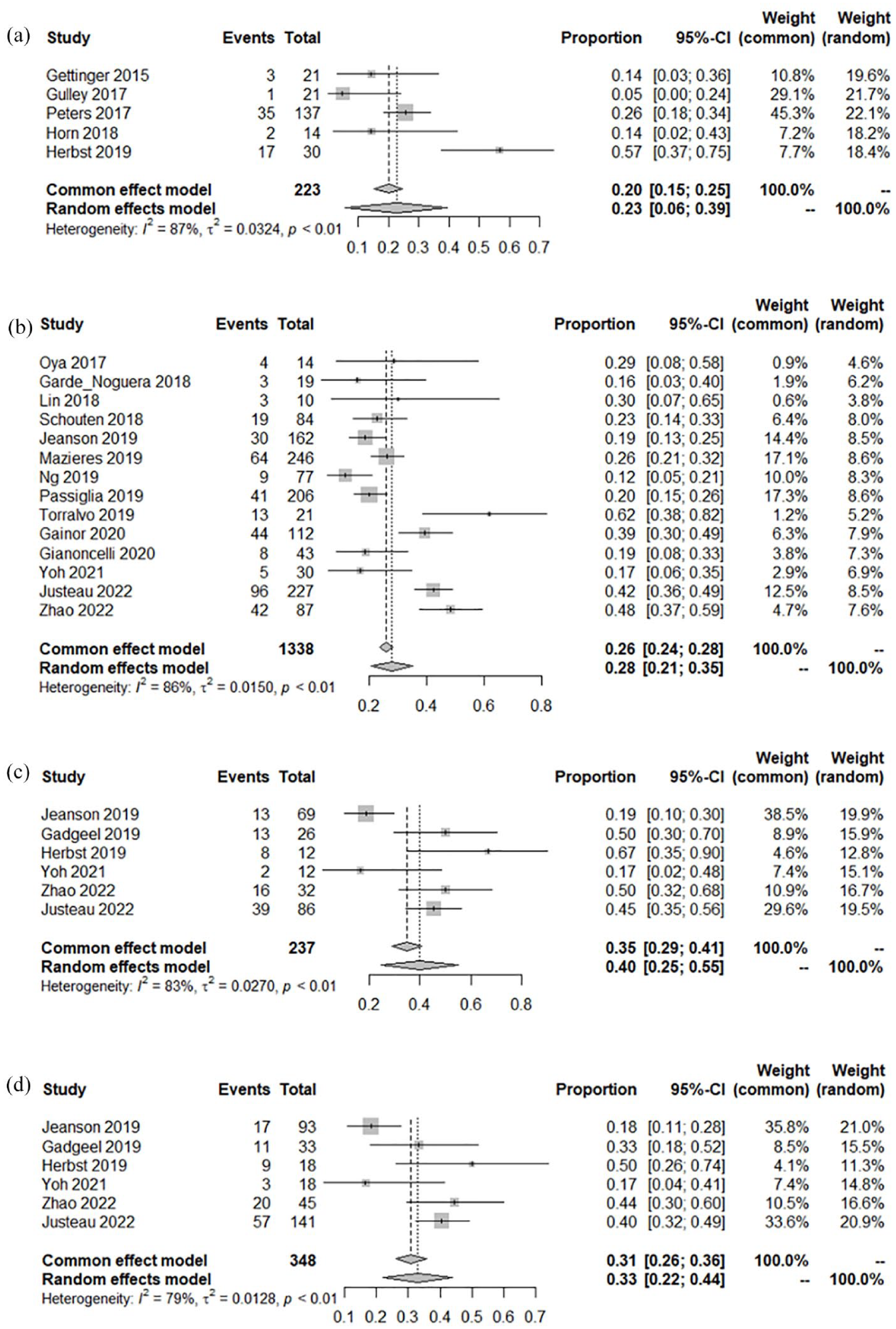
ORR of KRAS-mutant NSCLC patients with monoimmunotherapy. (a) Forest plots of ORR in (a) prospective studies of monoimmunotherapy; (b) retrospective studies of monoimmunotherapy; (c) KRAS G12C mutations; and (d) KRAS non-G12C mutations.

The mPFS of KRAS-mutant NSCLC patients with monoimmunotherapy. (a) Forest plots of mPFS in prospective studies of monoimmunotherapy and (b) Forest plots of mOS in prospective studies of monoimmunotherapy.
ALK-fusion NSCLC
The pooled ORR was 0% (95% CI: 0–8, I² = 0) of ALK-fusion-positive NSCLC treated with immunotherapy in three clinical trials, while 3% (95% CI: 0–13, I² = 21%) in eight retrospective studies (Table 4 and Figure 6).21,23,29,38,39,44,47,57,58,63,82

ORR of ALK-rearranged NSCLC patients with monoimmunotherapy. Forest plots of ORR in (a) prospective studies of monoimmunotherapy and (b) retrospective studies of monoimmunotherapy.
Other uncommon driving gene-positive NSCLC
Ten retrospective concerning BRAF-mutant NSCLC studies were included in our analysis, the results showed that the pooled ORR was 24% (95% CI: 18–31, I² = 0), and the subgroup analysis showed that the pooled ORRs of patients with BRAF V600E and non-V600E mutations were 20% (95% CI: 9–31, I² = 0) and 34% (95% CI: 19–49, I² = 0; Supplemental Figure S1 and Table S4), respectively.43,49,57,58,64,83–87 In seven retrospective studies concerning MET-altered NSCLC, the pooled ORR of them was 23% (95% CI: 12–33, I² = 55%; Supplemental Figure S2 and Table S5.)57,58,64,83,86,88,89. By subgroup analysis, the pooled ORR of MET-exon 14 skipping NSCLC patients treated with immunotherapy was 17% (95% CI: 5–28, I² = 28%), while the pooled ORR of MET-amplification population was 60% (95% CI: 17–100, I² = 77%). In addition, the pooled mPFS of monoimmunotherapy was 2.48 months (95% CI: 1.14–3.83, I² = 35.6%; Supplemental Figure S3). A total of 11 retrospective studies with HER2-mutant NSCLC were included in the analysis, the pooled ORR was 14% (95% CI: 9–19, I² = 0) in immunotherapy alone, while 37% (95% CI: 24–50, I² = 35%) in immunotherapy combined with other therapies (Supplemental Figure S4 and Table S6).43,50,57,58,69,83,86,90–93 The pooled ORRs of RET-rearranged and ROS1-rearranged NSCLC were 6% (95% CI: 0–16, I² = 29%; Supplemental Table S7) and 8% (95% CI: 0–17, I² = 0; Supplemental Table S8), respectively.57,58,63,83,86,94–97 Moreover, the pooled mPFS rates of HER2-mutant and RET-rearranged monoimmunotherapy were 2.31 months (95% CI: 1.61–3.01, I² = 0) and 2.68 months (95% CI: 1.60–3.76, I² = 0), respectively (Supplemental Figure S3).
Publication bias
There was an apparent asymmetry in the funnel plots, which suggested the presence of publication bias (Supplemental Figure S5). However, this finding can be explained by the high heterogeneity in each subgroup, which was an inevitable limitation of the single-arm or retrospective study. Therefore, we decided to include these studies in our analysis.
Discussion
At present, there is no sufficient evidence for immunotherapy for NSCLC with a positive driving gene. Herein, our study analyzed the efficacy of advanced oncogene-driven NSCLC patients treated with monoimmunotherapy or immune-combination therapy. In retrospective data, the pooled ORRs of EGFR, ALK, KRAS, BRAF, MET, HER2, RET, and ROS1 alteration in retrospective studies treated with monoimmunotherapy were 8%, 3%, 28%, 24%, 23%, 14%, 7%, and 8%, respectively. Among them, the pooled ORRs of KRAS non-G12C mutation, KRAS G12C mutation, BRAF V600E mutation, BRAF non-V600E mutation, MET-exon 14 skipping, and MET-amplification were 33% 40%, 20%, 34%, 17%, and 60%, respectively. In addition, the pooled mPFS rates of EGFR, KRAS, MET, HER2, and RET alteration were 2.77, 3.24, 2.48, 2.31, and 2.68 months, respectively. Moreover, the pooled mOS rates of EGFR and KRAS alteration were 9.98 and 12.29 months, respectively. In prospective data, the pooled ORRs of EGFR, ALK, and KRAS alteration were 6%, 0%, and 23%, respectively. The pooled ORR and mPFS of EGFR-mutant NSCLC treated with IC reached 38% and 6.20 months, while 58% and 8.48 months with ICA. In addition, the pooled mPFS and mOS of monoimmunotherapy were 2.33 months and 12.43 months in EGFR mutation, respectively.
The pooled ORR and mPFS of EGFR-mutant NSCLC are similar to or lower than those of KRAS, BRAF, MET, HER2, RET, and ROS1 alteration in studies treated with monoimmunotherapy, while those of EGFR mutation become higher with immuno-combination therapy than monoimmunotherapy. That means EGFR mutation may be biomarkers of poor response to monoimmunotherapy and good response to immune-combination therapy. The results of a meta-analysis of Yang’s, Lee’s, and Qian’s teams all showed that compared with conventional chemotherapy, monoimmunotherapy did not prolong the survival time of EGFR-mutant NSCLC patients.98–100 And meta-analysis performed by Rui’s and Lee’s teams both showed that ICI was more effective than chemotherapy in EGFR wild-type NSCLC, which may due to the low expression of PD-L1 in EGFR-mutant NSCLC.101,102 Now, there was no randomized controlled trial (RCT) concerning the effect of ICI on various subtypes of EGFR mutation. Hastings et al.’s retrospective study in 2019 showed that patients with G719X mutation received the best response (ORR = 28.6%, mPFS = 4.8 months), while L861Q mutation received the worst response (ORR = 0%, mPFS = 1.3 months) treated with monoimmunotherapy. And the ORR of the 19del mutation was lower than that of the L858R mutation, while the ORR of the L858R mutation was similar to that of the wild type. 55 Different subtypes of EGFR mutation have different responses to monoimmunotherapy, which may be due to the different immunogenicity, and further study is needed to be conducted. IMpower150 is the first study in a subgroup to show that ICIs have clinical benefits for EGFR-mutant patients. Further analysis of the EGFR-mutant subgroup showed that the PFS benefit in the ABCP (atezolizumab plus bevacizumab plus carboplatin plus paclitaxel) group was higher than that in the BCP (bevacizumab plus carboplatin plus paclitaxel) group (10.2 months versus 6.9 months). Similar results took place in a single-arm clinical trial, the ORR of that 40 TKI-resistant and EGFR-mutant NSCLC patients treated with combinational induction therapy of atezolizumab, bevacizumab, pemetrexed, and carboplatin reached 62.5%, and mPFS was 9.4 months. 34 Therefore, immunotherapy combined with other therapies can significantly improve the survival of patients with EGFR mutation, especially immunotherapy combined with chemotherapy and anti-angiogenesis therapy.
Of patients with NSCLC-harbored KRAS mutations, 35–45% were the G12C subtype, sotorasib and adagrasib are the only target drugs approved by the FDA for the treatment of patients with KRAS G12C mutation NSCLC.103,104 However, they could not meet the needs of all kinds of KRAS-mutant NSCLC treatment. In 2019, Mazieres et al. conducted a retrospective analysis of advanced NSCLC patients who received ICI monotherapy and found that patients with KRAS mutations had a higher ORR than other types of driving gene mutations in immunotherapy, which was consistent with our results. 57 According to a previous study, the activation of the KRAS signal pathway can inhibit the activity of tristetraprolin, stabilize PD-L1 mRNA, increase the synthesis of PD-L1, and upregulate the expression of PD-L1, so KRAS-mutant tumor was sensitive to ICIs therapy. 105 In 2021, Landre et al. reported a meta-analysis of anti-PD-(L)1 for KRAS-mutant advanced NSCLC, the results suggested that immunotherapy can prolong the OS of NSCLC patients with KRAS mutation whether or not combined with chemotherapy (HR = 0.59, 95% CI: 0.49–0.72; PP = 0.0003). 106 Gu et al. conducted a meta-analysis and found that patients with advanced NSCLC with KRAS mutations can benefit from ICIs but no difference between KRAS mutant subtypes was observed. 107 Generally speaking, monoimmunotherapy or immune-combination therapy might be the main treatment for non-G12C mutations until other more effective targeted drugs are available. More clinical trials are needed to evaluate the efficacy of immunotherapy in the KRAS-mutant population.
No ALK-rearranged NSCLC patients participated in clinical trials of ICI monotherapy to achieve objective remission. Almost all studies on the efficacy of ICIs in ALK-positive patients are small sample or subgroup analyses. ICIs alone did not show a good effect on ALK-rearranged patients, while ICIs combined with ALK-TKIs showed a little high efficacy. Therefore, further research is needed to explore better treatment options for ALK-rearranged patients. In the ATLANTIC study, the high expression of PD-L1 in ALK-rearranged patients was higher than that in patients with EGFR mutations but the effectiveness of durvalumab was only observed in patients with EGFR mutations. 23 Therefore, the role of ICIs in patients with ALK fusion is not prominent. In general, EGFR- and ALK-altered patients who would have better benefits from targeted therapy do not need to try immunotherapy and would not consider starting immune-combination therapy unless their EGFR/ALK-TKI resistance developed.
Immunotherapy with BRAF, HER2, MET, RET, and ROS1 alteration NSCLC is currently lacking prospective studies, and the baseline levels of patients analyzed in retrospective studies vary from study to study, so the results of the studies are varied. In our study, patients with BRAF non-V600E mutations showed better responsiveness than those with BRAF V600E mutation and the MET-amplification population showed a better response to MET-exon 14 skipping. Dabrafenib and trametinib are targeted in the treatment of BRAF V660E mutant NSCLC, while for non-V600E, there is no standard recommended medication. 108 Based on the literature we included, immunotherapy is a not bad option for patients with BRAF non-V600E mutations or MET amplification. We showed that pure immunotherapy has limited efficacy for HER2/RET/ROS1-altered NSCLC. Previous studies also showed that the activity of monoimmunotherapy in these tumors was modest and chemoimmunotherapy was more effective than monoimmunotherapy.92,97
Negrao’s team analyzed the effect of driving gene mutation on the expression of tumor mutation burden (TMB) and PD-L1 and the correlation with the efficacy of immunotherapy in 4017 NSCLC patients treated with ICI. The results showed that ALK/ROS1/RET fusion and MET-exon14 mutation group had high expression of PD-L1 and low TMB but did not translate into clinical benefit. It is suggested that in addition to TMB and PD-L1, the driving gene can also affect the clinical outcome of immunotherapy. 109 In addition, for different gene mutations, mutations at different sites can activate different signal pathways and lead to different downstream effects, which may lead to different responses to immunotherapy.
There are several limitations in this study. Most of the included studies were retrospective trials with small sample sizes or prospective with subgroup data, which may lead to selection bias. In addition, owing to the lack of PFS and OS data in ALK, BRAF, and ROS1 alterations, we evaluated the therapeutic efficacy by examining the ORR of the published data. Precision therapy is the trend of NSCLC treatment in the future but ICI therapy for oncogene-driven NSCLC patients still needs to be further studied to bring more survival benefits to more oncogene-driven NSCLC patients.
Conclusion
The efficacy of immunotherapy is different in patients with different oncogene-driven NSCLC. EGFR-, ALK-, HER2-, RET-, and ROS1-altered NSCLC patients have poor reactivity to monoimmunotherapy but the efficacy of immune-based combined therapy is significantly improved. KRAS G12C mutation, BRAF non-V600E mutation, and MET amplification have better responses to immunotherapy, and more prospective studies are needed for further research.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231225036 – Supplemental material for Efficacy of immunotherapy in patients with oncogene-driven non-small-cell lung cancer: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359231225036 for Efficacy of immunotherapy in patients with oncogene-driven non-small-cell lung cancer: a systematic review and meta-analysis by Jiayan Chen, Wanjun Lu, Mo Chen, Zijing Cai, Ping Zhan, Xin Liu, Suhua Zhu, Mingxiang Ye, Tangfeng Lv, Jiawen Lv, Yong Song and Dong Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231225036 – Supplemental material for Efficacy of immunotherapy in patients with oncogene-driven non-small-cell lung cancer: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359231225036 for Efficacy of immunotherapy in patients with oncogene-driven non-small-cell lung cancer: a systematic review and meta-analysis by Jiayan Chen, Wanjun Lu, Mo Chen, Zijing Cai, Ping Zhan, Xin Liu, Suhua Zhu, Mingxiang Ye, Tangfeng Lv, Jiawen Lv, Yong Song and Dong Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359231225036 – Supplemental material for Efficacy of immunotherapy in patients with oncogene-driven non-small-cell lung cancer: a systematic review and meta-analysis
Supplemental material, sj-docx-3-tam-10.1177_17588359231225036 for Efficacy of immunotherapy in patients with oncogene-driven non-small-cell lung cancer: a systematic review and meta-analysis by Jiayan Chen, Wanjun Lu, Mo Chen, Zijing Cai, Ping Zhan, Xin Liu, Suhua Zhu, Mingxiang Ye, Tangfeng Lv, Jiawen Lv, Yong Song and Dong Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
