Abstract
Background:
Induction chemotherapy (IC) regimens such as gemcitabine plus cisplatin (GP), docetaxel plus cisplatin plus fluorouracil (TPF), and paclitaxel plus cisplatin (TP) are optional clinical options for locoregionally advanced nasopharyngeal carcinoma (LA-NPC).
Objectives:
This study aims to evaluate the efficacy and toxicity profiles of the GP, TPF, and TP induction regimens in LA-NPC.
Design:
This was a retrospective study.
Methods:
This multicenter retrospective study enrolled 722 patients with stage III–IVA LA-NPC who received GP, TPF, or TP IC. Propensity score matching (PSM) was performed before comparing survival outcomes and acute grades 3–4 toxicities among the three groups. Survival outcomes included the overall survival (OS), failure-free survival (FFS), local recurrence-free survival (LRFS), and distant metastasis-free survival (DMFS).
Results:
The original cohort comprised 722 patients (247 in the GP group, 240 in the TPF group, and 235 in the TP group). After PSM analysis, the GP group showed a better 3-year OS rate than the TP group (p = 0.019), while the 3-year OS rate revealed no difference between the GP group and TPF group and between the TPF group and TP group. There were no significant differences in the 3-year FFS, 3-year LRFS, and 3-year DMFS rates between and within the three groups. During the induction period, the toxicity of the three regimens was generally acceptable and manageable.
Conclusion:
The GP induction regimen demonstrated superior efficacy in terms of OS, with its favorable safety profile. Compared with the TPF and TP regimens, the GP induction regimen represents a more clinically advantageous treatment option for LA-NPC.
Plain language summary
This study evaluated the efficacy and toxicity profiles of gemcitabine plus cisplatin (GP), docetaxel plus cisplatin plus fluorouracil (TPF), and paclitaxel plus cisplatin (TP) induction regimens in locoregionally advanced nasopharyngeal carcinoma to provide evidence for the selection of induction chemotherapy regimens.
Induction chemotherapy regimens such as GP, TPF, and TP are optional clinical options for locoregionally advanced nasopharyngeal carcinoma. This study compared the efficacy and toxicity profiles of the GP, TPF, and TP induction regimens in locoregionally advanced nasopharyngeal carcinoma.
The researchers conducted a multicenter retrospective study to evaluate the efficacy and toxicity profiles of three induction chemotherapy regimens—GP, TPF, and TP—in patients with locoregionally advanced nasopharyngeal carcinoma.
The original cohort comprised 722 patients (247 in the GP group, 240 in the TPF group, and 235 in the TP group). After PSM analysis, the GP group showed a better 3-year overall survival rate than the TP group, while the 3-year overall survival rate revealed no difference between the GP group and TPF group and between the TPF group and TP group. There were no significant differences in the 3-year failure-free survival, 3-year local recurrence-free survival, and 3-year distant metastasis-free survival rates between and within the three groups. During the induction period, the toxicity of the three regimens was generally acceptable and manageable.
This study has provided evidence supporting the use of GP induction chemotherapy for locoregionally advanced nasopharyngeal carcinoma. The GP induction chemotherapy regimen offers both efficacy and a manageable toxicity profile compared to the TPF and TP regimens.
Introduction
Nasopharyngeal carcinoma (NPC) is a highly prevalent malignancy arising from the epithelial lining of the nasopharynx, with a particularly high incidence in East and Southeast Asia. 1 Due to its marked radiosensitivity, radiotherapy remains the standard of care for early-stage disease. In cases of locoregionally advanced NPC (LA-NPC), combined chemoradiotherapy has become the cornerstone of treatment, significantly enhancing locoregional control and overall survival (OS).1,2
Recent large-scale, multicenter randomized trials have demonstrated that the addition of induction chemotherapy (IC) prior to concurrent chemoradiotherapy (CCRT) can effectively eradicate micrometastatic disease and further improve 5-year survival outcomes in patients with locoregionally advanced disease.3–9 Various induction regimens are currently available. Among them, gemcitabine plus cisplatin (GP) and docetaxel plus cisplatin plus fluorouracil (TPF) have shown promising efficacy in large, randomized clinical trials.3–7 Taxane (including docetaxel and paclitaxel) plus cisplatin (TP) has gained attention due to its favorable tolerability and survival benefits.8,10,11
Different IC regimens exhibit distinct efficacy profiles and toxicity patterns. Although TPF is effective, it is associated with considerable toxicities such as myelosuppression and hepatorenal dysfunction, which can increase treatment burden and reduce compliance. 12 TP, which omits fluorouracil, may reduce toxicity while maintaining similar survival benefits. 12 However, both TPF and TP require pretreatment, complicating clinical delivery. By contrast, GP has gained popularity owing to its favorable efficacy–toxicity balance and broader therapeutic window, despite challenges such as drug resistance, patient variability, and the need for intensive monitoring. 13
To date, many studies have been conducted to compare the efficacy and toxicity of different induction regimens. Lian et al. evaluated the treatment response and survival of patients receiving the GP, TPF, and TP regimens. The authors noted that this study had limitations, including a small sample size and imbalanced proportions of patients receiving different IC regimens. 14 Therefore, we conducted a propensity score-matched analysis to evaluate the comparative efficacy and toxicity of the GP, TPF, and TP regimens, aiming to identify the most advantageous IC approach for patients with LA-NPC.
Materials and methods
Eligibility criteria
This multicenter retrospective study included 722 patients treated at the First Affiliated Hospital of Guilin Medical University, Wuzhou Red Cross Hospital, and Nanxishan Hospital of Guangxi Zhuang Autonomous Region in China between December 2014 and December 2019. Eligible patients met the following criteria: histologically confirmed NPC; clinical stage III–IVA as defined by the 8th Edition of the American Joint Committee on Cancer/Union for International Cancer Control TNM classification system 15 ; absence of distant metastasis; Karnofsky Performance Status score ⩾70; adequate hematologic, renal, and hepatic function; receipt of IC (GP, TPF, or TP regimen) followed by either CCRT or radiotherapy alone; treatment with intensity-modulated radiation therapy (IMRT); and availability of complete clinical records and follow-up data.
Exclusion criteria included metastatic disease at diagnosis, prior anticancer treatment, severe comorbidities, incomplete radiotherapy, or missing follow-up information.
Treatment
Treatment strategies followed the National Comprehensive Cancer Network guidelines for locoregionally advanced disease. 2 Most patients received IC followed by CCRT. In some cases, concurrent chemotherapy was omitted due to patient refusal, intolerance from prior toxicity, or financial limitations. The IC regimens included the GP regimen (gemcitabine and cisplatin), TPF regimen (docetaxel, cisplatin, and 5-fluorouracil), and TP regimen (paclitaxel and cisplatin). Concurrent chemotherapy consisted of cisplatin or nedaplatin.
All patients received IMRT targeting the primary tumor and cervical lymph nodes. The prescribed dose to the gross tumor volume (GTV) exceeded 66 Gy, and >50 Gy was delivered to bilateral cervical lymph nodes and potential sites of microscopic infiltration. The dose per fraction was 2.0–2.27 Gy. GTV comprised the primary nasopharyngeal tumor (GTVnx) and metastatic cervical lymph nodes (GTVnd). The clinical target volume (CTV) was composed of both high-risk (CTV1) and low-risk (CTV2) volumes. CTV1 was defined as the nasopharyngeal GTV plus a 5- to 10-mm margin. The distance of this margin was determined by the adjacent tissue’s characteristics, but it always included the entire nasopharyngeal cavity mucosa and 0.5 cm of submucosa. CTV2 was created by expanding CTV1 with an additional 5- to 10-mm margin. This posterior margin was reduced to 2–3 mm adjacent to the spinal cord or brainstem to protect these critical organs. In addition to CTV1, the CTV2 volume also encompassed the posterior third of the nasal cavity, the parapharyngeal space, maxillary sinuses, pterygoid process, inferior sphenoid sinus, skull base, the anterior half of the clivus, petrous apex, and the cervical lymph drainage area.
Follow-up and evaluation
Information regarding chemotherapy and radiotherapy regimens, start dates, and dosages was collected. Acute toxicities were evaluated and graded using the Radiation Therapy Oncology Group criteria 16 and the National Cancer Institute’s Common Terminology Criteria for Adverse Events, Version 4.0. 17 Survival data were collected through electronic medical records, telephone interviews with patients or family members, and follow-up assessments. The last follow-up was completed on February 25, 2023.
Endpoints
The primary endpoint was OS, defined as the interval from the initiation of IC to death from any cause or the last follow-up. Secondary endpoints included failure-free survival (FFS), local recurrence-free survival (LRFS), distant metastasis-free survival (DMFS), and acute toxicity. FFS was defined as the time from the start of IC to locoregional recurrence, distant metastasis, or cancer-related death, whichever occurred first. LRFS and DMFS were defined as the times to the first loco-regional recurrence or the first distant metastasis, respectively. If loco-regional recurrence and distant metastasis occur simultaneously, the patient is considered to have an event for both LRFS and DMFS.
Statistical analysis
Categorical variables were compared using the chi-square test or Fisher’s exact test. Propensity score matching (PSM) was performed using the pm3 package in R (The R Foundation, Vienna, Austria) to balance age, sex, T category, N category, and TNM stage across the three treatment groups in a 1:1:1 ratio. OS, FFS, LRFS, and DMFS were estimated using the Kaplan–Meier method, and differences were analyzed using the log-rank test. Multivariate analysis was performed using the Cox proportional hazards model to identify independent prognostic factors and to calculate hazard ratios (HR) with 95% confidence interval (CI). Statistical analyses were conducted using SPSS version 27.0 and R version 4.4.2 (R Foundation, Vienna, Austria). All statistical tests were two-sided, with p < 0.05 considered statistically significant.
The reporting of this study conforms to the STROBE statement (Supplemental Material 1). 18
Results
Patient characteristics
A total of 748 patients were screened, and 722 with eligible characteristics were enrolled in the study (Figure 1). Of these, 247 patients received the GP regimen, 240 received TPF, and 235 received TP. Baseline demographic and clinical characteristics before and after PSM are presented in Table 1. Before PSM, imbalances were observed in the T category and TNM stage between treatment groups. After PSM, 210 matched cases per group (630 total patients) were retained, with well-balanced baseline characteristics across the three groups. The matched cohort included 459 men (72.9%) and 171 women (27.1%), with a median age of 48 years (range, 18–75). The median follow-up duration was 62 months (interquartile range, 45–67 months) as of the last follow-up date on February 25, 2023.

Flow chart of study patient inclusion.
Baseline characteristics before and after PSM.
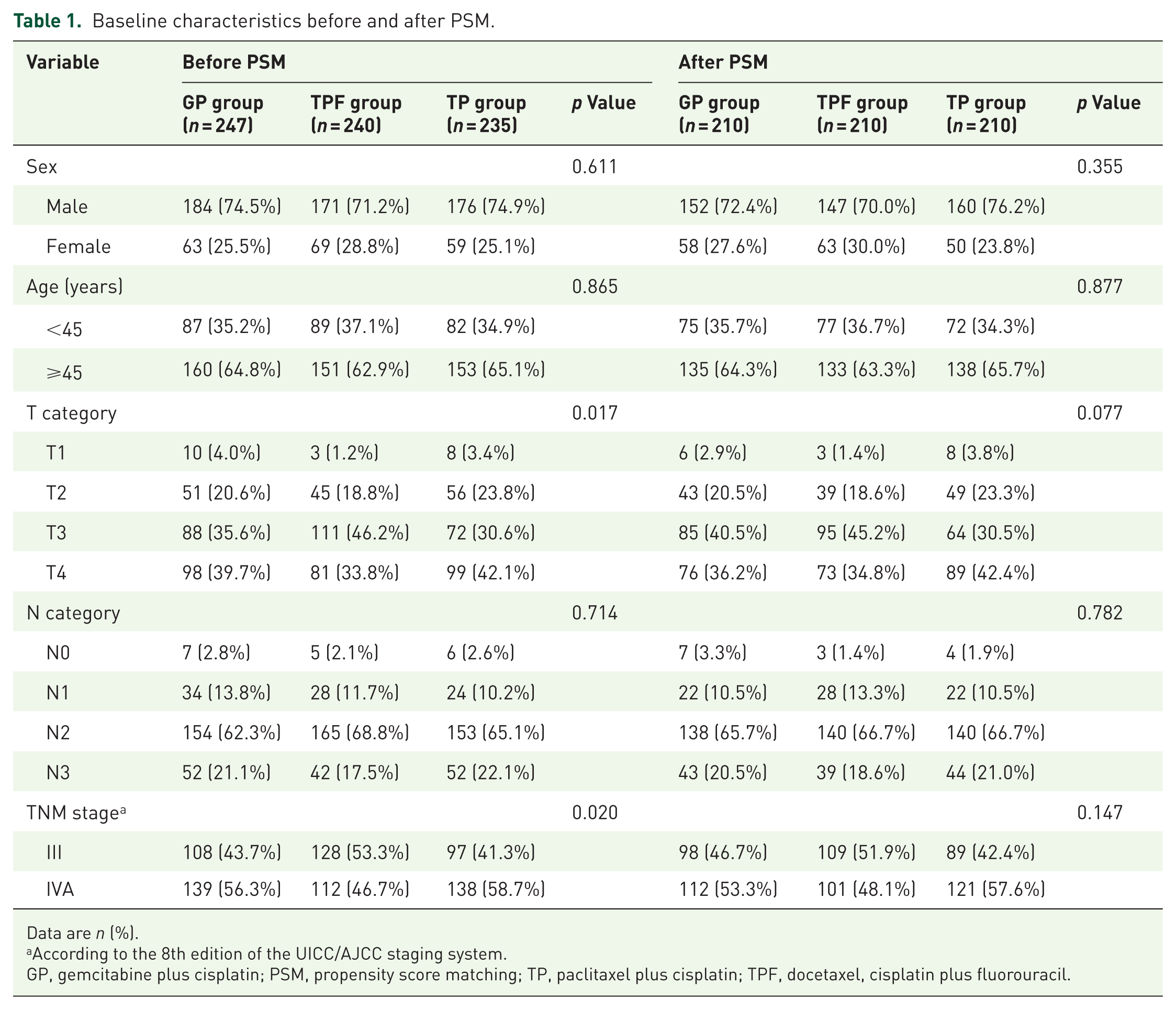
Data are n (%).
According to the 8th edition of the UICC/AJCC staging system.
GP, gemcitabine plus cisplatin; PSM, propensity score matching; TP, paclitaxel plus cisplatin; TPF, docetaxel, cisplatin plus fluorouracil.
All patients received IC. At least two cycles of IC were completed by 177 patients (84.3%) in the GP group, 206 (98.1%) in the TPF group, and 188 (89.5%) in the TP group. All patients completed radiotherapy. During the radiotherapy phase, 478 patients (75.9%) received concurrent chemotherapy, with 163 (77.6%) in the GP group, 164 (78.1%) in the TPF group, and 151 (71.9%) in the TP group. The remaining 152 patients (24.1%) received radiotherapy alone (22.4%, 21.9%, and 28.1%, respectively).
Survival and treatment failure events
In the matched population, the 3-year OS rates were 94.3% for the GP group, 90.5% for the TPF group, and 86.7% for the TP group(p = 0.076; Table 2, Figure 2(a)). Pairwise comparisons showed that the GP group resulted in significantly higher OS rates than the TP group (p = 0.019), but no significant differences were observed between the GP group and the TPF group (p = 0.236) or between the TPF group and the TP group (p = 0.299). The 3-year FFS rates were 80.5%, 77.6%, and 76.5% for the GP group, the TPF group, and the TP group, respectively (p = 0.483; Table 2, Figure 2(b)). The corresponding 3-year LRFS rates were 94.7%, 89.2%, and 90.9% (p = 0.439; Table 2, Figure 2(c)), and the DMFS rates were 84.7%, 83.7%, and 84.1% (p = 0.906; Table 2, Figure 2(d)). No significant differences were detected in any pairwise comparisons for FFS, LRFS, or DMFS. Overall, 73 locoregional recurrences, 119 distant metastases, and 95 deaths were recorded (Table 3).
Survival analyses after PSM.

DMFS, distant metastasis-free survival; FFS, failure-free survival; GP, gemcitabine plus cisplatin; LRFS, local recurrence-free survival; OS, overall survival; PSM, propensity score matching; TP, paclitaxel plus cisplatin; TPF, docetaxel, cisplatin plus fluorouracil.
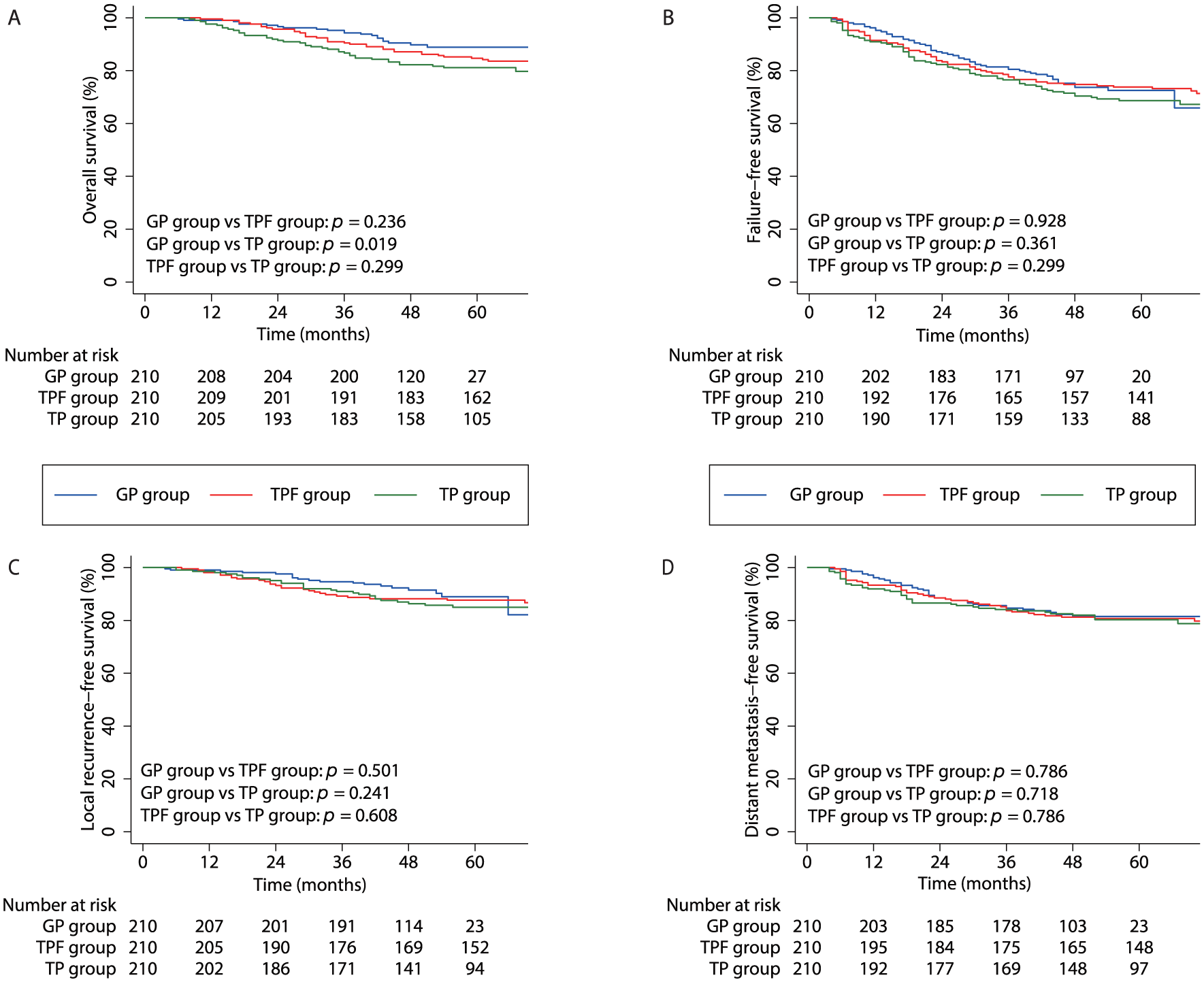
Kaplan–Meier survival curves after PSM: (a) OS, (b) FFS, (c) LRFS, and (d) DMFS.
Distribution of disease failure after PSM.

Data are n (%).
Three patients had distant lymph node metastasis, and one patient had parotid gland metastasis.
GP, gemcitabine plus cisplatin; PSM, propensity score matching; TP, paclitaxel plus cisplatin; TPF, docetaxel, cisplatin plus fluorouracil.
Multivariable analysis
Multivariate Cox regression analysis, adjusted for sex, age, T category, N category, TNM stage, and treatment group, identified the GP regimen as a significant independent predictor of OS compared with TP (HR, 0.573; 95% CI, 0.337–0.975; p = 0.040). GP regimen was not independently associated with FFS (HR, 0.869; 95% CI, 0.604–1.249; p = 0.448), LRFS (HR, 0.700; 95% CI, 0.390–1.256; p = 0.231), or DMFS (HR, 0.960; 95% CI, 0.614–1.501; p = 0.858). TPF regimen was not a significant predictor for any survival endpoint compared to TP: OS (HR, 0.814; 95% CI, 0.510–1.281; p = 0.381), FFS (HR, 0.879; 95% CI, 0.614–1.257; p = 0.479), LRFS (HR, 0.912; 95% CI, 0.531–1.566; p = 0.739), or DMFS (HR, 1.016; 95% CI, 0.656–1.572; p = 0.944). In addition, age ⩾45 years was an independent predictor of worse OS, FFS, and DMFS. TNM stage IV was independently associated with poorer outcomes across all endpoints (Table 4).
Multivariable analysis of prognostic factors after PSM.

p Values were calculated with an adjusted Cox proportional-hazards model. All HRs are adjusted for other covariates.
Male versus female.
≥45 years old versus <45 years old.
T3–4 versus T1–2.
N2–3 versus N0–1.
Stage IV versus stage III.
GP group versus TP group.
TPF group versus TP group.
CI, confidence interval; DMFS, distant metastasis-free survival; FFS, failure-free survival; GP, gemcitabine plus cisplatin; HR, hazard ratio; LRFS, local recurrence-free survival; OS, overall survival; PSM, propensity score matching; TP, paclitaxel plus cisplatin; TPF, docetaxel, cisplatin plus fluorouracil.
Toxic effects
Grade 3–4 toxicities during IC are summarized in Table 5. In the GP group, the most common severe toxicities were neutropenia (20.5%), leukopenia (6.2%), and anemia (3.8%). In the TPF group, the predominant toxicities were neutropenia (23.8%), leukopenia (11.0%), and vomiting (5.2%). In the TP group, neutropenia was the most frequent grade 3–4 event (6.7%). The incidence of grade 3–4 neutropenia and leukopenia in the GP group was intermediate between those in the TPF and TP groups (neutropenia: 20.5% vs 23.8% vs 6.7%; leukopenia: 6.2% vs 11.0% vs 0.5%). Grade 3–4 vomiting occurred in 1.4% of patients in both the GP and TP groups, compared to 5.2% in the TPF group. During radiotherapy, the GP group exhibited a significantly lower incidence of grade 3–4 stomatitis (10.5% vs 18.1% vs 10.5%; p = 0.028), leukopenia (13.3% vs 22.9% vs 10.0%; p = 0.001), and vomiting (1.9% vs 9.5% vs 3.8%; p = 0.001) compared with TPF or TP. Additional toxicity details are presented in Table 5.
Grade 3–4 acute toxicities according to treatment phase after PSM.
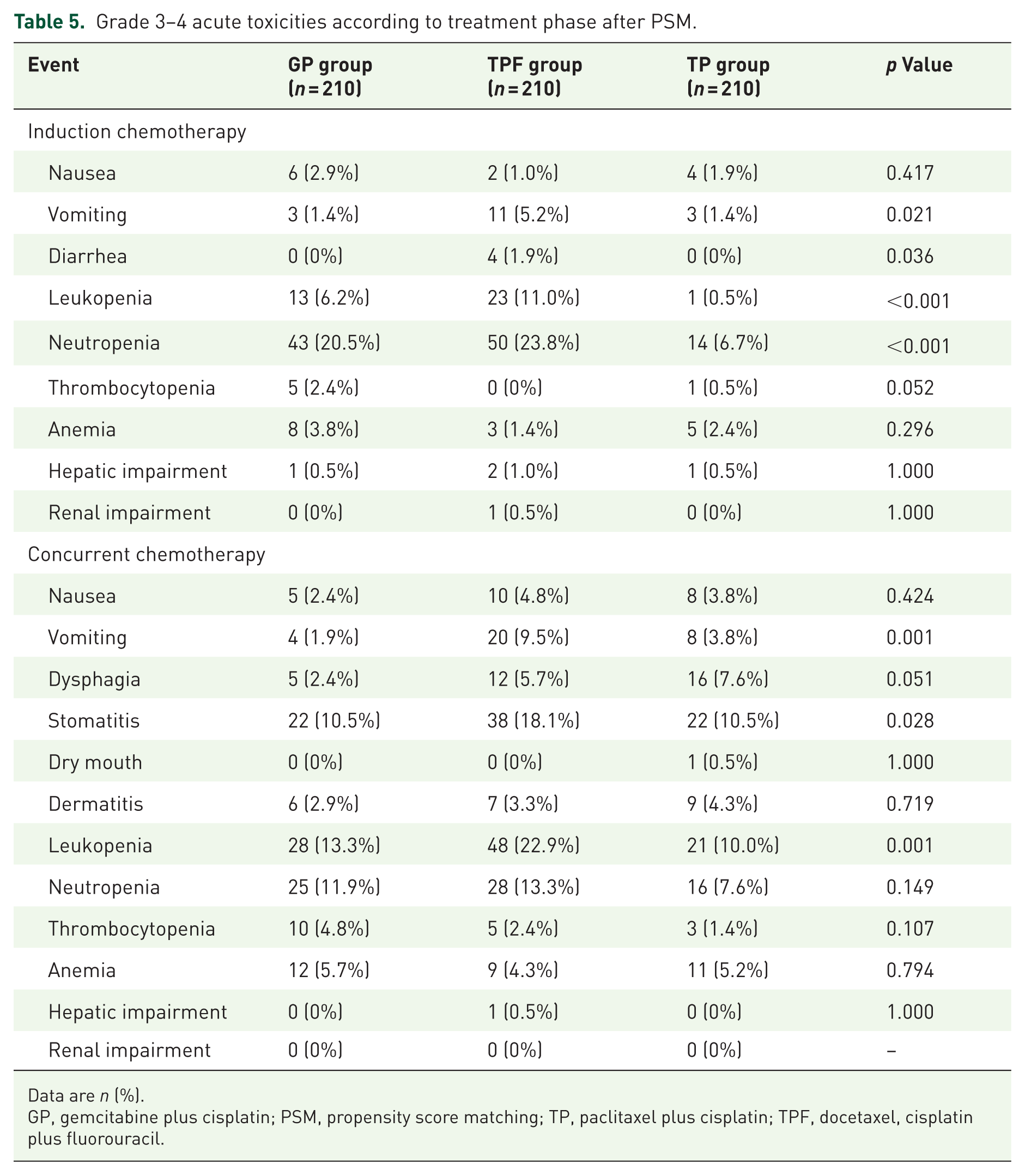
Data are n (%).
GP, gemcitabine plus cisplatin; PSM, propensity score matching; TP, paclitaxel plus cisplatin; TPF, docetaxel, cisplatin plus fluorouracil.
Discussion
This study compared the efficacy and toxicity profiles of the GP, TPF, and TP IC regimens for LA-NPC. Our findings indicate indicate that the GP group offers superior efficacy, with a significantly higher 3-year OS rate compared to the TP group and a comparable OS rate to the TPF group. No significant differences were observed among the three groups in terms of 3-year FFS, LRFS, and DMFS. Furthermore, the toxicity associated with the GP regimen was generally tolerable and manageable during the induction phase.
Despite advances in CCRT, local recurrence and distant metastasis continue to be the leading causes of treatment failure for LA-NPC. 19 Strategies such as incorporating IC or adjuvant chemotherapy have been explored to improve survival outcomes. IC followed by CCRT is now widely endorsed by clinical guidelines due to its favorable tolerability and efficacy in reducing distant metastases.1,10,11 Prior trials have demonstrated that adding GP, TPF, or TP IC to CCRT yields similar survival benefits.3–8 Evidence supports the superiority of the GP regimen over other regimens, so the GP regimen has been recommended as the category 1 preferred option for induction therapy. 2 One comparative study reported no significant differences in OS (p = 0.504) or PFS (p = 0.501) among GP, TPF, and TP regimens. 14 It is worth noting that this study included a total of 227 patients, and the distribution of patients across different IC regimens was notably uneven (37 (16.3%) in the GP group, 130 (57.3%) in the TP group, and 60 (26.4%) in the TPF group). 14 Thus, larger studies with better-balanced patient populations are needed.
Our multicenter study, which included a large sample size and used PSM to control for baseline differences, allows for a more reliable comparison of GP, TPF, and TP. Our findings in this paper confirm previously known studies from another point of view.12,13,20–23 These results support GP as an optimal IC regimen for LA-NPC, with the added benefit of simplifying treatment and avoiding unnecessary chemotherapeutic pretreatment.
The survival advantage of the GP regimen over the TP regimen may be attributed to superior tumor volume reduction post-IC. Greater tumor shrinkage translates to reduced residual disease burden, which could improve long-term outcomes.14,24 Post-IC tumor volume has been shown to hold prognostic significance in LA-NPC. 25 In one study, the GP regimen yielded a higher overall response rate than the TP regimen. 14 Another reported greater GTVnx reduction with the GP regimen compared to the TP regimen. 24 Our previous work also confirmed that tumor volume reduction rate after IC is an independent prognostic factor in patients undergoing IMRT. 26
Toxicities during the induction phase were consistent with prior studies and generally acceptable.3,7,8 Grade 3–4 neutropenia was the most frequent severe adverse event across all regimens. Among the three group, the TPF group was associated with the highest toxicity burden, while the GP group exhibited intermediate rates of neutropenia and leukopenia. Moreover, the incidence of severe vomiting and diarrhea was lower in the GP group compared to the TPF group. These results, coupled with its efficacy, further support the clinical viability of GP as a well-balanced IC option.
Nonetheless, this study has limitations. Although PSM was used to control for confounders, retrospective data collection may introduce bias. Furthermore, the absence of EBV DNA dynamics limited our ability to explore individualized risk stratification. While this study suggests that induction GP results in the best outcomes for patients with LA-NPC, both IC and adjuvant chemotherapy are applicable for different groups of NPC patients. The future points toward reinvestigating the role of adjuvant chemotherapy. This will result in better local control, similar or even better distant control, and OS benefit, less overtreatment, and less overall morbidity. This can even dovetail into the realm of precision medicine with the realization that the predictive approach has failed, and the future should be in adapting treatment to separately improve local and distant control, which can then together improve overall outcomes for patients with LA-NPC.
Conclusion
The preliminary results demonstrated that the GP group exhibited a significantly superior 3-year OS compared to the TP group, with a similar 3-year OS to the TPF group. Furthermore, in terms of other outcomes, the GP group was comparable to both the TPF and TP groups. Consequently, the GP regimen can be regarded as the most convenient and advantageous IC regimen for LA-NPC patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251413429 – Supplemental material for Comparing three induction regimens for nasopharyngeal carcinoma: a propensity score-matched analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251413429 for Comparing three induction regimens for nasopharyngeal carcinoma: a propensity score-matched analysis by Jinxuan Dai, Yu Pan, Yuanyuan Liu, Yufei Pan, Defeng Wang, Yi Liang, Yuejia Wei, Hengwei Chen, Bin Zhang, Xiaojuan Wu, Bing Liu, Xiangyun Kong, Yunyan Mo, Xiaolan Ruan, Shufang Liao, Xiaoping Lin, Chen Huang, Jinping Xu, Fei Mo and Wei Jiang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
