Abstract
The combined use of stereotactic ablative radiotherapy (SABR) and immune checkpoint inhibitors (ICIs) is an emerging treatment paradigm for oligometastatic non-small-cell lung cancer (NSCLC). Recent phase I and II trial data suggest that SABR to multiple metastases in addition to ICI use is safe and effective with promising progression-free survival and overall survival signals. There is great interest in capitalizing on combined immunomodulation from these two modalities for the treatment of oligometastatic NSCLC. Ongoing trials seek to validate the safety, efficacy, and preferred sequencing of SABR and ICI. This narrative review of the role of SABR when combined with ICI in oligometastatic NSCLC discusses the rationale for this bimodality treatment, summarizes recent clinical trial evidence, and proposes key principles of management based on the available evidence.
Keywords
Introduction
There has been an increasing interest in the use of stereotactic ablative radiotherapy (SABR) for non-small-cell lung cancer (NSCLC) patients with oligometastatic disease (OMD). It is postulated that definitive treatment of the primary as well as OMD in these patients may improve their overall survival (OS). 1 OMD is commonly defined as 1–5 metastatic lesions, in keeping with the recent European Society of Radiotherapy and Oncology (ESTRO) and American Society for Radiation Oncology consensus definition of OMD. 2 If discovered within 3–6 months of diagnosis, they are termed synchronous oligometastases (Figure 1). Alternatively, should OMD develop following definitive treatment of the primary malignancy, this is termed metachronous oligometastasis (Figure 2). 3 OMD is considered an intermediate state between local and polymetastatic disease wherein multiple clinical trials have demonstrated prolonged survival following SABR treatment to all sites of disease, particularly in NSCLC.4–6 Similarly, immunotherapy has transformed the landscape of NSCLC treatment by improving OS in both the locally advanced 7 and metastatic setting.8–11 However, most SABR trials for OMD were conducted in the pre-immunotherapy era. How, exactly, SABR and immunotherapy should be integrated in the treatment of oligometastatic NSCLC therefore remains an active area of investigation.

Synchronous oligometastatic disease. A patient with oligometastatic lung cancer at diagnosis with three metastases receives systemic therapy as well as SABR to all gross sites of disease.

Metachronous oligometastatic disease. A patient with localized lung cancer at diagnosis received local treatment and eventually develops OMD with three new metastatic deposits. The patient then receives systemic therapy as well as SABR to all gross sites of disease.
This review aims to explore the evolving role of radiotherapy in the treatment of NSCLC OMD in patients receiving immunotherapy, with an emphasis on rationale, recent data, and future research questions.
Methods
We performed a review of the literature based on the narrative literature review guidelines outlined by Green et al. 12 The PubMed (Medline) database was queried from its date of inception until June 2022 for MeSH terms as well as different keywords including but not limited to NSCLC, immunotherapy, SABR, stereotactic body radiotherapy, radiation, and OMD. Peer-reviewed clinical trials and large retrospective studies published in the English language that reported on SABR and immunotherapy treatment in NSCLC patients were included. Clinical trials and retrospective studies not exclusive to NSCLC or with ⩽5 patients were excluded. Results for studies which met inclusion criteria are summarized in Table 1.
Selected studies evaluating the safety of immunotherapy with SABR in oligometastatic and polymetastatic NSCLC.
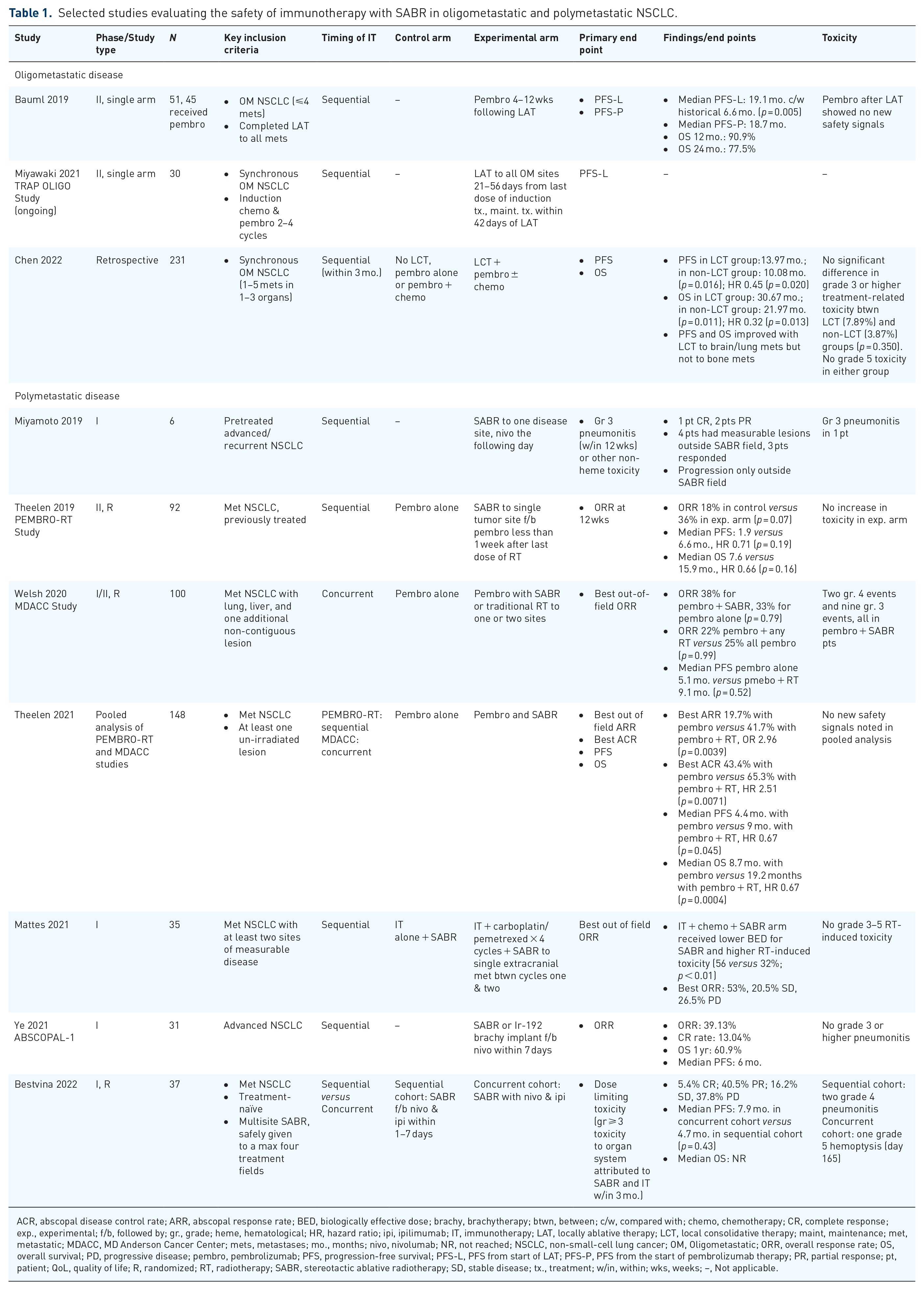
ACR, abscopal disease control rate; ARR, abscopal response rate; BED, biologically effective dose; brachy, brachytherapy; btwn, between; c/w, compared with; chemo, chemotherapy; CR, complete response; exp., experimental; f/b, followed by; gr., grade; heme, hematological; HR, hazard ratio; ipi, ipilimumab; IT, immunotherapy; LAT, locally ablative therapy; LCT, local consolidative therapy; maint, maintenance; met, metastatic; MDACC, MD Anderson Cancer Center; mets, metastases; mo., months; nivo, nivolumab; NR, not reached; NSCLC, non-small-cell lung cancer; OM, Oligometastatic; ORR, overall response rate; OS, overall survival; PD, progressive disease; pembro, pembrolizumab; PFS, progression-free survival; PFS-L, PFS from start of LAT; PFS-P, PFS from the start of pembrolizumab therapy; PR, partial response; pt, patient; QoL, quality of life; R, randomized; RT, radiotherapy; SABR, stereotactic ablative radiotherapy; SD, stable disease; tx., treatment; w/in, within; wks, weeks; –, Not applicable.
Discussion
Rationale and randomized evidence for treating oligometastatic NSCLC with SABR
When Weichselbaum and Hellman described the oligometastatic paradigm, they hypothesized that metastatic disease falls on a spectrum whereby OMD is considered clinically distinct from polymetastatic disease, presenting a unique therapeutic window during which the treatment of all oligometastases may result in long-term disease control and possibly cure in select cases.13,14 Such treatment may take the form of either metastectomy or SABR. SABR offers the advantages of being non-invasive, safe, and well-tolerated, even by frail patients. It also ablates multiple targets simultaneously, achieves good rates of local control, and requires no post-surgical healing time.4,6,15–17 The objectives of treating OMD using SABR include (1) ablating all sites of visible disease to reduce tumor burden; (2) preventing progression to a polymetastatic disease state; (3) relieving morbidity associated with metastases without a decline in quality of life 18 ; (4) delaying the start of systemic therapy and its associated toxicity19,20; and (5) providing cost-effective treatment. 21
There are a number of reasons to support the use of SABR in the OMD setting. First, systemic treatment alone does not eradicate the presence of all oligometastatic malignant cells. They have already demonstrated metastatic potential and are therefore prone to seed further metastases, leading to the less favorable polymetastatic state. 5 Ablative treatment in the form of SABR may decrease this risk by reducing the burden of proliferative malignant cells. Second, SABR is a histology-agnostic ablative technique which can eradicate systemic therapy-resistant disease. This, in turn, may render remaining cells more susceptible to further lines of systemic treatment. Lastly, SABR optimizes local control at the sites of OMD, thereby delaying or eliminating the morbidity and potential mortality associated with local and eventually distant progression of disease.
Several randomized clinical trials establishing the use of SABR in the oligometastatic setting have been published. Iyengar et al. randomized 29 oligometastatic NSCLC patients with up to five metastatic lesions to receive chemotherapy alone in the control arm or consolidative SABR to all OMD followed by maintenance chemotherapy in the experimental arm. All patients underwent induction chemotherapy with no evidence of progression of disease prior to enrollment. Maintenance systemic therapy began 1 week following the last fraction of radiotherapy. The primary end point of progression-free survival (PFS) was significantly longer in the SABR arm (9.7 months) compared to the control arm (3.5 months, p = 0.01) and as a result the trial was halted prematurely. Toxicity was similar in both arms. The first sites of recurrence in the control arm were observed at locations of original disease, whereas in the SABR arm, there were no in-field recurrences at sites of original disease and there were fewer distant recurrences. These findings support the treatment of all OMD sites as a means to reduce the risk of both local and distant failures.
A phase II trial by Gomez et al. randomized patients with oligometastatic NSCLC with 1–3 metastases to either receive observation or maintenance systemic therapy in the control arm or local consolidative therapy (LCT) in the form of SABR or surgery to all OMD ± maintenance treatment in the experimental arm. All patients were treated with systemic therapy upfront with no evidence of progression for a minimum of 3 months. A significant PFS benefit in the LCT arm triggered an early closure of the trial after only 49 patients were enrolled. The long-term primary outcome of median PFS was significantly longer in in the LCT arm (14.2 months) versus 4.4 months in the control arm (p = 0.022). An OS benefit was also observed in favor of the LCT arm (41.2 months) versus the control arm (17 months, p = 0.017).5,6 Not only did the authors find prolonged survival in the LCT arm (37.6 months) compared to the control arm (9.4 months, p = 0.034), but early LCT was favored over delaying LCT until progression. This suggests that the best therapeutic window for ablating OMD may be prior to disease progression,5,6 although ongoing trials are yet to determine the optimal timing of SABR with systemic therapy. 22
The SABR-COMET trial randomized 99 patients with a controlled primary tumor and up to five metastatic lesions in a 1:2 ratio to receive either palliative standard of care (SoC) or SoC in addition to SABR to all sites of disease. Eighteen of these patients had a primary lung cancer. Systemic therapy agents were not administered 4 weeks prior to SABR or 2 weeks after the last fraction. The primary end point of OS was met per the phase II screening design, with a significantly longer median OS in the SABR arm of 41 months compared to only 28 months in the SoC alone arm (p = 0.090). Of note, three grade five events were reported in the SABR arm of the SABR-COMET trial, two of which were related to pulmonary complications. 15 Extended long-term results were recently published confirming a prolonged OS benefit, with an 8-year OS of 27.2% in the SABR arm compared to 13.6% in the SoC only arm. PFS at 8 years was 21.3% in the SABR arm and 0.0% in the SoC only arm, mirroring OS. Fewer patients in the SABR arm received cytotoxic chemotherapy. Slightly higher low-grade toxicity was noted in the SABR arm with no difference in quality of life between arms. 23 Unlike the control arm, the SABR arm had 11 patients who were alive beyond 5 years without progression, of those, two had a primary lung tumor. These findings support the existence of an oligometastatic state given that a minority of patients survived 5 years following SABR treatment without evidence of disease recurrence.
Overall, the body of randomized evidence described seems to validate the oligometastatic paradigm in NSCLC, with a significant improvement in OS and PFS associated with SABR or LCT when compared with cytotoxic chemotherapy alone. These favorable results also highlight the importance of incorporating SABR into the SoC treatment for this patient population. Despite adequate local treatment however, OMD patients have a propensity for failing distantly.24,25 Moreover, the first-line SoC treatment for metastatic NSCLC has shifted from cytotoxic chemotherapy to upfront immunotherapy, chemo-immunotherapy, or targeted therapy. 26 For NSCLC patients with synchronous or metachronous OMD who are to receive immunotherapy upfront or at disease progression, it is imperative to carefully consider how and when SABR can be strategically incorporated into this treatment paradigm.
Rationale and randomized evidence for treating NSCLC with immune checkpoint inhibitors
Over the past decade, immunotherapy, more specifically immune checkpoint inhibitors (ICIs), has revolutionized anti-neoplastic treatment for several malignancies, including NSCLC. Mechanisms of immune evasion, namely the upregulation of specific proteins on the cell membrane [e.g. programmed death-ligand 1 (PD-L1)], are often developed by malignant cells. ICIs are antibodies targeting these proteins at key immune checkpoints which activate T cells and remove a method of immune evasion by neoplastic cells, thereby upregulating the immune system and encouraging an antitumor immune reaction. 27 The first class of ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) inhibitors (e.g. ipilimumab); the second class are programmed cell death 1 (PD-1) inhibitors (e.g. pembrolizumab, nivolumab, dostarlimab, and cemiplimab); and the third class are anti-PD-L1 antibodies (e.g. atezolizumab, durvalumab, and avelumab). 27 Other ICIs with emerging evidence in the treatment of several malignancies include LAG-3 (CD223), 28 T-cell immunoreceptor with immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGIT), 29 and B7H3. 30
Second-class and third-class ICIs have gained more favor given their superior toxicity profiles and more favorable outcomes. Several randomized studies have established the current use of ICIs in the treatment of NSCLC. In the locally advanced setting, the phase III PACIFIC trial randomized 713 unresectable stage III NSCLC after curative-intent chemoradiotherapy (CRT) without progression in a 2:1 ratio to either receive adjuvant durvalumab (experimental arm) or placebo (control arm) for 1 year. Median PFS was 16.8 months in the durvalumab arm versus 5.6 months in the placebo arm (p < 0.001). Although toxicity was similar in both arms, the durvalumab arm had a longer duration of response, higher response rate, and longer median time to death or distant metastasis. 7 Updated results confirmed an OS benefit in the durvalumab arm with a median OS of 47.5 months versus 29.1 months in the placebo arm. 31 This seminal trial cemented adjuvant durvalumab following concurrent CRT as the current SoC for unresectable locally advanced NSCLC.
Patients with metastatic NSCLC appear to respond favorably to immunotherapy. The KEYNOTE-024 phase III trial randomized 305 previously untreated patients with a PD-L1 proportion score ⩾50% without driver mutations to either pembrolizumab monotherapy or platinum-based chemotherapy. Median OS was significantly prolonged (30.0 months) in the pembrolizumab arm versus 14.2 months in the chemotherapy arm, with less frequent toxicity documented in the pembrolizumab arm. 11 Similarly, KEYNOTE-189 randomized 616 previously untreated metastatic non-squamous NSCLC patients in a 2:1 ratio to either pemetrexed with platinum and pembrolizumab or pemetrexed with platinum and placebo. Median OS was significantly longer at 22.0 months in the pembrolizumab arm versus 10.7 months in the placebo arm, regardless of PD-L1 status. 8 KEYNOTE-407 randomized 559 untreated metastatic squamous NSCLC patient to either pembrolizumab plus carboplatin/paclitaxel chemotherapy or chemotherapy plus placebo. Median OS in the pembrolizumab arm was significantly longer at 17.1 months versus 11.6 months in the placebo arm. 9 The superiority of pembrolizumab with or without chemotherapy compared with chemotherapy alone persisted in pooled analyses in patients with stable brain metastases 10 irrespective of PD-L1 expression, and in the elderly (⩾75 years of age). 32 Similarly, CheckMate 9LA demonstrated an OS benefit for nivolumab and ipilimumab with two cycles of chemotherapy when compared with chemotherapy alone (median 15.8 months versus 11 months; p = 0.00065). 33 The CheckMate 227 trial also showed an improvement in 4-year OS with nivolumab plus ipilimumab when compared with chemotherapy, regardless of PD-L1 status, for both squamous and non-squamous histologies. 34 These findings build a strong argument for the use of ICIs as a key treatment paradigm for metastatic NSCLC in the absence of a targetable driver mutation.
Despite the substantial improvement in outcomes associated with ICI use in NSCLC, approximately two-thirds of patients do not respond to immunotherapy and for those who do respond, most eventually progress due to acquired ICI resistance. 27 It has therefore been suggested that a combination of ICIs to target microscopic disease and SABR to cytoreduce gross metastases (potentially harboring ICI-resistant clones) may lead to more durable response rates in oligometastatic NSCLC. 35
Rationale and clinical evidence for treating metastatic NSCLC with ICI and SABR
T-cell activation within the tumor microenvironment plays a pivotal role in local radiation therapy (RT) and in systemic immunotherapy treatment. RT induces T-cell and lymphocyte infiltration, augmenting the antitumor reaction.36,37 Similarly, the level of T-cell infiltration has been shown to correlate positively with ICI response.27,38 However, preclinical syngeneic mouse models have indicated that fractionated radiotherapy can upregulate pro-tumor PD-L1 production resulting in immune evasion. This creates a form of radioresistance 39 which may be overcome by the administration of a concurrent PD-L1 antibody with RT.24,40 ICI use in combination with RT therefore has the potential to address the challenges of pro-tumor microenvironment changes as well as tumor radioresistance.
Several clinical trials evaluated the safety of ICI use with SABR delivered sequentially in polymetastatic NSCLC (Table 1). Miyamoto et al. conducted a phase I trial which enrolled six patients with pre-treated advanced and recurrent NSCLC to receive SABR to a single metastasis with nivolumab administered the following day. A single grade 3 pneumonitis was observed among these six patients and there were no cases of in-field progression. 41 Mattes et al. enrolled 35 patients in a phase I trial comparing immunotherapy and SABR given sequentially, with immunotherapy, chemotherapy, and SABR given between chemotherapy cycles. Although there were no cases of grade 3 or higher radiation-induced toxicity in either arm, more grade 1–2 radiation-induced toxicities (mostly pneumonitis) were observed in the combination of immunotherapy, chemotherapy, and SABR arm. 42 Ye et al. performed a phase I trial administering an Iridium-192 brachytherapy implant or SABR in 31 advanced NSCLC patients followed by nivolumab given within 7 days following RT. Median PFS was 6 months with no grade 3 or higher pneumonitis toxicity. 43 The PEMBRO-RT study by Theelen et al. randomized 92 previously treated metastatic NSCLC patients in a phase II design to pembrolizumab alone or SABR to a single tumor site followed by pembrolizumab within 1 week from RT. No significant difference in median PFS, median OS, or toxicity was found between treatment arms. 44 These studies suggest that ICI use in the polymetastatic NSCLC setting given sequentially with SABR appears to be relatively safe, although there is a paucity of PFS and OS data.
In addition, several clinical trials have explored concurrent ICI and SABR in polymetastatic NSCLC (Table 1), demonstrating an overall increase in toxicity associated with this strategy. The MD Anderson Cancer Center (MDACC) study by Welsh et al. randomized 100 metastatic NSCLC patients with lung, liver, and one other non-contiguous metastasis in a phase I/II design to receive pembrolizumab alone or pembrolizumab concurrently with SABR (or standard hypofractionated RT if SABR could not be safely delivered). There were two grade 4 events and nine grade 3 events in patients who received SABR with concurrent pembrolizumab in the phase II group. No grade 4 or 5 adverse events were observed in patients who received pembrolizumab alone or pembrolizumab with non-SABR RT. Median PFS was not significantly different between arms: 5.1 months for pembrolizumab versus 9.1 months for pembrolizumab with concurrent RT (p = 0.52). A pooled analysis of the PEMBRO-RT and MDACC studies comparing pembrolizumab alone to pembrolizumab and SABR in metastatic NSCLC patients was performed by Theelen et al. Although OS and PFS were not significantly different for each individual study, the pooled analysis revealed a median PFS in the pembrolizumab alone group of 4.4 months and 9 months in the pembrolizumab with RT group (p = 0.045). Similarly, median OS was 8.7 months in the pembrolizumab alone group and 19.2 months in the pembrolizumab with RT group (p = 0.0004). Toxicity outcomes were not among the pre-specified end points and thus were not evaluated within the pooled analysis. Bestvina et al. included 37 metastatic NSCLC treatment-naïve patients in a randomized phase I trial comparing SABR with nivolumab and ipilimumab given either concurrently or sequentially. Two grade 4 cases of pneumonitis reactions were observed in the sequential cohort prompting a 20% radiation dose reduction to organs at risk. No grade 3 or higher toxicity was noted in the concurrent cohort within 3 months of treatment. However, at 165 days from treatment, one patient in the concurrent cohort experienced a grade 5 toxicity in the form of hemoptysis. This was deemed to possibly be attributed to either SABR or nivolumab/ipilimumab. Median PFS in the concurrent cohort was 7.9 months compared to 4.7 months in the sequential cohort (p = 0.43). 45 Given the limited sample sizes and short follow-up, these findings imply that concurrent immuno-radiotherapy with SABR calls for an abundance of caution as we await more data to elucidate short- and long-term toxicity patterns.
Few clinical trials have evaluated the safety and efficacy of ICI use with SABR specifically in the oligometastatic NSCLC setting (Table 1), although larger retrospective analyses have been published demonstrating favorable outcomes. Bauml et al. completed the first single-arm phase II trial on the topic, enrolling 51 oligometastatic NSCLC patients with 4 or fewer metastases treated with local ablative therapy (LAT) and pembrolizumab given sequentially, 4–12 weeks thereafter. Patients were enrolled regardless of their biomarker status. In total, 45 patients received pembrolizumab, 30 of those (67%) received SABR. PFS from the start of LAT was 19.1 months, nearly triple the historical control of 6.6 months (p = 0.005). OS was also quite favorable at 90.9% (12 months) and 77.5% (24 months). One grade 4 pneumonitis was documented. There was no reduction in quality of life or new safety signals with this sequential treatment. 46 Chen et al. conducted a retrospective study of 231 patients with synchronous oligometastatic NSCLC and no targetable mutations. They compared PFS and OS for pembrolizumab ± chemotherapy with and without LCT, delivered sequentially, within 3 months. PFS was significantly longer in the LCT group: 13.97 months versus 10.08 months (p = 0.016). Median OS was also significantly higher in the LCT group: 30.67 months versus 21.97 months (p = 0.011). This PFS and OS benefit was reserved for LCT to brain and lung metastases, with no apparent benefit of LCT to bone metastases. There was no significant difference in grade 3 or higher toxicity rates between LCT and non-LCT groups (p = 0.350). 47
These findings indicate that oligometastatic NSCLC patients may indeed benefit from SABR to all metastases when combined with ICI. Promising OS and PFS signals favor this approach over ICI alone and warrant further study in prospective clinical trials. Combination of SABR and ICI also appears safe and well tolerated, but further clinical trials are required to confirm this in larger cohorts. 48 One such study is the TRAP OLIGO study by Miyawaki et al., a single-arm phase II study which will recruit 30 patients with synchronous oligometastatic NSCLC. Participants will receive induction chemotherapy and pembrolizumab followed by LAT within 21–56 days to all oligometastases, after which they will receive maintenance systemic therapy within 42 days of LAT. The primary end point of the study is PFS rate from the last dose of LAT. 48 While the results of this study are eagerly awaited, randomized data are also required to definitively assess the safety and efficacy of combining SABR and ICI in oligometastatic NSCLC.
Management principles for treating oligometastatic NSCLC using SABR and ICI
Considering the available data and acknowledging the limitations therein, we propose the following four guiding principles when combining SABR and ICI for the treatment of oligometastatic NSCLC: (1) careful patient selection; (2) cautious integration with concurrent ICI; (3) ablate all visible disease; and (4) minimize toxicity (Figure 3).

Proposed management principles for treatment of oligometastatic disease with SABR and ICIs. (1) Careful patient selection; (2) caution with concurrent SABR and ICI; (3) ablate all sites of disease; and (4) minimize toxicity.
Principle 1: Careful patient selection
The best candidates to receive combined immuno-radiotherapy are those who are most likely to tolerate this treatment with the lowest likelihood of experiencing unacceptable levels of toxicity. Jasper et al. described the ideal candidate for LAT among patients with NSCLC: good performance status (Eastern Cooperative Oncology Group 0–1) with a life expectancy greater than 6 months and limited comorbidities, low burden metachronous oligometastases (e.g. solitary metastasis), 49 and a previous history of response to systemic therapy with further lines of systemic therapy available.4,6,50 Immunotherapy is also thought to be more effective in patients with a low disease burden, in keeping with the oligometastatic paradigm. 51 We therefore suggest that combined SABR and ICI be strategically given to select oligometastatic NSCLC patients who embody these characteristics until further trials facilitate the selection of the ideal patient for SABR and ICI.
Principle 2: Cautious integration of SABR with concurrent ICI
The optimal timing between SABR and ICI treatment remains unclear, yet current studies can provide some guidance while we await results from future clinical trials. Bestvina et al. documented one grade 5 toxicity reported in a patient who received both ICI and SABR concurrently. Miyamoto et al. described a 1/6 (17%) risk of grade 3 pneumonitis when nivolumab was given on the day following the last SABR treatment. 41 The shortest time interval longer than this which was shown to be safe is approximately 1 week, 44 and the longest is 12 weeks.46,47 A pooled analysis of trials in the US Food and Drug Administration Database revealed no increased risk of serious adverse events with the administration of an ICI within 90 days of RT. 52 More studies are nevertheless required to narrow this window between ICI and SABR delivery to optimize treatment outcomes and maximize safety. In the interim, given the limited mature safety information on the delivery of concurrent SABR and ICI, we call for caution as we await further data. 52
Whether to give ICI or SABR first also requires clarification. The Iyengar et al. and Gomez et al. trials required systemic therapy administration with treatment response or at least stable disease to be eligible for enrollment and randomization. However, systemic therapy in these trials often took the form of cytotoxic chemotherapy. The SABR-COMET trial and the Gomez et al. trial did not find a significant difference in time to new metastases after LAT, implying that micro-metastatic disease had already seeded long before patients received LAT.4,6 It is possible that the opposite approach of delivering SABR first may have greater biological rationale in the context of ICI. 52 For example, it may allow SABR, which is receptor-agnostic, to ablate gross disease harboring treatment-resistant clones, thereby priming the tumor microenvironment for ICIs that could target other micro-metastatic foci of malignant cells.
However, initiating SABR first may delay ICI delivery given the time needed for simulation, contouring, treatment planning, and quality assurance prior to stereotactic treatment delivery. Some patients may not have the opportunity to start SABR upfront or may have already received immunotherapy when they present for a radiation oncology opinion. To balance these challenges, sandwiching SABR between ICI cycles may be a plausible alternative. Active doses of drug remain between cycles: the half-life of ICIs ranges between 6 and 27 days with cycles often administered every 2–4 weeks. 53 Nonetheless, an example of safe delivery of SABR between ICI cycles is found in the CHEERS trial presented at the 2021 ESTRO meeting. This phase II study enrolled 99 patients with locally advanced or metastatic disease, randomizing them to receive an anti-PD-L1 ICI alone, or combination of anti-PD-L1 ICI and SABR to a maximum of three metastases. SABR was delivered before the second or third ICI cycle. The primary end point of median PFS nearly doubled in the SABR arm (4.4 versus 2.8 months), although this did not reach statistical significance (p = 0.7). Similar grade 3 or higher toxicity rates were noted in both arms (17.8% versus 17.6%) with no grade 5 adverse events. Although only seven NSCLC patients were enrolled in this study, it implies that SABR may be delivered safely between ICI cycles. 54 To maximize safety, we propose that if SABR is delivered between ICI cycles, consideration could also be given to prolonging the time between ICI cycles by approximately 1–2 weeks to allow for the safe and efficient delivery of SABR.
We urge caution when integrating SABR within a course of ICI. Whenever possible, patients should be enrolled on a clinical trial of co-administration. For patients previously untreated with ICI, up-front SABR seems to be a reasonable option provided that SABR can be delivered without significant delay. For patients who are currently receiving or have previously received ICI (e.g. oligoprogressive disease), the long half-life of ICIs means that active drug remains present between cycles. Nonetheless, based on the data available, it is prudent to avoid same-day administration of SABR and ICI in favor of between-cycle SABR delivery. If oncologically appropriate, consideration can also be given to extending the between-cycle interval slightly (e.g. 1 week) to accommodate the efficient delivery of SABR. This aligns with the recently published European Organisation for Research and Treatment of Cancer (EORTC)-ESTRO OligoCare consortium consensus recommendations which counsel against same-day administration of SABR and ICI, with a minimum ICI interruption of 1 week before or after SABR. 55 Ongoing and future clinical trials will provide empirical guidance and help clarify the issue of timing.
Principle 3: Ablate all sites of disease
The basis of oligometastatic ablation lies in targeting all detectable metastatic deposits, theoretically collapsing clonal heterogeneity within metastases to prevent metastatic cascade(s) that precede polymetastatic dissemination. 17 Most studies evaluating SABR and immunotherapy have aimed to test the abscopal effect whereby local SABR to one or few metastases in polymetastatic patients, combined with immunotherapy, activates a systemic antitumor response. In the first patient-level data meta-analysis of this effect, NSCLC was the most common cancer type where abscopal responses were reported. 56 The abscopal objective response rate (ORR), although statistically significant in some studies, is rare and modest at best. It also remains questionable whether or not these ORRs are indeed clinically significant given the frequently non-significant OS and PFS when compared to ICI alone.41–45,57,58 Moreover, untreated metastases will continue to pose a threat both locally, as they are more likely to progress without adequate local treatment, 41 and systemically by potentially seeding further metastases. Studies of the abscopal effect do, however, demonstrate a level of synergy between SABR and immunotherapy which can be capitalized on in oligometastatic NSCLC. Ultimately, this too remains an active area of investigation.
Treatment time for SABR to all sites of OMD should be shortened as much as possible (ideally no more than 2 weeks), 59 to allow for the initiation or resumption of ICI at the earliest clinically acceptable time. This reduces the risk of progression from untreated micro-metastatic disease during LAT, which, in turn, may improve patient outcomes. Ablating all foci of metastatic disease using SABR within a short treatment interval is thus a key tenet of our treatment philosophy for oligometastatic NSCLC patients.
Principle 4: Minimize toxicity
Oligometastatic NSCLC patients often require several rounds of systemic and local treatments, each potentially associated with morbidity, in addition to malignancy-related morbidity and potential mortality. SABR and ICI treatment in this setting should therefore aim to minimize toxicity by maximizing the safety and tolerability of treatment whenever possible. Although not statistically significant, Chen et al. reported slightly higher rates of pneumonitis in patients who received systemic therapy in addition to LAT (7.89% versus 3.87% with systemic therapy alone; p = 0.350) and in patients who previously received RT. 46 This also appears to be higher than the incidence of pneumonitis with PD1 inhibitors (1.3%) and PD-L1 inhibitors (3.6%) when administered alone in metastatic NSCLC patients. 60 Individuals who have had previous RT with subsequent immunotherapy treatment may also have higher rates of recall radiation pneumonitis. 61 Bestvina et al. reported a 50% (2/4 patients) grade 4 pneumonitis rate attributable to the combination of SABR and ICI. This led to a 20% organ at risk (OAR) dose constraint reduction after which no grade ⩾ 3 toxicities were noted in the sequential arm. 45 A recent analysis of three prospective trials of SABR and ICI for polymetastatic disease (NSCLC being the most common cancer type) reported a grade 3+ pneumonitis rate of 8.8% at 12 months. 62 These findings suggest that, per the SABR-COMET protocols, OAR dose constraints should be prioritized over target coverage if necessary when planning SABR for OMD. 63 Strategies for ensuring this consist of (1) compromising the planning target volume while maintaining an ablative SABR dose; (2) dose reduction; and (3) increasing dose fractionation. Significant variability has been noted for SABR OAR dose constraints, 64 prompting an international effort to standardize practices. Examples include the 2022 UK SABR consensus guideline 65 and the Hypofractionated Treatment Effects in the Clinic document, 66 which can be used by clinicians to safely respect OAR tolerances when delivering SABR treatment.
What about the addition of chemotherapy to SABR & ICI? Mattes et al. compared immunotherapy and SABR ± chemotherapy in metastatic NSCLC with at least two sites of measurable metastases. Despite SABR being delivered to only a single extracranial metastasis, higher radiation-induced toxicity was reported in the immunotherapy, SABR, and chemotherapy arm (p < 0.01). This raises the question of whether chemotherapy can be safely added to ICI and SABR in OMD. The data informing the use of SABR with chemo-immunotherapy are scarce, posing unique challenges in determining who would benefit most from the addition of chemotherapy and when it can be safely delivered, given the added challenges of toxicity and timing.
Patients with any relative contraindications to SABR, including but not limited to active connective tissue disorders, significant overlap with a previously irradiated field, and interstitial lung disease for lung metastases, have a more severe toxicity profile at higher incidence rates. 67 The same also applies to patients with contraindications to immunotherapy, particularly for those who have active autoimmune disorders. 68 Close monitoring for ICI side effects is required to tailor treatment breaks, dose modification, and immunosuppressive therapy needs accordingly.
We maintain that minimizing toxicity for both SABR and ICI by prioritizing OARs, exercising caution with the addition of chemotherapy, and close monitoring, particularly in patients with relative contraindications, is critical in oligometastatic NSCLC patients receiving radio-immunotherapy.
Future directions
Many questions remain unanswered in oligometastatic NSCLC. The ongoing TRAP OLIGO phase II single-arm trial aims to determine the safety and efficacy of induction chemotherapy and pembrolizumab followed by LAT to all metastases in biomarker-negative oligometastatic NSCLC patients. Moreover, oligometastatic intracranial and extracranial diseases likely require a different treatment approach based on the effectiveness and central nervous system penetration of systemic agents.
Novel questions surrounding the use of blood-based or imaging biomarkers are also being explored. The use of liquid biopsy in cancer patients is a promising topic of ongoing research. It would offer a simple, non-invasive method of monitoring tumor response throughout treatment and subsequent follow-up. It would also help identify patients who would benefit most from immunotherapy and SABR, using circulating tumor cells and cell-free DNA as novel biomarkers.59,69 With respect to imaging biomarkers, the emergence of novel, immune-based positron emission tomography (PET) tracers has enabled ‘immuno-PET’. Patients can now undergo functional imaging to monitor response to ICI, potentially identifying poorly responding metastases to which SABR could then be directed. 70
The optimal sequence and timing of SABR with immunotherapy and whether this is influenced by biomarker status is evidently unknown. The SABRseq trial (NCT03307759) was launched to address this question, randomizing metastatic NSCLC patients to either receive SABR followed by pembrolizumab or pembrolizumab followed by SABR after the first cycle, assessing the safety and efficacy of both approaches.
Dosimetric questions also arise in the context of SABR and ICI treatment. Whether OAR dose constraints should be adjusted for patients receiving radio-immunotherapy in the oligometastatic setting, particularly when toxicity profiles overlap (e.g. pneumonitis), remains undetermined.
Ultimately, phase III randomized trials are needed to determine if combination of SABR and ICI in oligometastatic NSCLC is not only safe and effective, but also confers an OS and PFS benefit for patients. The ongoing LU-002 phase II/III randomized trial aims to answer this question by enrolling oligometastatic NSCLC patients who received induction immunotherapy ± platinum-based chemotherapy for a total of four cycles with stable disease or a partial response. Participants are randomized to receive maintenance systemic therapy alone (chemotherapy ± pembrolizumab) or SABR/surgery to all sites of metastases and RT to the primary site followed by maintenance systemic therapy. The phase II component has completed accrual and the trial is temporarily closed with data analysis underway to ascertain whether there is a signal for prolonged PFS with the addition of LCT. Should such a signal be found, the trial will re-open for the phase III component with OS as the primary end point.
We believe promoting accrual to clinical trials is vital while there is still equipoise. To this end, we recommend that combination of SABR and ICI treatment in oligometastatic NSCLC patients be delivered primarily in the context of a clinical trial whenever possible. This will generate high-quality data, empowering clinicians, and their patients, to making evidence-based, informed treatment decisions.
Conclusion
The advent of immunotherapy drugs and advanced radiation techniques such as SABR has revolutionized our approach to treating OMD. Oligometastatic NSCLC patients in particular are a unique population where emerging evidence supports the use of SABR in combination with ICIs to exploit their synergy, thereby optimizing both local and systemic disease control and improving patient outcomes. Further clinical trials are essential to characterize patients who would benefit most from this bimodality treatment and to establish the ideal timing as well as treatment approach for oligometastatic NSCLC.
