Abstract
Recently, attention has increasingly centered on non-small-cell lung cancer (NSCLC) with immune checkpoint inhibitors application. Numerous clinical studies have underscored the potential of immunotherapy in treating resectable NSCLC, highlighting its role in improving patient outcomes. However, despite these promising results, there is ongoing debate regarding the efficacy of immunological combination therapy strategies, the prevalence of treatment-related side effects, the identification of predictive biomarkers, and various other challenges within the neoadjuvant context. Careful consideration is essential to maximize the benefits of immunotherapy for patients with resectable NSCLC. This article offers a detailed overview of recent advancements in neoadjuvant immunotherapy for resectable NSCLC. By examining these developments, we aim to provide new perspectives and valuable insights into the benefits and challenges of applying neoadjuvant immunotherapy in clinical settings.
Keywords
Introduction
Lung cancer is the primary cause of cancer-related mortality globally, with non-small-cell lung cancer (NSCLC) comprising about 85% of cases. Stages I–IIIA represent early-stage lung cancer, and computed tomography (CT) is increasingly being used for early detection in high-risk populations. NSCLC patients typically undergo surgery, followed by adjuvant chemotherapy. 1 Despite surgical intervention, only 25%–30% of these patients successfully eradicate the lesions, leaving a significant threat of recurrence and mortality. 2 Platinum-based chemotherapy following surgery has been the standard treatment for stage II–IIIA NSCLC, but it only enhances 5-year survival rates by approximately 5%.3,4 Exploring novel therapies for NSCLC is crucial to improving patient prognosis, addressing the high risk of postoperative recurrence, and enhancing survival rates. Neoadjuvant chemotherapy shows promise in improving survival rates for stage IB–IIIA patients. However, further research is necessary to assess its efficacy3–7 fully. Immune checkpoint inhibitors (ICIs), such as PD-1/PD-L1 antibodies, are now advised for advanced NSCLC lacking driver mutations, representing a significant advancement in lung cancer treatment. 8
Perioperative immunotherapy includes strategies such as neoadjuvant immunotherapy, adjuvant immunotherapy, and neoadjuvant combined with surgery. Adjuvant immunotherapy refers to immunotherapy conducted after surgery. Neoadjuvant immunotherapy, given to cancer patients before surgery to strengthen their immune system, shows great promise. It can reduce tumor size and enhance the body’s antitumor response before surgical lymph node removal. After the primary lesion is excised, activated T cells can target potential metastatic lesions, improving cure rates. This method leverages dendritic cells to stimulate T cells, thereby reducing harm to healthy cells and enhancing the body’s innate cancer defenses. However, several challenges remain for neoadjuvant immunotherapy.9,10 This paper examines recent studies on neoadjuvant immunotherapy and compiles the findings to guide perioperative clinical diagnosis and treatment for patients with resectable NSCLC.
Mechanisms of neoadjuvant immunotherapy
Tumor cells stimulate innate immunity by influencing T cells, which activate antigen-presenting cells to produce antigens11,12 (Figure 1(a)). This process activates effector T cells through co-stimulatory molecules. 13 CTLA-4 and PD-1/L1 play crucial roles in cancer immunoevasion. 14 CTLA-4 counteracts CD28, but CTLA-4 inhibitors reduce this inhibition, effectively activating tumor-specific T cells 15 (Figure 1(b)). PD-L1 ligands, belonging to the B7-CD28 family, are abundant in most cancers.16,17 Tumor cells escape immune detection by suppressing T-cell activity through the PD-1/PD-L1 pathways. 18 CD8+ T cells, antigen-presenting cells, and tumor cells interact with ICIs, blocking immune regulatory molecules. 19 Neoadjuvant immunotherapy for resectable NSCLC shrinks tumors, targets micrometastases, and boosts T-cell activation by inhibiting PD-1/PD-L1 in the tumor microenvironment (TME). This process reaches lymph nodes and the bloodstream, removing undetectable metastases through lymphatic drainage. CTLA-4 and PD-1 collaborate to prevent tumors and maintain self-tolerance in T-cell responses.

(a) Binding of anti-PD-1 or anti-PD-L1 antibodies to PD-1/L1 lifts the inhibition of T-cell activation and proliferation, restores T-cell function, and promotes their attack on tumor cells. (b) CTLA-4 opposes CD28, but CTLA-4 inhibitors reduce this inhibition and activate tumor-specific T cells. (a + b) CTLA-4 and PD-1 collaborate to prevent tumors and maintain self-tolerance in T-cell responses. (a + c) Chemotherapy shrinks tumors by killing cancer cells, and chemotherapy enhances the effects of immunotherapy by inducing mutant cells and antigenic sites. (a + d) Radiotherapy enhances the body’s immune response, leads to cell death, and stimulates immune cell activity. (a + e) Anti-angiogenic drugs and ICIs can enhance immune function by normalizing blood vessels and activating immune cells, normalizing tumor blood vessels, and improving the immune response.
Chemotherapy reduces tumor size by killing cancer cells, achieving the treatment goal. Recently, immune drugs have been used to treat cancer. Chemotherapy enhances the effectiveness of immunotherapy by inducing mutated cells and antigenic sites20,21 (Figure 1(c)). Inducing and activating the immune system promotes apoptosis, reducing T-cell production and increasing apoptosis. 21 Neoadjuvant immunotherapy combined with chemotherapy may more effectively activate the human immune system. Combining them may enhance the antitumor effect compared to using them separately. Luis A. Godoy’s research indicated that over 80% of patients with resectable NSCLC achieved complete tumor removal following neoadjuvant immune-combination chemotherapy. Combining ICIs with chemotherapy yields a better pathological response than using ICIs alone. 22
Combining chemotherapy with ICIs is common, but neoadjuvant immune-combination therapy needs further research for patients with poor responses or side effects from platinum-based chemotherapy. Research shows that combining ICIs with anti-angiogenic agents is effective and safe for advanced NSCLC, providing valuable insights for neoadjuvant treatments in early-stage patients23,24 (Figure 1(e)). Tumor anti-angiogenic drugs boost antigen presentation, activate the immune response, and counteract VEGF-induced immunosuppression. They enhance T-cell activity, normalize nearby blood vessels, activate effector T-cells, increase IFN-γ, lower VEGF levels, and boost tumor-infiltrating lymphocyte density. Lung cancer exploits VEGF to create blood vessels, depriving the body of nutrients and oxygen. This hostile environment prevents immune cells from detecting and attacking tumor cells. Anti-angiogenic drugs normalize blood flow and reduce immunosuppression, allowing immune checkpoint inhibitors (ICIs) to reveal tumors and boost immune cell activity. Anti-vascular drugs increase CD8+ T-cells with IFN-γ in tumors, enhancing T-cell movement to antigens and boosting the immune response. ICIs boost effector T cells, increasing IFN-γ secretion and improving their ability to infiltrate and kill targets, which enhances drug delivery and may reduce dosage and side effects. Anti-angiogenic drugs and ICIs can work together to normalize tumor blood vessels and improve the immune response, addressing VEGF-induced immunosuppression in lung cancer. This dual approach boosts immune function by normalizing blood vessels and activating immune cells, creating a beneficial feedback loop.
Radiotherapy is a standard cancer treatment that boosts the body’s immune response, causes immunogenic cell death, and stimulates increased immune cell activity25,26 (Figure 1(d)). Combining radiotherapy with immunotherapy might enhance its effectiveness. Researchers at Zhongshan Hospital, Fudan University, found that radiotherapy can trigger immune escape in tumors by activating the cGAS-STING pathway. 27 This pathway, essential for the immune response, boosts PD-L1 expression, causing tumor immune suppression.
Neoadjuvant immunotherapy
Neoadjuvant immunization monotherapy
The initial breakthrough in PD-1/PD-L1 inhibitor research was using neoadjuvant single-agent immunotherapy. The U.S. Food and Drug Administration (FDA) has approved four PD-1/PD-L1 inhibitors for NSCLC treatment: nivolumab and pembrolizumab (PD-1 inhibitors), and atezolizumab and durvalumab (PD-L1 inhibitors). In 2018, Forde et al. reported a study involving 21 patients with stage I–IIIA NSCLC who received two courses of neoadjuvant nivolumab monotherapy (NCT02259621). Out of the total patients, 20 underwent R0 resection, yielding a primary pathological response (MPR) rate of 45%, with two patients (10%) achieving a pathological complete response (pCR). 28 Rosner et al. reported that the recently published 5-year trial results indicate a recurrence-free survival (RFS) rate of 60% and an overall survival (OS) rate of 80%. Among the patients who achieved MPR, 89% had no recurrence or death during the 5-year follow-up. 29 The publication of these results provided significant encouragement to researchers. However, subsequent studies could have replicated these findings, including LCMC3 and NEOSTAR. In the LCMC3 trial, the MPR was 20% with a pCR of 6%, whereas NEOSTAR’s nivolumab monotherapy arm reported an MPR of 22% and a pCR of 9%.30,31 According to the latest results published from the LCMC3 study at the 2023 European Lung Cancer Conference, the 3-year DFS rate and 3-year OS rate for patients receiving neoadjuvant treatment with atezolizumab in the assessable MPR population (n = 137) were 72% and 82%, respectively. 32 Numerous phase I and II single-arm studies (Table 1) have demonstrated high MPR rates (19%–45%) in patients with resectable stage IB–IIIA NSCLC while maintaining safety. However, since most studies lacked control groups, direct visual comparisons with other treatment regimens, such as chemotherapy, were impossible. Nonetheless, they demonstrated significant improvements in MPR and pCR compared to past neoadjuvant chemotherapy studies, which reported MPRs of about 20% and pCRs of around 4%. Neoadjuvant single-agent immunotherapy regimens appear feasible and practical. However, due to the small sample sizes in these phase II studies, optimizing combinations among various agents still needs to be addressed and requires further investigation.
Clinical trials of neoadjuvant immune monotherapy for non-small cell lung cancer.
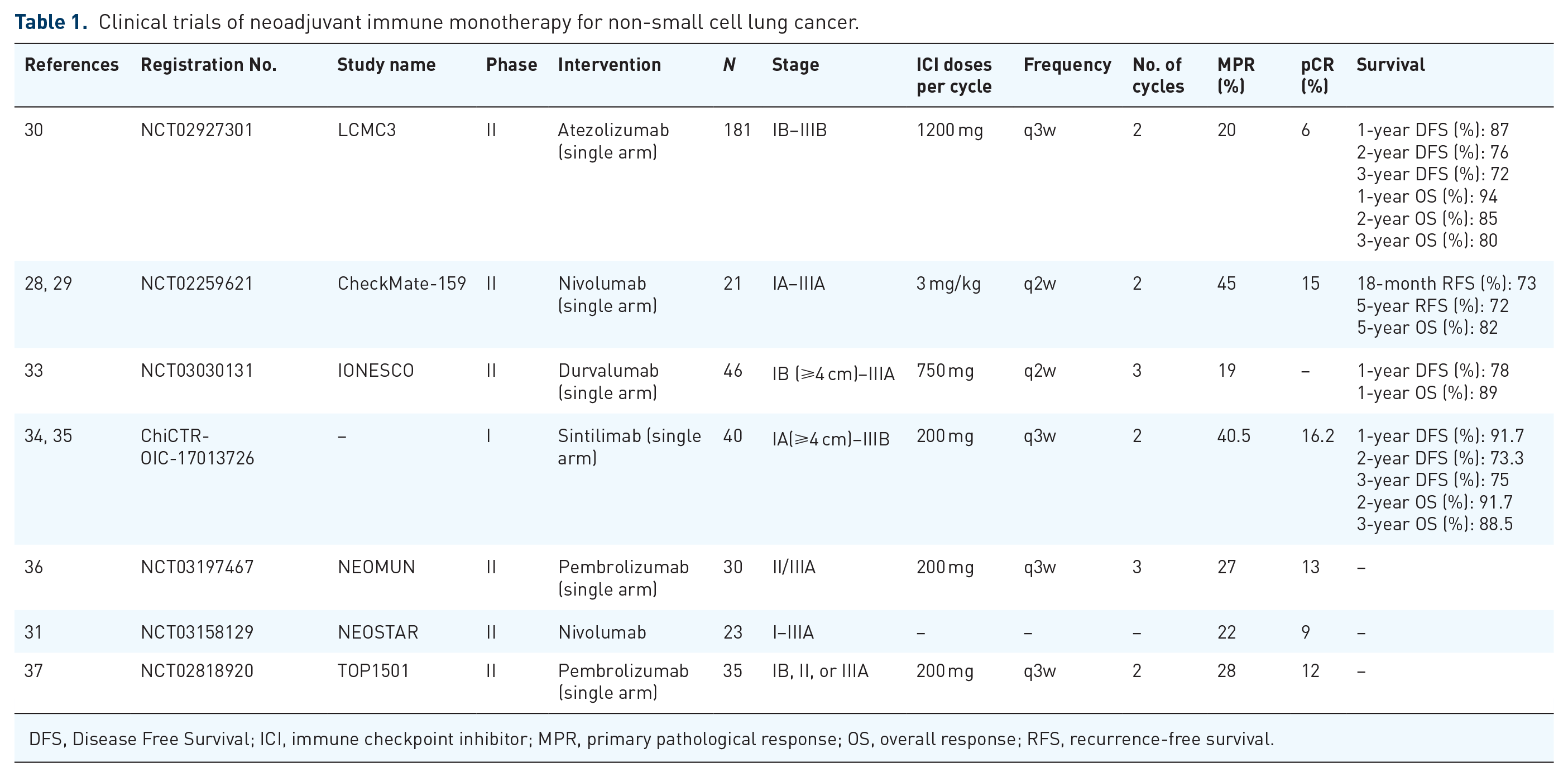
DFS, Disease Free Survival; ICI, immune checkpoint inhibitor; MPR, primary pathological response; OS, overall response; RFS, recurrence-free survival.
Neoadjuvant immune combination therapy
Neoadjuvant immune combination chemotherapy
Following advancements in immunotherapy, many studies are now exploring combination immunotherapy regimens with chemotherapy or other agents. Several phase II trials have shown the efficacy of preoperative neoadjuvant immunotherapy and chemotherapy in patients with stage IB–IIIA NSCLC. The NADIM trial, which included 46 patients with resectable stage IIIA NSCLC, showed promising outcomes: among the 41 patients who underwent surgery, 83% achieved an MPR, 63% attained a pCR, and 90% experienced pathological downstaging. Regarding safety, 30% of patients experienced grade 3 or higher treatment-related adverse events (TRAEs), predominantly elevated lipase and febrile neutropenia; significantly, no adverse events were associated with delayed surgery or death. 38 Recent research indicates a 2-year progression-free survival (PFS) rate of 77.1% (95% confidence interval (CI): 59.9–87.7) for the intention-to-treat (ITT) population, a 36-month OS rate of 81.9% (95% CI: 66.8–90.6), and a 36-month OS rate of 91.0% (95% CI: 74.2–97.0) for the per-protocol population. 39 For the ITT population, the 5-year PFS rate is 65.0% (95% CI: 49.4–76.9), and the OS rate is 69.3% (95% CI: 53.7–80.6). 40 The phase II study NCT02716038 included 30 patients with stage IB–IIIA NSCLC who received four cycles of atezolizumab in combination with carboplatin and albumin-bound paclitaxel preoperatively. Regarding efficacy, the 26 patients who underwent successful resection had an MPR of 57%. The most frequent grade 3 or higher TRAEs included neutropenia (50%), elevated alanine aminotransferase (7%), elevated aspartate aminotransferase (7%), and thrombocytopenia (7%, 2 cases). Notably, no deaths associated with the treatment were reported. 41
The phase III CheckMate-816 study has demonstrated the effectiveness of a neoadjuvant immunotherapy regimen combining nivolumab with platinum-containing doublet chemotherapy, in addition to the earlier phase II studies. This open-label, multicenter randomized trial included 358 patients diagnosed with stage IB–IIIA NSCLC. Participants were randomly assigned to receive either preoperative nivolumab (360 mg) with platinum-based doublet chemotherapy or the chemotherapy alone, administered every 3 weeks for up to three cycles. Partial results from April 2021 showed significantly higher MPR and pCR rates in the combination treatment group compared to the chemotherapy-only group, with MPR at 36.9% versus 8.9% and pCR at 24% versus 2.2% (p < 0.0001). 42 According to the latest data, in terms of the primary endpoint Event Free Survival (EFS), the O drug group combination treatment group achieved a 4-year EFS rate of 49% (compared to 38% in the control group) and a 4-year OS rate of 71% (compared to 69% in the control group). 43
The trials indicate that combining single-agent immunotherapy with platinum-based doublet chemotherapy is an effective preoperative treatment for patients with stage IB–IIIA resectable NSCLC. This approach shows significant benefits in OS, MPR, and pCR, with additional neoadjuvant immune-conjugate chemotherapy trials detailed in Table 2. The incidence remained tolerable despite an increase in grade 3 or higher TRAEs compared to neoadjuvant immune monotherapy regimens.
Clinical trials of neoadjuvant immunotherapy in combination with chemotherapy for the treatment of non-small cell lung cancer.
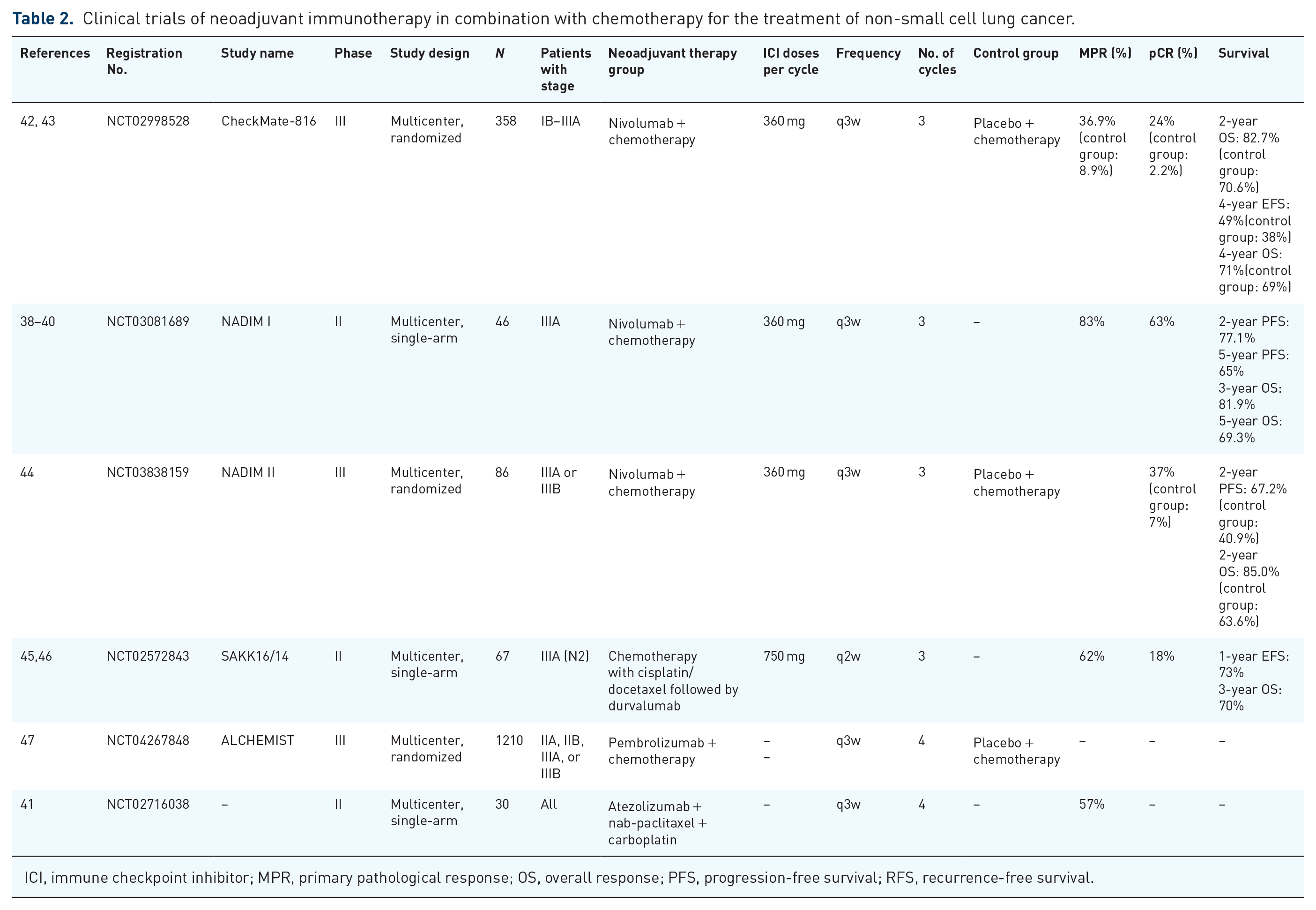
ICI, immune checkpoint inhibitor; MPR, primary pathological response; OS, overall response; PFS, progression-free survival; RFS, recurrence-free survival.
Dual ICIs (O + Y) neoadjuvant immunotherapy regimen
Clinical trials, including CheckMate-9LA, Hellmann MD, and CheckMate 568, have established the combination of nivolumab and ipilimumab (O + Y) as a recommended first-line treatment for NSCLC patients with PD-L1 expression ⩾1%.48–50 In the NEOSTAR study, some patients received nivolumab (O), while the remaining 21 patients were treated with nivolumab combined with ipilimumab (Y). The study found an MPR rate of 25% and a pCR rate of 18%. Notably, the MPR and pCR rates were higher in the combination group compared to the monotherapy group, with MPR at 50% versus 24% and pCR at 38% versus 10%, respectively. Subsequent safety evaluations indicated no notable difference in grade 3 or higher TRAEs between the combination regimen and nivolumab monotherapy (10% vs 13%), with no rise in perioperative morbidity or mortality. This trial demonstrates that the neoadjuvant nivolumab and ipilimumab offer benefits and maintain safety for patients with resectable stage I–IIIA NSCLC compared to nivolumab alone. 31
Neoadjuvant immunotherapy combined with anti-vascular drugs
Recent clinical trials have investigated the combination of neoadjuvant immunotherapy with antiangiogenic treatments. The EAST ENERGY phase II trial assessed the safety and efficacy of two cycles of neoadjuvant pembrolizumab and ramucirumab in resectable PD-L1-positive NSCLC patients. 51 Results from the EAST ENERGY trial showed a 50% MPR rate and a 25% pCR rate, with median RFS and OS not yet reached at 23.6 months of follow-up. In addition, an ongoing study is investigating the combination of anlotinib with pembrolizumab in neoadjuvant therapy (NCT04762030). The synergistic effects of anti-vascular drugs and ICIs may help address the lack of preoperative tumor shrinkage observed with single-agent immunotherapy.
Neoadjuvant immunotherapy combined with radiotherapy
A research study suggests that combining radiotherapy with immunotherapy can enhance treatment outcomes, leading to increased clinical trials investigating neoadjuvant immunotherapy alongside radiotherapy for resectable NSCLC. In the ACTS-30 phase IB trial, 26 NSCLC patients were enrolled, with 24 receiving a neoadjuvant regimen of durvalumab combined with chemotherapy and radiotherapy. Results indicated a treatment-related adverse reaction rate of 16.7% and no deaths, confirming safety. Among the 18 surgically treated patients, postoperative MPR and pCR rates were 77.8% and 38.9%, respectively, demonstrating efficacy. In addition, a phase II trial (NCT03217071) is ongoing to evaluate pembrolizumab with radiotherapy for neoadjuvant therapy in stage I–IIIA NSCLC, aiming to improve the understanding of the safety and feasibility of immune-combination radiotherapy regimens.
Limitations of clinical trials for neoadjuvant immunotherapy
Although neoadjuvant immunotherapy shows promising efficacy in NSCLC patients, existing research faces certain limitations. First, many clinical trials have small sample sizes, which may need to be increased to draw statistically significant conclusions. 52 Second, the selection criteria for patients often need to be more relaxed, leading to heterogeneity in the study results. For example, some studies fail to adequately consider patients’ tumor staging, histological types, and biomarker status, which may affect the assessment of treatment efficacy. 53
Moreover, the monitoring and reporting of immune-related adverse events in existing studies are not comprehensive enough, leading to biases in evaluating the safety of neoadjuvant immunotherapy. 54 Certain studies prioritize MPR or pCR, overlooking crucial endpoints like long-term survival rates and quality of life. 55 This makes it difficult to assess the clinical value of neoadjuvant immunotherapy comprehensively.
Finally, the lack of direct comparative studies of different immunotherapy regimens prevents us from determining the best treatment combinations and timing. 56 For instance, the effects of monotherapy versus chemotherapy combined with immunotherapy have not been sufficiently validated, limiting treatment options in clinical practice. 57
Problems in neoadjuvant immunotherapy
Pseudoprogression versus hyperprogression
Pseudoprogression can be misinterpreted as tumor growth on imaging due to immune cell infiltration or changes induced by treatment. 58 In the CheckMate-159 trial, two patients who initially showed tumor growth post-immunotherapy were later confirmed to have pseudoprogression, with MPR and pCR, upon further pathology analysis. Similarly, the NEOSTAR study identified nodal immune flare, a radiological phenomenon caused by inflammation rather than cancer. 59 Some patients experience rapid deterioration, known as hyperprogression, which necessitates different treatment approaches. Misidentifying these findings in NSCLC patients can delay or even prevent surgery. Therefore, accurately distinguishing between pseudoprogression, proper progression, and hyperprogression is crucial for effective treatment planning and improved patient outcomes. 60 PET/CT with specific contrast agents can aid in differentiating these conditions, but further research into immunoimaging that targets immune cell receptors is essential for enhancing accuracy.45,61–64
Immune tolerance and toxicities
Neoadjuvant immunotherapy carries risks, such as TRAEs, which can delay surgery and potentially increase disease progression. 65 Patients with early-stage cancer often exhibit a better immunological response compared to those with advanced stages. While immunotherapy side effects can complicate lesion removal, most studies indicate that adverse effects are generally manageable, with severe toxicities being rare. However, some studies have reported instances of fatal respiratory toxicity following surgery.31,34 Data indicate that surgery cancellations are primarily due to disease progression, with adverse events being a rare cause for cancellations. 66 Perioperative complications can include pneumonia and thromboembolic events. However, neoadjuvant immunotherapy has not demonstrated a significant increase in complications compared to control groups. 28
The advantages of sandwich-style perioperative immunotherapy are clear, but the target population requires more precision. Patients achieving a pCR following neoadjuvant therapy may benefit from postoperative adjuvant immunotherapy, though there is a potential risk of over-treatment with combination regimens. Imperfect criteria for determining pCR and potential errors in minimal residual disease (MRD) testing may necessitate perioperative immunotherapy for all patients. In addition, identifying the most effective adjuvant regimen for non-responders remains challenging. While neoadjuvant immunotherapy generally improves surgical outcomes, some patients still experience disease progression or miss the opportunity for surgery. In studies,31,67 a small percentage could not undergo surgery due to disease progression while on immunotherapy, posing a new challenge in perioperative care.
Standardization of cycles and regimens for neoadjuvant immunotherapy
Despite expert summaries, neoadjuvant immunotherapy for NSCLC needs more guideline standardization due to limited phase III trials. We raise concerns about the treatment cycle and regimen.
Neoadjuvant immunotherapy aims to shrink tumors and boost the success of surgery, improving long-term survival by eliminating hidden cancer cells. The ideal treatment duration is debated; too short may be ineffective, while too long could promote tumor growth. 68 Most cycles last two to four rounds, with some therapies varying. A study found that three cycles with sintilimab and chemotherapy improved outcomes without added surgery risks. 69 More research is needed to optimize treatment duration for maximum benefit in early-stage NSCLC.
Which is more effective in neoadjuvant treatment: immunotherapy alone or combined with other drugs? No direct clinical studies have compared their efficacy or toxicity. Unlike CheckMate-816, NADIM included adjuvant intravenous nivolumab post-surgery, resulting in higher PFS and OS rates for subsequent adjuvant patients than overall neoadjuvant patients. Further trials are needed to assess the benefits of postoperative immunoadjuvant therapy for neoadjuvant patients, akin to NADIM, and to compare it with adjuvant chemotherapy for long-term survival.
Perioperative immunotherapy includes neoadjuvant immunotherapy, adjuvant immunotherapy, and neoadjuvant combined surgery. Neoadjuvant immunotherapy aims to systematically treat the tumor before surgery to reduce tumor burden and improve pathological response rates, thereby increasing the success rate of surgery and patient survival rates. 70 By contrast, adjuvant immunotherapy is conducted after surgery to eliminate any potentially remaining microscopic lesions and reduce the risk of recurrence. 71 In addition, neoadjuvant combined surgical approaches integrate chemotherapy and immunotherapy to achieve better clinical outcomes. 72 The CheckMate 77T study demonstrated that for operable NSCLC patients, the neoadjuvant regimen of nivolumab combined with chemotherapy, followed by surgery and adjuvant nivolumab, significantly improved EFS compared to neoadjuvant chemotherapy alone (hazard ratio (HR) = 0.58, p < 0.001).
In addition, this regimen increased the pCR rate to 25.3% from 4.7% and the MPR rate to 35.4% from 12.1%. 73 In addition, compared to the chemotherapy group, the nivolumab group had a lower risk of worsening disease-related symptoms, and the surgical outcomes were similar between the two groups, with no new safety signals observed in the nivolumab group. The results of the three phase III studies are consistent. In terms of pCR, the pCR rate in the nivolumab group of the CheckMate 77T study was about five times that of the chemotherapy group (25.3% vs 4.7%, with the CheckMate 816 study showing 24.0%); in the phase III AEGEAN, KEYNOTE-671, and Neotorch studies, the pCR rates for the perioperative durvalumab group, pembrolizumab group, and toripalimab group ranged from 17.2% to 24.8% (the data from the Neotorch study only included resectable stage III NSCLC patients).73–76 The CheckMate 77T study confirmed the correlation between pCR and EFS benefits, aligning with other perioperative immunotherapy research findings.
When selecting an appropriate perioperative immunotherapy strategy, multiple factors must be considered, including the patient’s specific pathological characteristics, tumor staging, genetic mutation status (such as EGFR or ALK mutations), and PD-L1 expression levels. These factors affect the patient’s response to different types of immunotherapy and may also influence their OS prognosis.77,78 Patients with high PD-L1 expression may benefit more from ICIs alone, whereas those with specific gene mutations might require a combination with targeted therapies for effective management.79,80
Perioperative immunotherapy presents promising prospects for NSCLC patients. When selecting neoadjuvant, adjuvant, or neoadjuvant combined regimens, it is essential to comprehensively consider the principles of personalized medicine and develop the best plan based on the patient’s specific circumstances to maximize efficacy and minimize the risk of adverse reactions. Future research will continue to explore the effectiveness of these strategies in different populations and their potential mechanisms, providing more evidence for clinical practice.70,81
Approaches to evaluating the effectiveness of neoadjuvant immunotherapy for ICIs
Clinical studies indicate that neoadjuvant immunotherapy offers prolonged efficacy and diverse response patterns. Accurate assessment of its effectiveness is crucial for optimal patient treatment and long-term benefits. Current assessment methods include imaging and liquid biopsy. Imaging, particularly PET/CT scans, shows promise in evaluating treatment effectiveness, with studies linking decreased Standard Uptake Value (SUV) to favorable outcomes.36,48,82–84 However, the cost and potential discrepancies with pathology warrant consideration.85,86 Liquid biopsy, detecting ctDNA, offers a noninvasive, repeatable method to monitor treatment response, aiding in treatment strategy formulation. Further research is needed to refine ctDNA screening. 87 In addition, a >30% decrease in SUV max on PET/CT can be an evaluation criterion for neoadjuvant immunotherapy.
Biomarkers
ICI has changed lung cancer treatment, but not all patients benefit. Driver-negative NSCLC monotherapy has MPR rates of 19%–45% and pCR rates under 15%, while neoadjuvant immune-combination chemotherapy shows MPR rates of 20%–85% and pCR rates of 8%–57%. This drives the search for predictive biomarkers to identify PD-1/PD-L1 inhibitor beneficiaries and avoid futile treatments. 88
PD-L1
Ongoing research on immunotherapy markers faces challenges in screening populations for neoadjuvant benefits due to a need for quality data. High PD-L1 levels in tumor cells and PD-1 in TILs suggest immunotherapy success, as shown in KEYNOTE-024 and KEYNOTE-042.89,90 Pembrolizumab is FDA approved for advanced NSCLC with ⩾50% PD-L1 expression, and PD-L1 is recommended for driver-negative advanced NSCLC. Higher pre-treatment PD-L1 correlates with better response rates, but inconsistencies in studies indicate the need for further investigation into PD-L1 testing variables.28,31,42,91
Tumor mutational load as well as commonly mutated genes
High tumor mutational load (TMB) levels suggest effectiveness. CheckMate-026 and CheckMat-568 showed better ORR and PFS with TMB ⩾243 and ⩾10 mut/Mb, respectively.92,50 KEYNOTE-158 supported pembrolizumab for high TMB, leading to FDA approval for TMB ⩾10 mut/Mb. Recent studies question TMB’s reliability in immunotherapy. 93
The research shows that patients with EGFR or ALK mutations gain little from ICI treatments and are often excluded from studies.94,95 However, the IMpower150 trial found that atezolizumab with Bevacizumab + carboplatin + paclitaxel (BCP) improved OS for EGFR-sensitive patients compared to standard treatment, indicating a need for further exploration in NSCLC patients post-targeted therapy. NSCLC patients with KRAS and BRAF V600E mutations may respond better to immunotherapy.
Early data on immunotherapy for NSCLC with driver mutations are limited. STK11 and KEAP1 mutations worsen outcomes, as shown by the MYSTIC study, which showed poor results for durvalumab. However, KEYNOTE-042 found no significant impact of STK11 mutations with pembrolizumab, indicating unclear roles.89,96,97 The NADIM study reported median PFS for NSCLC patients with mutations in STK11, KEAP1, RB1, and EGFR who received neoadjuvant immunotherapy. 38
Relevant biomarkers in peripheral blood
Peripheral blood offers noninvasive, easily accessible samples, allowing comprehensive immune status assessment. The research explores peripheral blood indicators for anti-PD-(L)1 treatment response, including ctDNA, blood cell counts, circulating immune cells, and T-cell receptors. 98
CtDNA predicts lung cancer treatment outcomes; its clearance post-surgery correlates with better results. 99 Studies like LUNGCA and NADIM show its importance, while CheckMate 816 and AEGEAN highlight its impact on treatment response.42,100 Current analysis methods are limited, necessitating improved tests for MRD detection.100–102
Routine blood tests indicate blood cell counts and ratios that may reflect tumor response to ICIs. In advanced NSCLC, higher NLR and M/L ratios are linked to poor survival with PD-1 treatment. 103 The NADIM study showed decreased specific blood cell levels after neoadjuvant immune combination chemotherapy. More trials are necessary to confirm the predictive value of blood cell markers for neoadjuvant immunotherapy.
Flow cytometry analysis found that ILT2 and NKG2A expressing cells, like NK and NK-like T cells, predict immunotherapy response pre-surgery.
TCRs recognize neoantigens for immune antitumor effects, assessed by deep sequencing; studying blood TCR pools as immunotherapy markers is nascent.
Higher T-cell density in blood correlates with better OS in early lung adenocarcinoma. 28 However, TCR characteristics did not predict neoadjuvant ICI therapy success, with a dominant clonal TCR linked to response and PFS, necessitating further trials to confirm TCR’s predictive value.
TME-related markers
Neoadjuvant therapy in advanced NSCLC enables tissue analysis before and after treatment, comparing immune responses to immunological drugs via histological staining and genomic testing.
Tumor immunity relies on cellular immunity, particularly T lymphocytes, with CD3+ T lymphocyte balance being crucial. Higher CD4+ and CD8+ T-cell levels in tumors improve immunotherapy responses, while lower levels increase mortality risk.103–105 The NADIM study linked specific T cells and macrophages to better treatment outcomes. 38 Increased tissue-resident memory T cells indicate a better prognosis in early NSCLC. 106 Neoadjuvant treatments raise T-cell levels in tumors, and ILT2 expression in immune cells relates to treatment response, while specific tumor-associated macrophages can impede immunotherapy. 107 Cancer-associated fibroblasts create a suppressive microenvironment, affecting treatment efficacy. 108 Factors like ILT2, IL-6, IL-8, and IFN-γ levels may influence immunotherapy responses, and high TMB correlates with improved outcomes.28,109 Different tumor immune cell types can predict neoadjuvant immunotherapy responses, necessitating further research on biomarkers for personalized treatment.
Studies suggest that the diversity and clonality of immunohistochemical libraries can forecast recurrence in resectable stage IIIA (N2) NSCLC following neoadjuvant chemotherapy with durvalumab. 45 Post-treatment TCR libraries in tumors correlate with EFS, MPR, and nodal clearance. In the NEOSTAR trial, resected tumors from both therapy groups showed increased TCR diversity and reactivity compared to baseline, with higher abundance and clonotypes than paraneoplastic lung tissue.
Examination of immune cells in tumors showed dysfunction and poor recognition, hindering immune response. The NADIM study found that neoantigen-specific immune cells in non-MPR patients had higher exhaustion markers. 110 In MPR patients, neoantigen-specific T cells expressed more memory and effector genes. By contrast, unresponsive mutation-specific T cells with high HOBBIT levels had reduced TCR signaling and increased immune checkpoints. These findings suggest strategies to overcome resistance to PD-1 inhibitors.
Homologous recombination deficiency can predict immunotherapy response in NSCLC patients by identifying individuals with DNA repair gene mutations. This identification can improve treatment responses and prolong survival. 111
The focus has been on discovering biomarkers in the TME, especially changes in immune cell phenotypes before and after neoadjuvant therapy, with studies on CD4+, CD8+ T-cells, CD68+ macrophages, and PD-1+ lymphocytes, highlighting CD4+ PD-1+ and CD3+ PD-1+ T-cells as predictors of immunotherapy efficacy.
In addition, researchers are examining the diversity of immunomic libraries, including the clonality and diversity of TCR libraries in tumor tissue specimens. Furthermore, other genomic markers, such as the relationship between the transcriptional profiles of neoantigen-specific TILs and MPR, are still under investigation.
Gut microbiota
The gut microbiome influences the immune system and immunotherapy, balancing bacterial threats as the largest immune organ. Cancer can evade immune responses through bacterial interactions, while the immune system alters bacterial signals, affecting tumor response. 112 Gut bacteria diversity impacts immunotherapy effectiveness.113–118 Patients with diverse flora respond better, while those with dysbiosis or antibiotic history may have poorer outcomes, suggesting microbiome characteristics can predict treatment responses.
One study showed that antibiotics in metastatic NSCLC patients hindered tumor response to ICIs by changing intestinal flora. Other studies in Chinese populations linked gut microbiome diversity to anti-PD-1 immunotherapy effectiveness, with higher diversity correlating to longer PFS.113,114 NSCLC patients on ICIs had increased gut bacteria levels associated with treatment efficacy and diversity. 119 NEOSTAR study analyses found specific gut bacteria abundance linked to treatment response and fewer side effects. 66 A 2021 study highlighted gut flora’s role in neoadjuvant immunotherapy effectiveness, suggesting more research is needed. 120
Neural network modeling
Immunotherapy’s complexity can make biomarkers unreliable. Shen Lin’s team created a model to predict gastric cancer responses, aiding precision treatment. 121 Researchers improved their understanding of immunotherapy factors using diverse data and machine learning. Our center also developed a predictive model for squamous lung cancer efficacy, achieving high validation accuracy. 122
Various studies link the efficacy of PD-1/PD-L1 inhibitors to biomarkers such as PD-L1 expression and TMB, yet trial outcomes differ. According to the International Expert Consensus on Neoadjuvant Immunotherapy for NSCLC, no definitive efficacy markers exist, and marker-guided drug strategies are not recommended. 123
Challenges facing adjuvant immunotherapy in the future
Research on preoperative immunotherapy for NSCLC is progressing swiftly. Future clinical trial designs need to consider several key factors to ensure the effectiveness and safety of the studies. Here are some recommendations to guide future research:
(1) Future research should refine patient selection criteria by considering tumor staging, molecular characteristics like PD-L1 expression and tumor mutation burden, and the patient’s overall health status. This approach will pinpoint the patient group most likely to benefit from neoadjuvant immunotherapy. 124
(2) Optimization of treatment regimens: The research should explore different immunotherapy regimens, including monotherapy combined with chemotherapy and combinations of different ICIs. These combinations may enhance immune responses and improve the rate of pCR. 125
(3) Biomarker research: Future research should focus on developing and validating biomarkers to predict patient responses to neoadjuvant immunotherapy. These biomarkers can assist physicians in assessing patient prognosis before treatment and formulating personalized treatment plans. 126
(4) Clinical trial design: Future clinical trials should adopt randomized controlled designs to ensure the reliability of results. Furthermore, trials should incorporate novel assessment criteria, including pCR rates and EFS, to enhance the evaluation of treatment efficacy. 127
(5) Long-term follow-up: Since the effects of immunotherapy may take a long time, future research should include long-term follow-up to assess the impact of neoadjuvant immunotherapy on patient survival and quality of life. 128
(6) Multicenter collaboration: Encouraging multicenter collaborative research to increase sample size and diversity will enhance the generalizability of research findings. This will help to gain a more comprehensive understanding of the effects and safety of neoadjuvant immunotherapy. 129
Implementing these recommendations will enhance the evaluation of neoadjuvant immunotherapy in NSCLC clinical trials, offering improved patient treatment options.
Conclusion
Previously, platinum-based chemotherapy was the mainstay for treating NSCLC patients. The discovery of gene mutations such as EGFR and ALK fueled research into effective antitumor targets. The discovery of immune checkpoints such as PD-1/PD-L1 and CTLA-4 has generated considerable interest in immunotherapy, establishing ICIs as an effective means for sustained tumor management.
Neoadjuvant immunotherapy enhances surgical outcomes and reduces recurrence in NSCLC without driver mutations, with combination therapies proving more effective. However, further research is needed for precision therapy, particularly in patient selection and biomarker identification. A multidisciplinary approach is essential for personalized treatment.
Upcoming clinical studies are expected to explore perioperative systemic treatment options to optimize biomarkers for improved patient selection and survival outcomes.
