Abstract
Biliary tract cancers (BTCs) are rare and heterogeneous malignant tumours including cholangiocarcinoma and gallbladder cancer. They are very aggressive, often refractory to chemotherapy and associated with an overall poor prognosis. Surgical resection remains the only potentially curative treatment option but less than 35% present with resectable disease. Adjuvant treatments have been widely used but until recently, supportive data were limited to non-randomised, non-controlled retrospective studies. Recent evidence from the BILCAP trial has established adjuvant capecitabine as the standard of care. But there are still unanswered questions as to the role of adjuvant therapy. Further prospective data and translational research with reproducible evidence of clinical benefit are needed. In this review of adjuvant therapy in resectable BTCs, we will summarise the latest evidence setting current treatment standards and highlight future prospects.
Keywords
Background and current practice
Biliary tract cancers (BTCs) are rare and whilst being the second most common primary hepatic malignancy, they account for less than 1% of all cancers, 3% of all gastrointestinal cancers, and typically present in the seventh decade with a slight male predominance. 1 They represent a distinct and heterogeneous group of cancers arising from the bile duct epithelium and are subdivided into gallbladder cancer (GBC) and cholangiocarcinoma (CCA). CCAs are classified by anatomical origin as intrahepatic CCA (iCCA) and extrahepatic CCA (eCCA) – the latter further subdivided into perihilar CCA (pCCA) and distal CCA (dCCA) (Figure 1). 2

Anatomical classification of cholangiocarcinoma.
These subgroups demonstrate distinct epidemiological variations with a low incidence in Western countries (0.3–6 cases per 100,000 per year) but higher in specific regions of Southeast Asia (7.1–14.5 per 100,000 per year). 3 Overall incidence is rising specifically for iCCA, whereas rates are stable or falling for eCCA. Worldwide mortality rates have increased over the last 20 years with mortality higher in men and the Asian population. 4 These shifts are probably explained by differences in risk factor exposure and genetic predispositions. Identified risk factors differ relative to anatomical origin but are largely based on the principle of chronic inflammation and bile stasis such as chronic parasitic infection (in regions of high endemicity), primary sclerosing cholangitis and hepatolithiasis. Other potential risk associations include hepatitis and smoking with iCCA 5 ; and obesity, inflammatory bowel disease and diabetes with GBC. 6 But most patients do not have an identifiable risk factor nor do these factors offer a clear etiologic explanation for global trends and so largely, the underlying pathogenesis remains unknown. 7 At a molecular level, through comprehensive whole-exome and transcriptome sequencing, heterogeneity is again observed in the different molecular profiles of iCCA, eCCA and GBC. Genetic alterations in potential therapeutic targets occur in nearly 40%, particularly in iCCAs and most commonly as fibroblast growth factor receptor (FGFR) fusions and isocitrate dehydrogenase 1 mutations. 8 Correlation of molecular profiling with prognosis and/or potential benefits from adjuvant treatment are currently lacking, but translational analysis of the large randomised adjuvant trials is on the way.
The only curative management and the best chance for prolonged survival is radical surgical resection. Unfortunately, less than 35% of patients present with resectable disease at diagnosis, and postoperative recurrence and relapse rates are around 50–70%.9–11 Despite advances in diagnostics and therapeutic interventions, prognosis remains poor with an overall 5-year survival of less than 20%. 12 As most patients present with advanced or metastatic disease, treatment strategies currently used include systemic chemotherapy, targeted therapy or locoregional management. 1 First-line chemotherapy with gemcitabine and cisplatin (GemCis) was standardised based on the ABC-02 phase III clinical trial, with a median overall survival (OS) of 11.7 months and an objective response rate (ORR) of 26.1%. 13 More recently, the TOPAZ-1 trial has reported an improvement of all efficacy endpoints with the addition of durvalumab to GemCis and the immuno-oncology-based combination will most likely be the primary choice in the first-line setting once approved by European Medicines Agency (EMA; already approved by Food and Drug Administration). 14 Pemigatinib, the FGFR2 inhibitor, is the first approved molecularly targeted therapy and is recommended for patients harbouring FGFR2 fusions or rearrangements who have progressed after one or more lines of therapy. 15
For the favourable patients presenting with resectable disease, support and recommendations for adjuvant therapy followed the 2012 Horgan
Randomised trials since 2017
A literature search was performed using PUBMED, American Society of Clinical Oncology (ASCO) Gastrointestinal, European Society of Medical Oncology (ESMO) and ClinicalTrials.gov. The following search terms were used in combination: ‘biliary tract cancer’, ‘cholangiocarcinoma’, ‘gallbladder cancer’ AND ‘adjuvant’ from inception to the last search on 26th July 2022. Figure 2 reports the flow chart of trials included in this review. The previously mentioned meta-analysis included only one randomised phase III trial, 18 and so this search identified four completed phase III randomised trials for discussion since this time point.

Flow chart of literature search.
The BCAT open-label multicentre phase III trial was the first published trial to explore adjuvant chemotherapy in BTC with long-term follow-up over 5 years.
19
Set up in Japan, patients with resected eCCA were randomised to gemcitabine

Kaplan–Meier estimates of (a) OS and (b) RFS in gemcitabine and observation groups. (a)
Similar negative findings were reported in the PRODIGE 12-ACCORD 18 (UNICANCER GI) phase III trial comparing adjuvant chemotherapy to observation.
21
A total of 196 patients diagnosed with iCCA/eCCA/GBC were enrolled following R0 or R1 surgery and randomised 1:1 to 12 cycles of gemcitabine oxaliplatin (GEMOX) (

(a) RFS and (b) OS Kaplan–Meier curves.
Both the BCAT and PRODIGE trial findings contrast with the BILCAP trial published in 2019 which is so far the largest prospective phase III randomised adjuvant BTC study.
17
The BILCAP trial ran over 8 years and recruited from 44 centres. A total of 447 patients were randomised to eight cycles of adjuvant oral capecitabine

Long-term outcomes and exploratory analyses of the randomised phase III BILCAP Study. (a) Kaplan–Meier OS curves for ITT population, (b) OS for PP population, (c) RFS for ITT, and (d) RFS for PP.
These three randomised controlled trials differ in sample size calculation, statistical power of study design, maturity of data and follow-up. Whilst attempting comparison is complex, these differences, as outlined in Table 1, may explain the conflicting positive results of the capecitabine-based BILCAP trial and the negative results of gemcitabine-based BCAT and PRODIGE trials. First comparing study design, all three were similarly randomised open-label phase III trials. The PRODIGE-12 trial was ambitiously designed to detect an improvement in RFS with GEMOX at an HR of 0.60 and therefore a 40% reduction in risk. No statistical difference was found; however, as significantly underpowered, a role for GEMOX in this setting cannot be excluded. When comparing time to treatment, it is noted that although not consistently reported, for pre-2017 studies the time from surgery to adjuvant therapy was within 12 weeks, with one trial giving adjuvant chemotherapy at the time of resection. 18 The ‘modern’ trials started adjuvant therapy up to 12 weeks post-surgery and continued for up to 6 months. The BCAT trial enrolled patients within 10 weeks of surgery and the median interval between surgery and randomisation was 50 days. Gemcitabine was discontinued in 53 patients (45.3%) for reasons such as intolerance, patient refusal, disease relapse, physician decision and dosing delays. So, 61 patients (52.1%) completed protocol therapy. The PRODIGE trial similarly enrolled patients who had curative resections less than 3 months before randomisation and started adjuvant treatment within 7 days of random assignment. The included patients received a median of 12 cycles of gemcitabine and 10 cycles of oxaliplatin. In the BILCAP trial, the original start to chemotherapy was 8 weeks but this extended to 16 weeks following multiple protocol amendments, and 55% of included patients completed the protocolled eight cycles of capecitabine. Next comparing study patient selection, the BCAT trial patients have exclusively eCCA tumours, whereas the PRODIGE trial was predominantly iCCA (44%). As well as differences in the BTC subtypes, there was heterogeneity in the populations enrolled in these three adjuvant trials based on nodal involvement and resection margins. Both the BCAT and PRODIGE trials had similar-sized node-positive populations of 35% and 36%, respectively, but this was higher at 47% in the BILCAP study. The genomic profile varies based on tumour location and so therefore could explain differences in chemotherapy response rates and prognosis. Also, R0 clear resection margin was again similar in BCAT and PRODIGE at 89% and 86%, respectively, but lower in the BILCAP trial at 62%. Notably, the BILCAP trial included the largest number of patients, and so statistical power, as well as a high proportion with poor prognostic factors which partly explains why it was the only positive study. Primary endpoints differed between the trials, with PRODIGE-12 powered for RFS. Although a treatment effect on RFS would be expected to show an effect on OS, it is not an optimal surrogate and ideally, studies should be adequately powered to detect significance in both to appropriately determine clinically meaningful benefits.
Adjuvant therapy randomised trials in resectable BTC: published trials since 2017.

BTC, biliary tract cancer; CCA, cholangiocarcinoma; dCCA, distal CCA; DFS, disease-free survival; GEMOX, gemcitabine oxaliplatin; ECOG PS, Eastern Cooperative Oncology Group performance status; iCCA, intrahepatic CCA; pCCA, perihilar CCA; OS, overall survival; QoL, quality of life; RFS, relapse-free survival.
The OS in the control arms was clearly different between the three studies with BCAT at 63.8 months, PRODIGE 50.8 months and BILCAP 36.0 months. Although there were also differences in the control arms’ survival rates, it is not clear whether these differences affected trial outcomes and statistical power. Of the modern randomised phase III trials, only the BILCAP trial indicated a survival benefit. It did not meet its primary endpoint and disease recurrence was high at 65% in the observation cohort and 60% in the treatment cohort. Despite this, it changed clinical practice and current ASCO and recently updated ESMO guidelines recommend adjuvant capecitabine as the standard of care for resectable BTC22,23.
Included in Table 1 for comparison are the more recently completed phase III ASCOT and phase II STAMP trials. A favourable outcome with adjuvant therapy has been described in the unpublished Japanese JCOG1202 (ASCOT) [UMIN00001166888] open-label multicentre randomised phase III trial comparing adjuvant S-1 (oral fluoropyrimidine derivative only used in Japan) with observation alone in resected CCA, GBC and ampullary cancer.
24
Presented at ASCO Gastrointestinal Symposium 2022, 440 patients were enrolled and randomised to observation (
In contrast, the relatively small STAMP multicentred randomised phase II trial [NCT03079427] comparing adjuvant GemCis with capecitabine for resected lymph node-positive pCCA and dCCA was statistically negative. 25 A total of 101 patients were included and randomised within 12 weeks of resection. The 2-year disease-free survival was 25.1% in the capecitabine cohort (17.4–33.5) compared to 38.5% in the GemCis cohort (29.5–47.4), HR: 0.96 (90% CI: 0.71–1.30) after a median follow-up of 33 months. There was no improvement in secondary endpoints including OS. The clinical implications of this small trial in a high-risk eCCA cohort are not yet clear, but it is the first randomised trial to include GemCis.
In the ongoing work to determine the value of adjuvant therapy, further insights are also awaited from the largest adjuvant phase III trial ACTICCA-1 [NCT02170090] which is currently enrolling patients with CCA and GBC in seven countries (Australia, Austria, Denmark, Germany, Italy, the Netherlands and the United Kingdom).
26
The trial initially randomised biliary tract patients to observation with or without GemCis and was paused after the initial presentation of the BILCAP results with 187 patients randomised. After protocol adjustment, the trial was continued and randomised a further 594 patients to GemCis
Role of neoadjuvant treatment
Although not a standard approach in resectable BTCs, early systemic therapy as neoadjuvant treatment is already standard of care in other cancers such as oesophageal, gastric and rectal. The rationale is to control systemic spread as early as possible, optimise surgery by downstaging the tumour and increasing the likelihood of resectable disease, reducing recurrence rates, and thus improving OS.
27
Evidence so far supporting neoadjuvant treatment for BTCs comes from retrospective analyses indicating a higher likelihood of attaining an R0 resection in those who receive neoadjuvant chemotherapy, but the evidence is conflicting and insufficient.
28
There are no completed phase III randomised controlled trials assessing the survival benefit of neoadjuvant treatment in BTC. Several single-arm or randomised trials are currently recruiting with neoadjuvant chemo- and /or immunotherapy or transarterial approaches in iCCA (Table 2). The phase II single-arm NEO-GAP trial administered preoperative combination gemcitabine, cisplatin and nab-paclitaxel in 30 patients with resectable high-risk iCCA.
29
The primary outcome of feasibility was met with 77% of patients completing all pre-operative chemotherapy and surgical resection. The partial response rate was 23%, the disease control rate was 90% and there is ongoing follow-up for RFS and OS. Other trials underway include the randomised phase II/III trial ECOG-ARCIN (EA2197) evaluating the role of neoadjuvant GemCis before re-resection of incidental GBC compared to adjuvant only GemCis after re-resection [NCT04559139]; the phase II DEBATE trial of preoperative GemCis with or without durvalumab [NCT04308174]; phase III GAIN trial on perioperative GemCis
Ongoing randomised phase II/III clinical trials including adjuvant and neoadjuvant therapy for resectable BTC.
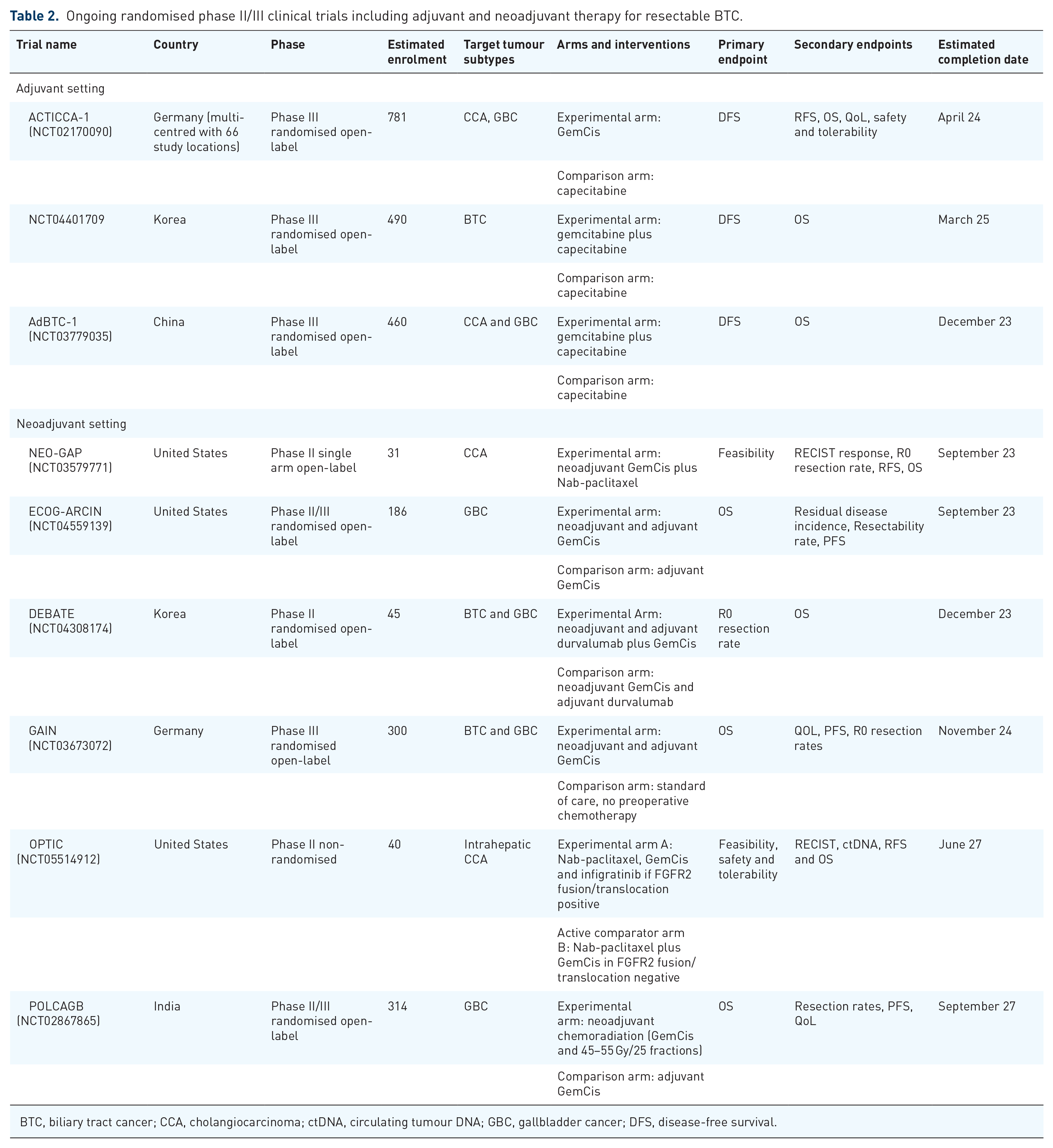
BTC, biliary tract cancer; CCA, cholangiocarcinoma; ctDNA, circulating tumour DNA; GBC, gallbladder cancer; DFS, disease-free survival.
Role of immunotherapy
Immunogenic features of BTC include programmed cell death ligand 1 (PD-L1) upregulation, mismatch repair deficiency (dMMR/MSI-H) and HLA class I antigen downregulation.
30
dMMR is associated with sensitivity to immune checkpoint inhibition due to the accumulation of somatic mutations.
31
In BTC, dMMR is reported in 5–10% of tumours, being more common in iCCA than eCCA or GBC. This implies a potential role for checkpoint inhibition, but data are limited, and efficacy has only really so far been assessed in advanced/metastatic settings. Recently, the EMA approved the programmed cell death protein 1 (PD-1) inhibitor pembrolizumab in pre-treated dMMR/MSI-H BTC based on the results of the phase II KeyNote 158 basket trial.
32
This is one of the largest published immunotherapy trials assessing the efficacy of pembrolizumab in solid tumours. Subgroup analysis of BTC MSI-H subgroup found an ORR of 40.9%, median PFS 4.2 months and mOS 24.3 months. In considering the role of immunotherapy in systemic therapy, the TOPAZ-1 phase III trial was presented at the ASCO Gastrointestinal symposium 2022, showing a significant albeit modest improvement in survival with anti-PD-1 durvalumab as first-line treatment in combination with GemCis.
33
Now published, 685 patients were randomised 1:1 double-blind to GemCis plus durvalumab or placebo.
14
Durvalumab treatment was well tolerated, and median OS was 12.8 months compared to 11.6 months with GemCis alone (HR: 0.80, 95% CI: 0.66–0.97,
Role of radiotherapy
No prospective randomised trials have compared adjuvant sequential radiotherapy and chemotherapy to chemotherapy alone. The feasibility of such a regimen was established in a single-arm trial; however, due to the lack of comparative data, this has not become a standard of care.
SWOG S0809, a single-arm phase II multi-centre study, recruited 79 patients with eCCA and GBC post-resection who were deemed high risk due to advanced T stage, nodal status and/or involved resection margins. 34 Patients received four cycles of gemcitabine–capecitabine chemotherapy followed by chemoradiotherapy 45 Gy/25 fractions to regional lymph node bed and 54–59 Gy to the preoperative tumour bed. Three-dimensional planning and intensity-modulated radiotherapy was included and found to be effective, tolerable and safe. The predetermined threshold of activity was exceeded by the 65% 2-year OS (67% for R0 and 60% for R1) and median OS of 35 months. This trial established a protocol of sequential chemotherapy and chemoradiation and showed good survival rates, particularly in R1 resected patients. However, without a control group, it is of limited application as the efficacy of radiotherapy remains unclear. Its relevance in the current and evolving perioperative treatment landscape remains open. Future randomised prospective studies are needed to evaluate the role of radiotherapy in the perioperative setting.
Role of locoregional treatment
Locoregional treatment strategies for selected patients with locally advanced or liver-predominant disease are being increasingly explored including transarterial embolisation, hepatic artery infusion, transarterial chemoembolisation and radiofrequency ablation (RFA). Edeline
Patient stratification and biomarkers for adjuvant chemotherapy
The currently accepted classification of BTC is anatomical but does not offer predictions of the natural history of the disease or its underlying pathogenesis, therefore nor does it offer insight into treatment response. Treatment decisions so far are based on clinical and pathological features such as tumour location and subtype, staging and the patient’s performance status and comorbidities. There is a well-established emphasis on pathology in predicting likely outcomes by considering the recognised negative prognostic indicators of tumour size, positive resection margins and nodal involvement.
37
Lymph node involvement is independently associated with a high risk of early relapse and adequate lymphadenectomy should be standard in patients undergoing liver resection due to the survival benefit, even in clinically node negative iCCA.
38
Recently, Lamarca
Serum tumour markers such as carcinoembryonic antigen and carbohydrate antigen (CA) 19-9 have been used for diagnosis, prognostic value and disease monitoring. 41 Although a non-specific diagnostic marker, CA 19-9 is the most studied and widely used in predicting resectability. 42 CA 19-9 levels are indicative of unresectable disease with a sensitivity of 76.3% and a specificity of 70.8%. 43 It can also act as a surrogate marker for treatment efficacy as persistently elevated CA 19-9 levels post-curative resection are a predictor of worse survival. 44 During gemcitabine chemotherapy, a reduction in CA19-9 equal to or more than 50% correlated with improved therapy response and increased survival but its sensitivity and specificity is 72% and 84%, respectively. 45 Changes in serum CA 19-9 could therefore be used to identify high-risk patients and therefore those who are more likely to benefit from adjuvant therapy. 44 Rather than absolute levels, an observed change may be more applicable but approximately 10% of the general population are Lewis blood group antigen negative and unable to produce CA 19-9. 46 Another potentially useful serum tumour marker, cytokeratin-19 fragment (CYFRA 21-1), is significantly upregulated in BTC and is negatively correlated with postoperative outcomes in iCCA and GBC. 47 Concentrations of CYFRA 21-1 vary according to tumour size and vascular invasion; and as an independent predictor of RFS and OS, it is therefore a potentially suitable biomarker. The prognostic value of other serum biomarkers for example, preoperative inflammatory indices such as cytokines, circulating nucleic acids including microRNA and other molecular factors are being investigated but so far in small subsets and as of yet, none with clinical applicability. 48
In view of the standardisation of adjuvant capecitabine, few available publications so far have explored potential predictors of capecitabine response. Thymidine phosphorylase (TP) is the rate-limiting enzyme in the activation of 5′-deoxy-5-fluorouridine and capecitabine. Retrospective studies have analysed its potential as a predictor of response. Higher TP expression, although associated with a worse prognosis, is seen predominantly in GBC and may be linked to their higher response rates seen in clinical trials. 49
Tumour tissue biomarkers in resectable disease can have both prognostic and predictive implications. 45 Pre-clinical and early-phase research have identified molecular alterations with potential prognostic implications and therapeutic options including B-raf proto-oncogene serine/threonine kinase (BRAF) V600E, neurotrophic tyrosine receptor kinase fusions, dMMR, tumour mutational burden (TMB), human epidermal growth factor-2 (HER2/ERBB2) amplifications and mutations, BRCA1/2 and DNA damage repair mutations, PIK3C, Akt, RSPO fusions, RNF43 mutations and RET fusions. 50 Overexpression of p53, KRAS mutations, amplifications of HER2, and overexpression of EGFR are associated with recurrence and poor survival. 48 But none can predict response to chemotherapy.
Predictors of response to immunotherapy in specific tumour types includes PD-L1 status, TMB and MSI. Although not yet used in the adjuvant setting, for BTCs the only current relevant marker is dMMR/MSI-H. But the role of immunotherapy is yet to be defined and available data on biomarker predictors are conflicting and limited. 30
Overall, no predictive or prognostic biomarkers are currently validated for use in clinical practice with exploration so far limited to a few studies in small cohorts with inconclusive results. CA19-9 remains the most used serum biomarker. Large cohort prospective biomarker-embedded studies in early-stage disease are needed to establish clinically applicable predictors of treatment response, gain understanding of primary and acquired resistance and identify suitable future lines of therapy including immunotherapy and targeted. As an adjunct to clinical trials, pre-clinical models could also further clarify the underlying molecular pathogenesis, discovery of biomarkers as well as testing therapeutic efficacy. 51 In clinical practice, this is all crucial for stratifying decision-making regarding likelihood of surgical response, timing and duration of adjuvant treatment, follow-up schedules and identifying likely poor responders who may benefit from neoadjuvant therapy. Evidence-supported patient selection is important in improving survival and directing treatment strategies to those most likely to benefit. Results of translational studies from clinical trials, including the use of circulating tumour DNA (ctDNA) sequencing for relapse mechanisms are awaited, but an affordable, sensitive and specific biomarker for BTC is yet to be found.
Efforts to improve patient selection face the well-known constraint of a lack of sufficient tissue. Emerging use of liquid biopsies overcomes this challenge and offers the potential to guide treatment selection for example in detecting post-resection residual disease or early relapse; tracking therapeutic response and emerging acquired resistance mechanisms. 52 Liquid biopsies are a method of detecting and analysing tumour-derived material such as circulating tumour cells (CTCs), ctDNA, circulating exosomes and microRNAs from biofluids. The tumour genome is isolated from accessible sources of serum, urine, bile or saliva, and then sequencing can be done from a range of whole-genome sequencing to targeted or mutation-specific analyses. The ABC-03 trial was the first to demonstrate CTCs and to correlate this with a poor prognosis in patients with BTC. 53 Subsequent genotyping studies have also found ctDNA shares over 70% concordance with tumour tissue and is an independent predictor of survival.54,55 The clinical validation and relevance of liquid biopsies for more accessible profiling are still being defined but promising evidence so far advocates already for integration alongside standard tumour biopsies.
Summary of recommendations and conclusion
The scope of treatment for BTCs has made slow progress over the last 10 years. Surgical resection is only a beneficial option for the small proportion of those with resectable BTC and the approved standard of care remains 6 months of adjuvant capecitabine after the positive RFS and OS results of the BILCAP trial, despite its limitations. Adjuvant therapy offers the best opportunity to impact patient outcomes with systemic therapy; however, several issues and inconsistent trial outcomes remain, most likely due to a lack of consideration for the heterogeneity of BTC seen in its epidemiology, aetiology, molecular characteristics, surgical intervention and prognosis. This complexity calls for personalised surveillance, treatment and de-escalation strategies matched to tumour subtype, molecular alterations and high-risk cohorts. To get there, better patient selection and further studies of the role of combination therapy, immunotherapy, targeted treatment and radiotherapy in neoadjuvant, adjuvant and oligoprogression settings are necessary. Neoadjuvant strategies could address the issue of early recurrence. The application of immunotherapy from basket cohort trials has the potential to bring durable clinical responses into the adjuvant setting. Although consideration of chemoradiotherapy is reasonable when addressing those at high risk of recurrence, such as R1 disease, there is so far low-level evidence supporting this. Optimal large prospective randomised trials need to be rationally designed for biologically selected patients and adequately powered with standardised risk factors, relevant pre-specified analyses and sufficient follow-up to provide evidence of clinical efficacy. With patients stratified, it is possible to have a ‘low-risk’ observation arm and randomised ‘high-risk’ comparative active treatment arms. They also need to be open to potentially significant amendments as new data become available.
Molecular profiling, understanding its relevance and identifying suitable robust biomarkers are integral to precision oncology and should be offered to all patients. And whilst being validated in an advanced setting, the incorporation of molecular profiling will be informative to subgroup analyses, predictive biomarkers, prognostic variability and drive a move into targeted therapeutic options. For this, accessible liquid biopsies and fast and affordable sequencing technologies should be adopted. BTC is a rare and challenging cancer, so encouraging trial participation, multicentre cross-study collaboration and dedicated expert centres are essential for the evaluation and development of meaningful evidence-based recommendations that go beyond OS. Internationally collaborative databases of the unprecedented wealth of information from sequencing such as NGS are needed to determine clinical significance and actionability. A dynamic synchronicity is needed between broadening our understanding of the natural history and genomic spectra of BTC alongside translational research and new drug development.
Footnotes
Acknowledgements
Not applicable.
