Abstract
Background:
Targeted therapy with EGFR tyrosine-kinase inhibitors (TKIs) is the preferred first-line treatment for EGFR-mutated advanced non-small-cell lung cancer (NSCLC), but acquired resistance inevitably occurs in almost all responding individuals.
Objectives:
We aimed to comprehensively review the literature to investigate the efficacy and safety of distinct regimens in the subsequent-line setting, thereby identifying the optimal regimen for these TKI-resistant NSCLC patients.
Design:
A systematic review and network meta-analysis (NMA) using a Bayesian framework.
Data sources and methods:
The PubMed, Embase, Cochrane Library databases, and abstracts of ASCO, ESMO, and WCLC were searched from database inception to November 3, 2024, to identify eligible randomized controlled trials (RCTs) that assessed distinct regimens for individuals with advanced EGFR-mutated NSCLC who progressed on TKIs. The outcomes of progression-free survival (PFS), overall survival (OS), objective response rate (ORR), disease control rate (DCR), and grade 3 or higher adverse events (⩾3AEs) were compared and ranked in overall patients and various subgroups among eight regimens by NMA and the surface under the cumulative ranking curve, respectively. The protocol is registered with PROSPERO, CRD42024601619.
Results:
In total, 14 RCTs, involving 3177 participants and 8 treatment regimens (chemotherapy plus ivonescimab (programmed cell death protein 1/vascular endothelial growth factor inhibitor; chemotherapy + ivonescimab (CT + IVO)); CT + amivantamab + lazertinib (CT + AMI + LAZ), CT + immunotherapy + bevacizumab (CT + IO + BEV), CT + AMI, CT + BEV, CT + IO, CT, and IO), were included. Overall, in patients, the most pronounced PFS benefit was observed with the “CT + IVO,” followed by “CT + AMI + LAZ,” “CT + IO + BEV,” and “CT + AMI,” ranked second, third, and fourth, respectively. In terms of OS, the regimen of “CT + AMI” ranked the best, followed by “CT + IVO.” However, the comparisons of OS among different regimens did not reach statistical significance, possibly due to immature data. The results for ORR and DCR were similar to those for OS, with “CT + AMI” topping the rankings, followed by “CT + AMI + LAZ.” In terms of safety, the incidence of ⩾3AEs was highest in “CT + AMI + LAZ,” followed by “CT + AMI.” In subgroup analysis, “CT + IVO” demonstrates stable PFS benefits across clinicopathological characteristics, ranking first in most subgroups. Due to the unavailability of OS subgroup data in most RCTs, many regimens were missing in the OS subgroup analysis.
Conclusion:
Integrating the results of different clinical outcomes and subgroup analyses, we conclude that “CT + IVO” is the optimal treatment option with an acceptable safety profile for patients with advanced EGFR-mutated NSCLC who have progressed on TKIs. “CT + AMI + LAZ” and “CT + AMI” are alternative subsequent line options as well, with superior efficacy compared to immunotherapy-based or chemotherapy regimens, yet elevated toxicity profiles requiring vigilant management.
Background
Lung cancer is one of the malignant tumors with the highest incidence and mortality. 1 Non-small-cell lung cancer (NSCLC) is the most prevalent subtype, accounting for 85%–90% of all cases.2,3 Surgical resection stands as the most effective treatment for NSCLC. However, a significant proportion of patients, upon initial diagnosis, present with advanced or metastatic disease, thereby often precluding the possibility of surgical intervention, and with chemotherapy being the standard first-line treatment. 4 However, the 5-year survival rate for standard treatments is under 5%. 5
Advances in the past decade have substantially expanded treatment options for patients with advanced or metastatic NSCLC, notably with the advent of targeted therapies. 6 The treatment of NSCLC has evolved significantly from pathology-based to genotype-driven therapies, with a marked shift toward precision medicine. EGFR is the most frequently mutated driver gene in NSCLC, particularly among East Asian, non-smoking women, with mutation rates exceeding 50%.7,8 Targeted therapy with tyrosine kinase inhibitors (TKIs) has become standard care for advanced EGFR-mutated NSCLC in the first-line setting.9,10 Although the therapy is initially effective, acquired drug resistance is inevitable.11,12 Post-resistance, therapeutic options are indeed limited, and chemotherapy emerges as the primary alternative for patients who have developed resistance to EGFR-TKIs, but with limited efficacy. 13
The introduction of immunotherapy in lung cancer is another major milestone beyond targeted therapies.14,15 Multiple clinical trials have confirmed the survival benefit of immunotherapy for advanced NSCLC, making immunotherapy-based combination regimens the standard first-line regimen. 16 However, immunotherapy is limited in EGFR-mutated NSCLC. 17 Notably, some trials have indicated that immunotherapy can provide a survival benefit for EGFR-mutated NSCLC after TKI resistance. Results from the IMpower150 trial showed that the addition of immunotherapy to “chemotherapy + bevacizumab” as a later-line regimen significantly prolonged the survival of EGFR-mutated NSCLC. 18 The ORIENT-31 study demonstrated that a similar regimen (cisplatin, pemetrexed, sintilimab, and IBI305) was generally effective and well-tolerated in patients with EGFR-mutated NSCLC who had progressed after receiving EGFR-TKIs. 19 Inconsistently, there was no clinically meaningful survival improvement for patients receiving the regimen of “atezolizumab + bevacizumab + chemotherapy” versus “bevacizumab + chemotherapy” in the phase III trial of IMpower151. 20 Current guidelines endorse the use of immune checkpoint inhibitors (ICIs) for late-line treatment in EGFR-mutant NSCLC, yet their efficacy remains a contentious issue. The latest meta-analysis conducted by Zhao et al. 21 comprehensively included clinical trials and concluded that the four-drug regimen “ICI + anti-angiogenesis + chemotherapy” is indeed the optimal treatment option for individuals with advanced EGFR-mutated NSCLC who have progressed on EGFR-TKIs. However, the median progression-free survival (PFS) for patients who received the four-drug regimen was only approximately 6 months, leaving an urgent need for more effective regimens.
In recent years, numerous clinical trials have been exploring novel therapeutic agents for EGFR-mutated NSCLC patients who have developed resistance to TKIs. The MARIPOSA-2 trial is a paradigmatic study that compared the efficacy of amivantamab, an EGFR-mesenchymal–epithelial transition (MET) factor bispecific antibody, in combination with chemotherapy versus chemotherapy alone for patients with EGFR-mutated advanced NSCLC who experienced disease progression on osimertinib. Reports from the 2024 ESMO congress have indicated that the addition of amivantamab to chemotherapy significantly extended the survival of the patient. 22 The HARMONi-A trial evaluated the therapeutic efficacy and safety profile of another novel agent, ivonescimab, a first-in-class humanized tetravalent bispecific antibody targeting both programmed cell death protein 1 (PD-1) and vascular endothelial growth factor (VEGF). It was demonstrated that the addition of ivonescimab to chemotherapy achieved a median PFS of 7.1 months, significantly outperforming the 4.8 months observed with placebo plus chemotherapy. 23 Despite numerous therapeutic strategies having been investigated in the context of EGFR-mutated NSCLC patients post-TKI resistance, the optimal regimen remains elusive due to the lack of comparative studies directly assessing these alternatives.
In this paper, a comprehensive network meta-analysis (NMA) was conducted, synthesizing data from the most extensive clinical trials available, to evaluate the efficacy and safety of various treatment regimens for EGFR-mutated NSCLC patients who have progressed on EGFR-TKIs. This study aims to discern the most effective treatment option to guide clinical decision-making.
Methods
This NMA was performed by the Preferred Reporting Items for Systematic Review and Meta-analysis guidelines (Table S1). 24 The protocol was registered in the Prospective Register of Systematic Reviews (PROSPERO CRD42024601619).
Data sources and searches
PubMed, Embase, and the Cochrane Central Register of Controlled Trials were systematically searched for trials up to November 9, 2024, across all languages, using a combination of key search terms “non-small-cell lung cancer” and “EGFR,” limited to “randomized controlled trials.” To ensure the inclusion of comprehensive and updated data, abstracts and presentations from major international conferences (American Society of Clinical Oncology, European Society of Medical Oncology, European Cancer Conference, and World Conference on Lung Cancer) from 2020 to 2024 were also reviewed. The detailed search strategy is outlined in Table S2. The inclusion criteria and exclusion criteria were as follows:
Inclusion criteria
(1) Trials enrolled patients with histologically confirmed advanced (stage III/IV/relapsed) NSCLC harboring EGFR mutations.
(2) Trials compared at least two treatment arms for subsequent-line therapy in patients who had progressed on EGFR tyrosine-kinase inhibitors.
(3) Trials reported at least one outcome measure, such as PFS, overall survival (OS), objective response rate (ORR), or grade 3 or higher adverse events (⩾3AEs).
Exclusion criteria
(1) Trials in which the treatment was administered as adjuvant or neoadjuvant therapy.
(2) Trials that involved first-line treatment.
(3) Trials in which the number of patients in any single arm was less than 30.
Data extraction and risk of bias assessment
The primary endpoint of our analysis was PFS, with secondary endpoints including OS, ORR, and the incidence of ⩾3AEs. Data extraction and quality assessment were conducted independently by two researchers (Wengang Zhang and Yujie Li), with discrepancies resolved through consultation with a third, independent reviewer. The extracted data from the original studies comprised the study title, identifier, phase, publication year, patient characteristics, treatment regimens, and clinical outcomes such as PFS, OS, ORR, and ⩾3AEs. When multiple publications reported on the same trial, the most updated data were extracted. To reduce potential bias in the assessment, survival data from independent reviews took precedence. In this NMA, we utilized the Cochrane Risk of Bias Tool to assess potential biases within individual trials across several domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, handling of incomplete outcome data, selective reporting, and other sources of bias. 25
Inconsistency assessment
The node-splitting method was utilized to evaluate the consistency within the network, with a p-value threshold of less than 0.05 being considered indicative of significant inconsistency. 26
Data synthesis and statistical analysis
All statistical analyses for this study were conducted using R software, version 4.1.3, and Stata software, version 16.0. Stata was utilized to generate network diagrams that visually depicted direct comparisons among different treatment regimens. We pooled hazard ratios (HRs) for PFS and OS, as well as odds ratios (ORs) for the ORR, each with their corresponding 95% confidence intervals (CIs) from the randomized controlled trials (RCTs), to evaluate the relative efficacy of various treatment strategies. For safety assessments, ORs for ⩾3AEs and their 95% CIs were employed. The NMA was performed using the Markov Chain Monte Carlo simulation within the GEMTC package and the JAGS interface in R. 27 For PFS and OS, a dataset with 150,000 iterations was established, with the first 100,000 iterations discarded as burn-in and a thinning interval of 1 to enhance sample efficiency. For ORR and ⩾3AEs, the thinning interval was increased to 10 to reduce autocorrelation effects. The deviance was computed for each model. By comparing the deviance information criterion (DIC) values of different models, we chose the model with the lowest DIC value, as it indicates the best trade-off between model fit and complexity. 28 A fixed-effects consistency model was chosen for the NMA to ensure model stability and reliability. Convergence adequacy, ensuring a stable equilibrium distribution, was assessed through visual inspection of trace plots and computation of the Brooks–Gelman–Rubin statistic values. 29 Once convergence was confirmed, posterior distributions for the model parameters were derived. This allowed us to determine the probability of each treatment regimen being ranked in any given position. The surface under the cumulative ranking curve (SUCRA) was employed to rank and compare treatment regimens. A higher SUCRA score indicates superior efficacy for regimens regarding PFS, OS, and ORR, whereas a higher SUCRA score for ⩾3AEs implies an increased probability of these events occurring. 30
Results
Studies’ characteristics
Following a comprehensive selection process illustrated in Figure 1, our NMA included a total of 15 RCTs—12 phase III and 3 phase II—which involved 3177 patients and encompassed eight treatment regimens (Table 1). The specific regimens are chemotherapy + ivonescimab (CT + IVO), chemotherapy + amivantamab + lazertinib (CT + AMI + LAZ), chemotherapy + amivantamab (CT + AMI), chemotherapy + immunotherapy + bevacizumab (CT + IO + BEV), chemotherapy + bevacizumab (CT + BEV), chemotherapy + immunotherapy (CT + IO), chemotherapy (CT), and immunotherapy (IO), respectively. As depicted in Figure S1, the assessment of risk of bias generally indicated a low risk of bias across the enrolled RCTs.

Flowchart of study selection.
Baseline characteristics of studies.

NA, not available.
NMA of survival in overall patients
The network plots for PFS and OS, as presented in Figure 2(a) and (b), encompassed all patients (3177 individuals) from the enrolled RCTs for PFS and 3019 patients from 13 RCTs for OS. In terms of PFS, “CI + IVO” provided significant advantages compared with “CT + BEV” (HR 0.66, 95% CI 0.44–0.99), “CT + IO” (HR 0.60, 95% CI 0.43–0.82), “CT” (HR 0.46, 95% CI 0.34–0.62), and “IO” (HR 0.35, 95% CI 0.24–0.51; Figure 2(c)). Meanwhile, there was a discernible tendency favoring greater efficacy of “CI + IVO” over “CI + IO + BEV,” “CT + AMI,” but no statistical significance was observed (all HR < 1 but their 95% CI crossed 1; Figure 2(c)). “CT + AMI + LAZ” is superior to the regimen with the exception of “CI + IVO,” especially over “CT + IO” (HR 0.62, 95% CI 0.45–0.86; Figure 2(c)). A similar trend was observed for “CT + AMI.” Concerning “CT + IO + BEV,” it was significantly better than “CT + IO” (HR 0.71, 95% CI 0.58–0.89), and a tendency toward superiority over “CT + BEV” was also observed (HR < 1, 95% CI crossed 1), but did not reach a statistical difference (Figure 2(c)). All regimens were superior to “CT” and “IO” (all HR < 1 and their 95% CI did not cross 1; Figure 2(c)). When compared with “IO,” “CT” is more effective (HR 0.77, 95% CI 0.62–0.95), and the combination of “CT” and “IO” further extended PFS compared to “CT” alone (HR 0.77, 95% CI 0.67–0.88; Figure 2(c)). In terms of OS, comparisons among all regimens did not reach statistical differences. A slight tendency was observed for “CT + IVO” to be superior to other regimens, including “CT + IO + BEV” (HR < 1 but 95% CI crossed 1; Figure 2(c)).
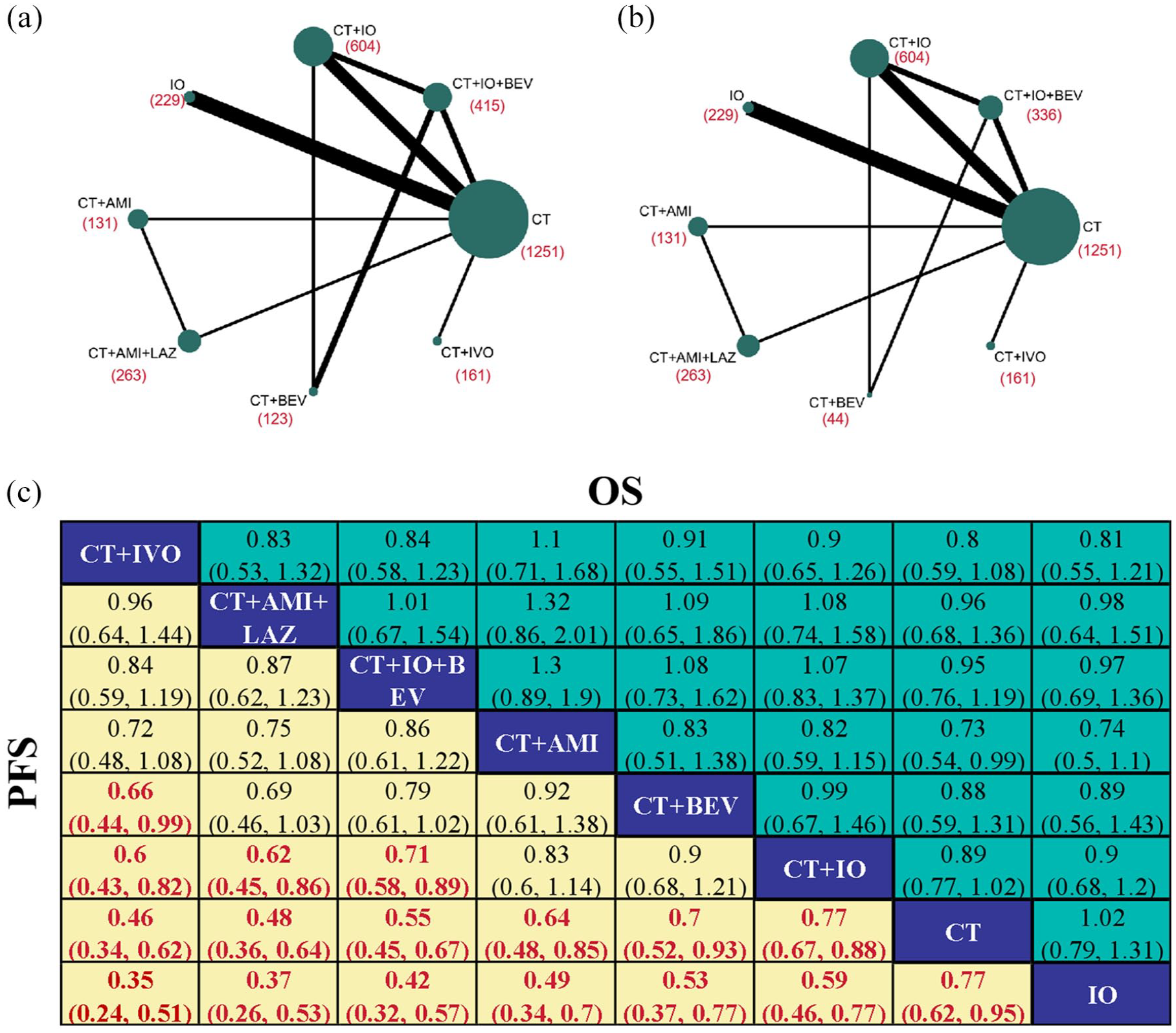
Pooled OS and PFS of multiple comparisons in network meta-analysis. (a and b) Network plots illustrating the direct and indirect comparisons for PFS (a) and OS (b). (c) Pooled HR (95% CI) for OS (upper triangle) and PFS (lower triangle). Data in each cell are HR (95% CI) for the comparison of the row-defining regimen versus the lower column-defining regimen. HR < 1 favors row-defining regimen. Significant results were bolded and highlighted in red.
NMA of response and safety in overall patients
The network plots included a total of 2826 participants from 11 RCTs for ORR and 2432 participants from 10 RCTs for DCR, as shown in Figure 3(a) and (b), respectively. In terms of ORR, the regimens “CT + AMI + LAZ,” “CT + AMI,” “CT + IO + BEV,” and “CT + IVO” were comparable, with no significant differences observed among them (Figure 3(c)). Of note, these four treatment regimens exhibited superior ORR versus “CT + IO” and “CT + BEV” (all OR > 1, most 95% CI did not cross 1; Figure 3(c)). When compared with “CT” and “IO,” the ORR was significantly higher in all regimens except for “CT + IO” and “CT + BEV” (all OR > 1, and all 95% CI did not cross 1; Figure 3(c)). In addition, the ORR of CT was significantly higher than that of “IO” (OR 4.45, 95% CI 2.17–9.81; Figure 3(c)). With respect to DCR, most of the tendency is similar to that of ORR. What was not observed in the ORR was that “CT + AMI + LAZ” (OR 2.26, 95% CI 1.07–4.85) and “CT + AMI” (OR 2.05, 95% CI 1.06–3.94) had significantly higher DCR than “CT + IO + BEV” (Figure 3(c)).
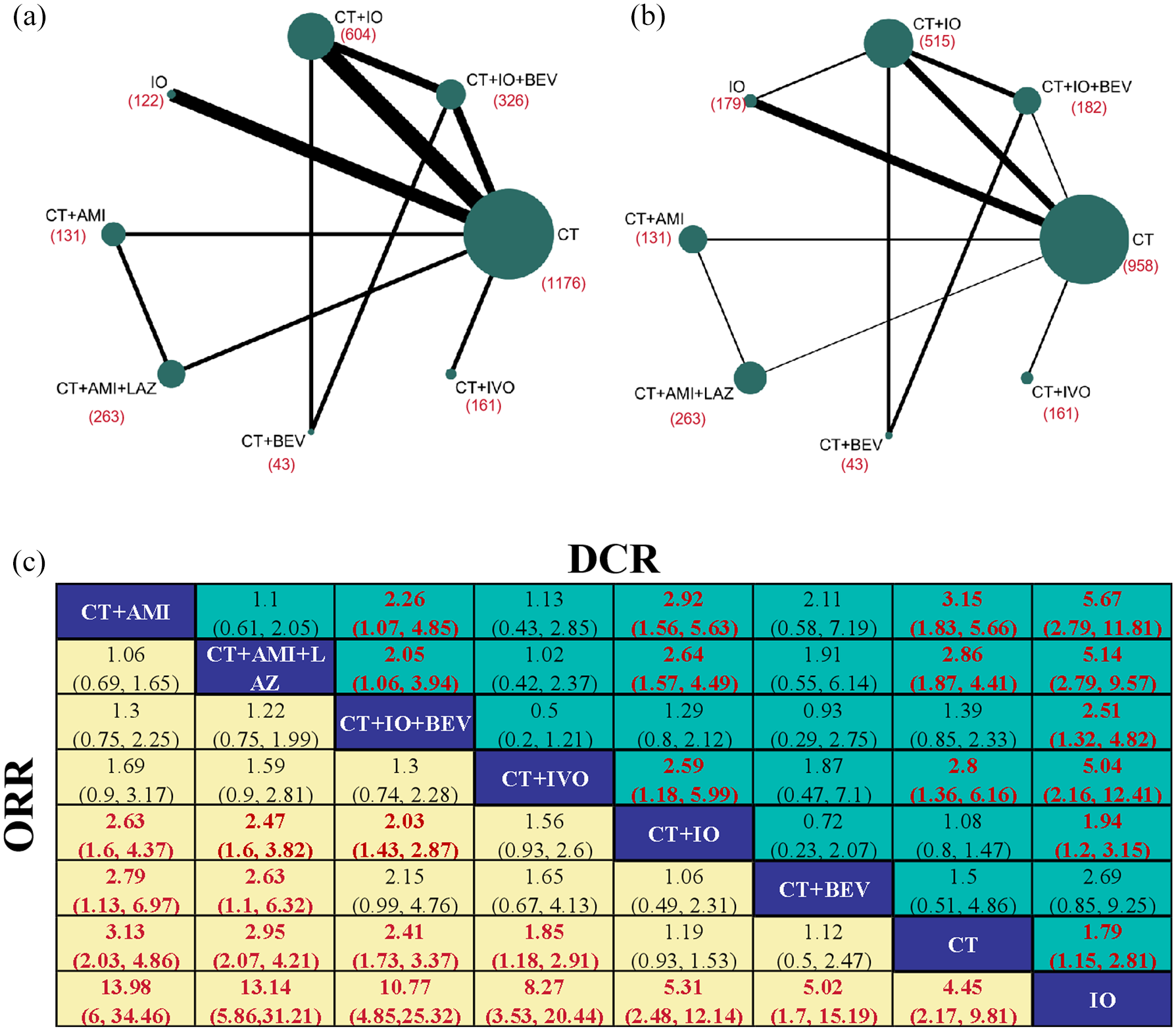
Pooled ORR and DCR of multiple comparisons in network meta-analysis. (a and b) Network plots illustrating the direct and indirect comparisons for ORR (a) and DCR (b). (c) Pooled OR (95% CI) for DCR (upper triangle) and ORR (lower triangle). Data in each cell are OR (95% CI) for the comparison of the row-defining regimen versus the lower column-defining regimen. OR more than 1 favors row-defining treatment. Significant results were bolded and highlighted in red.
As shown in Figure S2, the analysis of ⩾3AEs included 2432 patients from 10 trials. Of particular concern, AMI-based regimens (CT + AMI + LAZ and CT + AMI) had a significantly higher incidence of ⩾3AEs than other regimens. Besides, there were higher ⩾3AEs for “CT + IVO” (OR 1.66, 95% CI 1.07–2.6) and “CT + IO + BEV” (OR 1.37, 95% CI 1–1.88) versus “CT.” Nevertheless, no significant differences in ⩾3AEs were observed among “CT + IVO,” “CT + IO + BEV,” “CT + IO,” and “CT + BEV.”
Subgroup analysis based on EGFR mutation types
First, the efficacy of various regimens was compared based on EGFR-sensitive mutation subtypes. In terms of PFS, consistent with the overall analysis, the “CT + IVO” regimen was superior to others in both the 19DEL subgroup and the L858R subgroup (all HR < 1, and most 95% CI did not cross 1; Figure 4(a)). Compared with “CT” or “IO” alone, the combined regimens demonstrated a significant advantage in both the 19DEL and L858R subgroups (all HR < 1, and most 95% CI did not cross 1; Figure 4(a)). In terms of OS, four regimens were available: “CT + IO + BEV,” “CT + IO,” “CT,” and “CT,” which showed similar efficacy, aligning with the overall analysis (Figure S3(A)).

Subgroup network meta-analysis of PFS in EGFR-mutated NSCLC based on EGFR mutation subtypes. (a) Pooled HR (95% CI) for PFS of patients with 19DEL (lower triangle) and 21L858R (upper triangle) NSCLC. (b) Pooled HR (95% CI) for PFS of patients with T790M-negative (lower triangle) and T790M-positive (upper triangle) NSCLC.
Second, NMA was conducted based on T790M status. Significant disparities in PFS benefit were observed between the T790M positive and T790M negative across regimens, particularly for “CT + IVO” and “CT + IO + BEV” (Figure 4(b)). Specifically, compared to T790M-negative patients, “CT + IVO” more effectively extends PFS in T790M-positive patients (“CT + IVO” vs “CT + IO + BEV,” “CT + IO,” and “CT”: all HR < 1 and all 95% CI did not cross 1; Figure 4(b)). By contrast, “CT + IO + BEV” is more effective for T790M-negative NSCLC (“CT + IO + BEV” vs “CT + IO,” “CT,” and “IO”: all HR < 1 and all 95% CI did not cross 1; Figure 4(b)). OS results were consistent with the overall analysis, without significant differences among regimens (Figure S3(B)).
Subgroup analysis based on various clinicopathological characteristics
Age
Age-stratified analysis mirrored the overall results, showing that “CT + IVO” was superior in extending PFS across both age subgroups, followed by “CT + IO + BEV” (Figure S4(A)), while OS among regimens was comparable (Figure S4(B)).
Sex
Irrespective of gender, “CT + IVO” and “CT + IO + BEV” showed markedly better PFS outcomes compared to other regimens (all HR < 1 and most 95% CI did not cross 1; Figure S5(A)), while no statistical differences were found among the regimens in terms of OS (Figure S5(B)).
Eastern Cooperative Oncology Group
The results of the ECOG subgroup analysis were consistent with the overall analysis, with “CT + IVO” and “CT + IO + BEV” offering the best PFS prolongation both in ECOG PS = 0 and ECOG PS = 1 (Figure S6(A)), while the efficacy among various regimens was comparable regarding OS (Figure S6(B)).
Smoking
The subgroup NMA stratified by smoking history yielded results consistent with the overall analysis, demonstrating that combination therapies were superior to monotherapy in terms of PFS benefit, with “CT + IVO” being the most effective (Figure S7(A)), while the efficacy was comparable among various regimens with respect to OS (Figure S7(B)).
Brain metastasis
For both “CT + IVO” and “CT + IO + BEV,” stable prolongation of PFS was maintained across patients with and without brain metastases, outperforming “CT + IO,” “CT,” and “IO” (Figure S8(A)). By contrast, “CT + IO” showed variable PFS efficacy in different brain metastases subgroups, being more effective in those without brain metastases (Figure S8(A)). In line with the results of PFS, “CT + IO” exhibited divergent OS efficacy in patients with and without brain metastases, with the potential to be the best option for brain-free metastases (Figure S8(B)).
Live metastasis
The subgroup analysis focusing on liver metastasis indicates that the presence of liver metastasis is a pivotal determinant of therapeutic outcomes. To elaborate, the effectiveness of multiple regimens is markedly diminished in patients with liver metastasis as opposed to those without. In patients without liver metastasis, the regimens “CT + IVO” (HR 0.44, 95% CI 0.32–0.61), “CT + IO + BEV” (HR 0.52, 95% CI 0.38–0.72), and “CT + IO” (HR 0.69, 95% CI 0.53–0.90) showed enhanced PFS extension over CT alone, whereas in patients with liver metastasis, the efficacy of all regimens was found to be indistinguishable (Figure S9).
Rank probabilities
The Bayesian ranking probabilities for various regimens in overall patients and distinct subgroups regarding PFS, OS, ORR, DCR, and ⩾3AEs are illustrated in Figure 5, with outcomes aligning with those from the NMAs. Regarding PFS, “CT + IVO” emerged as the top-ranked regimen in the overall analysis, with its ranking remaining consistently stable across all subgroups (Figure 5(a)). Meanwhile, “CT + IVO” exhibited a manageable safety profile (ranking third for ⩾3AEs). “CT + AMI + LAZ” and “CT + AMI” were the top-ranked treatments for ORR and DCR, yet they also had the highest prevalence of ⩾3AE, securing the first and second ranks, respectively (Figure 5(a)). Subgroup data for “CT + AMI + LAZ” and “CT + AMI” were not available, thus precluding subgroup ranking. “CT + IO + BEV” ranked third in PFS, only after “CT + IVO” and “CT + AMI + LAZ,” and placed third in ORR, fifth in DCR, and fifth in the prevalence of ⩾3AE (Figure 5(a)). Subgroup analysis indicated that “CT + IO + BEV” is particularly suitable for T790M-negative patients (ranking first) and those with liver metastasis (ranking first; Figure 5(a)).

Bayesian ranking profiles of comparable treatments on efficacy and safety for patients with advanced EGFR-mutated NSCLC. (a) Number in each cell indicates the probability of each treatment being ranked from first to last on overall PFS, ORR, ⩾3AEs, and PFS for subgroups. (b) Number in each cell indicates the probability of each treatment being ranked from first to last on overall OS, ORR, ⩾3AEs, and OS for subgroups.
In terms of OS, “CT + AMI” unexpectedly secured the top rank, followed by “CT + IVO,” “CT + IO,” “CT + BEV,” “CT + IO + BEV,” and “CT + AMI + LAZ,” which were ranked second, third, fourth, fifth, and sixth, respectively (Figure 5(b)). Due to the unavailability of subgroup OS data for various regimens, the OS ranking was limited to only four treatment options: “CT + IO + BEV,” “CT + IO,” “CT,” and “IO” (Figure 5(b)).
Inconsistency assessment
The consistency model fit was superior to that of the inconsistency model across most comparisons, as detailed in Table S3. Inconsistency was evaluated using the node-splitting approach, which did not reveal significant differences in comparisons, except “CT” versus “IO” and “CT + IO” versus “IO” concerning DCR (Table S4). The trace plot and the Brooks–Gelman–Rubin diagnostic plot confirmed stable model convergence (Figures S10–S29).
Discussion
Currently, effective therapeutic options are lacking for patients with EGFR-mutated NSCLC after TKI resistance. Many clinical trials have explored the efficacy and safety of various treatment strategies, including chemotherapy, chemotherapy combined with anti-angiogenic therapy, immunotherapy, and the combination of chemotherapy with anti-angiogenic and immunotherapy, in the setting of TKI-resistant NSCLC. 41 In addition, RCTs such as MARIPOSA-2 and HARMONi-A have recently reported outcomes of novel therapeutic strategies.22,23 However, direct comparisons of efficacy and safety among these regimens are lacking, making it challenging for clinicians in clinical decision-making. In the study, we systematically collected and synthesized data from 14 RCTs to identify the optimal regimen for EGFR-mutated NSCLC patients who were resistant to TKIs. To the best of our knowledge, our study stands out as the most comprehensive to date, incorporating the broadest range of treatment regimens and pioneering the inclusion of the MARIPOSA-2 and HARMONi-A trials within an NMA to identify the most effective second-line treatment options.
The optimal regimen for EGFR-mutated NSCLC after TKI resistance remains a contentious issue, particularly regarding the benefits of an immunotherapy-based regimen for the patient. EGFR-mutated NSCLC is characterized by an immunosuppressive phenotype with higher infiltration of Tregs, myeloid-derived suppressor cells (MDSCs), and upregulation of PD-L1 expression. 42 Clinical trials corroborate this observation, demonstrating that EGFR-mutated NSCLC exhibits a limited response to immunotherapy. 43 Therefore, targeted therapy is preferred as the first-line treatment for EGFR-mutated NSCLC, while immunotherapy is not considered. 41 Interestingly, it was found that the tumor microenvironment of EGFR-mutated NSCLC dynamically evolves with targeted therapy, with a tendency to shift to an activated tumor immune microenvironment with higher immunogenicity following TKI treatment.44 –46 The IMpower150 subgroup analysis demonstrates that adding ICIs to the treatment regimen can significantly prolong survival for EGFR-mutated NSCLC patients who have progressed on TKI. 18 However, the subsequent phase III RCT, IMpower151, revealed that there was no significant difference in survival between patients who received ICIs and those who did not. 20 In this NMA, it was shown that the survival of patients treated with the four-drug regimen “CT + IO + BEV” was significantly longer than that of patients treated with “CT + BEV,” suggesting that immunotherapy does provide a survival benefit for TKI-resistant NSCLC patients, which is in line with the result of previous analysis. 21 Notably, the survival of patients receiving ICI alone was found to be inferior to that of those undergoing chemotherapy alone, reinforcing the evidence that the efficacy of immunotherapy is diminished in EGFR-mutated NSCLC compared to EGFR wild type. Therefore, ICIs should not be used alone for EGFR-mutated NSCLC, whether in the first-line setting or after TKI resistance.
Over recent years, the development of novel drugs has provided patients with additional therapeutic options. Angiogenesis is a hallmark of cancer, characterized by chaotic and leaky blood vessels that contribute to the formation of a hypoxic, acidic tumor microenvironment, which can promote tumor growth and metastasis. 47 Furthermore, the abnormal tumor vasculature can regulate cytokine release and immune cell infiltration, thereby creating an immunosuppressive microenvironment that limits the effectiveness of immunotherapy. 48 Consistently, evidence has confirmed that VEGF inhibitors can promote the infiltration of anti-tumor immune cells, thereby exerting an immune-enhancing effect. 45 The hypothesis is corroborated in EGFR-mutated NSCLC, with our NMA finding that the anti-angiogenic drug bevacizumab indeed augments the efficacy of ICIs. In addition to bevacizumab, novel bispecific antibody drugs that target VEGF have been developed. Ivonescimab (AK112/SMT112), the first-in-class humanized IgG1 bispecific antibody targeting PD-1 and VEGF, is a representative example. 49 The HARMONi-A (AK112-301) study has explored the efficacy and safety of ivonescimab combined with chemotherapy versus chemotherapy alone in patients with advanced EGFR-mutated NSCLC who experienced disease progression after EGFR-TKIs treatment. 23 The results demonstrated that the addition of ivonescimab to chemotherapy significantly improved survival outcomes with a tolerable and manageable safety profile. 23 Notably, our NMA revealed that “ivonescimab plus chemotherapy” is optimal among all regimens, outperforming the four-drug “CT + IO + BEV” regimen. Compared to the four-drug regimen, the “ivonescimab plus chemotherapy” combination involves fewer medications, is more convenient, imposes a lighter psychological burden on patients, and has a similar safety profile.
The MET factor is a key contributor to TKI bypass resistance mechanisms.50,51 Therefore, extensive research has been devoted to the development of drugs against MET. Amivantamab, possessing the ability to direct immune cell activity as an EGFR-MET bispecific antibody, stands out as the most successful case. 22 The MARIPOSA study has demonstrated that the combination of amivantamab and lazertinib exhibited superior efficacy compared to osimertinib in previously untreated EGFR-mutated advanced NSCLC. 22 The MARIPOSA-2 trial investigated the efficacy and safety of “amivantamab ± Lazertinib plus chemotherapy” as a second-line treatment for EGFR-mutated NSCLC patients who had progressed on osimertinib. The results demonstrated that the addition of “amivantamab ± Lazertinib” to chemotherapy significantly extended patients’ survival compared to chemotherapy alone. The result of our NMA revealed that the regimen of “amivantamab + Lazertinib + chemotherapy” ranked second in terms of PFS, inferior only to “CT + IVO” but superior to the four-drug regimen “CT + IO + BEV.” Besides, “amivantamab + chemotherapy” ranked first in both ORR and DCR. Equally of special concern is the fact that ⩾3AEs occurred more frequently in amivantamab-based regimens than in other regimens, with “amivantamab + Lazertinib + chemotherapy” ranking first and “amivantamab + chemotherapy” ranking second. Accordingly, “amivantamab ± Lazertinib + chemotherapy” does exhibit strong anti-tumor effects, but its safety profile warrants further evaluation.
Available evidence points to EGFR mutation subtypes as a critical factor influencing immunotherapy efficacy for EGFR-mutated NSCLC.52,53 Patients with T790M mutations were found to respond dramatically lower to immunotherapy compared to the EGFR-sensitive mutation. In our study, there was indeed a phenomenon that the T790M mutation affected the therapeutic effect of different regimens, with “CT + IO + BEV” exhibiting superior anti-tumor effects in T790M-negative NSCLC, whereas there was no superior survival benefit compared to “CT + IO” or “CT” in T790M-positive patients. By contrast, “CT + IVO” is superior in T790M-positive NSCLC. Interestingly, distinct EGFR-sensitive mutation subtypes were characterized by unique microenvironmental features, with a higher number of activated T cells observed in patients with EGFR L858R compared to those with EGFR 19 DEL. 54 Correspondingly, disparities in immunotherapeutic efficacy between 19DEL and L858R mutant NSCLC were observed in clinical practice. 52 By contrast, both “CT + IVO” and “CT + IO + BEV” provide relatively stable PFS benefits compared to other regimens in both 19DEL and L858R NSCLC populations in this NMA. To summarize, the type of EGFR mutation is a pivotal determinant of treatment efficacy, with the T790M mutation being particularly influential. For T790M-positive NSCLC, “CT + IVO” emerges as the preferred option, whereas “CT + IO + BEV” should be prioritized for T790M-negative NSCLC.
Growing evidence indicates that specific clinicopathological features, including brain and liver metastases, ECOG performance status, gender, and smoking history, are critical determinants of prognosis and treatment outcomes. 44 In the NMA, we performed subgroup analyses based on diverse clinicopathological characteristics, which demonstrated stable PFS benefits for “CT + IVO” and “CT + IO + BEV” across subgroups. However, the unavailability of subgroup data from multiple included trials precluded subgroup analyses for regimens such as “CT + AMI + LAZ” and “CT + AMI.” Consequently, the influence of specific pathological features on the efficacy of different treatment strategies requires further investigation in future studies.
There are still some limitations in our study. First, given that the primary endpoint in all included RCTs was PFS, and that OS outcomes were immature or not disclosed in some RCTs, our NMA prioritized PFS as the main outcome, with the result and discussion sections predominantly focusing on PFS. However, the prolongation of OS is the most critical indicator for patients and the ultimate objective for clinicians. Consequently, the efficacy of various treatment regimens in terms of OS merits deeper validation in future studies. Second, in this study, we categorized diverse chemotherapy regimens, encompassing single-agent and combinations of doublet chemotherapy, under a single regimen “CT,” which may introduce bias into the final NMA results. Third, there are regimens (i.e., there was only one trial available in terms of “CT + IVO,” “CT + AMI + LAZ,” and “CT + AMI”), with very few RCTs available, which can undermine the reliability of the analysis results.
Conclusion
Integrating the results of different clinical outcomes and subgroup analyses, we conclude that “CT + IVO” is the optimal treatment option with an acceptable safety profile for patients with advanced EGFR-mutated NSCLC who have progressed on TKIs. “CT + AMI + LAZ” and “CT + AMI” are alternative subsequent line options as well, with superior efficacy compared to immunotherapy-based or chemotherapy regimens, yet elevated toxicity profiles requiring vigilant management.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251338046 – Supplemental material for Efficacy and safety of distinct regimens for individuals with advanced EGFR-mutated non-small-cell lung cancer who progressed on EGFR tyrosine-kinase inhibitors: a systematic review and network meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251338046 for Efficacy and safety of distinct regimens for individuals with advanced EGFR-mutated non-small-cell lung cancer who progressed on EGFR tyrosine-kinase inhibitors: a systematic review and network meta-analysis by Wengang Zhang, Jian Xiong, Yujie Li, Jing Nie, Wencheng Zhao, Zhiyi Guo, Xinyue Liu, Qianqian Zhang, Xuyang Chen, Li Ye, Zhimin Chen, Hao Wang, Kandi Xu, Lishu Zhao, Yujin Liu, Lihua Huang, Yuhang Li and Yayi He in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251338046 – Supplemental material for Efficacy and safety of distinct regimens for individuals with advanced EGFR-mutated non-small-cell lung cancer who progressed on EGFR tyrosine-kinase inhibitors: a systematic review and network meta-analysis
Supplemental material, sj-pdf-1-tam-10.1177_17588359251338046 for Efficacy and safety of distinct regimens for individuals with advanced EGFR-mutated non-small-cell lung cancer who progressed on EGFR tyrosine-kinase inhibitors: a systematic review and network meta-analysis by Wengang Zhang, Jian Xiong, Yujie Li, Jing Nie, Wencheng Zhao, Zhiyi Guo, Xinyue Liu, Qianqian Zhang, Xuyang Chen, Li Ye, Zhimin Chen, Hao Wang, Kandi Xu, Lishu Zhao, Yujin Liu, Lihua Huang, Yuhang Li and Yayi He in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
