Abstract
Background:
Neoadjuvant (NE), adjuvant (AD), and perioperative (PE) immunotherapies have gained validation in early-stage non-small cell lung cancer (NSCLC) trials. However, a comprehensive assessment of their comparative efficacy and safety is lacking.
Objectives:
To compare the efficacy and safety of NE, AD, and PE immunotherapies in early-stage NSCLC.
Design:
A systematic review and network meta-analysis using a Bayesian framework.
Data sources and methods:
We searched PubMed, Embase, and Cochrane databases for randomized controlled trials (RCTs) of immune checkpoint inhibitors plus chemotherapy (CT) for early-stage NSCLC. Hazard ratios (HRs) and odds ratios (ORs) for binary endpoints with 95% confidence intervals (CIs) were calculated.
Results:
We included 10 RCTs involving 5569 NSCLC patients, categorized as NE, PE, or AD immunotherapy. Indirect comparisons highlighted differences in efficacy between PE and AD immunotherapy, specifically in event-free survival (EFS)/disease-free survival (DFS) (HR = 0.72, 95% CI: 0.53–0.96). NE/PE immunotherapies improved pathologic complete response (pCR) (OR = 7.56, 95% CI: 5.24–10.92), major pathologic response (MPR) (OR = 5.46, 95% CI: 3.97–7.51), and EFS (HR = 0.58, 95% CI: 0.52–0.65), while AD immunotherapy enhanced DFS (HR = 0.78, 95% CI: 0.69–0.90). Overall survival (OS) benefits were seen only with PE immunotherapy (HR = 0.66, 95% CI: 0.55–0.81). PE treatment improved EFS across various subgroups (PD-L1 < 1%, IIIB, squamous, female, without MPR/pCR, epidermal growth factor receptor (EGFR) mutant-negative), except EGFR mutant-positive NSCLC (HR = 0.54, 95% CI: 0.21–1.43). AD (OR = 1.81, 95% CI: 1.20–2.73) and PE (OR = 1.28, 95% CI: 1.10–1.50) immunotherapies were associated with higher grade ⩾3 adverse events.
Conclusion:
In the three treatment modalities, PE immunotherapy appears to be more effective than AD immunotherapy, with PE showing significant advantages in certain subgroups that NE does not. NE and PE immunotherapy significantly improved pCR, MPR, and EFS, while AD immunotherapy significantly improved DFS in NSCLC patients compared to the control group. However, only PE immunotherapy significantly improved OS. Differences in efficacy between NE and PE across the entire population of resectable NSCLC remain to be explored in additional studies.
Keywords
Introduction
Patients with early or locally advanced non-small cell lung cancer (NSCLC) have the opportunity to be treated with surgical resection by undergoing perioperative therapy. However, even after successful complete tumor resection, patients have a higher rate of recurrence and risk of distant metastasis, which affects long-term survival outcomes.1–3 Studies suggest that traditional perioperative chemotherapy (CT) utilizing cytotoxic agents provides a marginal survival advantage of 5.4% compared to surgery alone. However, this benefit comes at the cost of a 60% increase in the occurrence of grade 3 or higher adverse events (AEs). Moreover, preoperative neoadjuvant CT demonstrates inferior outcomes compared to postoperative adjuvant CT.4–6 Recent advancements in immune checkpoint inhibitor (ICI) therapies, specifically those involving monoclonal antibodies targeting the programmed death-1 (PD-1) signaling pathway, have generated considerable attention in the fields of neoadjuvant, adjuvant, and perioperative treatment. These therapies are designed to achieve objectives such as tumor size reduction, increased likelihood of complete resection, and the eradication of micrometastases.7,8
The PD-1 signaling pathway and its ligand (PD-L1) have a central role in facilitating tumor immune evasion by suppressing T-cell immune function. This inhibition is achieved through the blockade of T-cell proliferation, chemotaxis, and cytokine release, facilitating tumor cell escape from immune surveillance. 9 Conversely, ICIs potentiate T-cell-mediated antitumor responses by inhibiting the endogenous regulatory molecules of the immune system. This action preserves the dynamic interplay among CD8+ T cells, antigen-presenting cells, and tumor cells, essential for effective immune surveillance against tumors. 10 Several ICIs have received approval from the U.S. Food and Drug Administration and in various countries for use in neoadjuvant, adjuvant, and perioperative therapies for individuals with early or locally advanced NSCLC. 11
In the phase III randomized controlled trial (RCT) CheckMate 816, Nivolumab in combination with CT as neoadjuvant therapy improved event-free survival (EFS), compared to CT alone.12,13 Furthermore, the IMpower 010 trial demonstrated that adjuvant therapy with atezolizumab, following CT, resulted in a significant enhancement of disease-free survival (DFS) compared to optimal supportive care.14,15 Moreover, recent advancements in clinical research on perioperative therapy, utilizing a sandwich regimen (Neoadjuvant therapy + Surgery + Adjuvant therapy), have rapidly increased, showing marked improvements in EFS.16–21 However, the differences in efficacy and safety among neoadjuvant, adjuvant, and perioperative therapy require further clarification.
In this systematic review and meta-analysis, our primary objective was to assess the efficacy and safety of neoadjuvant, adjuvant, or perioperative ICI therapy in combination with CT compared to CT alone for NSCLC. Additionally, we aimed to investigate the effectiveness of these three treatment sequences within specific subpopulations and indirectly compare their efficacy, providing valuable insights for the design of future head-to-head clinical studies.
Methods
This systematic review and meta-analysis are based on the Preferred Reporting Items for Systematic Reviews and the Network Meta-Analysis (PRISMA) extended statement (Supplemental Table 1). 22 This study was registered in the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY202420024). 23
Data sources and search strategy
A comprehensive search was conducted for all RCTs pertaining to NSCLC. Databases including PubMed, Embase, and the Cochrane Library were systematically queried, spanning from the inception of these databases up to January 15, 2024. In addition, manual searches were meticulously carried out for reviews, abstracts, and conference reports originating from major international conferences on lung cancer. Search terms included PD-1 inhibitors, PD-L1 inhibitors, and specific drugs such as pembrolizumab, atezolizumab, toripalimab, nivolumab, and so on (Supplemental Table 2). We only included RCTs reported in English. The two authors conducted the search independently, and any discrepancies were resolved by mutual discussion to reach consensus.
Selection criteria
Eligible studies for inclusion met the following criteria: (1) RCTs; (2) investigations of neoadjuvant, adjuvant, or perioperative ICIs combined with CT versus CT alone; (3) inclusion of adult patients diagnosed with early-stage I–III NSCLC; (4) included studies were those that reported any of the following endpoints: pathologic complete response (pCR), major pathologic response (MPR), AEs of any grade, AEs of grade 3 or higher, immune-related adverse events (irAEs) of any grade, irAEs of grade 3 or higher, EFS/DFS, and overall survival (OS). EFS is defined as the time from randomization to the first occurrence of local progression that precludes planned surgery, unresectable tumor, progression or recurrence, or death from any cause. DFS is defined as the time from randomization to the first occurrence of progression or recurrence, or death from any cause.
Excluded from consideration were studies exhibiting the following characteristics: (1) patients with a history of prior systemic immunosuppressive therapy and active autoimmune disease; (2) studies lacking outcomes of interest; and (3) investigations that incorporated radiotherapy or dual immunotherapy interventions.
Data extraction and quality assessment
Two reviewers independently assessed the titles, abstracts, full texts, and Supplemental Materials, extracting data in accordance with the PRISMA guidelines. Data extraction encompassed study name/title, national clinical trials identification number, publication year, study phase, blinding status, protocol details for trial and control groups, sample size, patient demographics, and outcome data.
Risk of bias assessment
The quality assessment of individual RCT was executed employing the Cochrane Collaboration’s tool for evaluating the risk of bias in randomized trials (RoB 2) (Cochrane Collaboration, London, UK). 24 Two authors independently conducted the assessment, and any discrepancies were resolved through consensus. Red represents high risk, yellow represents some concern, and green represents low risk. Publication bias was evaluated using funnel plots, and their symmetry was assessed through Egger’s and Begg’s tests (significance set at p < 0.05, indicating substantial asymmetry). Funnel plots served as a tool to detect potential publication bias in the study.
Statistical analysis
For survival outcomes, hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated, while odds ratios (ORs) with 95% CIs were used for dichotomous variables. Due to the heterogeneity among the studies, random-effect models were applied across the analysis. Comparisons were deemed not statistically significant if the 95% CI included the value 1. The network meta-analysis employed a Bayesian framework to perform indirect comparisons among perioperative therapy, neoadjuvant therapy, and adjuvant therapy. We assessed the efficacy of these treatment regimens using the surface under the cumulative ranking curve (SUCRA), where higher SUCRA scores approaching 1 indicate greater effectiveness, while lower scores approaching 0 indicate lesser effectiveness. Subgroup analyses were conducted based on several key factors, including PD-L1 expression, baseline disease stage, histology, epidermal growth factor receptor (EGFR) mutation status, gender, and the presence or absence of MPR or pCR. Sensitivity analyses validated the meta-analysis results through the sequential exclusion of individual studies. Statistical analyses utilized R software (version 4.3.0) and R Studio, integrating the gemtc and rjags packages. A two-sided p-value was employed, with p < 0.05 considered statistically significant.24–27
Results
Study overview and characteristics
The initial search yielded 916 results, from which duplicates were removed, leading to the screening of titles and abstracts. Subsequently, 73 full-text articles, including gray literature, were meticulously reviewed against predetermined inclusion criteria. Ultimately, 11 RCTs were selected for inclusion, consisting of 2 neoadjuvant, 2 adjuvant, and 6 perioperative studies, encompassing a total of 5569 patients with NSCLC. The screening process is depicted in Figure 1, while Table 1 summarizes the baseline characteristics of the included studies, and Supplemental Table 3 provides supplementary baseline data.

Study flowchart: literature search and selection.
Basic characteristics of the included studies.

CT, chemotherapy; Ctrl, control; DFS, disease-free survival; EFS, event-free survival; Exp, experimental; NA, not applicable; NCT, national clinical trials; OS, overall survival; pCR, pathologic complete response; Q3W, every 3 weeks.
Efficacy analysis
In two neoadjuvant RCTs, significant improvements were observed in ICI + CT group versus the CT-alone group. These improvements included EFS (HR = 0.66, 95% CI: 0.49–0.89), MPR (OR = 6.81, 95% CI: 4.07–11.40), and pCR (OR = 8.66, 95% CI: 3.18–23.62) (Figure 2(a), (d), and (e)). However, there was no significant difference in OS (HR = 0.62, 95% CI: 0.36–1.05, based on 1 RCT) (Figure 3(c)).
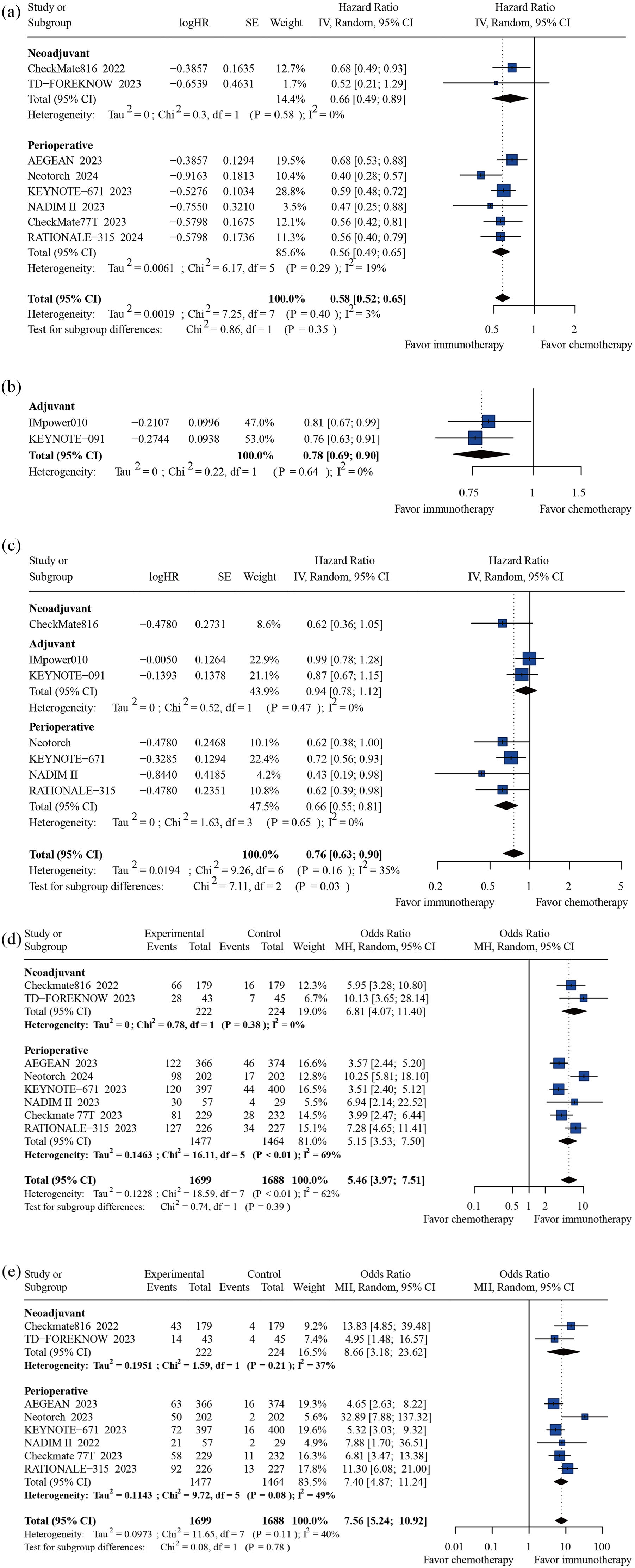
Forest plots presenting pooled hazard ratio and odds ratio analysis for efficacy of neoadjuvant, adjuvant, and perioperative immunotherapy in the population of resectable NSCLC. (a) Event-free survival. (b) Disease-free survival. (c) Overall survival. (d) Major pathologic response. (e) Pathologic complete response.
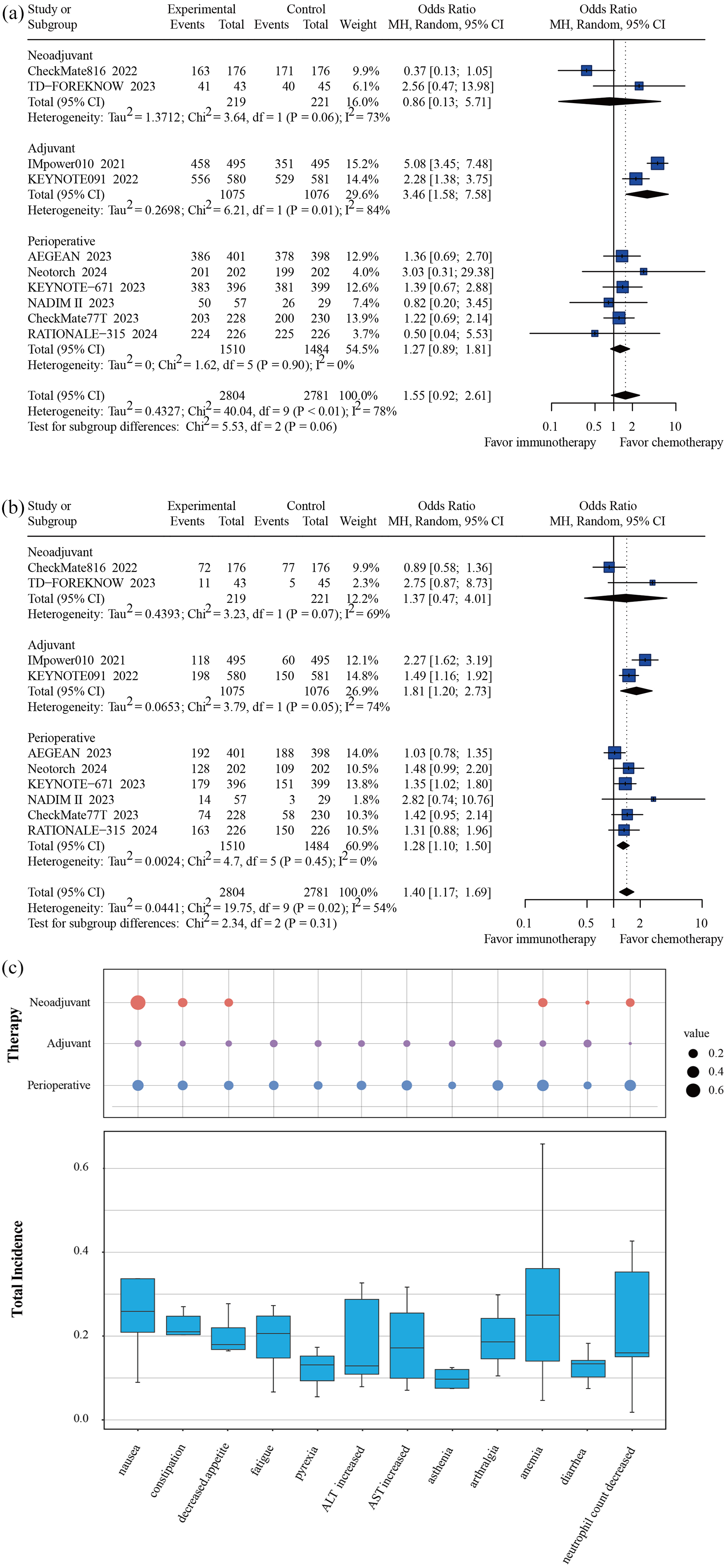
Forest plots presenting pooled odds ratio analysis for toxicity of neoadjuvant, adjuvant, and perioperative immunotherapy in the population of resectable NSCLC. (a) Any grade AEs. (b) Greater than or equal to level 3 AEs. (c) Bubble chart and box plot presenting the incidence of any grade AEs.
Within the 2 adjuvant RCTs, the adjuvant ICI group demonstrated a significant benefit in DFS (HR = 0.78, 95% CI: 0.69–0.90) and a nonsignificant trend toward improved OS (HR = 0.94, 95% CI: 0.78–1.12) compared to the control group (Figure 2(b) and (c)).
Compared with the CT group, the perioperative ICI + CT group showed significant improvements in EFS/PFS (HR = 0.56, 95% CI: 0.49–0.65), OS (HR = 0.66, 95% CI: 0.55–0.81, including 4 RCTs), MPR (OR = 5.15, 95% CI: 3.53–7.50, including 6 RCTs), and pCR (OR = 7.40, 95% CI: 4.87–11.24, including 6 RCTs) (Figure 2(a), (c)–(e)).
Safety analysis
There was no statistically significant difference in the incidence of AEs between the neoadjuvant and perioperative ICI + CT groups and their respective control groups. However, the adjuvant ICI group showed a significant difference compared to the control group (OR = 3.46, 95% CI: 1.58–7.58) (Figure 3(a)). However, AEs of grade 3 or higher were more frequent in both the adjuvant ICI group (OR = 1.81, 95% CI: 1.20–2.73) and the perioperative ICI + CT group (OR = 1.28, 95% CI: 1.10–1.50) (Figure 3(b)).
The ICI groups had a significant increase in any grade of alanine aminotransferase (ALT) increased (OR = 1.76, 95% CI: 1.31–2.36), aspartate aminotransferase increased (OR = 1.88, 95% CI: 1.24–2.85), arthralgia (OR = 1.61, 95% CI: 1.21–2.13), rash (OR = 2.64, 95% CI: 1.74–4.02), pruritus (OR = 3.01, 95% CI: 1.39–6.49), hypothyroidism (OR = 6.38, 95% CI: 4.74–8.59), hyperthyroidism (OR = 3.58, 95% CI: 2.50–5.13), and pneumonitis (OR = 2.60, 95% CI: 1.88–3.58) (Supplemental Figures 1 and 2).
Nausea was the most common any grade AEs for neoadjuvant ICI + CT (68.95%), arthralgia was the most common any grade AEs for adjuvant ICI (12.59%), and neutrophil count decreased was the most common any grade AEs for perioperative ICI + CT (35.60%) (Figure 3(c)), whereas rash was the most common any grade irAEs for neoadjuvant and perioperative ICI + CT (10.50%, 13.22%). Pruritus was the most common any grade irAEs for adjuvant ICI (16.37%) (Supplemental Figure 5).
It is noteworthy that nausea was the most frequently reported grade 3 or higher AE in the neoadjuvant ICI + CT group (26.94%). Conversely, neutrophil count decreased was predominantly observed in perioperative ICI + CT groups (22.56%), while the adjuvant ICI group most commonly reported an increase in ALT as a grade 3 or higher AE (1.12%). Furthermore, it is important to highlight that pneumonitis emerged as the predominant grade 3 or higher irAE in perioperative and adjuvant therapies (2.27%, 0.84%). Rash was the common grade 3 or higher irAE in neoadjuvant therapy (1.37%) (Supplemental Figures 3, 4, and 6).
Subgroup analysis
Based on tumor PD-L1 expression level
To assess the impact of PD-L1 expression levels on the clinical outcomes of NSCLC patients, individuals were stratified into three subgroups based on PD-L1 tumor proportion score (TPS): <1%, 1–49%, and ⩾50%.
For patients with PD-L1 TPS <1%, the pooled analysis indicated a significant benefit in EFS/PFS for perioperative ICI + CT over CT alone (HR = 0.76, 95% CI: 0.62–0.92, across 6 RCTs). However, no significant differences were observed with neoadjuvant ICI + CT and adjuvant ICI treatments compared to controls (Figure 4(a)).

Forest plots presenting subgroup analyses targeting event-free survival. (a) PD-L1 <1%. (b) PD-L1 1%–49%. (c) PD-L1 ⩾50%. (d) Without pCR. (e) With pCR. (f) EGFR mutant type. (g) EGFR wild type.
In the PD-L1 TPS 1%–49% subgroup, both adjuvant ICI (HR = 0.75, 95% CI: 0.58–0.97, across 2 RCTs) and perioperative ICI + CT (HR = 0.53, 95% CI: 0.53–0.73, across 5 RCTs) demonstrated significant DFS/EFS benefits compared to control groups. Neoadjuvant ICI + CT did not exhibit a significant advantage over CT alone in this subgroup (Figure 4(b)).
Patients with PD-L1 TPS ⩾50% showed substantial EFS benefits with both neoadjuvant ICI + CT (HR = 0.24, 95% CI: 0.10–0.61, including 1 RCT) and perioperative ICI + CT (HR = 0.47, 95% CI: 0.35–0.63, across 5 RCTs) compared to controls. Adjuvant ICI did not confer a significant advantage (Figure 4(c)).
Based on clinical stage
Patients with varying clinical stages were divided into 4 subgroups: II, IIIA, and IIIB for analysis (Supplemental Figure 7).
In stage II patients, significant DFS/EFS benefits were observed with adjuvant ICI therapy (HR = 0.72, 95% CI: 0.59–0.87, across 2 RCTs) and perioperative ICI + CT (HR = 0.64, 95% CI: 0.49–0.82, across 4 RCTs) compared to the control group.
For stage IIIA patients, substantial EFS improvements were seen with both neoadjuvant ICI + CT (HR = 0.54, 95% CI: 0.37–0.80, including 1 RCT) and perioperative ICI + CT (HR = 0.55, 95% CI: 0.47–0.66, across 4 RCTs) relative to CT alone, whereas adjuvant ICI did not demonstrate a significant DFS advantage.
In the stage IIIB cohort, perioperative ICI + CT conferred a notable EFS benefit over CT alone (HR = 0.54, 95% CI: 0.31–0.94, including 3 RCTs).
Based on histology
Patients were divided into two subgroups, non-squamous and squamous, for different histologic types (Supplemental Figure 8).
In the non-squamous lung cancer population, neoadjuvant ICI + CT, adjuvant ICI, and perioperative ICI + CT showed significant EFS/DFS benefit. In the squamous lung cancer population, perioperative ICI + CT had a significant EFS benefit compared to CT alone (HR = 0.51, 95% CI: 0.42–0.63 including 6 RCTs) but no significant difference was seen between neoadjuvant ICI + CT and adjuvant ICI.
Based on EGFR mutation status
In the EGFR wild-type population, neoadjuvant (HR = 0.68, 95% CI: 0.49–0.93), adjuvant ICI (HR = 0.73, 95% CI: 0.57–0.95), and perioperative ICI + CT (HR = 0.50, 95% CI: 0.41–0.60) were shown to be effective in terms of EFS/DFS benefit compared to controls. In the EGFR-mutated population, none of the adjuvant or perioperative ICI groups demonstrated significant benefit compared to the control group (Figure 4(f) and (g)).
Based on sex
In the male and female population, significant DFS/EFS benefit was observed in both adjuvant ICI and perioperative ICI + CT compared to controls (Supplemental Figure 9).
Based on MPR/pCR status
In this study, we exploratively analyzed the EFS benefit of those who did or did not achieve MPR/pCR, categorized into four groups: with MPR, without MPR, with pCR, and without pCR.
Only the perioperative ICI + CT studies reported EFS in both with/without MPR subgroups, and the pooled analysis showed that perioperative ICI + CT benefited this population compared to CT alone regardless of whether MPR was obtained or not (Supplemental Figure 10(A) and (B)).
In addition, perioperative ICI + CT resulted in better EFS benefit regardless of with or without pCR, but the EFS benefit of neoadjuvant ICI + CT treatment did not show statistically significant differences in the population without pCR compared to CT alone (HR = 0.84, 95% CI: 0.61–1.17) (Figure 3(d) and (e)). We similarly analyzed the difference in efficacy with ICI + CT in the population with or without pCR and found that the efficacy with ICI + CT was better in the population with pCR (HR = 0.14, 95% CI: 0.07–0.30) (Supplemental Figure 10(C)).
Indirect comparison of neoadjuvant, adjuvant, and perioperative therapies
Using a Bayesian framework, we conducted an indirect comparison of the three treatment sequences. As illustrated in the Network Diagram (Figure 5(a)), we found that perioperative ICI + CT was significantly superior to adjuvant ICI (HR = 0.72, 95% CI: 0.53–0.96) and control group (HR = 0.57, 95% CI: 0.47–0.67), and neoadjuvant ICI + CT was significantly superior to CT alone in terms of EFS/DFS benefit (HR = 0.65, 95% CI: 0.44–0.95). But no statistical difference was observed between neoadjuvant and perioperative ICI + CT. In terms of OS benefit, only perioperative ICI + CT was significantly superior to the control group (HR = 0.65, 95% CI: 0.46–0.85) (Figure 5(b)). SUCRA rank showed that perioperative ICI + CT was ranked first in the order of both EFS and OS benefit (Figure 5(c) and (d)).

Indirect comparison of neoadjuvant, adjuvant, and perioperative immunotherapy efficacy differences via a Bayesian framework. (a) Network diagram comparing the three treatment modalities. (b) Blue triangles show HRs and 95% CIs for EFS comparisons of the three treatment modalities, and purple triangles show HRs and 95% CIs for OS comparisons, with HRs <1.00 providing a better survival benefit; SUCRA ranking profiles for three treatment modalities: (c) EFS; (d) OS.
Bias test and sensitivity analysis
Risk of bias was assessed using the available information in the full text, and four studies were open-label clinical studies that were rated as high risk (Supplemental Figure 11). Funnel plots were used to assess publication bias (Supplemental Figure 12). To further assess publication bias, significant bias was seen using Egger’s test (p = 0.0605) analysis but not seen using Begg’s test (p = 0.1074). Sensitivity analysis demonstrated that the sequential exclusion of individual studies did not significantly impact the combined results for EFS/DFS and OS (Supplemental Figure 13(A) and (B)).
Discussion
Challenges
With the disclosure of results from various clinical trials involving ICIs for resectable NSCLC, the growing significance of perioperative treatment in NSCLC management has been underscored. However, the selection among adjuvant, neoadjuvant, and perioperative treatment modalities for distinct patient populations presents several critical challenges. These include determining the potential benefits for patients with PD-L1 TPS < 1%, selecting the optimal regimen for individuals with locally advanced disease, assessing the efficacy of ICI treatments across driven gene mutant-positive, and exploring if perioperative ICI + CT is preferable for eradicating micrometastases in patients who did not achieve MPR or pCR.
Implications
In this study, a systematic review and meta-analysis of 10 RCTs found that neoadjuvant/perioperative ICI + CT significantly improved pCR, MPR, and EFS compared with CT alone, and that adjuvant ICI significantly improved DFS, but only perioperative ICI + CT significantly improved OS.12–21,28–31
Several RCTs of neoadjuvant, adjuvant, and perioperative ICI therapy with published study data have suggested that the efficacy of ICI + CT in patients with PD-L1 <1% is not significant compared to CT alone.12,14–18,20,21,28,29 Meanwhile, following the publication of CheckMate816, IMpower010, and KEYNOTE-671 results, the National Comprehensive Cancer Network guidelines have now included Nivolumab, Atezolizumab, and Pembrolizumab in the guideline recommendations for perioperative treatment. 32 The IMpower010 study used adjuvant atezolizumab to target patients with PD-L1 TPS > 1%, while the efficacy of the perioperative use of Pembrolizumab-CT for the treatment of patients with PD-L1 TPS < 1% is unclear, and the guideline therefore suggests that PD-L1 status be tested to assess the likelihood of benefit prior to administering perioperative systemic therapy. However, in our meta-analysis, we showed that patients with PD-L1 TPS < 1% still benefited significantly from perioperative ICI + CT, which may be due to the expanded sample size, and therefore, PD-L1 TPS < 1% patients cannot be excluded from perioperative immune system therapy solely on the basis of PD-L1 status, which may lead to the loss of their chances of achieving good survival.
Those who achieve MPR/pCR are usually those who have pre-established a stronger immune infiltrate prior to treatment, and benefit better from neoadjuvant ICI + CT.33,34 However, whether the subgroup of the population without MPR/pCR can benefit from ICI + CT treatment has become a general concern. We compared whether there was any difference in prognosis between the ICI + CT treatment groups with or without pCR, and the pooled analysis of the two studies showed that in the experimental group (using ICI + CT), patients with pCR had a more significant EFS benefit compared to those without pCR, which was similar to the results of previous studies. In addition, we also found benefit from perioperative ICI + CT compared to CT, regardless of whether MPR/pCR was obtained or not. Only one neoadjuvant EFS data has been published for the without pCR population, which unfortunately failed to observe a significant benefit in this population. Thus, for the without MPR/pCR subgroup population, perioperative ICI + CT may be a better therapy, but this result still needs to be confirmed by more clinical studies publishing data from subgroup analyses.
We conducted an analysis stratified by EGFR mutation status, distinguishing between wild-type and mutant-type EGFR. Our findings indicate that perioperative, neoadjuvant ICI + CT, and adjuvant therapy provide a substantial EFS/DFS benefit in the EGFR wild-type population. However, patients with EGFR mutations do not experience significantly improved outcomes with ICI + CT compared to the control group. This suggests that immunotherapy may not be the optimal choice for the majority of patients with EGFR mutations. In patients with EGFR mutant type, postoperative targeted therapy or CT remains the primary treatment strategy. Further clinical studies are warranted to explore the potential survival benefits of employing targeted therapy or immunotherapy in the neoadjuvant and adjuvant phases within this subgroup. 35
In addition, we found that both adjuvant and perioperative ICI treatments were effective in improving EFS in stage II patients, and the phase IB–II data published in the CheckMate816 study showed that no significant benefit was seen in ICI + CT treatment compared with the CT group, which may be related to the longer survival of stage IB patients. For stage IIIA patients, adjuvant ICI therapy did not perform well, and neoadjuvant and perioperative ICI + CT showed significant benefit compared to the CT group, suggesting the importance of preoperative ICI + CT therapy to reduce the extent of the tumor and eliminate micrometastases. For stage IIIB patients, no neoadjuvant ICIs studies have disclosed survival data in this subgroup, but our analysis showed that perioperative ICI + CT could significantly benefit this subgroup of the population, which was different from the results of previous meta-analysis, probably due to the increase in sample size. 36 Therefore, neoadjuvant and perioperative ICI + CT are optional for stage IIIA patients, and perioperative ICI + CT therapy is an effective option for stage IIIB patients undergoing surgical resection.
In addition, indirect comparisons showed that perioperative ICI + CT was significantly better than adjuvant ICI in improving EFS/DFS in the overall population, but did not have a significant advantage over neoadjuvant ICI + CT, which may suggest that neoadjuvant ICI + CT therapy has provided considerable clinical benefit, and that in order to avoid escalating costs and toxicity, perioperative ICI + CT needs to be used to screen for a more precise population of benefit by incorporating additional biomarkers or other clinical features.
Limits
Our study has several shortcomings. First, the small number of studies with published subgroup data on neoadjuvant ICI + CT may have resulted in limitations in the analysis. Second, our exclusion of single-arm studies may have introduced bias by potentially omitting some negative results. This could impact the overall findings, as these studies often report adverse outcomes not captured in RCTs. Third, in our analysis, we used EFS to represent both EFS and DFS endpoints. This approach was necessary to compare the efficacy of neoadjuvant, adjuvant, and perioperative immunochemotherapy for resectable NSCLC. However, due to differences in the definitions of EFS and DFS across trials, combining these endpoints might introduce heterogeneity and potential bias. Therefore, the results should be interpreted with caution.
Conclusion
Perioperative ICI + CT seems to be more effective than neoadjuvant ICI + CT and adjuvant immunotherapies ICI in these three treatment settings. Neoadjuvant and perioperative ICI + CT significantly improved pCR, MPR, EFS, and adjuvant ICI significantly improved DFS in early-stage NSCLC patients compared with the control group. However, only perioperative ICI + CT significantly improved OS. Meanwhile, perioperative ICI + CT showed long-term benefits in different key subgroups (PD-L1 < 1%, IIIB, squamous, female, without MPR/pCR, EGFR mutant-negative), at the expense of some safety. In the subgroup of EGFR mutant-positive, perioperative ICI + CT did not show significant benefit in EFS. Differences in efficacy between neoadjuvant and perioperative treatments across the entire population of resectable NSCLC remain to be explored in additional studies, and a more accurate combination of biomarkers and clinical features is needed to develop personalized treatment plans.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359241284929 – Supplemental material for Efficacy and safety of perioperative, neoadjuvant, or adjuvant immunotherapy alone or in combination with chemotherapy in early-stage non-small cell lung cancer: a systematic review and meta-analysis of randomized clinical trials
Supplemental material, sj-pdf-1-tam-10.1177_17588359241284929 for Efficacy and safety of perioperative, neoadjuvant, or adjuvant immunotherapy alone or in combination with chemotherapy in early-stage non-small cell lung cancer: a systematic review and meta-analysis of randomized clinical trials by Yunchang Meng, Qingfeng Zhang, Ranpu Wu, Huijuan Li, Zhaofeng Wang, Yang Yao, Xinjing Li, Zhangxuan Chen, Yanzhuo Gong and Hongbing Liu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank all the reviewers who participated in the review for their assistance during the preparation of this manuscript. This work was supported by Jinling Hospital Management Project grants 22LCYY-XH2.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
