Abstract
Background:
Biliary tract cancers (BTCs) are heterogenous, highly aggressive tumors that harbor a dismal prognosis for which more effective treatments are needed. The role of cancer immunotherapy in BTC remains to be characterized. The tumor microenvironment (TME) of BTC is highly immunosuppressed and combination treatments are needed to promote effective anticancer immunity. Vascular endothelial growth factor (VEGF) drives immunosuppression in the TME by disrupting antigen presentation, limiting T-cell infiltration, or potentiating immune-suppressive cells. Many VEGF-regulated mechanisms are thought to be relevant to repressed antitumor immunity in BTC, making dual targeting of VEGF and programmed cell death protein 1 (PD-1)/PD-L1 pathways a rational approach. Gemcitabine and Cisplatin (Gem/Cis) can also modulate anticancer immunity through overlapping and complementary mechanisms to those regulated by VEGF. Anti-PD-L1/VEGF inhibition, coupled with chemotherapy, may potentiate antitumor immunity leading to enhanced clinical benefit.
Methods:
IMbrave 151 is a randomized, double-blind, placebo-controlled, multicenter, international phase II study to evaluate atezolizumab (a PD-L1 inhibitor) in combination with chemotherapy (gemcitabine and cisplatin) and bevacizumab (an anti-VEGF monoclonal antibody) as a first-line treatment for advanced BTC. Approximately 150 patients with previously untreated, advanced BTC will be randomized to either Arm A (atezolizumab + bevacizumab + Gem/Cis) or Arm B (atezolizumab + placebo + Gem/Cis). Randomization is stratified by the presence of metastatic disease, primary tumor location, and geographic region. The primary efficacy endpoint is investigator-assessed progression-free survival (PFS) per RECIST 1.1. Secondary endpoints include objective response rate (ORR), duration of response (DoR), disease control rate (DCR), overall survival (OS), and safety and patient reported outcomes (PROs). Tissue, blood, and stool samples will be collected at baseline and on-treatment in order to perform correlative biomarker analyses.
Discussion:
IMbrave 151 represents the first randomized study to evaluate combined PD-L1/VEGF blockade on a chemotherapy backbone in BTC.
Trial registration:
NCT identifier: NCT04677504; EUDRACT number: 2020-003759-14
Background
Biliary tract cancer (BTC) comprises a group of rare, anatomically distinct epithelial tumors arising in the biliary tree. Based on their anatomical location, BTCs are classified as gallbladder carcinoma (GBC), intrahepatic cholangiocarcinoma (iCCA), or extrahepatic cholangiocarcinoma (eCCA).1–3 In addition, eCCA can be further divided into perihilar (pCCA) and distal (dCCA), depending on origination above or below the cystic duct. 1 Anatomical subtypes possess distinct demographics, natural history, clinical presentations, risk factors, molecular backgrounds, treatment options, and prognosis.1,2,4,5 Inoperable, recurrent, or metastatic CCA and GBC are collectively referred to as advanced BTC. Despite being rare, BTCs are an important source of cancer-related mortality and harbor a dismal prognosis (3-year survival approximately 1% for patients with advanced disease).4,6 iCCA is the second most common primary hepatic malignancy after hepatocellular carcinoma (HCC), comprising 10–20% of all newly diagnosed primary liver tumors and 3% of gastrointestinal cancers.2,7 Approximately 50% of patients with iCCA have liver-only disease. 4 The incidence and mortality of iCCA has risen in most regions over recent years, whereas the incidence of eCCA and GBC has tended to decline.8–12
BTCs are often asymptomatic in their early stages and, as a result, are often first diagnosed when the tumor is at an advanced stage when therapeutic options are limited. Localized BTC can be managed with surgical resection and adjuvant chemotherapy, 13 however, only approximately 20% are eligible for curative resection at presentation and a significant number of initially resectable cases at diagnosis are subsequently found to be inoperable; local and distant relapses following surgery are frequent.12,14 Most patients (approximately two thirds) initially present at an advanced stage when palliative chemotherapy is the only feasible treatment option. 15 The results of the phase III ABC-02 and Japanese BT22 phase II studies have established gemcitabine and cisplatin (Gem/Cis) doublet chemotherapy as the standard of care for the first-line management of advanced BTC.16–18
Targeted agents have been extensively investigated in BTC. Earlier studies targeting classical oncogenic pathways implicated in BTC carcinogenesis such as angiogenesis and epidermal growth factor receptor (EGFR) in unselected patient populations failed to show superiority to standard chemotherapy.19–25 With respect to antiangiogenesis, the addition of either cediranib [a vascular endothelial growth factor (VEGF) multikinase inhibitor] or ramucirumab (a VEGFR2 monocolonal antibody) to Gem/Cis as first-line treatment failed to improve PFS or survival.20,21 The addition of bevacizumab (a VEGF monoclonal antibody) to gemcitabine/oxaliplatin demonstrated good tolerability and antitumor activity in advanced BTC in a single arm phase II trial. 26 In the second-line setting, regorafenib (a multikinase VEGF inhibitor) significantly improved PFS and disease control compared with best supportive care. 27 Based on improved understanding of the molecular basis of BTC, focus has shifted to ‘precision medicine’ strategies involving targeted therapies for use in biomarker-selected patient subgroups.19,28–30 Inhibitors of fibroblast growth factor receptor (FGFR) fusions, isocitrate dehydrogenase 1 (IDH1/2) mutations, and BRAFV600E mutations, which are found almost exclusively in iCCA, represent the most promising targets to date.28,31–34 The modest efficacy of chemotherapy (median survival ~12 months), coupled with the limited applicability of molecularly-targeted agents, emphasizes the urgent need to develop more effective treatments to improve patient outcomes.
Cancer immunotherapy (CIT) with immune checkpoint inhibitors (CPIs), most notably anti-PD-1/PD-L1 antibodies, has joined surgery, chemotherapy, targeted agents, and radiation therapy as a standard treatment modality for a variety of malignancies. PD-1 and PD-L1 inhibitors as monotherapies or in combination with other treatments are now considered mainstream treatments for lung cancer, melanoma, and hepatocellular carcinoma (HCC), as well as other maligancies. 35 For most cancers, only a minority of patients respond to PD-1/PD-L1 inhibitor monotherapy due to the immunosuppressive mechanisms operating in both the tumor and surrounding tumor microenvironment (TME) that result in primary and acquired resistance.36,37 By identifying and therapeutically targeting resistance mechanisms, treatment regimens can be developed to improve clinical outcomes. 37
PD-L1 expression in either tumor cells or infiltrating immune cells has been observed in up to 70% of BTCs38,39 and high PD-L1 expression has been associated with poor prognosis, suggesting a potential role for PD-1/PD-L1 inhibition. 39 To date, however, clinical data for PD-1 or PD-L1 inhibitors in BTC are limited to small studies or sub analyses from basket trials focusing on pre-treated patients.40–42 The antitumor activity of single-agent anti-PD-1/PD-L1 antibodies, including atezolizumab, in BTC patients, is modest, and in most cases response rates have not exceeded 10%.43–49 Consistent with other solid tumors, responses were durable in a small subgroup of responsive patients. 48 At present, anti-PD-1 treatment is indicated for approximately 2% of patients with CCA harboring DNA mismatch repair (MMR) defects or microsatellite instability (MSI). 1 Despite relatively low response rates, the quality and durability of responses to PD-1/PD-L1 inhibitors are similar to other tumor types, thereby warranting the development of combination treatment strategies to improve response rates and clinical outcomes.47,48
The goal of CIT combinations is to increase the potential of the immune system to eliminate cancer by disrupting immunosuppressive mechanisms in both the tumor and the TME. 50 In CCA, the TME is dominated by a desmoplastic stroma made up of a complex network of malignant cells, stromal cells, extracellular matrix components, and blood vessels, as well as immune cell subsets. 51 These components of the TME operate individually, or in combination, to directly or indirectly orchestrate tumor growth, treatment resistance, antitumor immunity, and sensitivity to CIT.50,51 Therefore, drug combinations targeting immunosuppressive components of the TME represent a rational approach in BTC.
One way to target TME immunosuppression is to combine PD-1/PD-L1 inhibitors with chemotherapy and/or anti-angiogenics which both possess immunomodulatory capabilities.50,52–54 In addition to its well characterized role in mediating angiogenesis, VEGF is a potent driver of tumor immune evasion. 54 The immunosuppressive effects of VEGF include inhibition of dendritic cell (DC) function and maturation, impaired CD8+ T-cell infiltration and function, upregulation of inhibitory immune checkpoints, and the accumulation of immunosuppressive cell types such as tumor-associated macrophages (TAM), myeloid-derived suppressor cells (MDSC), and regulatory T cells (Treg).53–56 Each of these mechanistic components provides a therapeutic target to reprogram an immunosuppressive TME. Preclinical and clinical studies have demonstrated that antiangiogenic drugs can revert VEGF-driven immunosuppression and augment the activity of PD-1 and PD-L1 inhibitors. 54 In the clinical setting, combinations of PD-1 or PD-L1 inhibitors, along with anti-VEGF agents, (either multikinase inhibitors or monoclonal antibodies) have proven superior to standard treatments in non-small cell lung cancer (NSCLC), renal cell carcinoma (RCC), and more recently HCC.57–60 Similarly, multiple phase III studies have shown that PD-1 and PD-L1 inhibitors combined with chemotherapeutic regimens are superior to chemotherapy alone in a variety of solid tumors.61–64 Triplet combination therapy with atezolizumab, bevacizumab, and platinum-based chemotherapy (carboplatin plus paclitaxel) has been evaluated in phase III studies in patients with advanced NSCLC (IMpower 150) and ovarian cancer (IMagyn050)58,65 and is under investigation in multiple randomized studies across different cancer types with various chemotherapy backbones. 54 While this combination was found to be effective in NSCLC, it was not efficacious in ovarian cancer. These disparate findings suggest that response to this regimen may differ depending on the malignancy under study and it remains to be seen if the combination is effective in BTC. In both phase III studies, the toxicity of the triplet regimens were found to be tolerable and manageable with no new safety signals identified.65,66 A detailed analysis of the safety data from IMpower 150 demonstrated that the addition of atezolizumab to bevacizumab and chemotherapy did not result in premature termination of chemotherapy and the adverse events associated with the triplet combination were mostly grade 1 or 2 and manageable. 66 In addition, the incidence of immune-related adverse events – mainly grade 1 or 2, was similar in the atezolizumab/chemotherapy and atezolizumab/bevacizumab/chemotherapy arms. 66
Many of the critical immunosuppressive mechanisms identified in the BTC TME are regulated to some degree by VEGF, thereby providing justification to study dual PD-L1/VEGF blockade in this setting. Antitumor immunity can also be augmented with cytotoxic chemotherapies, including gemcitabine and cisplatin, through mechanisms complementary to those that are regulated by VEGF. Therefore, dual inhibition of PD-L1 and VEGF coupled with the immunomodulatory effects of Gem/Cis could create an immune-favorable TME in biliary cancers, leading to enhanced clinical benefit over and above standard of care Gem/Cis.
In this paper, we describe the design of IMbrave 151, a randomized phase II study, that will be the first trial to evaluate combined anti-PD-L1/VEGF inhibition in tandem with chemotherapy in patients with advanced BTC. We also briefly discuss the scientific and clinical rationale for this combination approach in BTC.
Methods
Study design
IMbrave 151 is a randomized, double-blind, placebo-controlled multicenter phase II study designed to evaluate the efficacy and safety of atezolizumab with bevacizumab in combination with Cis/Gem, compared with atezolizumab plus Cis/Gem, in patients with advanced BTC (iCCA, eCCA, and GBC) who have not received systemic therapy for advanced disease.
The study will enroll approximately 150 patients who will be randomized, in a 1:1 ratio, to one of two treatments arms (see Table 1). Randomization will be stratified according to the anatomical location of the primary tumor (iCCA versus eCCA versus GBC), presence or absence of metastatic disease, and geographic region (Asia versus rest of world). The study design is shown in Figure 1. Recruitment will be competitive.
Eligibility criteria.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; BP, blood pressure; BTC, biliary tract carcinoma; COPD, chronic obstructive pulmonary disease; CT, computed tomography; eCCA, extrahepatic cholangiocarcinoma; ECOG, Eastern Cooperative Oncology Group; EGD, esophagogastroduodenoscopy; FFPE, formalin-fixed paraffin-embedded; GBC, gallbladder cancer; GI, gastrointestinal; HBV, hepatitis B virus; HCV, hepatitis C virus; iCCA, intrahepatic cholangiocarcinoma; MRI, magnetic resonance imaging; NCI CTCAE, National Cancer Institute Common Terminology Criteria for Adverse Events; NSAID, non-steroidal anti-inflammatory drugs; OS, overall survival; ULN, upper limit of normal.

IMbrave 151 study design.
Study endpoints
The primary efficacy endpoint of IMbrave 151 is PFS, defined as the time from randomization to the first occurrence of disease progression as determined by the investigator according to RECIST v1.1 67 or death from any cause (whichever occurs first).
Secondary efficacy endpoints include OS, confirmed ORR, duration of response, and disease control rate according to investigator-assessed RECIST 1.1, time to confirmed deterioration (TTCD) in patient-reported physical functioning, role functioning, quality of life (QoL), and safety.
Exploratory endpoints include PFS and OS rates at specific timepoints, additional patient-reported outcomes (PROs), biomarkers, pharmacokinetics, and the evaluation of anti-drug antibodies (ADAs).
Key eligibility criteria
Key inclusion and exclusion criteria for IMbrave 151 are shown in Table 1. Briefly, IMbrave 151 will enroll patients aged 18 years or older with a histopathological or cytological diagnosis of advanced BTC (intrahepatic or extrahepatic cholangiocarcinoma), or gallbladder carcinoma. Patients with ampullary carcinomas are not eligible. Previous systemic treatment for locally advanced or metastatic disease is not allowed. Patients may have received neoadjuvant or adjuvant therapy provided that was completed at least 6 months prior to Day 1 of Cycle 1. All patients must have adequate biliary drainage with no evidence of ongoing infection. A baseline tumor specimen (accompanied by a pathology report) should be provided unless one is either unavailable or a biopsy is not clinically feasible. All patients at high risk of esophageal varices must undergo an esophagogastroduodenoscopy (EGD), either during screening or within 6 months of cycle 1/day 1. Variceal risk criteria are listed in the study protocol. Any varices must be assessed and treated per local standard of care prior to enrollment.
Study treatments
The study treatment schedule is shown in Table 2. During the induction phase, patients will receive up to 8 cycles of Cis/Gem in combination with either atezolizumab and bevacizumab (arm A) or atezolizumab plus placebo (arm B). Following the cessation of chemotherapy, atezolizumab and bevacizumab (arm A) or atezolizumab with placebo (arm B) will be continued until unacceptable toxicity, disease progression (per RECIST 1.1) or loss of clinical benefit.
Study treatments.

Treatment during the chemotherapy combination phase will be administered on a 21-day cycle until completion of eight cycles, loss of clinical benefit, or unacceptable toxicity, whichever occurs first.
Atezo, atezolizumab; Bev, bevacizumab; CisGem, cisplatin and gemcitabine; CIT, cancer immunotherapy; d, day; IV, intravenous; PBO, placebo.
In the absence of unacceptable toxicity, patients who meet criteria for disease progression while receiving atezolizumab alone or in combination with bevacizumab will be permitted to continue the study treatment if they meet all of the following criteria; evidence of clinical benefit, as determined by the investigator following a review of all available data, absence of symptoms and signs (including laboratory values, such as new or worsening hypercalcemia) indicating unequivocal progression of disease, absence of decline in Eastern Cooperative Oncology Group (ECOG) Performance Status that can be attributed to disease progression, and the absence of tumor progression at critical anatomical sites (e.g., leptomeningeal disease) that cannot be managed by protocol-allowed medical interventions.
Study assessments
Patients will sign a written informed consent prior to any study related procedures. Screening evaluations, including medical history, physical examination, laboratory assessments, and ECG must be completed and reviewed to confirm that patients meeting all eligibility criteria prior to enrollment. Once enrolled and randomized, all patients will return to the clinic on day 1 and 8 of each 21-day cycle for the first 8 cycles, then on day 1 of each 21-day cycle thereafter for study treatment and assessments. Patients will undergo tumor assessments at baseline and every 9 weeks following treatment initiation until radiographic disease progression per RECIST v1.1 or (for patients who continue atezolizumab plus bevacizumab or placebo, after radiographic disease progression) loss of clinical benefit, as determined by the investigator.
Patient reported outcomes
Patient experience with advanced BTC is complex and effected by both disease- and treatment-related symptoms. To this end, an innovative PRO endpoint strategy has been developed for this study and includes using a digital platform to uniquely and directly capture the patient voice in a robust manner. The PRO assessments include the following questionnaires:
European Organization for Research and Treatment of Cancer (EORTC) quality-of-life questionnaire (QLQ-C30) – a validated and reliable self-reported measure that assesses aspects of patient functioning, overall health, and generic cancer symptomatology, during the preceding week. 68
EORTC quality-of-life questionnaire for cholangiocarcinoma and cancer of the gallbladder (QLQ-BIL21) – a validated and reliable self-reported measure that assesses BTC specific symptomology during the preceding week. 69
Selected items from the EORTC Item Library (IL77) – a reduced version of the QLQ-C30 that was created for this study, and which assesses aspects of patient functioning and generic cancer symptomology.
Patient Global Impression of Change and its Importance (PGI-CI) – a self-reported measure that was adapted for this study and which assesses patients’ impression about changes to their overall health because of their cancer and the associated importance compared with when they began the study.
Patient Global Impression of Severity (PGI-S) – a self-reported measure that was adapted for this study and which assesses patients’ impression about how severely their overall health has been impacted because of their cancer during the preceding week
Patient-Reported Outcomes Common Terminology Criteria for Adverse Events (PRO-CTCAE) – a validated item bank of 78 patient-reportable symptomatic treatment toxicities from which four symptoms deemed most applicable to the treatments being evaluated for this study were selected. 70
The PRO assessments will be completed before patients start treatment, at specified timepoints over the course of treatment, and at treatment discontinuation.
Translational research
An extensive program of translational research is planned, using both tissue, blood, and stool samples from enrolled patients in order to identify potential biomarkers (predictive and/or prognostic) and mechanisms of treatment resistance. Tissue (either archival or fresh), if available, will be collected at baseline with an option to provide an on-treatment tissue specimen. Given the challenges associated with tissue harvesting in BTC, the provision of tumor tissue is strongly encouraged, but not mandated in our study. 71
Blood samples [serum, plasma, and peripheral blood mononuclear cells (PBMCs)] will be collected from enrolled patients at baseline, during treatment, and at disease progression. Patients will be given the option of consenting to provide stool samples that will be collected at baseline and at one additional timepoint between Cycle 2 and Cycle 3 of treatment.
All biomarker analyses are exploratory and will be performed retrospectively to assess their association with clinical endpoints in order to identify potential mechanisms of response and resistance to atezolizumab and bevacizumab combination therapy. Key translational science objectives of IMbrave 151 include:
Evaluating the association between PD-L1 expression, tumor mutation burden (TMB), and MSI/MMR status and clinical outcomes.
Exploring the relationship between genomic markers and clinical benefit.
Studying the association between circulating biomarkers and clinical outcomes.
Key biomarker analyses are described below.
Tissue-based biomarker analysis
Achieved tumor tissue samples or fresh biopsies at baseline will be collected to assess PD-L1 expression levels on both tumor and immune cells by immunohistochemistry assay (SP263).
Whole exome sequencing (WES) will be performed to determine TMB, mismatch repair (MMR) deficiency, microsatellite instability (MSI), as well as frequent driver mutations found in BTC, such as IDH1/2 mutations and FGFR fusions. These genetic aberrations will be correlated with clinical outcomes in this trial to inform their potential roles in immunotherapy in BTC patients. Transcriptome analysis will be performed on tumor tissues by RNA sequencing. Gene expression signatures on T-effectors, regulatory T cells, myeloid inflammation, and angiogenesis, as well as other immune subsets and biological pathways, will be evaluated and associated with clinical outcomes.
Blood-based biomarker analysis
Peripheral blood samples for exploratory biomarker studies are collected at baseline and at several time points during treatment (C2D1, C3D1, C4D1, C8D1, C12 D1, and C16D1) and at disease progression. Circulating tumor DNA (ctDNA) will be evaluated in the plasma by SignateraTM 16-plex multiplex PCR next-generation sequencing assay specific to each patient’s tumor mutational signatures. Single cell RNA sequencing or Flow Cytometry panels will be performed in PBMC samples collected at baseline and post-treatment to explore the proportion and kinetics of different immune subsets and their association with responses to the treatment. Circulating cytokines and chemokines [i.e., interleukin-6 (IL6), interleukin-8 (IL8), and c-reactive protein(CRP)] will be evaluated by an ELISA-based immunoassay in the serum. Correlations between these biomarkers and safety and efficacy endpoints will be explored to identify blood-based biomarkers that might predict which patients are more likely to benefit from atezolizumab in combination with bevacizumab.
Microbiome
Stool sample collection, in order to study the effect of the gut microbiome on treatment efficacy and safety, is optional for patients in IMbrave 151. The gut microbiome has been shown to be a key determinant of carcinogenesis and in anticancer immunity, in part by influencing T-cell driven anti-tumor responses. 72 The role of microbial dysbiosis in the development and immune regulation of hepatobiliary tumors is not well characterized. 73 Furthermore, antibiotic treatment, which is commonly used in BTC management, is associated with poor survival outcomes to anti-PD-1 therapy in a variety of solid tumors. 74 The effect of antibiotics on the microbiome of patients with BTC undergoing immunotherapy will be evaluated.
Statistics
The primary objective of this study is to estimate the treatment effect in each of the treatment arms. No formal hypothesis testing will be conducted. The primary efficacy endpoint is investigator-assessed PFS according to RECIST v1.1. A planned sample size of approximately 150 patients will be randomized at a 1:1 ratio to either arm A or arm B. The final PFS analysis will occur when approximately 90 events have been observed. The number of PFS events is deemed adequate to provide sufficient data and precision to estimate the PFS hazard ratio (HR) point estimate and its 95% confidence interval (CI).
Key secondary endpoints include OS, confirmed ORR, duration of response, and disease control rate according to investigator-assessed RECIST 1.1 and safety.
Efficacy analyses will be performed based on the intent-to-treat (ITT) principle stating that all randomized patients, regardless of whether they receive the assigned treatment or not, will be included and grouped according to the treatment assigned at randomization. All subjects who received any amount of study treatment will be included in the safety-evaluable population. HRs and associated 95% CIs for PFS and OS will be estimated by a stratified Cox proportional hazards regression model. The differences in PFS and OS between the two treatment arms will be further estimated by use of the stratified log-rank test. The Kaplan–Meier method will be used for computing medians for time-to-event endpoints including PFS, OS, DOR, and TTCD. The Brookmeyer–Crowley method will be used to calculate the 95% CI for each median time to event. Objective response and disease control rates will be calculated along with 95% CIs estimated by the Clopper–Pearson method. Safety analyses will be conducted using descriptive statistics. One interim analysis will be performed at the time once 100 randomized patients have been followed for at least 6 months.
Ethics
This study will be conducted in accordance with the 1964 Declaration of Helsinki and Ethical Guidelines for Medical and Health Research Involving Human Subjects. Patients will provide written informed consent for participating in the study and for allowing the collection of tissue and blood samples. This study is carried out in accordance with International Conference on Harmonization Good Clinical Practice (ICH-GCP). The study is under review by Institutional Review Board or Ethics Committee (IRB/EC) at participating institutions and, if applicable, an appropriate regulatory body.
Discussion
BTC is a highly lethal tumor, for which new and effective treatments, such as immunotherapies, are urgently needed. The role of CIT in the treatment of BTC is at a nascent stage, and, given the modest response rates reported with PD-1/PD-L1 antibodies, combination approaches are likely needed to unleash effective antitumor immunity in biliary tumors. IMbrave 151 seeks to build on the Gem/Cis regimen and will evaluate the efficacy and safety of atezolizumab combined with Gem/Cis with or without bevacizumab.
IMbrave 151 is a randomized phase II study with two parallel experimental arms. The study is not designed to directly compare the arms but rather to evaluate the efficacy of each arm independently in order to inform future phase III studies and/or novel combination regimens. We believe this design is justified and appropriate, in light of both the inherent complexity and heterogeneity of BTC, as well as the availability of relevant combination safety data from other advanced solid tumors. IMbrave 151 does not include a Gem/Cis treatment arm as the clinical outcomes for this standard regimen are well described in the literature.4,16,17 The clinical and molecular heterogeneity of BTC make interpretation of data, especially time-to-event endpoints such as PFS, from single-arm studies challenging. 75 Historically, ORR, a typical phase I study efficacy endpoint, has proven to be an unreliable surrogate endpoint in BTC.76,77 However, ORR and its association with PFS or OS has not yet been evaluated in the setting of immunotherapy where antitumor responses are often more durable than responses to chemotherapy or targeted agents. A randomized study design allows for stratification based on critical clinical parameters. 75 While the combination of atezolizumab, bevacizumab, and Gem/Cis has not been studied in BTC, similar combinations have been, or are currently being evaluated in phase III studies in other advanced solid tumors.58,65
The immune phenotype of tumors can be broadly characterized as immune inflamed (‘hot’), immune-excluded or immune-desert according to the type, location, and density of the immune-cell infiltrate.78–80 These frameworks can be used to predict the likelihood of response to a CPI and to guide combination treatment strategies. 78 Tumors harboring immune-excluded and immune-desert phenotypes are considered poorly responsive to PD1/PD-L1 blockade. 78 Relative to other cancers, the TME of BTC is not well characterized, but recent data indicate that the majority of CCAs harbor either immune-excluded (T-cell effectors confined to the tumor margin unable to penetrate the tumor bed) or immune-desert phenotypes (absence or scarce T-cell infiltration in the stroma and tumor bed).81,82 Approximately 11% of both iCCA and eCCA harbor an immune-inflamed phenotype defined using integrative genomic analysis, which may portend to response to PD-1 or PD-L1 monotherapy.29,82 TME composition is an important determinant of patient prognosis in BTC.83–85 and the accumulation of immunosuppressive cell types e.g. tumor associated macrophages (TAMs) and regulatory T-cells (T-regs), poor infiltration of cytotoxic CD8+ T-cells, or defective MHC class 1 expression is linked with poor prognosis as well as gemcitabine resistance.83,84,86 Basic and clinical studies indicate that many of these immunosuppressive mechanisms within the TME of CCAs are subject to regulation by VEGF, or can be modulated with platinum-based chemotherapy, thereby providing a mechanistic rationale for IMbrave 151 (Table 3).
Immunomodulatory effects of gemcitabine, cisplatin, and VEGF inhibitors.

↑, increase; ↓, decrease;?, data mixed or inconclusive ✓, drug shown to modulate mechanism; ✘, drug does not modulate mechanism; MDSC, myeloid derived suppressor cell; MHC, major histocompatibility complex; PD-L1, programmed death ligand 1; TAM, tumor-associated macrophage; Treg, T-regulatory cell.
VEGF blockade can restrain the immune-suppressive effects of VEGF, which include inhibition of DC function and maturation, defective T-cell function and infiltration, the accumulation of immunosuppressive cells (TAMs, MDSCs and Tregs), as well as upregulated expression of PD-L1. 54 Known immunomodulatory effects of cisplatin and gemcitabine include increased MHC class 1 expression, recruitment and proliferation of immune effectors, increased PD-L1 expression, and downregulation of immune-suppressive cell types. VEGF inhibitors, gemcitabine, and cisplatin exhibit varied and overlapping immunomodulatory effects that may promote therapeutic synergy with anti-PD-L1 treatment (Table 3).
Clinical evidence from randomized trials has demonstrated the benefit of PD-1/PD-L1 inhibitors combined with anti-VEGF agents in both primary and secondary liver cancers.57,98,99 Atezolizumab combined with bevacizumab significantly improved OS, PFS, and ORR compared with sorafenib in patients with unresectable HCC. 57 The combination of atezolizumab and bevacizumab was superior to atezolizumab alone in terms of PFS in a randomized unresectable HCC phase 1b cohort. 98 An exploratory analysis from this study demonstrated that PFS was prolonged in patients with high expression of VEGR2, Treg, and myeloid inflammation signatures treated with atezolizumab and bevacizumab compared with atezolizumab alone. 100 Atezolizumab combined with bevacizumab and platinum-based chemotherapy significantly improved PFS, OS, and ORR in patients with NSCLC adenocarcinoma with liver metastases at baseline, whereas neither atezolizumab or bevacizumab in tandem with chemotherapy were effective in this subgroup. 99 Together, these data indicate that VEGF is an important driver of hepatic immunosuppression and dual VEGF PD-1/PD-L1 blockade can be exploited clinically to improve treatment outcomes. Although combined PD-1/PD-L1 and VEGF inhibition has proven effective in HCC and NSCLC, it failed in advanced ovarian cancer, 65 which suggests that tumor histology may be an important factor.
Therapeutic combinations of PD-1/PD-L1 antibodies with VEGF inhibitors or chemotherapy have not been well studied in BTC (Table 4). It should be noted that no study has yet evaluated all three treatment modalities in patients with BTC. In the first-line setting, doublet regimens comprising VEGF inhibitors and Gem/Cis have failed to demonstrate superiority to Gem/Cis;20,21 while the efficacy of PD-1/PD-L1 blockade in tandem with chemotherapy is unclear (Table 4).
Clinical data for PD-(L)1 antibodies combined with Gem/Cis or anti-VEGF agents in BTC.
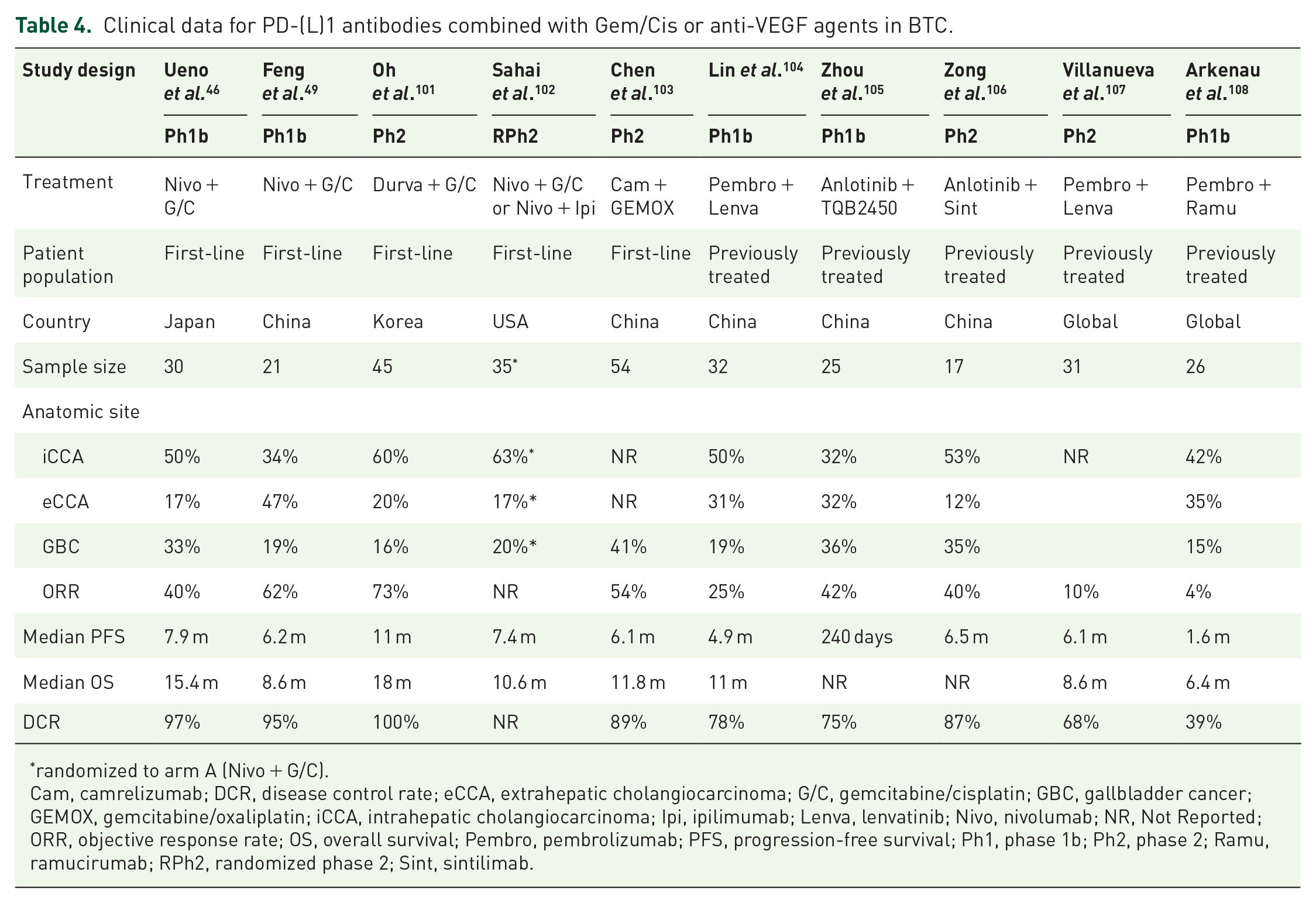
randomized to arm A (Nivo + G/C).
Cam, camrelizumab; DCR, disease control rate; eCCA, extrahepatic cholangiocarcinoma; G/C, gemcitabine/cisplatin; GBC, gallbladder cancer; GEMOX, gemcitabine/oxaliplatin; iCCA, intrahepatic cholangiocarcinoma; Ipi, ipilimumab; Lenva, lenvatinib; Nivo, nivolumab; NR, Not Reported; ORR, objective response rate; OS, overall survival; Pembro, pembrolizumab; PFS, progression-free survival; Ph1, phase 1b; Ph2, phase 2; Ramu, ramucirumab; RPh2, randomized phase 2; Sint, sintilimab.
Overall, data for PD-1 and PD-L1 inhibitors in combination with Cis/Gem as a first-line treatment suggest encouraging response rates and manageable toxicity, but PFS and OS similar to that reported for Gem/Cis alone (Table 4).46,49 Phase III studies are ongoing to evaluate PD-1 or PD-L1 antibodies in combination with Gem/Cis.109,110
To date, combined PD-1/VEGF inhibition has been evaluated in several small single-arm phase I and phase II studies in patients with previously treated advanced BTC with mixed results (Table 4).108,111 These studies suggest that dual PD-1/VEGF blockade is clinically feasible in advanced BTC but the antitumor activity may be limited. Based on the limited clinical benefit seen so far with PD-1/PD-L1 antibodies in combination with either Gem/Cis or anti-VEGF agents, it is possible that these doublet combinations may be unable to sufficiently overcome immunosuppression in the TME to unleash an anticancer immune response. Given the overlapping and complementary immune-modulating effects of VEGF and chemotherapy, co-targeting of PD-L1 and VEGF pathways coupled with Gem/Cis, could unlock more effective antitumor immunity in BTC by more effectively targeting relevant immune mechanisms within the TME.
This proof-of-concept randomized, double-blind, placebo-controlled phase II study will investigate the efficacy and safety of atezolizumab in combination Gem/Cis chemotherapy with or without bevacizumab as first-line treatment for advanced BTC. The combination of atezolizumab, bevacizumab, and Gem/Cis is a unique combination approach in BTC and the results of IMbrave 151, along with the accompanying translational studies and other ongoing clinical trials with other checkpoint inhibitor combination regimens, will provide important insights into the role of immunotherapy in the treatment of BTC.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is funded by F. Hoffmann-La Roche Ltd.
Conflict of interest statement
Stephen Hack: Full-time employee of Roche/Genentech; ownership of Roche stocks. Wendy Verret: Full-time employee of Roche/Genentech; ownership of Roche stocks. Sohail Mulla: Full-time employee of Roche/Genentech; ownership of Roche stocks. Bo Liu: Full-time employee of Roche/Genentech; ownership of Roche stocks. Yulei Wang: Full-time employee of Roche/Genentech; ownership of Roche stocks. Teresa Macarulla: Consulting or Advisory Role: Sanofi/Aventis, Shire, Celgene, Roche, Baxalta, QED Therapeutics, Baxter, Incyte, Servier, Lilly, Ipsen Research Funding: Celgene, Agios, ASLAN Pharmaceuticals, Bayer, Roche, Genentech, Astra Zeneca, Halozyme, Immunomedics, Lilly, Merrimack, Millennium, Novartis, Novocure, Pfizer, Pharmacyclics Travel, Accommodations, Expenses: Merck, H3 Biomedicine, Sanofi, Celgene, Servier. Anthony El-Khoueiry: Research support from Astex, received personal fees from Merrimack, and served as an adviser for Bristol-Myers Squibb, AstraZeneca, Bayer, Genentech, and Novartis. Zhenggang Ren: None Andrew Zhu: Consulting and advisory roles for Lilly, Bayer, Merck, Sanofi, Eisai, Exelixis, and Roche
