Abstract
Background:
The evolution of therapeutic landscape of human epidermal growth factor receptor-2 (HER2)-positive breast cancer (BC) has led to an unprecedented outcome improvement, even if the optimal sequence strategy is still debated. To address this issue and to provide a picture of the advancement of anti-HER2 treatments, we performed a large, multicenter, retrospective study of HER2-positive BC patients.
Methods:
The observational PANHER study included 1,328 HER2-positive advanced BC patients treated with HER2 blocking agents since June 2000 throughout July 2020. Endpoints of efficacy were progression-free survival (PFS) and overall survival (OS).
Results:
Patients who received a first-line pertuzumab-based regimen showed better PFS (p < 0.0001) and OS (p = 0.004) than those receiving other treatments. Median PFS and mOS from second-line starting were 8 and 28 months, without significant differences among various regimens. Pertuzumab-pretreated patients showed a mPFS and a mOS from second-line starting not significantly affected by type of second line, that is, T-DM1 or lapatinib/capecitabine (p = 0.80 and p = 0.45, respectively). Conversely, pertuzumab-naïve patients receiving second-line T-DM1 showed a significantly higher mPFS compared with that of patients treated with lapatinib/capecitabine (p = 0.004). Median OS from metastatic disease diagnosis was higher in patients treated with trastuzumab-based first line followed by second-line T-DM1 in comparison to pertuzumab-based first-line and second-line T-DM1 (p = 0.003), although these data might be partially influenced by more favorable prognostic characteristics of patients in the pre-pertuzumab era. No significant differences emerged when comparing patients treated with ‘old’ or ‘new’ drugs (p = 0.43), even though differences in the length of the follow-up between the two cohorts should be taken into account.
Conclusion:
Our results confirmed a relevant impact of first-line pertuzumab-based treatment and showed lower efficacy of second-line T-DM1 in trastuzumab/pertuzumab pretreated, as compared with pertuzumab-naïve patients. Our findings may help delineate a more appropriate therapeutic strategy in HER2-positive metastatic BC. Prospective randomized trials addressing this topic are awaited.
Introduction
The overexpression of the human epidermal growth factor receptor-2 (HER2) and inherent gene amplification is reported in 15–20% of breast cancers (BCs). HER2-amplified BCs are denoted by a biologically aggressive behavior, which translates into more frequent relapse and poor survival rates. 1 The monoclonal antibody trastuzumab has dramatically changed the natural history of HER2-positive BC, and has revolutionized the management of this subgroup of patients, both in the early and advanced settings.2,3 Subsequently, several anti-HER2 targeted therapies, such as pertuzumab, lapatinib, and ado-trastuzumab emtansine (T-DM1) have been developed and are now routinely used in the therapeutic management of HER2-positive BC patients. 4
The currently available guidelines in advanced HER2-positive BC recommend the sequential use of a first-line double blockade using trastuzumab plus pertuzumab and a taxane, according to the CLEOPATRA trial results,5–7 and a second-line treatment with T-DM1 based on the EMILIA trial results.8,9 Until recently, there has been no standard third-line treatment and patients have been usually treated with a combination of lapatinib/capecitabine, or a combination of trastuzumab and chemotherapy, or trastuzumab and lapatinib. 10 Despite the outstanding improvement in survival following the introduction of anti-HER2 therapies in the standard treatment of advanced disease, virtually, all these patients develop progressive disease and die. Several novel therapeutics are under development and have offered encouraging results, among them, the most representative are tucatinib or trastuzumab deruxtecan.11,12
When globally considered, results from registrative and prospective randomized trials are referred to extremely selected patients’ populations, who entered these trials if compliant with highly stringent inclusion criteria. Indeed, patients who receive treatments in the real-world setting may significantly differ from those enrolled in randomized clinical trials. Real-world patients may exhibit an older age, suffer with one or more relevant co-morbidities, and, within the advanced setting, present with a greater disease burden. These latter key patient- and disease-related features may at least partly explain the lack of full consistency between the evidence from several real-world studies and results from randomized prospective trials. Moreover, the rapid changes in standard treatments occurred over the last few years have left many unanswered questions, mainly concerning the best treatment sequence in real-world patient populations. In more detail, the amount of benefit obtained from the administration of a pertuzumab-based regimen in patients pretreated with adjuvant trastuzumab is not fully understood, since only 23% of the CLEOPATRA patients had received adjuvant trastuzumab.13–16 Another relevant issue is represented by the apparently lower T-DM1 efficacy when this agent is administered after a first-line pertuzumab-based regimen, since all patients enrolled in the EMILIA trial were pertuzumab-naïve.17–19
Unfortunately, prospective randomized studies evaluating the best sequence in advanced HER2-positive BC patients are lacking, and sequential treatments are delivered according to approval and registration timing.
In order to discern these issues, we performed a large multicentric, observational study with a retrospective design of HER2-positive advanced BC patients treated with HER2 blocking agents since June 2000 throughout July 2020.
Patients and methods
Study approval
The PANHER study is a multicenter, observational trial with a retrospective design including a large case series of HER2-positive advanced BC patients recruited in a 20-year time window.
This study was approved by the Institutional Review Board (IRB) of the coordinating center, that is, the IRCCS Regina Elena National Cancer Institute of Rome, Italy [reference number Rs1331/20(2370)], as well as from the IRBs of all the participating centers (Supplementary Material). The PANHER trial was conducted according to Helsinki Declaration. A specific consent form was conceived for patients who contributed data to our study, and individually administered to those who were still alive at the time we performed our analysis. Consent administration took place either in the course of follow-up visits or over telephone calls. For patients who had died at the time of data analysis or who were missed to the follow-up, attempts were made to reach the patients’ relatives and request permission to proceed.
As previously pointed out, the PANHER trial is fed by some relevant and thus far unanswered questions. Our trial primarily aimed to explore the efficacy of the anti-HER2 agents and their administration sequence and, secondarily, to provide a picture of the evolution of anti-HER2 treatments in advanced BC in a real-world population.
Patients’ selection
We retrospectively identified HER2-positive advanced BC patients who received anti-HER2-based therapies for advanced disease from June 2000 to July 2020, according to routine practice at 34 Italian oncologic centers.
We evaluated the efficacy of anti-HER2 agents with respect to the line of treatment administration and in relation to the specific sequence in which they were delivered. The anti-HER2 agents’ efficacy was also estimated by dividing patients into a cohort A, including those who were treated exclusively with ‘older drugs’, such as trastuzumab and lapatinib, and a cohort B, which included patients who received also ‘new drugs’, such as pertuzumab and T-DM1.
Chemotherapy, anti-HER2 agents, and endocrine therapy were administered according to national guidelines, outside of clinical trials. Treatment efficacy was evaluated according to standard practice, every 3 months, and responses defined by clinician interpretations of imaging reports and symptomatic criteria, not strictly and always applying RECIST criteria.
All included patients were treated for a HER2-positive advanced disease. Median follow-up was calculated starting from the diagnosis of metastatic disease to death or date of last follow-up. Endpoints of efficacy were progression-free survival (PFS) and overall survival (OS). Progression-free survival was calculated for any treatment lines from the time of treatment start to the date of disease progression, interruption for toxicity, death, or loss to follow-up. Overall survival was calculated from the diagnosis of metastatic disease to death or last follow-up, and, for some specific treatments, from the start of the treatment to death, or date at the last follow-up.
Data collection
Medical records were retrieved on demographic, clinical, and molecular data, as well as information regarding the treatments received, their sequence and related outcomes. Following anonymization, data were entered into an ad hoc database with an operating interface with an SPSS software for subsequent analysis. Pathology assessment was locally performed in surgical specimens of primary tumors or in biopsies of metastatic lesions, as per national standard. When missing, molecular features were centrally evaluated by the coordinator center in formalin-fixed, paraffin-embedded tissue sections. Estrogen and progesterone receptor (ER and PgR) status were determined at each center by immunohistochemistry (IHC) according to the local standards. Positivity was considered at a cut-off of ⩾1%. HER2 status was evaluated by IHC and an expression level 3+ was defined positive. Fluorescence, chromogenic, or silver in situ hybridization was performed to identify HER2 amplification in case of HER2 level 2+ staining at IHC. All the evaluations were performed according to the most updated ASCO-CAP guidelines at the time of diagnosis or at the re-evaluation whenever feasible.20–22
Statistical analysis
A descriptive analysis including clinical and demographic characteristics of the patients was performed by estimating the median and the range for continuous variables and the absolute value and relative frequencies for categorical variables. Chi-square, Kruskal–Wallis, and Fisher exact tests were used to evaluate possible associations between categorical variables. Overall survival and PFS were calculated by the Kaplan–Meier product-limit method. Log-rank or Tarone-Ware tests were used to assess differences between subgroups. Follow-up was estimated with Kaplan–Meier reverse method. The SPSS® (21.0) statistical software was used for all analyses.
Results from the overall patient population
Patient and disease characteristics
From June 2000 to July 2020, 1,328 HER2-positive advanced BC patients who received HER2-targeted treatments at 34 Italian oncologic centers were retrospectively identified. The main baseline characteristics of our study patients are listed in Table 1. Briefly, median age at initial diagnosis was 52 years (range, 24–88), median Eastern Cooperative Oncology Group (ECOG) PS status was 1 (range, 0–3), 564 (42.5%) patients were premenopausal, and 764 (57.5%) were postmenopausal. Numbers and percentages of patients whose tumors expressed hormonal receptors are first reported by single, specific receptor, that is, ER or PgR, and then by contemporary presence/lack of both receptors, that is, defining the IHC subtype. Overall, in 680 patients (51.2%), HER2-positive tumors also expressed both hormonal receptors positive, that is, triple positive (TP) disease, while in 202 patients (15.2%) the disease exclusively showed either ER or PgR. In 397 cases (29.9%), both hormone receptors were not expressed. Among the 830 non-metastatic patients at their first diagnosis, 484 (58.3%) had previously received a neo-/adjuvant trastuzumab-based regimen. Four hundred and ninety-eight (37.5%) patients were first diagnosed with metastatic disease. Two hundred and twenty-nine patients had re-biopsy of metastatic lesions. Concerning sites and number of metastases at the diagnosis of metastatic disease, briefly 293 (22.1%) patients had metastatic involvement of three or more sites, 786 (59.2%) had visceral metastases, 168 (12.7%) bone-only metastases, and 126 (9.5%) had brain metastases at first diagnosis.
Clinicopathological characteristics of the study participants (N = 1,328).

ECOG, Eastern Cooperative Oncology Group; ER, estrogen receptor; N, Number; PgR, progesterone receptor; TP, triple positive.
For patients with early disease at diagnosis (N: 830 patients).
Treatments received are summarized in Table 2. Most patients had received multiple treatment lines for advanced disease, the majority of which including a HER2-blocking agent. The median number of treatment lines for advanced disease was 2 (range, 1–12). All patients received a trastuzumab-based regimen at some point in their clinical history. Overall, 404 patients also received a lapatinib/capecitabine treatment. Of them, 362 (89.6%) were treated in first, second, or third line of treatment, while the remaining part (N = 42; 10.4%) received this treatment in fourth or subsequent line. Starting from 2014, patients could also have received T-DM1 and pertuzumab-based therapies. In more detail, 532 (40.1%) patients were treated with T-DM1, mostly in second line, and 755 (56.9%) of them received a pertuzumab-based regimen almost exclusively in first-line setting.
Treatments administered in first, second, and third line (N = 1328).
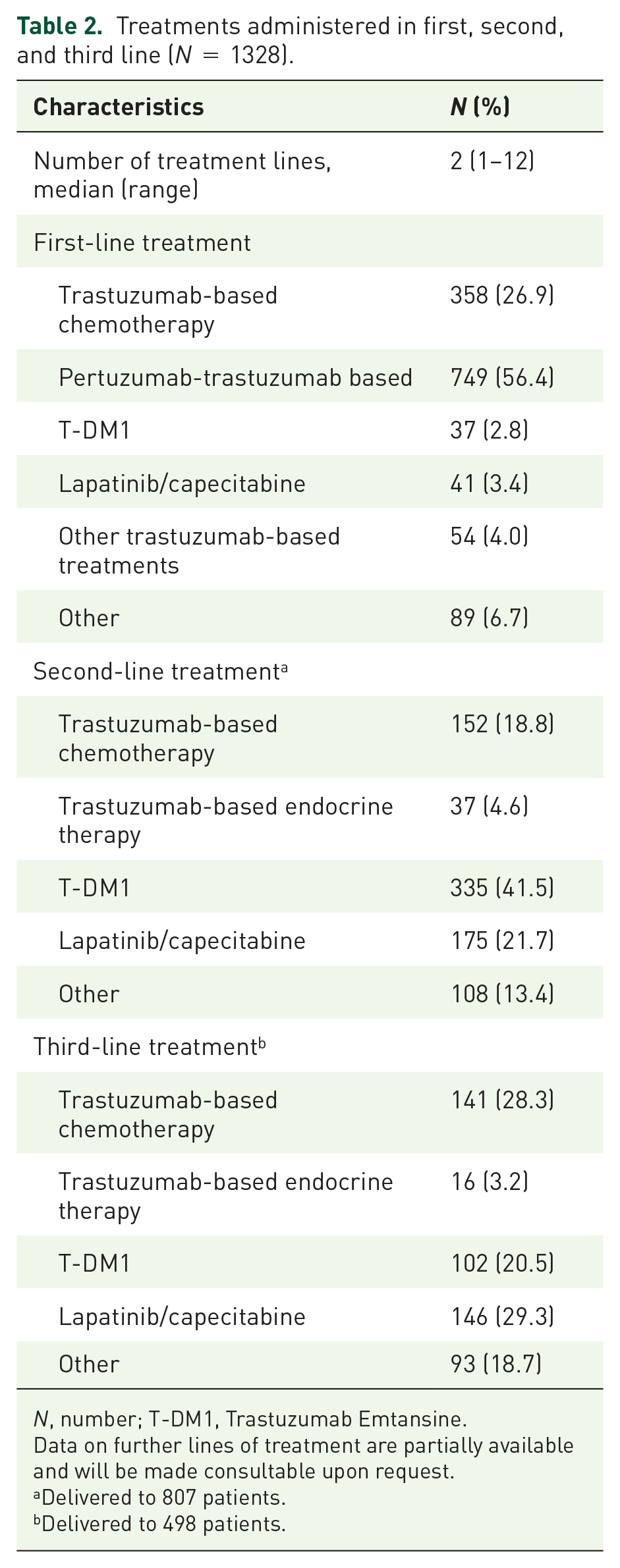
N, number; T-DM1, Trastuzumab Emtansine.
Data on further lines of treatment are partially available and will be made consultable upon request.
Delivered to 807 patients.
Delivered to 498 patients.
First-line treatment, available for all patients, was trastuzumab chemotherapy in 358 (26.9%) patients, pertuzumab/trastuzumab/taxane in 749 (56.4%) patients, T-DM1 in 37 (2.8%) patients, lapatinib/capecitabine in 41 (3.4%) patients, and other treatments, for example, endocrine therapy + trastuzumab, in 143 (10.7%) patients. Among the 749 patients treated with first-line pertuzumab-based regimen, 419 (55.9%) did not have metastasis at diagnosis and 265 (35.4%) of them had previously received trastuzumab in the early setting.
A second-line therapy was delivered to 807 patients (60.8%), and it was represented by T-DM1 in 335 (41.5%) patients, lapatinib/capecitabine in 175 (21.7%) patients, trastuzumab plus chemotherapy in 152 (18.8%) patients, trastuzumab plus endocrine therapy in 37 (4.6%) patients, and other treatments in 108 (13.4%) patients.
A third-line therapy was recorded in 498 (37.5%) patients. Lapatinib/capecitabine was the most frequently delivered regimen (146, 29.3%), while T-DM1 was administered in third line to 102 (20.5%) patients. Fourth and later lines of therapy were extremely heterogeneous.
In Table 3, we also report data on baseline patients’ characteristics by subgroups defined upon treatment sequence. A special focus is on the exposure to a pertuzumab-including regimen in first line, followed by second-line T-DM1 or lapatinib/capecitabine.
Clinicopathological characteristics of the study participants by treatment in first and second line (N: 510).

Group 1, first-line pertuzumab-based treatment and second-line T-DM1; Group 2, first-line trastuzumab-based treatment and second-line T-DM1; Group 3, first-line pertuzumab-based treatment and second-line lapatinib/capecitabine; Group 4, first-line trastuzumab-based treatment and second-line lapatinib/capecitabine; N, number; Pts, patients.
Groups were as it follows:
Kruskal–Wallis test.
For patients with early disease at diagnosis (510 patients).
Results from stratified analysis
The median follow-up for the whole patient population was 52 months (95% CI, 48–56).
Concerning first-line treatment, 749 patients who received a pertuzumab/trastuzumab/taxane treatment showed a 2-year PFS of 50.5% and a median PFS of 25 months (95% CI, 20.8–29.2). Conversely, 412 patients who received a trastuzumab-based first-line treatment had a 2-year PFS of 30.5% and a median PFS to first line of 14 months (95% CI, 12.2–15.6, p = 0.0001) (Figure 1(a)). In addition, when comparing the outcomes of 749 patients treated with a first-line pertuzumab-based regimen with the 358 patients treated with trastuzumab added to chemotherapy, the 2-year PFS was 50.5% and 34.6%, and the median PFS values were 25 (95% CI, 21–29) and 17 (95% CI, 15–19) months, respectively (p < 0.0001, Figure 1(b)).
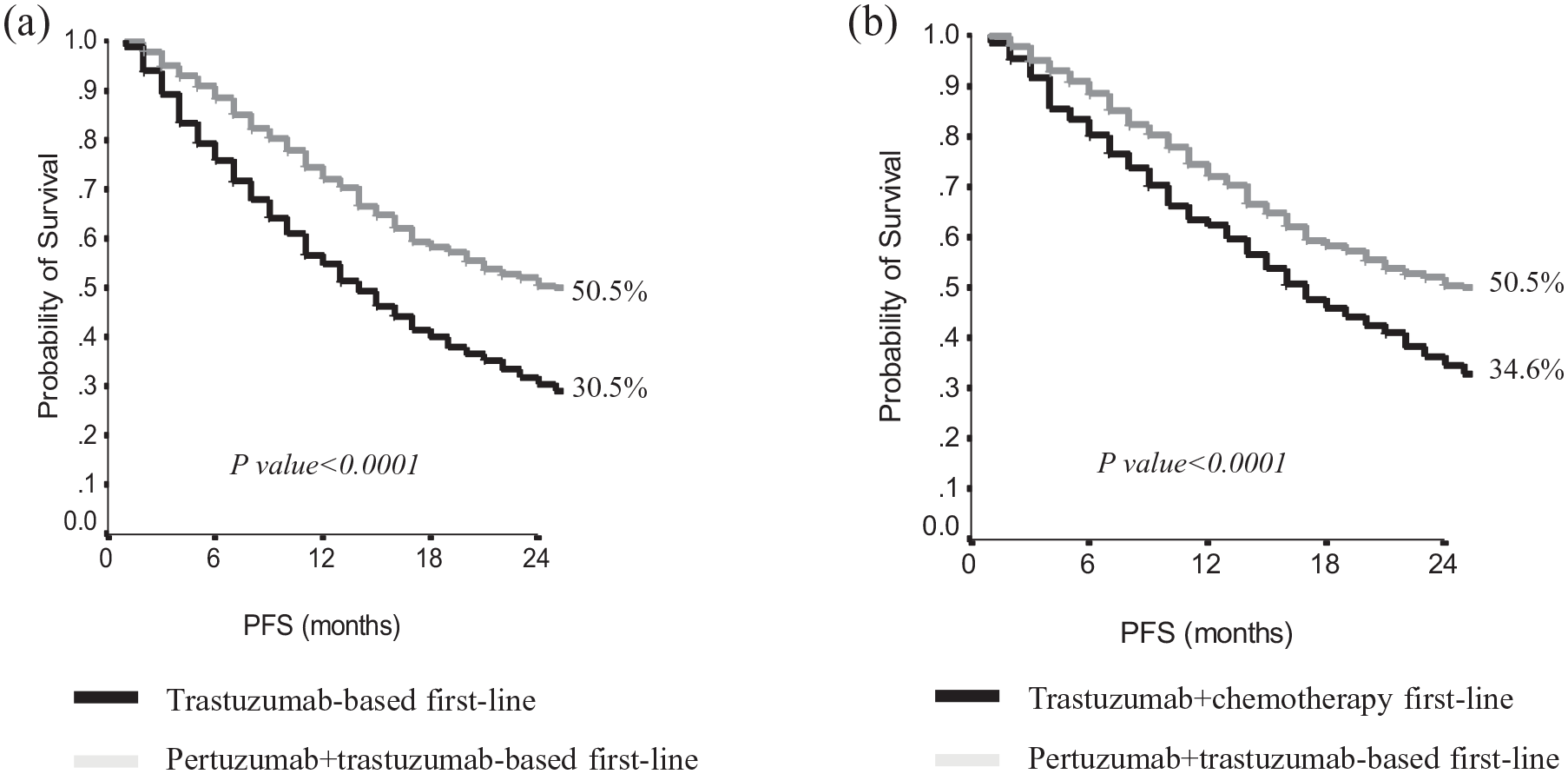
First-line progression-free survivalwhen comparing pertuzumab + trastuzumab – based treatment to trastuzumab – based chemotherapy (a), and to any trastuzumab – based treatment (b).
Among the 419 patients who received a pertuzumab-based first-line therapy following disease recurrence, the 2-year PFS and the median PFS were 43.8% and 19 months (95% CI, 14–24) in patients pretreated with neo-/adjuvant trastuzumab and 49.9% and 24 months (95% CI, 15–33) in trastuzumab-naïve patients (p = 0.10), respectively.
Second-line treatment, whichever it was, was administered in 807 patients, with a 2-year PFS of 20% and a median PFS of 8 months (95% CI, 7.1–8.7). Considering the therapeutic agents administered as second line, patients treated with T-DM1 showed a 2-year PFS of 24.4% and a median PFS of 8 months, compared with 17% and a 9 months in patients who received other second-line treatments. The difference observed was not statistically relevant (p = 0.22). When focusing on second-line T-DM1, pertuzumab-pretreated patients showed a 2-year PFS of 22.6% with a median PFS to T-DM1 of 7 months (95% CI, 5.1–8.9), versus 30.3% and 12 months (95% CI, 5.8–18.2) in pertuzumab-naïve patients (p = 0.01, Figure 2).
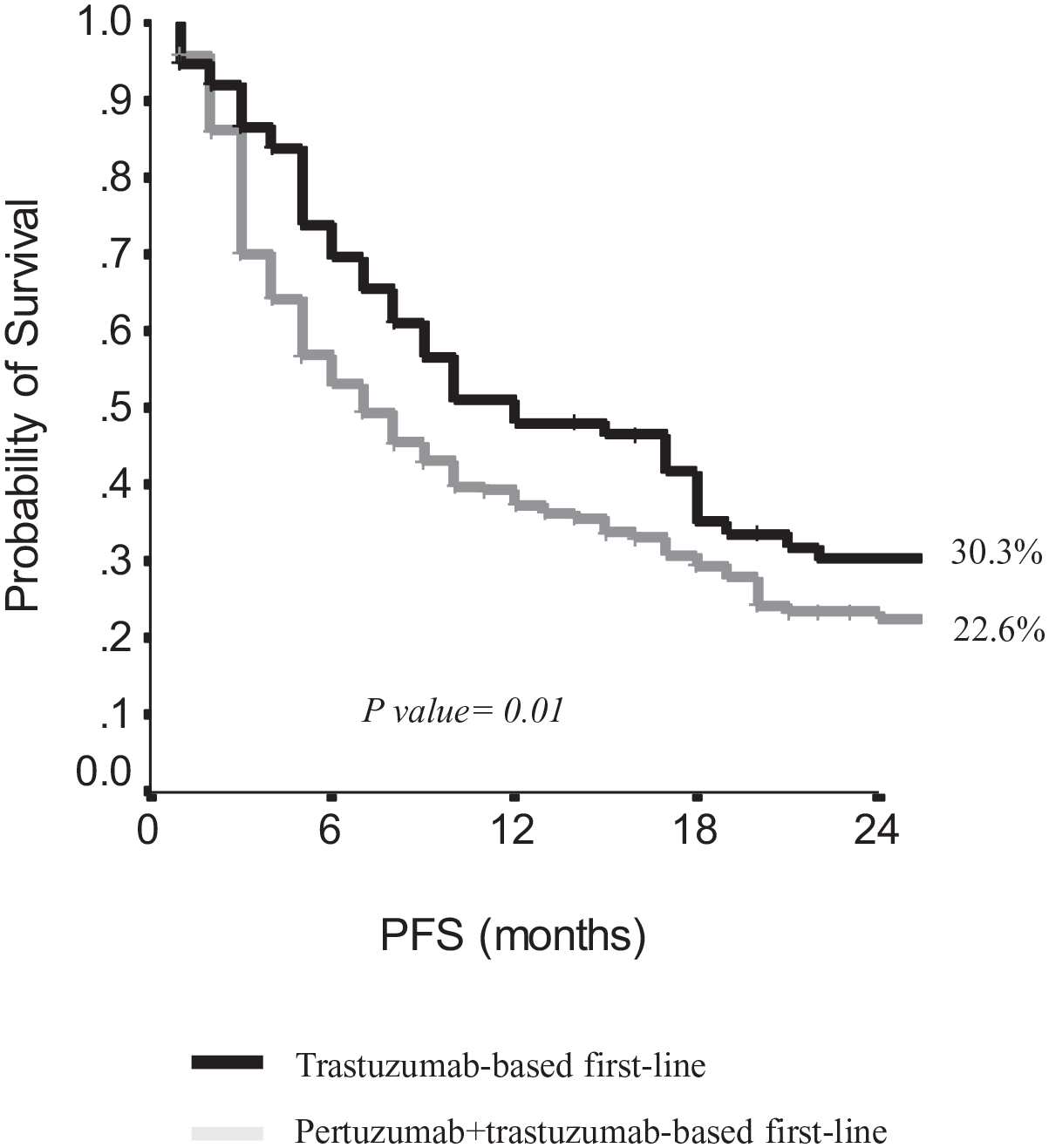
Progression-free survival to second-line TDM-1 in pertuzumab-naïve or pretreated patients.
Patients treated with first-line pertuzumab-based regimens and second-line lapatinib/capecitabine or T-DM1 showed a median PFS to second line of 7 months in both subgroups (95% CI, 2.1–11.9 and 5.1–8.9, respectively; p = 0.80). Conversely, patients who received a trastuzumab-based first-line regimen followed by T-DM1 as second line showed a median PFS to second line of 12 months (95% CI, 5.8–18.2) compared with a median PFS to second line of 8 months (95% CI, 6.4–9.6; p = 0.004) observed in patients who received second-line lapatinib/capecitabine (Figure 3).
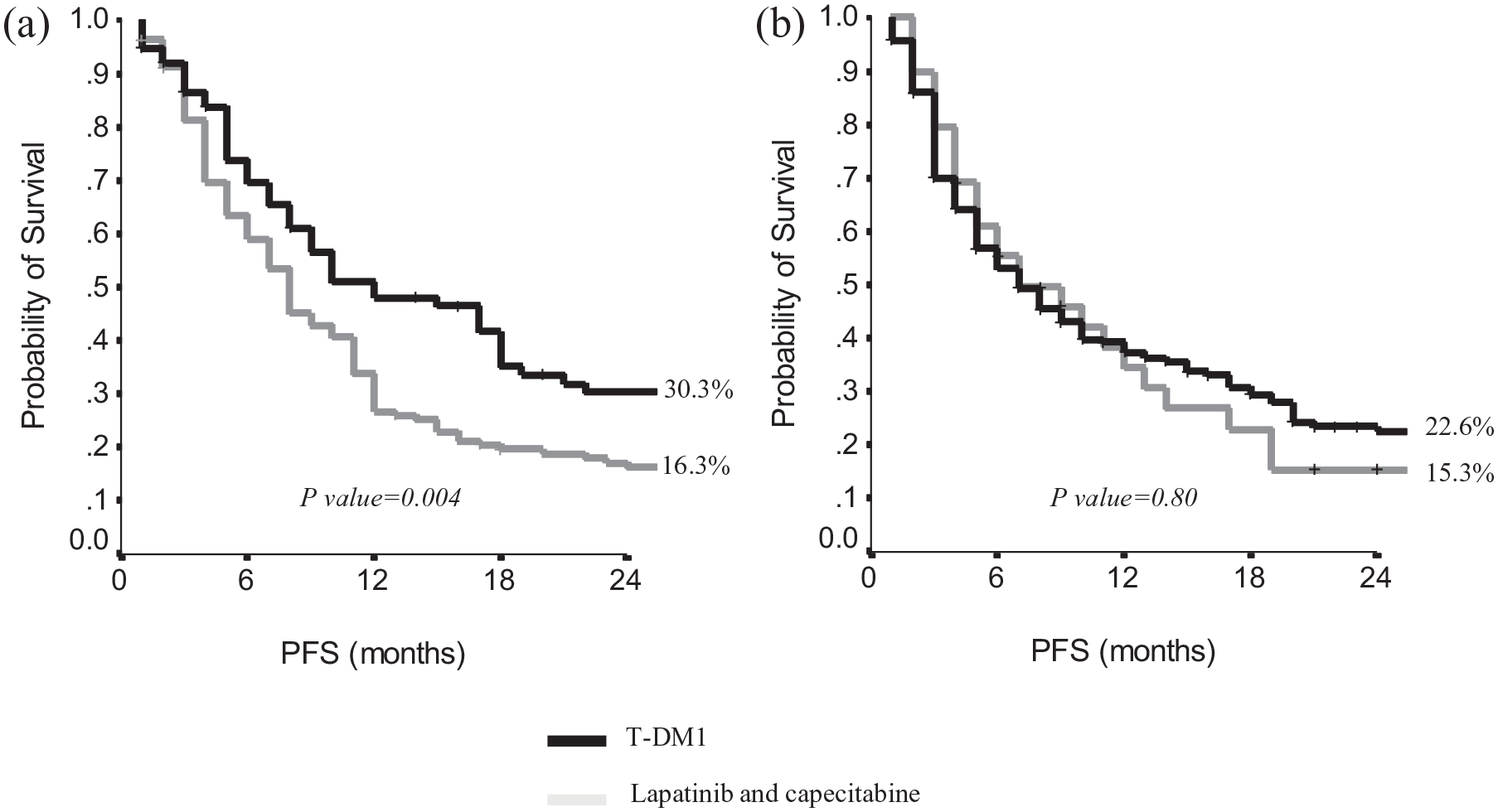
Progression-free survival to second-line T-DM1 or lapatinib/capecitabine in pertuzumab-naïve patients (a) versus pertuzumab-pretreated patients (b).
Overall, third-line treatment showed a 2-year PFS of 8.5% and a median PFS of 7 months (95% CI, 6.3–7.7), with no significant differences by treatments administered.
In the whole patient population, the median OS was 60 months (95% CI, 55–65), with a 3-year OS of 68.4% and a 5-year OS of 49.7%.
We then computed OS following stratification of the entire cohort by time period, and, consequently, types of treatments received. The following cohorts were obtained: (a) ‘old’ drugs, that is, trastuzumab and lapatinib, for the time-window June 2000–June 2014. This cohort included 478 patients; and (b) ‘new’ drugs, that is, pertuzumab and T-DM1, for the time-window July 2014–July 2020. This cohort included 850 patients. The 3-year OS for patients within the first and second cohort was 69% (cohort A) and 67.9% (cohort B), respectively, without significant differences (p = 0.43). However, the median follow-up of the cohort B is still considerably short (35 months, 95% CI = 32–38) compared with that of cohort A (109 months, 95% CI = 99.3–118). This significantly impairs our ability to reliably report on late recurrences for patients in the cohort B.
Among the 419 patients with early BC at diagnosis and treated with a pertuzumab-trastuzumab-based first-line therapy for advanced disease, no statistically significant difference in OS emerged by trastuzumab pretreatment. We observed a 3-year OS, 5-year OS, and median OS from the diagnosis of metastatic disease of 67.5%, 56.9%, and 95 (95% CI, 47–143) months in trastuzumab-pretreated patients versus 65%, 52%, and 63 (95% CI, 43–83) months in trastuzumab-naïve patients (p = 0.79).
Among the 498 patients with de novo metastases, the 3-year and 5-year OS were 71.4% and 52.3%, with a median OS of 62 months (95% CI, 55.5–68.5). When analyzing the subgroup of 830 patients with recurrent disease, 3- and 5-year OS were 66.7% and 48.1%, with a median OS of 57 months (95% CI, 49.2–64.8). Overall, results are not statistically different (p = 0.33). If stratifying according to type of first-line treatment received, patients with de novo metastases who had received pertuzumab showed a 3- and 5-year OS of 77% and 58.7%, with a median OS of 71 months (95% CI, 64.5–77.5). Among patients having received trastuzumab-based first-line, 3- and 5-year OS were 64.6% and 46.2%, with a median OS of 56 months (95% CI, 47.5–64.5). The difference is statistically significant (p = 0.01). Similarly, patients whose disease recurred and who had received first-line pertuzumab showed a 3- and 5-year OS of 66.9% and 54.6%, with a median OS of 70 months (95% CI, 52.5–87.5). This latter estimate was significantly more favorable than that observed in patients who received trastuzumab-based first line, showing 3- and 5-year OS of 65.8% and 45.2%, with a median OS of 55 months (95% CI, 47.4–62.6) (p = 0.01).
Among the 1328 patients who provided data to our analysis, when focusing on outcomes from the diagnosis of metastatic disease, the 3-year OS, 5-year OS, and median OS were 71.4%, 56.4%, and 70 months (95% CI, 60–80) in patients who received first-line pertuzumab/trastuzumab/taxane versus 65.4%, 45.5%, and 55 months (95% CI, 49–61) in patients treated with regimens not including pertuzumab (p = 0.004; Figure 4).

Overall survival from diagnosis of metastatic disease by specific first-line treatment.
Overall, among the 807 patients who received second-line treatment, median OS from second-line starting was 28 months (95% CI, 24.8–31.1), with a 41.0% 3-year OS and 24.8% 5-year OS. Among the 749 patients who had received first-line pertuzumab, those who received second-line T-DM1 (N: 259) had a 3-year OS of 63.6% and a 5-year OS of 39.5%, with a median OS from the diagnosis of metastatic disease of 48 months (95% CI, 42–54). Patients who received, after first-line pertuzumab, a lapatinib/capecitabine treatment had a 3-year OS of 51.8% and a 5-year OS of 32.2%, with a median OS of 41 months (95% CI, 28–54; p = 0.45; Figure 5). Moreover, in pertuzumab-naïve patients receiving second-line T-DM1, 3-year OS was 78%, 5-year OS 55.4%, with a median OS from the diagnosis of metastatic disease of 72 months (95% CI, 44–100), statistically significantly different from pertuzumab-pretreated patients (p = 0.003, Figure 6).

Overall survival from second-line starting of pertuzumab-pretreated patients according to type of second-line treatment.
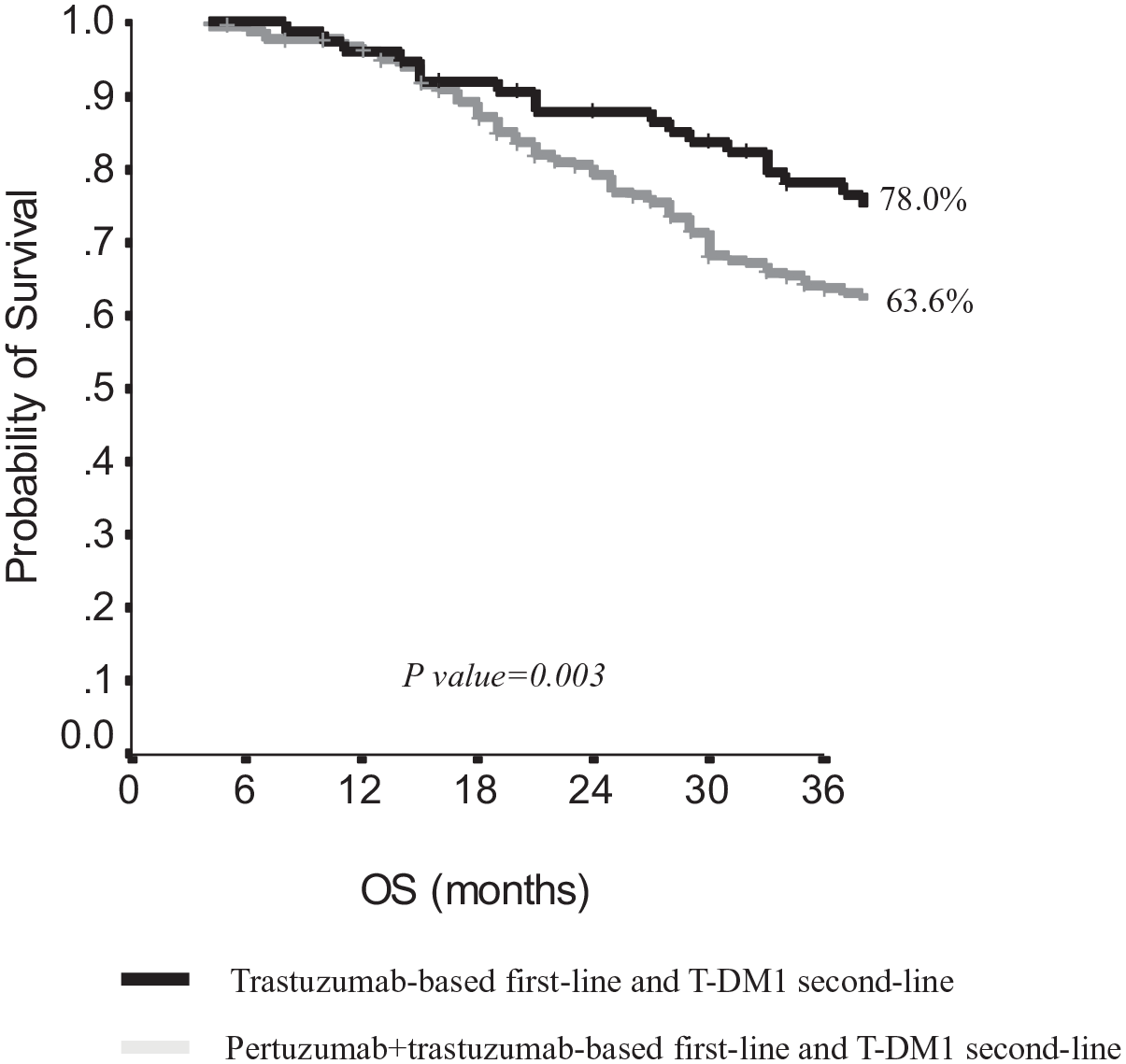
Overall survival from the diagnosis of metastatic disease of patients treated with second-line T-DM1 according to first-line treatment.
When analyzing OS calculated from second-line starting, independently on the type of first-line treatment, patients treated with second-line T-DM1 (325 patients) had a 3-year OS of 40.8%, a 5-year OS of 26.9%, with a median OS of 29 months (95% CI, 23–35). Patients treated with second-line lapatinib/capecitabine had a 3-year OS of 39.7% and a 5-year OS of 23.4%, with a median OS of 30 months (95% CI, 24.6–35.4). The difference observed was not statistically relevant (p = 0.59).
When OS was computed from starting of second-line T-DM1 according to previous pertuzumab or not, in pertuzumab pretreated patients 3-year OS was 32.2% and 5-year OS was 24.4%, with a median OS of 24 months (95% CI, 19–29); conversely, in pertuzumab-naïve patients, the 3-year OS was 58.9%, 5-year OS was 38.5%, with a median OS of 45 months (95% CI, 36–54, p = 0.002) (Figure 7). Moreover, in patients who received following first-line pertuzumab, a lapatinib/capecitabine regimen, the 3-year OS was 25.8%, the 5-year OS was 25.8%, with a median OS of 25 months (95% CI, 21–29), without significant difference with respect to patients who received T-DM1 in the second-line setting (p = 0.92).

Overall survival from second-line T-DM1 according to first-line treatment.
We then focused on treatment outcomes by hormonal receptor status. Overall survival calculated from the diagnosis of metastatic disease was as it follows: in patients with ER- and PgR-negative disease, the 3-year OS was 62.4%, the 5-year OS was 42.2%, with a median OS of 53 months (95% CI, 47–59). Conversely, in patients with ER- and PgR-positive tumors, the 3-year OS was 70.7%, the 5-year OS was 52.2%, with a median OS of 64 months (95% CI, 58–70, p = 0.004).
Among the 1328 patients recruited, 786 (59.2%) had visceral involvement when first diagnosed with advanced disease. Median OS starting from metastatic disease diagnosis was 53 months (95% CI, 47–59) in patients with visceral metastases versus 70 months (95% CI, 63–77) in the absence of visceral involvement (p = 0.001).
Discussion
The introduction of anti-HER2 agents in clinical practice has dramatically changed the outcomes of patients with HER2-positive disease. Currently, the standard first-line therapy for patients with HER2-positive metastatic BC is HER2 double-block with pertuzumab/trastuzumab plus a taxane. Following progression, the standard of care is currently trastuzumab emtansine (T-DM1). More recently, promising new drugs have being emerging as effective options for later lines, including tucatinib and trastuzumab deruxtecan. Finding the best treatment sequence at the individual patient level is of utmost importance to maximally optimize patient outcomes and quality of life. The present study provides a reliable ‘picture’ of the anti-HER2 treatment strategy choices starting from 2000 throughout 2020 at 34 Italian oncologic centers. All the 1,328 HER2-positive patients included in this analysis had received at least one line of HER2-based treatment for metastatic disease. Data analysis was performed to explore the effectiveness of anti-HER2 treatments in a real-world context across a remarkably wide time window, and to assess whether the choice of treatment sequence, mainly in first and second lines, was associated with significant differences in terms of treatment outcomes.
As expected, within our study population, patients who received a pertuzumab-based regimen as first-line treatment showed better PFS (p < 0.0001) and OS (p = 0.004) than those receiving other treatments (Figures 1 and 4, respectively). Second-line treatment was administered to 807 patients, with a median PFS of 8 months and a median OS from second-line starting of 28 months, without significant differences among the numerous schemes considered. Third-line treatment was delivered to 498 patients, with a median PFS of 7 months. Focusing on pertuzumab-pretreated patients, we found that median PFS to second-line therapy and median OS from second-line starting did not significantly differed by type of second-line treatment, that is, T-DM1 or lapatinib/capecitabine (p = 0.80 and p = 0.45, respectively; Figures 3(b) and 5). Conversely, pertuzumab-naïve patients treated with T-DM1 as second line showed a significantly better median PFS to second line with respect to those treated with lapatinib/capecitabine (p = 0.004, Figure 3(a)). Interestingly, median OS from diagnosis of metastatic disease was higher in patients treated with trastuzumab-based first-line followed by second-line T-DM1 in comparison to those who received pertuzumab-based first-line treatment and second-line T-DM1 (p = 0.003, Figure 6). This latter result could be at least partially explained by the possibly intrinsic less aggressive characteristics of the disease of patients in the first group, since patients treated in the pre-pertuzumab era, and more precisely, treated before 2014, seem to have a longer disease course which may have allowed treatment in second-line with T-DM1 after its approval.
Among patients treated in third line with T-DM1, we found a statistically significant PFS advantage in pertuzumab-naïve patients with respect to pertuzumab-pretreated (8 versus 4 months, p = 0.009).
Furthermore, we analyzed patients’ outcomes following stratification by time period and types of treatments received. No significant differences emerged when comparing patients treated with ‘old’ or ‘new’ drugs (p = 0.43), presumably due to the fact that the median follow-up of the second group is still considerably short. Moreover, no significant PFS or OS differences emerged when we analyzed data concerning patients who received neo-/adjuvant trastuzumab.
As expected, patients with hormonal receptor positive tumors had more favorable outcomes with respect to patients with hormonal receptor negative tumors. Moreover, patients with non-visceral metastases showed higher OS in comparison to patients with visceral disease.
In recent years, we have witnessed radical changes in the therapeutic landscape of HER2-positive metastatic BC. The CLEOPATRA trial is a cornerstone of the inherent literature, whose results support the use of pertuzumab and trastuzumab in combination with a taxane as the gold-standard in first line.5–7 Thus far, a quite limited number of studies have evaluated the use of double-block in combination with taxanes in the real-world setting. A real-world oncology practice analysis including 221 patients treated with pertuzumab, trastuzumab, and docetaxel as first line has been recently published. 23 The median PFS was 16.9 months (95% CI, 13.9–20.1), and the estimated PFS rate at 1 year was 62.5% (95% CI, 54.2–69.7). These results are fairly similar to those reported in the pivotal trial. Our collaborative group has carried out a real-world, multicentric retrospective observational study (RePER) on first-line treatment with pertuzumab-trastuzumab and taxane in 264 advanced HER2-positive BC patients. 15 Overall, our results are consistent with those from the CLEOPATRA trial, with a response rate of 77.3% and a median PFS of 21 months.
An additional real-world study, based on the French ESME cohort, reported on survival trends in advanced BC including the time period where pertuzumab and T-DM1 were introduced. 24 They observed steadily improving survival rates in patients diagnosed from 2008 through 2014. The investigators hypothesized that their findings might have been related to the market release of new HER2-targeted drugs, although they did not formally test this. Indeed, the impact of pertuzumab and T-DM1 may have been at least partly minimized by the extremely recent introduction of these two drugs at the time of results’ evaluation. Recently, a collaborative group from the Netherlands has examined whether the real-world OS improved in patients with HER2-positive advanced BC since the market release of pertuzumab and T-DM1. 25 The SOutheast Netherlands Advanced BREast cancer (SONABRE) registry included 493 systemically treated patients, who had been consecutively diagnosed with HER2-positive metastatic BC between 2008 and 2017. Outcome data confirmed that the survival of patients with HER2-positive advanced BC had significantly improved since the introduction of pertuzumab and T-DM1.
The RegistHER is a multicenter, observational, prospective study including more than 1,000 newly diagnosed HER2-positive advanced BC from US community and academic settings treated between the years 2003 and 2006 in a real-world setting. Data on outcome from a subset of 530 patients with HR positive tumors were analyzed, and patients having received as first-line treatment trastuzumab plus endocrine therapy had significantly more favorable median PFS with respect to patients exclusively receiving endocrine therapy (HR = 0.37). The subgroup receiving first-line chemotherapy and trastuzumab plus endocrine therapy had longer median PFS (HR = 0.53) and OS (HR = 0.50) in comparison with patients not receiving endocrine therapy. The sequential use of chemotherapy followed by endocrine therapy was associated with a remarkable advantage in OS compared with concomitant administration (HR = 0.48). 26 Among the various reports from the RegistHER study, interesting results have come from the analysis of patients with de novo versus recurrent HER2-positive BC. Patients with de novo metastatic disease showed more favorable clinical outcomes than those with recurrent disease. 27 In the PANHER study, the OS outcomes do not significantly differed between de novo metastatic and patients with recurrent disease (p = 0.33). However, the administration of a pertuzumab-based first-line treatment positively influenced OS in both these subgroups compared with patients having received trastuzumab-based first-line treatment (p = 0.01).
The SystHER is a further US multicentric prospective observational cohort study enrolling patients with HER2-positive advanced BC, starting in 2012 throughout 2016. 28 Nine hundred forty-eight patients received a first-line treatment, including only trastuzumab, double-block with trastuzumab and pertuzumab, or none of the available anti-HER2 agents, with or without chemotherapy and endocrine therapy. Most patients (711) received first-line treatment with trastuzumab, pertuzumab, and taxane, showing a median OS of 53.8 months and a median PFS of 15.8 months, thus resembling results from the CLEOPATRA trial. 29 In the present study, the subset of 749 patients having received pertuzumab/trastuzumab/taxane as first-line treatment showed an apparently more favorable outcome than those recruited in the SystHER study, with a median PFS of 25 months, and a median OS of 70 months. This may at least partially relate to the more favorable disease characteristics of the PANHER study participants. In more detail, in our patients, visceral metastases were less represented (59.2% versus 67.1%) and the disease burden was lower, with 22.1% of patients showing at least three metastatic sites in the PANHER study versus 32.6% in the SystHER study.
Current guidelines indicate T-DM1 as the preferred second-line treatment in HER2-positive advanced BC patients, according to EMILIA trial results. However, the impact of sequence of administration of the drugs in current use remains a matter of debate.
The issue of a possible decrease in T-DM1 efficacy if given immediately after the double pertuzumab-based HER2 block has not been exhaustively addressed in previous studies.8,26 The EMILIA 8 and TH3RESA 30 trials were the two pivotal randomized phase III clinical studies that brought T-DM1 as a standard of care as second-line or beyond for patients with HER2-positive advanced BC that progressed to standard first-line treatments. The TH3RESA trial showed clinical advantage by using T-DM1 compared with treatment of choice by the clinician also in patients that had received lapatinib/capecitabine, while the EMILIA trial showed superiority of T-DM1 in second-line when compared head to head with the lapatinib/capecitabine regimen. Unfortunately, the patient population of the EMILIA and TH3RESA trials does not reflect current clinical cohorts, since in both these randomized clinical trials, patients enrolled were pertuzumab-naïve.
To date, there are very few reported studies describing the use of T-DM1 in the real-world setting. Dzimitrowicz et al. 18 assessed the efficacy of T-DM1 in routine clinical practice in a patient population identified via electronic pharmacy records. Overall, 96% of patients received prior trastuzumab and pertuzumab, and 23% were also treated with lapatinib before T-DM1. Among the 78 patients who contributed data to this analysis, 32% received T-DM1 as first or second-line, 21% as third-line, and 48% as fourth-line or beyond. Median duration on therapy was 4 months, and response rate was 18%. These results are less favorable than those reported in the EMILIA trial, 8 although more comparable with the findings from the TH3RESA, the TDM4258 g, and TDM4374 g trials, which enrolled more heavily pretreated patients.30–32
The T-DM1 Patient Access Study (T-PAS) reported data for the first 215 enrolled patients. T-DM1 was given after a median number of 8 prior systemic therapies for HER2-positive metastatic BC (range: 3–23). 33 Median T-DM1 treatment duration was 5 months, and overall response rate was 26%. Overall, 19% of patients received more than 18 cycles. Authors concluded that T-DM1 was efficacious in the real-world setting, with no new safety concerns.
A retrospective mono-institutional analysis including 128 patients treated with T-DM1 in a real-world setting at The Royal Marsden has been recently published. 34 Overall, survival outcomes were less favorable (20.4 months) compared with those from the registrative trials (EMILIA: 29.9 months; TH3RESA: 22.7 months) as would be expected in a less selected population. This study included almost one third of patients who had received prior pertuzumab-based first-line treatment.
Few years ago, our group carried out a real-world, multicenter, observational study with a retrospective design. 17 In this latter trial, we aimed at testing T-DM1 efficacy in a non-selected cancer patient population of 250 HER2-positive metastatic BC to yield evidence in support of its use in real-world practice. In this analysis, T-DM1 efficacy seems independent on the line of treatment in pertuzumab-naïve patients, whereas, in pertuzumab-pretreated patients, T-DM1 seems to have lower effectiveness when administered immediately after pertuzumab-based treatment.
Within this same research pipeline, we carried out a further study (SePHER) which also included experiments on the exposure to different drug sequences including pertuzumab and T-DM1 in HER2-positive cell lines. The study confirmed the apparently lower effectiveness of T-DM1 when given after pertuzumab in 371 retrospectively recruited patients, and results from HER2-positive cell lines support the hypothesis that the trastuzumab/pertuzumab double-block might reduce the amount of available plasma membrane HER2 receptor, so limiting the binding of T-DM1 in cancer cells. 19 Another possible explanation of the lower efficacy of T-DM1 when given after pertuzumab-based combinations might be a bias of patient selection, since patients treated with trastuzumab/chemotherapy first-line before pertuzumab approval and having received subsequently T-DM1 might have had an intrinsically less aggressive disease, which allowed to control disease evolution until the approval of T-DM1.
The loss of HER2 receptor expression has been reported in surgical samples after HER2-targeted neoadjuvant treatment in tumors that were HER2-positive at the initial biopsy in up to 33% of the cases, and this phenomenon does not seem related to worst long-term outcomes.35–37 In the Katherine study, the outcome of patients treated with adjuvant T-DM1 for residual disease after neoadjuvant treatment does not seem less favorable in patients with HER2-negative residual tumors with respect to HER2-positive residual tumors, so indirectly suggesting an efficacy of T-DM1 even in this patient subgroup. 38 However, we have to consider that the settings analyzed are different, as well as treatment sequences, since in the papers reporting HER2 receptor changes none of patients have been treated with previous pertuzumab and subsequently T-DM1, whereas in the present study and in the SePHER study the administration of T-DM1 shortly follows HER2 double-block including pertuzumab.
The main limitations of the present study rely on its observational retrospective nature, which implies that patients’ heterogeneity and differences in timing and methods of response evaluation could have introduced bias in response and PFS analysis. Still, such a study design conferred the ability to span over an extremely wide time window and to analyze data from a considerable number of patients diagnosed and treated for a HER2-positive metastatic BC. The issue related to the lack of centralization in HER2 status assessment deserves mentioning. As previously reported, HER2 status assessment was generally performed locally, that is, at each of the 34 centers involved. Inter-pathologists agreement on HER2 scoring has been demonstrated to be potentially suboptimal, especially for 1+ and 2+ IHC score. 39 In addition, interpretation of in situ hybridization results has changed through time.20–22 We considered the possibility to retrospectively reassess all samples for HER2 status according to the latest ASCO/CAP guidelines. Unfortunately, the pre-existing logistic difficulties emerged in the attempt of coordinating all the centers involved became totally unsolvable due to the current pandemic. However, as in our prior work, we are confident in our data quality due to the quality controls in place at the labs involved. 22 In conclusion, the analysis of real-world data to answer clinical and policy-relevant questions that cannot be directly or completely answered using data from randomized clinical trials has gained increasing interest in recent years.40–43 Notably, while data coming from clinical trials are prospectively collected and verified with well-established rules and procedures, which should guarantee acceptable quality of data and minimize bias, the collection of real-world data poses several methodological problems, for instance, in terms of both data sources and data verification. In our view, among the numerous applications for analysis of real-world data proposed in recent years, the most interesting is to produce useful data in terms of treatment sequence. Indeed, one of the major limitations of the evidence produced by randomized clinical trials is that most trials are focused on the comparison of treatments within a specific line of therapy and are not designed to allow comparisons of sequences. Patients treated in a second-line trial could have not necessarily received the current first-line standard treatment, and patients treated in a first-line trial could have not necessarily received, after disease progression, the currently available second-line standard treatment. On this basis, real-world analysis could integrate the evidence of randomized clinical trials, especially in those treatment settings characterized by the recent introduction of therapeutic news.
Our study also has considerable strengths. The collaborative effort of numerous Italian oncologic centers has led to the inclusion of over a thousand patients, making this trial one of the largest ever conducted in this area.
Overall, our findings confirmed a clinically relevant impact of first-line pertuzumab-based treatment. At the same time, our results showed lower second-line T-DM1 efficacy in terms of long-term outcomes in patients who received trastuzumab/pertuzumab in first-line, as compared with pertuzumab-naïve patients.
Although results from prospective randomized trials addressing this topic are eagerly awaited, our findings, as well as those from similar studies previously conducted, are fundamental to optimally delineate the appropriate therapeutic sequence in patients with HER2-positive BC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359211059873 – Supplemental material for PANHER study: a 20-year treatment outcome analysis from a multicentre observational study of HER2-positive advanced breast cancer patients from the real-world setting
Supplemental material, sj-docx-1-tam-10.1177_17588359211059873 for PANHER study: a 20-year treatment outcome analysis from a multicentre observational study of HER2-positive advanced breast cancer patients from the real-world setting by Laura Pizzuti, Eriseld Krasniqi, Isabella Sperduti, Maddalena Barba, Teresa Gamucci, Maria Mauri, Enzo Maria Veltri, Icro Meattini, Rossana Berardi, Francesca Sofia Di Lisa, Clara Natoli, Mirco Pistelli, Laura Iezzi, Emanuela Risi, Nicola D’Ostilio, Silverio Tomao, Corrado Ficorella, Katia Cannita, Ferdinando Riccardi, Alessandra Cassano, Emilio Bria, Maria Agnese Fabbri, Marco Mazzotta, Giacomo Barchiesi, Andrea Botticelli, Giuliana D’Auria, Anna Ceribelli, Andrea Michelotti, Antonio Russo, Beatrice Taurelli Salimbeni, Giuseppina Sarobba, Francesco Giotta, Ida Paris, Rosa Saltarelli, Daniele Marinelli, Domenico Corsi, Elisabetta Maria Capomolla, Valentina Sini, Luca Moscetti, Lucia Mentuccia, Giuseppe Tonini, Mimma Raffaele, Luca Marchetti, Mauro Minelli, Enzo Maria Ruggeri, Paola Scavina, Olivia Bacciu, Nello Salesi, Lorenzo Livi, Nicola Tinari, Antonino Grassadonia, Angelo Fedele Scinto, Rosalinda Rossi, Maria Rosaria Valerio, Elisabetta Landucci, Simonetta Stani, Beatrice Fratini, Marcello Maugeri-Saccà, Michele De Tursi, Angela Maione, Daniele Santini, Armando Orlandi, Vito Lorusso, Enrico Cortesi, Giuseppe Sanguineti, Paola Pinnarò, Federico Cappuzzo, Lorenza Landi, Claudio Botti, Federica Tomao, Sonia Cappelli, Giulia Bon, Fabio Pelle, Flavia Cavicchi, Elena Fiorio, Jennifer Foglietta, Simone Scagnoli, Paolo Marchetti, Gennaro Ciliberto and Patrizia Vici in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank Dr Alessandro Zennaro and the ‘Consorzio Inter-Universitario Nazionale per la Bio-Oncologia’ (CINBO) for administrative and technical support.
Author contributions
LP and PV conceived and designed the study. EK, TG, MM, MMaz, EMV, IM, RB, FSDL, CN, MP, LI, ER, NDO, ST, CF, KC, FR, AS, EB, MAF, GB, AB, GDA, AC, AM, AR, BTS, GS, FG, IP, RS, DM, DC, EMC, VS, LM, LMe, GT, MR, MMi, EMR, PS, NS, LL, NT, AG, AFS, RR, MRV, EL, SS, BF, MMS, MDT, AMa, DS, AO, VL, EC, FC, LL, CB, SC, FP, FCav, OB, EF, JF, SS, and PM made substantial contributions to the acquisition of data. IS and MB analyzed the data. LP, EK, MB, and PV interpreted the data and drafted the manuscript. LM, GS, PP, FT, GB, and GC critically revised the manuscript. All authors reviewed the work for important intellectual content.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EK, IS, MB, MM, MMaz, EMV, IM, RB, FSDL, MP, LI, ER, NDO, ST, CF, KC, FR, AC, MAF, GB, AB, GDA, AC, AR, BTS, GS, FG, RS, DM, DC, EMC, VS, LMe, GT, MR, LM, MMi, EMR, PS, NS, LL, NT, AG, AFS, RR, MRV, EL, SS, BF, MMS, MDT, AM, AO, VL, EC, GS, PP, FC, LL, CB, FT, SC, GB, FP, FCav, OB, EF, JF, SS, and GC declare no conflicts of interest. LP received travel grants from Eisai, Roche, Pfizer, and Novartis; speaker fees from Roche, Pfizer, Novartis, and Gentili. TG received travel grants from Eisai, Roche, Pfizer, and Novartis; speaker fees/advisory boards from Roche, Pfizer, Novartis, Gentili, and Lilly. CN received travel grants/personal fees from Pfizer, Eisai, Novartis, Merck Sharp & Dohme, and AstraZeneca. EB is supported by the Italian Association for Cancer Research AIRC-IG 20583; he was supported by the International Association for Lung Cancer (IASLC), the LILT (Lega Italiana per la Lotta contro I Tumori), and Fondazione Cariverona; he received speakers’ and travels’ fee from MSD, AstraZeneca, Celgene, Pfizer, Helsinn, Eli Lilly, BMS, Novartis, and Roche; consultant’s fee from Roche, Pfizer; institutional research grants from AstraZeneca and Roche. AM received travel grants from Eisai, Celgene, and Novartis Ipsen; personal fees, advisory boards from Eisai, Novartis, AstraZeneca, Teva, Pfizer, and Celgene. IP received personal fees/advisory boards from Roche, Pfizer, Novartis, Italfarmaco, Gentili, and Pierre Fabre. LM received personal fees/advisory board from Roche, Novartis, Eisai, and Pfizer. GT and DS: advisory board Novartis, Pfizer, Eisai, Roche, and Eli Lilly. PM has/had a consultant/advisory role for BMS, Roche Genentech, MSD, Novartis, Amgen, Merck Serono, Pierre Fabre, and Incyte. PV received travel grants from Eisai, Roche, Pfizer, and Novartis; speaker fees/advisory boards from Roche, Pfizer, Novartis, Gentili, and Eli Lilly.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board in each patient’s enrolling institution.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
