Abstract
Background:
Detectable Epstein–Barr virus (EBV) DNA levels and unsatisfactory tumor response to induction chemotherapy (IC) could be used to guide the risk-adapted treatment strategy of locoregionally advanced nasopharyngeal carcinoma (LANPC) before concurrent chemoradiotherapy. We aim to compare the efficacy and safety of concurrent chemotherapy using taxane plus cisplatin [double-agent concurrent chemotherapy (DACC) group] with those of cisplatin alone [single-agent concurrent chemotherapy (SACC) group] in high-risk LANPC.
Methods:
Overall, 197 LANPC patients with detectable EBV DNA or stable disease (SD) after IC were retrospectively included. Potential confounders between the DACC and SACC groups were adjusted by propensity score matching. Short-term efficacy and long-term survival were assessed in the two groups.
Results:
Although the objective response rate of the DACC group was marginally higher than that of the SACC group, the difference was not significant (92.7% versus 85.3%, p = 0.38). Concerning long-term survival, DACC did not show superiority to SACC after patient matching: 3-year progression-free survival: 87.8% versus 81.7%, p = 0.80; overall survival: 97.6% versus 97.3%, p = 0.48; distant metastasis-free survival: 87.8% versus 90.5%, p = 0.64, and; locoregional relapse-free survival: 92.3% versus 86.9%, p = 0.77. The incidence of grade 1–4 hematological toxicities was significantly higher in the DACC group.
Conclusion:
Due to the small sample size, we do not have sufficient evidence that concurrent chemotherapy using taxane plus cisplatin provides additional survival benefits in LANPC patients with an unfavorable response (detectable EBV DNA levels or SD) after IC. But concurrent taxane and cisplatin chemotherapy is associated with a higher rate of hematologic adverse events. Further clinical trials will be required to establish evidence and identify more effective treatment modalities for high-risk LANPC patients.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a head and neck cancer closely correlated with the Epstein–Barr virus (EBV) and is highly prevalent in South China. Owing to its nonspecific symptoms in the early stages, 60−80% of newly diagnosed patients are locoregionally advanced nasopharyngeal carcinoma (LANPC), which has a poor prognosis and a high risk of distant metastasis. 1 Induction chemotherapy (IC) followed by platinum-based concurrent chemoradiotherapy (CCRT) is the first choice of treatment for LANPC.1,2 Previous studies demonstrated that tumor response to IC and EBV DNA were complementary and could be used to preidentify populations at high risk of treatment failure before CCRT for LANPC. 3 Particularly, complete response (CR) or partial response (PR) to IC indicates sensitivity to chemotherapy, and thus, the deintensification of IC plus radiotherapy is sufficient for patients with these responses. 4 Meanwhile, a suboptimal response [stable disease (SD) or disease progression (PD)] after IC or detectable EBV DNA (>0 copies/mL) indicates a poorer outcome [3-year progression-free survival (PFS): 71.1% versus 85.9%, p = .005]. 3 Thus, identifying effective treatment strategies for this high-risk subgroup is crucial for individualized treatment in LANPC.
However, little attention has been paid to the treatment strategies of these high-risk patients with suboptimal responses to IC. Adding another agent, such as paclitaxel/docetaxel, to cisplatin-based concurrent chemotherapy may be a potential strategy to improve treatment efficacy. A meta-analysis indicated that for advanced NPC, compared with 5-FU plus cisplatin, paclitaxel plus cisplatin was associated with a higher CR and lower incidence of adverse reactions. 5 A phase II study of LANPC patients also found that although the long-term survival rates were not significantly better compared to those receiving cisplatin alone, patients receiving raltitrexed plus cisplatin concurrent chemotherapy had a short-term objective response rate (ORR) of 98%. 6 Moreover, a recent study comparing concurrent dual-agent chemotherapy (conventional paclitaxel/5-FU plus cisplatin) and single-agent (cisplatin alone) chemotherapy has shown that the dual-agent regimen was not effective in prolonging survival and had greater hematologic toxicities. 7 Whether double-agent concurrent chemotherapy (DACC) could provide additional survival benefits and the optimal DACC regimens and patient subsets warrants investigation.
Taxanes (docetaxel, paclitaxel liposome, and nab-paclitaxel) are increasingly used in solid tumors such as breast cancer and nonsmall cell lung cancer (NSCLC).8–10 Studies have shown that radiotherapy combined with paclitaxel plus cisplatin/carboplatin chemotherapy is well tolerated and improves the survival rate in patients with inoperable locally advanced esophageal cancer and with NSCLC.11,12 In a phase I/II study of the combination of paclitaxel and cisplatin regimen, the ORR was 66% in patients with relapsed and metastatic NPC. 13 However, concurrent paclitaxel chemotherapy failed to improve efficacy in pancreatic cancer and had similar toxicity to the standard regimen. 14 Meanwhile, in LANPC, concurrent chemotherapy with a taxane plus cisplatin can ensure treatment efficacy while avoiding excessive toxicity is still unclear. Therefore, this study aimed to evaluate the efficacy and safety of concurrent chemotherapy using taxane plus cisplatin compared to that of cisplatin alone for LANPC patients with suboptimal responses (detectable EBV DNA or SD) to IC. While waiting for more convincing evidence, we attempted to modify the dual-agent chemotherapy regimen with taxane and cisplatin.
Methods
Study design and patients
This retrospective study evaluated 197 patients with LANPC who received taxane plus cisplatin or cisplatin alone during radiotherapy at Sun Yat-sen University Cancer Center between April 2013 and December 2020 (Figure 1). The inclusion criteria were as follows: (1) newly pathologically diagnosed stage III–IVA LANPC (T3-4NanyM0 or TanyN2-3M0) based on the Union for International Cancer Control/American Joint Committee on Cancer eighth edition staging system; (2) received IC and CCRT; (3) completed two to four cycles of IC; (4) available magnetic resonance images of the nasopharynx and neck before and after IC for tumor response evaluation; (5) detectable EBV DNA or SD response after two to four cycles of IC, as evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1), and; (6) no additional immunotherapy or adjuvant chemotherapy. The exclusion criteria were as follows: (1) any history of malignancy; (2) pregnancy or breastfeeding, and; (3) the presence of severe coexisting illness.
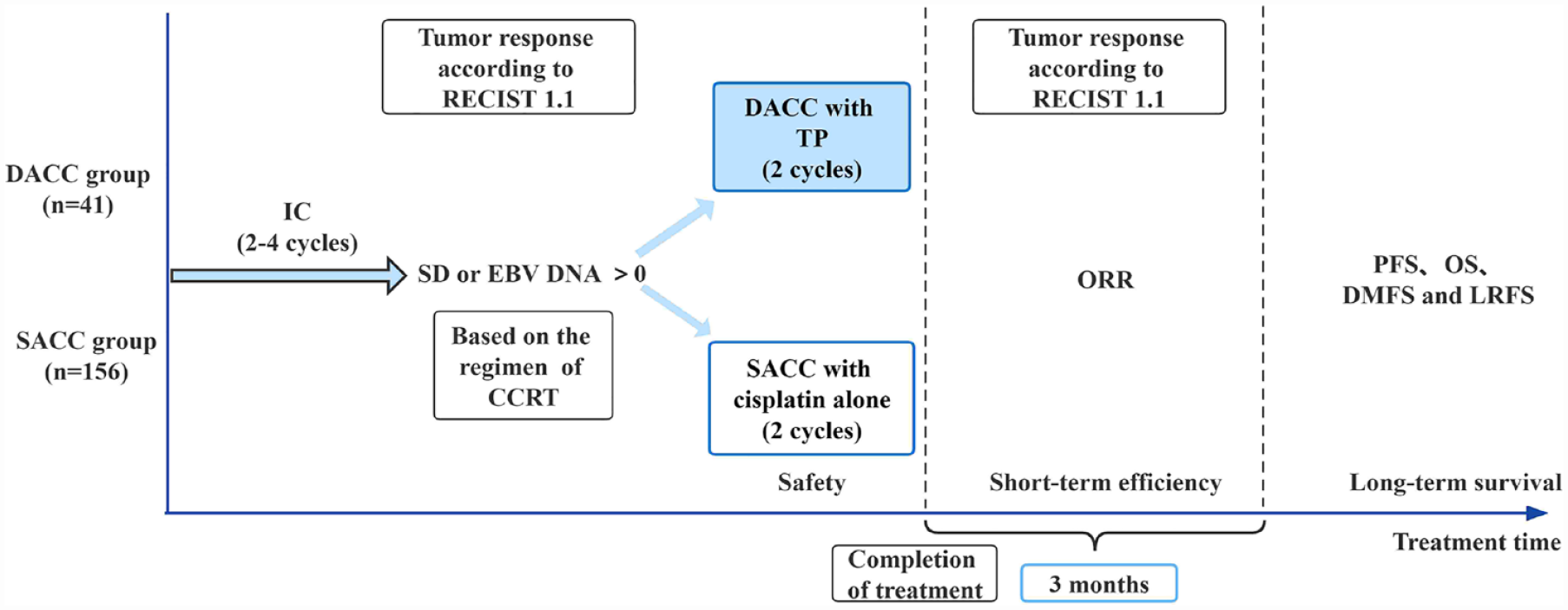
Treatment scheme of the two groups. All the included patients received two to four cycles of IC and were deemed resistant to the initial IC regimen (with detectable EBV DNA or SD according to RECIST 1.1). In addition, 41 patients received concurrent chemotherapy of taxane plus cisplatin regimen (the DACC group), and 156 patients received cisplatin alone (the SACC group).
The patients were divided into two groups based on their concurrent chemotherapy: those who received taxane-containing DACC and cisplatin [single-agent concurrent chemotherapy (SACC)] groups. This study was approved by the Clinical Research Committee of Sun Yat-sen University Cancer Center (approved number: B2022-674-01) and was waived of informed consent. The study was conducted following the principles of the Helsinki Declaration.
Assessment of plasma level of EBV DNA
Plasma EBV DNA concentrations were determined using a real-time quantitative polymerase chain reaction assay before treatment, after IC, and 3 months after the completion of CCRT. The cut-off values of pretreatment and posttreatment EBV DNA levels were 4000 copies/mL and 0 copies/mL, respectively. These cut-off values have been identified as a predictive marker of tumor burden and tumor response to treatment in previous studies.15–17
Treatment and tumor response evaluation
All of the patients received IC plus CCRT. IC regimens included TP, TPF, or TP/TPF combined with targeted therapy. T indicated taxane in any form, including docetaxel (75 mg/m2, day 1), paclitaxel liposomal (135–175 mg/m2, day 1), and nab-paclitaxel (260 mg/m2, day 1). P indicated cisplatin (80–100 mg/m2, day 1). F referred to fluorouracil (300–500 mg/m2, continuous intravenous infusion for 3–5 days). Targeted therapy indicated nimotuzumab (200 mg, day 1) or cetuximab (initial dose of 400 mg/m2, day 1 and 200 mg/m2, day 1 on the subsequent course of treatment). Every regimen was administered at 3-week intervals of two to four cycles. In the DACC group, the concurrent chemotherapy regimen comprised taxane (docetaxel/paclitaxel liposomal/ nab-paclitaxel) and cisplatin (80–100 mg/m2), which were administered on days 1 and 22 during radiotherapy combined with targeted therapy [nimotuzumab (200 mg, weekly during radiotherapy)].
All patients underwent intensity-modulated radiotherapy (IMRT) five times per week for 6–7 weeks at 2.00–2.34 Gy per fraction. The planning target volume dose was 68–70 Gy. Other details of the IMRT plan were in line with previous studies. 18 All patients underwent magnetic resonance imaging (MRI) of the nasopharynx and neck post-IC and 3 months after treatment completion, and images were independently evaluated by two physicians according to RECIST 1.1. Tumor responses were divided into the following four categories: CR, PR, SD, and PD.
Follow-up and end points
The primary end point was PFS, defined as the first day of IC to any treatment failure or death from any cause. The secondary end points were overall survival (OS, defined as the first day of IC to death from any cause), distant metastasis-free survival (DMFS, defined as the first day of IC to documented distant metastasis), and locoregional relapse-free survival (LRFS, defined as the first day of IC to documented locoregional recurrence). After treatment, patients were followed up every 3 months for the first 2 years, every 6 months for the next 3–5 years, and annually thereafter or until death. The routine evaluation included fiberoptic pharyngorhinoscopy, an MRI of the nasopharynx and neck, and a serological test for EBV. Any suspected recurrence or distant metastasis was further evaluated by biopsy or imaging techniques.
Statistical analysis
Between-group comparisons were performed using the chi-square test or Fisher exact test for categorical variables. Kruskal–Wallis H test was applied to analyze grade 1–4 toxicities in both groups. Propensity score matching (PSM) was performed to minimize bias in evaluating the curative effect. A logistic regression model was used to calculate propensity scores based on smoking, overall stage, pretreatment EBV DNA, IC regimen, and IC cycles. The dependent variable was the regimens of concurrent chemotherapy. Then, all the included patients were matched without replacement by two methods: (i) 1:2 nearest-neighbor matching and (ii) caliper matching, which utilized a caliper size of 0.2. Survival curves were generated using the Kaplan–Meier method and compared using the log-rank test. A Cox proportional hazard model was used for the multivariate analyses of survival outcomes. The hazard ratios (HRs) with their 95% confidence intervals (CIs) were calculated using the Cox proportional hazards model (treatment group as a single covariate), and further interaction analyses were performed to assess whether treatment effects differed among subgroups. All statistical analyses were performed using Statistical Package for the Social Sciences (SPSS) software (version 25.0) and R (version 4.1.3). Two-sided p values >0.05 were considered statistically significant.
Results
Patient characteristics
The SACC and DACC groups included 156 (79.2%) and 41 (20.8%) patients, respectively. The baseline patient characteristics are presented in Table 1. Overall, the median patient age was 48 years (range, 15–76 years), and the male:female ratio was 3.2:1. The median copies of pretreatment EBV DNA was 4370 (range, 0–1,400,000 copies/mL). The chemotherapy plus targeted therapy rate was 55.3% concerning the IC regimen. After 1:2 nearest-neighbor matching, 116 (75 versus 41) patients remained in the entire cohort. During the matching process, only one object was matched to each of the seven patients, so the final matching results did not fully satisfy the 1:2 ratio. After PSM, the standardized mean difference (SMD) of covariates, including smoking, overall stage, pretreatment EBV DNA, IC regimen, and IC cycles, is 0.07, 0.03, 0.07, 0.05, and 0.00 (all SMD < 0.1). In the matched cohort, stage IV patients made up 89%. The level of pretreatment EBV DNA ⩾ 4000 copies/mL was higher than that before matching, accounting for 57.8% of the matched population. In addition, in the DACC and SACC groups, the maximum number of copies of post-IC EBV DNA is 36,800 and 17,900, respectively, and the minimum is zero. For IC, the ratio of chemotherapy alone to chemotherapy in combination with targeted therapy was 2.74, with all matched patients receiving three to four courses of IC.
Baseline characteristics of the entire cohort before and after matching.

Bold indicates p value < 0.05.
Patients were divided into two groups according to the regimen of concurrent chemotherapy.
Patients were matched with smoking, overall stage, pretreatment EBV DNA, IC regimen, and IC cycles, which used a method of PSM.
TNM stage was based on the eighth edition of UICC/AJCC staging system.
No statistics are computed because IC cycles after matching is a constant.
EBV DNA, DNA load of Epstein–Barr virus; IC, induction chemotherapy; NPC, nasopharyngeal carcinoma; PSM, propensity score matching.
Treatment efficacy
Short-term efficacy was evaluated by MRI of the nasopharynx and neck and plasma EBV DNA level 3 months after the completion of CCRT (Table 2). Among all patients, magnetic resonance images were unavailable in seven patients, and EBV DNA data was inaccessible in nine patients. The ORR of the SACC and the DACC groups was 85.3% (133/156), and 92.7% (38/41), respectively. For the patients who experienced unsatisfactory responses (101 of 197 were SD) to IC, 18 of 197 (9.1%) patients still had a post-CCRT SD response. One patient was evaluated for PD in the entire cohort. The number of patients in the SACC and DACC groups who still had detectable EBV DNA after CCRT was 21 of 156 (13.5%) and 5 of 41 (12.2%). The comparison of PFS, OS, DMFS, and LRFS survival curves between the unmatched and matched cohorts are presented in Figures 2 and 3, respectively. The median follow-up was 54.5 months (range, 1–107 months). In general, 15 (7.6%) patients experienced locoregional relapse, 16 (8.1%) experienced distant metastasis, and 9 (4.6%) patients died. Before matching, there was no significant between-group difference for 3-year PFS (86.6% versus 87.8%, p = 0.56), OS (95.2% versus 97.6%, p = 0.77), DMFS (89.6% versus 87.8%, p = 0.60), and LRFS (90.6% versus 92.3%, p = 0.64). After matching, the 3-year PFS of the DACC group was not significantly better than that of the SACC group. There were also no significant differences in other endpoints between the two groups (all p > 0.05). More specifically, the 3-year PFS, OS, DMFS, and LRFS in the SACC and DACC groups were (81.7% versus 87.8%, p = 0.80), OS (97.3% versus 97.6%, p = 0.48), DMFS (90.5% versus 87.8%, p = 0.64), and LRFS (86.9% versus 92.3%, p = 0.77) after matching. Of all the patients, multivariate analysis indicated that the IC regimen was an independent prognostic factor of PFS (TP/TPF versus TP/TPF + target: HR = 0.58; 95% CI: 0.34–0.97; p = 0.036), LRFS (HR = 0.41; 95% CI: 0.20–0.83; p = 0.013), and DMFS (HR = 0.33; 95% CI: 0.15–0.70; p = 0.004). Overall stage showed independent significance for PFS (HR = 0.21; 95% CI: 0.05–0.88; p = 0.032). Patients with a pretreatment EBV DNA level ⩾4000 copies/mL had a higher risk of distant metastasis than those with an EBV DNA <4000 copies/mL (HR = 3.41; 95% CI: 1.10–10.60; p = 0.034) (Table 3). In addition, subgroup analysis in the matched patients did not show significant advantages in PFS in the DACC group (Figure 4). Unfortunately, 95% CIs contained 1.0 in both the general population (HR = 0.79; 95% CI: 0.27–2.29; p = 0.66) and other subgroups, indicating that there was no statistically significant difference between the two therapeutic strategies.
Short-term efficacy was assessed 3 months after the completion of treatment.
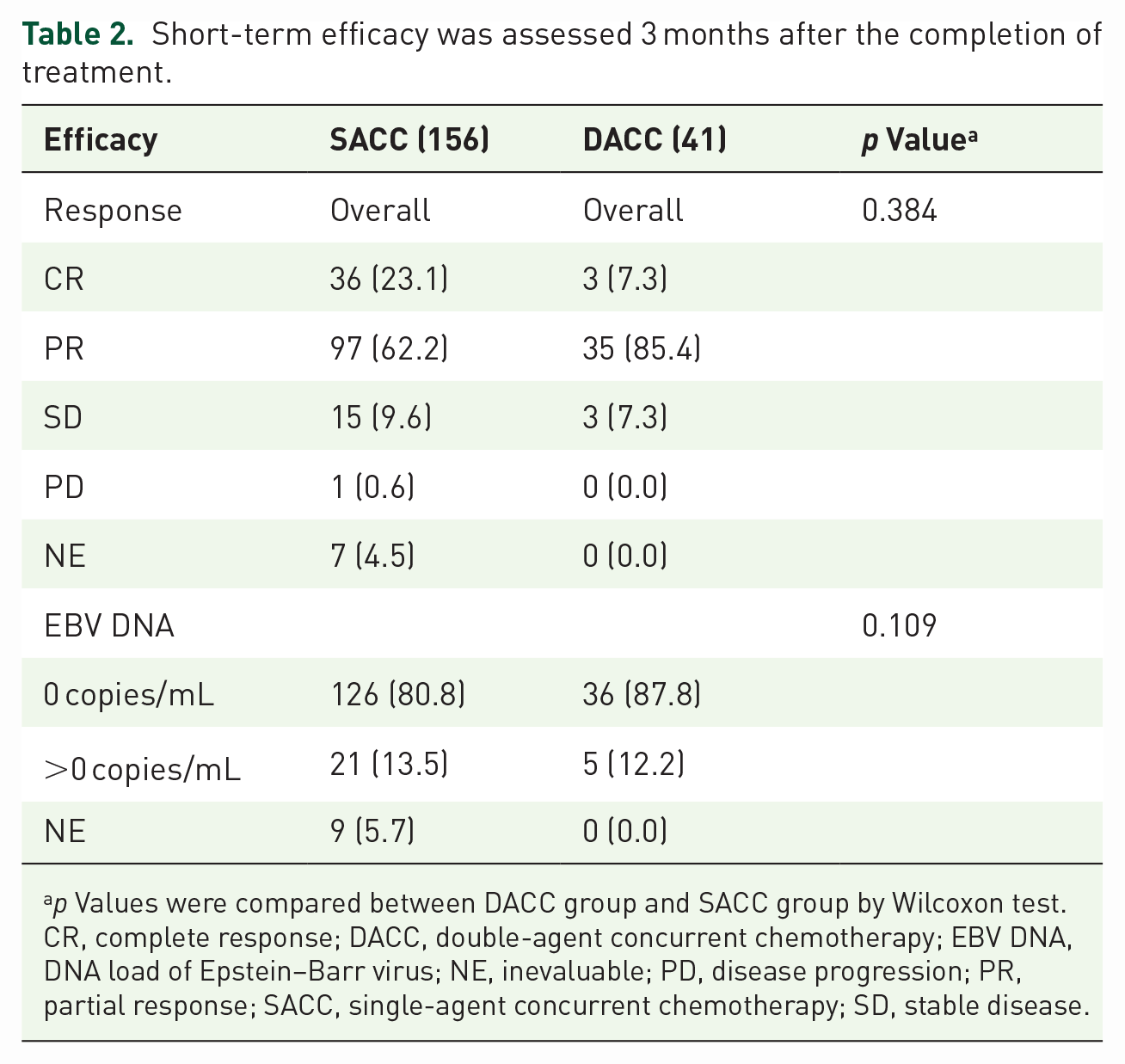
p Values were compared between DACC group and SACC group by Wilcoxon test.
CR, complete response; DACC, double-agent concurrent chemotherapy; EBV DNA, DNA load of Epstein–Barr virus; NE, inevaluable; PD, disease progression; PR, partial response; SACC, single-agent concurrent chemotherapy; SD, stable disease.

Kaplan–Meier survival curves of two groups before matching. Survival curves of (a) PFS, (b) OS, (c) DMFS, and (d) LRFS in all the included patients.

Kaplan–Meier survival curves of two groups after PSM. Survival curves of (a) PFS, (b) OS, (c) DMFS, and (d) LRFS in the two groups after matching with smoking, overall stage, pretreatment EBV DNA, IC regimen, and IC cycles.
Multivariate analysis of prognostic factors in the entire cohort.

Bold indicates p value < 0.05.
p Values were calculated by multivariate Cox proportional hazards regression analysis.
DMFS, distant metastasis-free survival; EBV DNA, DNA load of Epstein–Barr virus; HR, hazards ratio; IC, induction chemotherapy; LRFS, locoregional relapse-free survival; NPC, nasopharyngeal carcinoma; OS, overall survival; PFS, progression-free survival; 95% CI, 95% confidence interval.

Subgroup analysis of PFS in the matched patients. A Cox proportional hazards model was used to calculate HRs and 95% CIs and to conduct an interaction test, which included the interaction term (e.g., age × treatment), a covariate of interest (e.g., age), and the trial group. An HR of more than 1.0 implies a higher risk of disease progression with DACC than with SACC.
Toxicities
As shown in Table 4, grade 4 adverse events occurred in 6 (3.8%) patients in the SACC group and 13 (31.7%) patients in the DACC group. No other grade 5 adverse events or treatment-related deaths were reported in both groups. The most common adverse event in the entire cohort was hematologic adverse events, especially anemia, which occurred in 155 (78.7%) patients. Notably, DACC treatment was significantly associated with an increased incidence of grade 1–4 hematological toxicities including leukocytopenia [34 (82.9%)], neutropenia [23 (56.1%)], and thrombocytopenia [18 (43.9%)]. However, the incidence of grade 1 and grade 2 nephrotoxicity was higher in the SACC group [141 (90.4%)]. Meanwhile, the incidence of other adverse events was similar between the two groups.
Grade 1–4 acute toxicities between the two groups during concurrent chemotherapy.

Bold indicates p value < 0.05.
p Values were calculated by Kruskal–Wallis H test.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; DACC, double-agent concurrent chemotherapy; SACC, single-agent concurrent chemotherapy.
Discussion
In this study, we compared the feasibility and efficacy of DACC (taxane plus cisplatin) with those of SACC (cisplatin alone) in LANPC patients with suboptimal response to IC and found that concurrent chemotherapy using double agent did not provide any significant survival benefit when compared with cisplatin alone before and after PSM. In addition, the incidence of grade 1–4 hematological toxicities including leukocytopenia, neutropenia, and thrombocytopenia was higher in the DACC group than in the SACC group.
For patients with stage III–IVA (except T3N0) NPC, IC followed by CCRT is the recommended treatment modality in the American Society of Clinical Oncology (ASCO) and Chinese Society of Clinical Oncology (CSCO) guidelines. 19 IC plays an essential role in the IMRT era, mainly by eliminating distant metastasis to improve patient outcomes. In the updated MAC-NPC meta-analysis, which included 19 trials and 4806 patients from endemic and nonendemic areas, the results confirmed that the addition of concomitant chemotherapy to radiotherapy significantly improved OS (HR = 0.79; 95% CI: 0.73–0.86; p < 0.001).20,21 However, 30% of patients have a suboptimal response (SD or PD) to IC, resulting in poor outcomes and treatment failure. 3 Previous studies have shown that tumor response and EBV DNA after IC are reliable prognostic factors for risk stratification of NPC.22,23 A study of a nonendemic area (in France) reported that 5-year OS for patients with post-IC SD was 60%, while post-IC CR/PR was 100%/79.4%. 24 Luo et al. 4 found that concurrent cisplatin chemotherapy appears to be inadequate for patients with a suboptimal response to IC, and intensive therapy is needed. A recent study by Italian scientists has shown that negative pretreatment of EBV DNA can be considered a prognostic biomarker for longer disease-free survival (DFS) and OS in NPC in nonendemic regions. Unfortunately, the study lacked information on posttreatment testing for EBV DNA viral load. 25 However, the optimal strategy for increasing the intensity of treatment for these high-risk patients based on concurrent cisplatin chemotherapy is still to be clarified. A phase II study reported that concurrent dual-agent chemotherapy (raltitrexed plus cisplatin) was a safe and effective regimen in NPC, with an ORR of 98% and a 2-year OS rate of 92%. 6
In a combined analysis of six randomized trials in NPC, concurrent TP (paclitaxel + cisplatin) was associated with a significantly higher CR (OR = 2.60, 95% CI: 0.37–18.17, p < 0.05) and a significantly lower incidence of grade 3 and grade 4 gastrointestinal injury (OR = 0.38, 95% CI: 0.16–0.93, p < 0.05) than was PF (cisplatin + 5-FU). 5 However, Qin et al. 7 found that concurrent double-agent chemotherapy did not provide survival benefits and was associated with higher hematological toxicities than a single-agent regimen. Collectively, these findings support that concurrent dual-agent chemotherapy may be a potential treatment option for high-risk LANPC patients. However, it remains unclear whether this regimen is safer and more effective. Therefore, we compared concurrent chemotherapy using taxane plus cisplatin with cisplatin alone in patients with a suboptimal response (detectable EBV DNA, or SD) to IC. The results showed that the addition of taxane to cisplatin-based concurrent chemotherapy did not improve survival benefits and increased acute toxicities. Although the ORR of the DACC group was slightly higher than that of the SACC group, there were no significant differences between the two groups. Further, DACC did not show an advantage in the 3-year PFS, OS, DMFS, and LRFS.
There were several possible explanations for the nonsignificant difference in survival between the two groups. First, the potential survival benefit of DACC is decreased in the IMRT era. Studies have shown that concurrent chemotherapy helped improve local control and increased the opportunity for sensitizing radiotherapy in LANPC.26,27 However, with the development of radiation technology, there was evidence that the advantage of IMRT was mainly associated with local control when compared with conventional radiotherapy.28–30 Therefore, the application of IMRT may weaken the local control benefits of concurrent chemotherapy, and the radio-sensitization benefit of DACC may be similar to that of SACC. Second, the suboptimal responders to IC in the current study may be insensitive to the taxane-containing chemotherapy regimen. Thus, the use of the same or similar drugs in CCRT may reduce the efficacy of concurrent chemotherapy to some extent. Compared to the good prognosis of the landmark gemcitabine and cisplatin IC trial, we found that in the landmark trial, 97.1% of the patients (235 of 242) in the IC group had a CR, as did 96.6% of the patients (230 of 238) in the standard therapy group 1 month after radiotherapy. 31 Of the 480 patients, only two had PD and none had SD. In the current study, CR and SD rates in the SACC and DACC groups were 23.1% (36 of 156) versus 7.3% (3 of 41) and 9.6% (15 of 156) versus 7.3% (3 of 41), respectively. 31 A total of 3 months after treatment, patients with detectable EBV DNA were 13.5% (21 of 156) versus 12.2% (5 of 41). 31 Previous studies have shown that both SD and EBV DNA load can indicate a poor prognosis after treatment.22,23 In addition, it is worth noting that in the landmark trial, a total of 480 patients were enrolled, of which 11.2% (27 of 242) and 10.1% (24 of 238) N3 stages were in the induction and standard treatment groups, respectively. 31 In our study, the proportion of N3 patients in the SACC group and the DACC group was 30.1% (47 of 156) and 46.3% (19 of 41), respectively. Therefore, the majority of patients included in this study had poor prognostic features of N2 or N3 disease or bulk primary tumors (T3 or T4), all of which were surrogate indicators of latent metastasis. In terms of long-term survival, PFS, OS, DMFS, and LRFS in the IC arm in the landmark trial were comparable or a little superior to those in our study: 3-year PFS (85.3% in the landmark trial versus 86.6–87.8% in this study), 3-year OS (94.6% in the landmark trial versus 95.2–97.6% in this study), 3-year DMFS (91.1% in the landmark study versus 87.8–89.6% in this study), and 3-year LRFS (91.8% in the landmark study versus 90.6–92.3% in this study). 31 Given all this, clinical outcomes in high-risk LANPC patients were less than ideal even with aggressive treatment, including chemotherapy followed by CCRT. Finally, although the toxicities of concurrent chemotherapy were tolerated, severe acute toxicities associated with DACC may affect patient outcomes. Severe neutropenia has been reported as a negative independent prognostic factor for PFS in head and neck squamous cell carcinoma, 27 although it has not been reported in NPC. The incidence of grade 3 and grade 4 leukopenia and neutropenia was significantly higher in the DACC group (41.5% and 31.7%, respectively), consistent with previous reports.7,32
In the future, considering that the incidence of disease recurrence is the highest within 2 years after chemoradiotherapy, strategies for providing additional benefits for high-risk patients with large tumor volumes after IC need to be explored. There is growing evidence that cetuximab or nimotuzumab administered delivered during IC or radiotherapy is effective for long-term survival.33–35 Analysis of preliminary clinical results from two phase II trials showed that radiotherapy combined with weekly nimotuzumab produced excellent survival benefits and tolerable toxicities in LANPC.36,37 The results of our multivariate analysis are consistent with the reported studies, suggesting that chemotherapy combined with targeted therapy can improve survival benefits for PFS, LRFS, and DMFS. In addition, preclinical studies have shown that radiotherapy combined with immunotherapy has a synergistic for inhibiting tumor growth synergistically. In a small, single-arm, phase II trial, reported at the ASCO annual meeting, neoadjuvant chemotherapy plus toripalimab followed by CCRT in LANPC achieved an ORR of 88.9%, and the CR rate was 41.3%. The phase III trial is ongoing (NCT05211232).
Adjuvant therapy is also a robust treatment option available for high-risk populations; however, its use is limited by its poor tolerance, with 40−50% of patients unable to complete the standard cisplatin-based adjuvant regimens.38–40 A multicenter phase III randomized controlled trial reported that the addition of metronomic adjuvant capecitabine was associated with better failure-free survival (FFS) than observation alone, with the 3-year FFS being significantly higher in the metronomic capecitabine group (85.3% versus 75.7%). 41 Accordingly, the treatment model for high-risk recurrent or metastatic patients with LANPC is updated in the latest CSCO guidelines based on these findings. 42 Collectively, the addition of adjuvant therapy rather than the intensification of concurrent chemotherapy may be a more suitable strategy for high-risk patients with LANPC.
Several limitations should be considered. First, this was a retrospective study, and thus, inherent biases could not be avoided. Although we tried to minimize selection bias by using PSM, treatment-related factors were well-balanced between the groups after matching, while some confounding factors could not be adequately matched. Due to the removal of some mismatched data, the data involved in the analysis may no longer be representative of the population as a whole and should be interpreted with caution. Second, the included patients underwent different regimens and cycles of IC. A previous study showed that survival rates did not significantly differ according to the regimens between different regimens (e.g., gemcitabine and cisplatin versus docetaxel and cisplatin versus docetaxel, cisplatin, and fluorouracil) and cycles (e.g., 2 versus 3) of IC. The optimal number of IC cycles is still controversial, with one study showing that two or three cycles have a similar effect on survival rate, while four cycles of IC not only increase toxicities but also do not improve survival. 43 Further randomized trials are needed to establish a more effective chemo-radiotherapeutic strategy to improve the prognosis of high-risk LANPC patients after IC.
Conclusion
From this study, we do not have sufficient evidence due to the small sample size that concurrent taxane and cisplatin chemotherapy provides additional survival benefits in LANPC patients with an unfavorable response (detectable EBV DNA levels or SD) after IC. It appears that increasing the intensity of concurrent chemotherapy is not an appropriate option for patients with an unsatisfactory response to IC. Further studies are needed to identify more effective treatment modalities for high-risk LANPC patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231177016 – Supplemental material for Concurrent chemotherapy using taxane plus cisplatin versus cisplatin alone in high-risk nasopharyngeal carcinoma patients with suboptimal response to induction chemotherapy
Supplemental material, sj-docx-1-tam-10.1177_17588359231177016 for Concurrent chemotherapy using taxane plus cisplatin versus cisplatin alone in high-risk nasopharyngeal carcinoma patients with suboptimal response to induction chemotherapy by Ya-Ni Zhang, Yu-Pei Chen, Ji-Bin Li, Tai-Xiang Lu, Fei Han and Chun-Yan Chen in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
