Abstract
Objectives:
Several therapies are available for the treatment of advanced/metastatic prostate cancer (PC). However, the systematic assessment of evidence pertaining to the use of these therapies in Asian patients is lacking.
Methods:
A systematic literature review (SLR) was conducted using PubMed/Medline search in May 2021 to identify the randomized/nonrandomized controlled trials (RCTs/non-RCTs) and real-world observational studies (prospective/retrospective). Only studies published as full manuscripts in English were included if reporting the efficacy, effectiveness, and/or safety of treatments in Asian patients with advanced/metastatic PC.
Results:
Of the 1,898 retrieved publications, 24 studies were included. These studies had patients with nonmetastatic castration-resistant PC (n = 2), metastatic castration-sensitive PC (n = 4), and metastatic castration-resistant PC (n = 18). Study designs included RCTs (n = 7), non-RCTs (n = 2), and real-world studies (n = 15). Treatments used in included studies were abiraterone acetate plus prednisone (AAP; n = 6), enzalutamide, lutetium-177 prostate-specific membrane antigen (177Lu-PSMA; n = 4 each), docetaxel (n = 3), apalutamide, radium-223 (n = 2 each), darolutamide, cabazitaxel, and pembrolizumab (n = 1 each). The evidence from RCTs (i.e., ARAMIS, SPARTAN, ARCHES, TITAN, LATITUDE, PREVAIL) demonstrated the clinical benefits of apalutamide, darolutamide, enzalutamide, and AAP in terms of overall, disease-free, and metastasis-free survival in Asian patients. These treatments were reported to be well tolerated, with no new safety signals identified in Asian population. The efficacy and safety profiles in Asian patients were consistent with the overall trial population. Data from real-world studies supported the effectiveness and tolerability of AAP, enzalutamide, radium-223, docetaxel, cabazitaxel, 177Lu-PSMA, and pembrolizumab in patients with advanced/metastatic PC.
Conclusions:
This SLR of the Asian data on therapies for advanced PC from the pivotal and real-world studies confirms similar efficacy and safety outcomes, consistent with the results from the pivotal clinical trials. These findings will help clinicians make better treatment decisions in clinical practice for patients with advanced/metastatic PC.
Keywords
Background
Globally, prostate cancer (PC) is the third most common cancer of all cancer types, and second most frequently diagnosed cancer in men. 1 In 2018, there were an estimated 1,276,106 new diagnoses and 358,989 new deaths from PC. 1 Of the 1.2 million new PC cases in 2018, 80% presented with localized disease whereas 20% had advanced or metastatic disease. The incidence of PC has been projected to increase significantly to 2.3 million by 2040, indicating a substantial burden of PC. 1 The incidence of PC varies widely between geographies due to differences in ethnic origin, potential genetic polymorphism as well as varying early screening and detection practices, and is highest in North America (97.2/100,000) and Northern Europe (85/100,000) and lower in southeastern Asia (11.2/100,000) and eastern Asia (10.5/100,000).2,3 While in the Western region, majority of diagnoses occur when the disease is still localized or locally advanced, in Asian countries a significant proportion of patients are found to have metastasis at diagnosis. Thus, incidence is lower in Asia, but mortality rates are higher than other parts of the world. 4
PC is a hormonally driven disease and androgen receptor plays a key role in its progression. Thus, many agents targeting androgen signaling pathway form the backbone of PC treatment, with hormonal therapy also known as androgen deprivation therapy (ADT) being the first-line treatment for advanced PC.5,6 Patients with PC have good prognosis if diagnosed and treated early. Patients with metastatic castration-sensitive PC (mCSPC) may have clinical benefits with ADT alone but castration resistance generally develop after 1–2 years.6–8 The 5-year relative survival for localized PC is 100%, whereas for mCSPC it is 30.6% only. 9 Castration-resistant PC (CRPC) is characterized as nonmetastatic CRPC (nmCRPC) in the absence of detectable metastases, and these patients are at significant risk of developing metastatic CRPC (mCRPC).10,11 Most Asian patients present with advanced and symptomatic disease where the standard treatment is surgical or medical castration. 4 About two-thirds experience biological recurrence, which leads to CRPC and eventually to mCRPC. 12 mCRPC being an aggressive disease, prognosis remains poor, and no curative therapies are available.
The efficacy and safety of ADTs in combination with other agents in the treatment of advanced PC have been demonstrated in several clinical trials. 13 Some of these treatments are novel androgen-receptor targeting agents (abiraterone acetate plus prednisone (AAP), apalutamide, darolutamide, and enzalutamide), radiopharmaceutical agents (radium-223, lutetium 177 prostate-specific membrane antigen (177Lu-PSMA)), chemotherapy (docetaxel, cabazitaxel), poly(ADP-ribose) polymerase (PARP) inhibitors (olaparib, rucaparib, niraparib, talazoparib), and immunotherapy (pembrolizumab and sipuleucel-T). 13 However, Asian population is often underrepresented in global clinical trials, and real-world studies have shown differential toxicity patterns in this population as well. The systematic assessment of evidence pertaining to the use of these therapies in Asian patients is lacking. Therefore, we conducted a systematic literature review (SLR) to collate and summarize the evidence on efficacy and safety of the treatments for advanced PC in Asian patients.
Methods
Data sources and searches
This SLR was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines. 14 A systematic PubMed/Medline search was conducted in May 2021 using both Medical Subject Headings and free-text words for disease condition (PC or advanced PC or metastatic PC) and treatments (abiraterone, AAP, apalutamide, darolutamide, enzalutamide, androgen receptor inhibitor, antiandrogen, androgen biosynthesis inhibitor, docetaxel, cabazitaxel, radium, radium-223, radium 223, olaparib, rucaparib, niraparib, talazoparib, PARP inhibitor, PARP, pembrolizumab, Lu-PSMA, or PSMA). PubMed/Medline searches were limited to a publication date between January 2016 and May 2021. The detailed search strategy is provided in Supplementary Table S1. A bibliographic search of relevant reviews/SLR was also performed to identify additional publications.
Study selection
Supplementary Table S2 presents the details of the inclusion and exclusion criteria. Only studies published as full manuscripts in English were included if reporting the efficacy, effectiveness, and/or safety of treatments in patients with PC in Asian countries. Both controlled clinical trials (randomized controlled trials (RCTs) or non-randomized controlled trials (non-RCTs)) and prospective or retrospective observational studies providing real-world evidence were included. Studies published as conference abstracts, with no outcomes of interest, and from countries other than Asia were excluded. Two independent reviewers screened all retrieved publications based on the title and abstract; any discrepancies between the two reviewers were resolved by a third independent reviewer, by consensus after discussion. Full manuscripts were then screened, and those satisfying the inclusion criteria were used for data extraction. Multiple publications from the same study were linked.
Data extraction and quality assessment
Data extraction of the included studies was performed by one reviewer. The quality check of the data was performed by the second reviewer, with reconciliation of any differences by the third reviewer. Data were extracted into an extraction grid in Microsoft Excel for various parameters, including study design, country of publication, population, treatment(s), follow-up, patient characteristics (e.g., age, Eastern Cooperative Oncology Group Performance Status (ECOG PS), Gleason score, prior treatments, etc.), and treatment outcomes (overall survival (OS), progression-free survival (PFS), time to progression, response rate, adverse events (AEs), serious AEs, etc.). Each included full manuscript was critically appraised for methodological quality using the Cochrane Risk of Bias tool for RCTs 15 and the Newcastle–Ottawa Scale for non-RCTs and observational studies. 16 Data were analyzed qualitatively, and the findings are reported as numbers and/or percentages.
Results
The literature search provided 1,898 citations in total. Forty-seven were removed as duplicates due to overlapping among databases. After screening the titles and abstracts and then the full texts, 27 publications17–43 were shortlisted. No additional studies were identified from the bibliographic search. After linking multiple publications, 24 studies were included in this review (Figure 1).
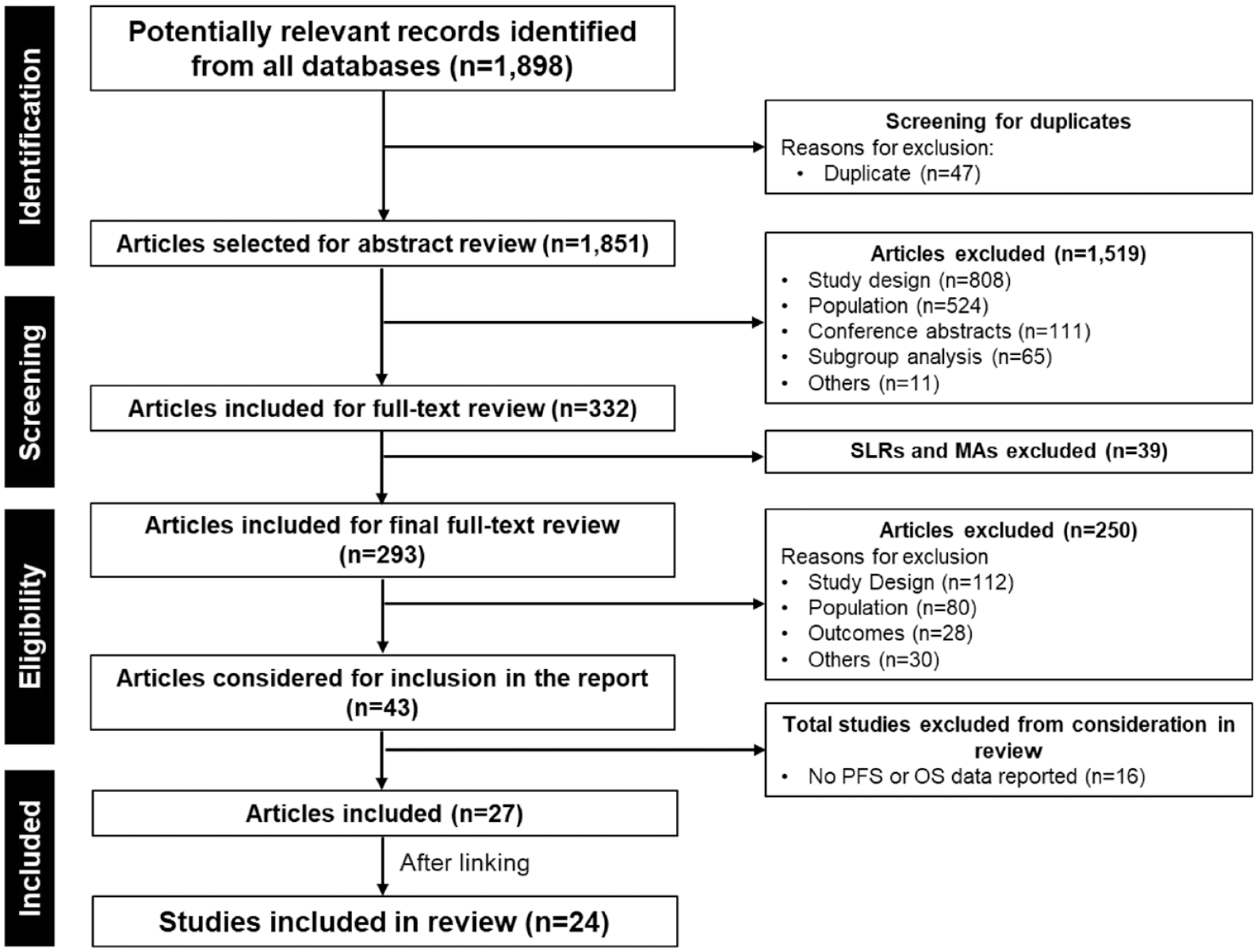
PRISMA diagram.
Overview of studies
Supplementary Table S3 presents the summary of included studies, which comprised RCTs (n = 7),17–21,26,27 non-RCTs (n = 2),27,28 and real-world prospective/retrospective observational studies (n = 15).23,29–43 Studies were from Japan (n = 8), India, Singapore, Hong Kong (n = 3 each), China (n = 2 each), and Taiwan (n = 1), whereas four studies were multi-Asian country studies. Most studies (n = 16) were published between 2019 and 2021, highlighting the recently published evidence.
Patients
The included studies covered the whole spectrum of PC disease, including patients with nmCRPC (n = 2),17,18 mCSPC (n = 4),19–21,23 and mCRPC (n = 19). Population included in all 15 real-world studies was patients with mCRPC. Number of patients enrolled in RCTs and non-RCTs varied from 49 28 to 313, 26 with 6 (out of 9) having less than 100 patients. Number of patients included in real-world studies ranged between 10 42 and 660, 38 with 9 of 15 studies having more than 100 patients. The median age of patients was 63 years or greater. The majority of patients had an ECOG PS of 0 or 1, and Gleason score of ⩾8 (Supplementary Table S3).
Therapies and doses
Therapies in the included studies were AAP (n = 6), enzalutamide, 177Lu-PSMA (n = 4 each), docetaxel (n = 3), apalutamide, radium-223 (n = 2 each), darolutamide, cabazitaxel, and pembrolizumab (n = 1 each) (Supplementary Table S3). Apalutamide (240 mg once daily), enzalutamide (160 mg once daily), and darolutamide (600 mg twice daily) were given orally. AAP was given as AA (1000 mg once daily) with prednisone (5 mg once- or twice-daily depending on the study quoted) orally. Radium-223 at the dose of 55 kBq/kg was given intravenously every 4 weeks for six cycles.27,28 Docetaxel was given as 60–75 mg/m2 intravenously every 3 weeks and 20–35 mg/m2 weekly along with prednisone 5 mg twice daily orally.23,36,37 Cabazitaxel was administered 20–25 mg/m2 intravenously every 3 weeks in combination with prednisolone once daily orally. 38 177 Lu-PSMA was administered at a mean dose of approximately 7 GBq or 814 MBq per cycle; patients were administered up to four cycles, with an interval of ⩾6 weeks.39–42 Pembrolizumab 200 mg was administered intravenously every 3 weeks. 43
Outcomes and follow-up
Supplementary Table S4 lists the outcomes assessed across the included studies. The most assessed outcomes were safety (n = 22), OS (n = 20), rPFS/PFS (n = 15), PSA response rate (n = 15), time to skeletal-related events (n = 9), time to PSA progression, changes in PSA (n = 7 each), time to pain progression, and time to chemotherapy initiation (n = 6 each). Some studies also reported the assessment of patient-reported outcomes, including Brief Pain Inventory Short Form (n = 5), Visual Analogue Scale (n = 3), Functional Assessment of Cancer Therapy–Prostate (n = 2), Analgesic Quantification Scale (n = 2), and European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (QLQ-PR25) (n = 1). The median duration of follow-up in these studies ranged from 6.3 34 to 56.6 months. 21
Evidence on efficacy and effectiveness
Table 1 provides the overview of PFS and OS reported in RCT and observational studies of advanced or metastatic PC. Subsequent sections describe these in detail by indications and treatments.
PFS and OS reported in RCTs, non-RCTs, and real-world studies of advanced/metastatic PC included in this review.

N is the total number of patients in the study; n is the number of patients on a particular treatment.
Metastasis-free survival.
Radiographic PFS.
Time to PSA progression.
Biochemical PFS.
OS given in days.
Time to castration-resistance.
AA, abiraterone acetate; ADT, androgen deprivation therapy; APA, apalutamide; CBT, cabazitaxel; CI, confidence interval; CN, China; DAR, darolutamide; DOC, docetaxel; EA, East Asia; ENZA, enzalutamide; HK, Hong Kong; HT, hormonal therapy; IN, India; JP, Japan; 177Lu-PSMA, lutetium-177 prostate-specific membrane antigen; mCRPC, metastatic castration-resistant prostate cancer; mCSPC, metastatic castration-sensitive prostate cancer; nmCRPC, non-metastatic castration-resistant prostate cancer; NR, not reported; NYR, not yet reached; MY, Malaysia; OS, overall survival; P, prednisone; PBO, placebo; PC, prostate cancer; PEMB, pembrolizumab; PFS, progression-free survival; PSA, prostate-specific antigen; Q3W, every 3 weeks; RCT, randomized controlled trial; RU, Russia; SG, Singapore; TH, Thailand.
Nonmetastatic CRPCApalutamide
Evidence for apalutamide in nmCRPC was available from one RCT (SPARTAN). 18 This trial reported the results of Japanese subpopulation in which 55 patients with nmCRPC with ongoing ADT were randomized to apalutamide (n = 34) or placebo (n = 21). In the primary end point metastasis-free survival (MFS), median was not reached for apalutamide compared with 18.23 months for placebo. The hazards ratio (HR) for metastasis or death was 71% lower for apalutamide (HR 0.29; 95% confidence interval (CI) 0.06–1.48), which was consistent with the global population. Thirty-three percent patients on placebo versus 6% on apalutamide treatment developed distant metastasis or died. Median OS was not reached for both groups but improved with apalutamide treatment. 18
Darolutamide
Only one RCT (ARAMIS) provided data for darolutamide in nmCRPC. 17 In this sub-analysis report, 95 Japanese patients with histologically/cytologically confirmed nmCRPC were randomized to darolutamide (n = 62) or placebo (n = 33) with continued ADT. Twenty MFS events were observed (9 in the darolutamide group and 11 in the placebo group). Median MFS was not reached for darolutamide compared with 18.2 months for placebo (HR 0.28; 95% CI 0.11–0.70). The improvement in MFS observed with darolutamide in the Japanese subgroup was consistent with the overall global population. Median OS was not reached in both groups but favored darolutamide in the Japanese subgroup (HR 0.72; 95% CI 0.12–4.31). Time to pain progression, time to PSA progression, and PSA response also favored darolutamide. 17
Metastatic castration-sensitive PC
Apalutamide
Data for apalutamide in mCSPC was available from one RCT (TITAN), 20 including 51 Japanese patients with mCSPC randomized to apalutamide (n = 28) or placebo (n = 23) in addition to ADT. Patients who did not experience radiographic progression or death were 82% in apalutamide group and 72% in placebo group, and HR for radiographic PFS (rPFS) favored treatment with apalutamide (HR 0.712; 95% CI 0.205–2.466). Also, 86% of patients in apalutamide group and 82% in placebo group were alive at 24 months, and HR for OS favored apalutamide (HR 0.840; 95% CI 0.210–3.361). The median rPFS and OS were not reached in apalutamide group and time to cytotoxic chemotherapy was delayed following apalutamide treatment. 20
Enzalutamide
One RCT (ARCHES) contributed evidence for enzalutamide in mCSPC. 19 In ARCHES trial, 92 Japanese patients with confirmed diagnosis of metastatic hormone-sensitive PC (mHSPC) were randomized to enzalutamide (n = 36) or placebo (n = 56) plus ADT. Treatment with enzalutamide reduced the risk of radiographic progression or death in Japanese patients by 61% compared with placebo (HR 0.39; 95% CI 0.30–0.50), similar to the overall population. OS data were immature for both groups, however, favored treatment with enzalutamide plus ADT (HR 0.92; 95% CI 0.15–5.52). 19
Abiraterone acetate plus prednisone
Evidence for AAP in mCSPC was available from one RCT.21,22 In the LATITUDE trial, 70 Japanese patients with metastatic hormone-naïve PC (mHNPC) were randomized to AAP or matching placebo (n = 35 each group). Median OS was not reached in both treatment groups. At the time of analysis, there were 26 and 40% deaths in AAP and placebo group, respectively. The overall 5-year survival rate was 69 and 54% in the AAP and placebo, respectively. The risk of death was 39% lower in the AAP group (HR 0.61; 95% CI 0.27–1.42), which was consistent with the overall population. Median PFS was not reached for AAP group versus 30.4 months for placebo. Treatment with AAP was associated with an improvement in the PFS (HR 0.32; 95% CI 0.167–0.620). Other Asian (non-Japanese) patients were also included in LATITUDE trial but the number was too small for a meaningful analysis. 21
Docetaxel
One real-world study provided evidence for docetaxel in mCSPC.23,24 In this study from Hong Kong, the medical records of 32 patients with mHNPC treated with chemohormonal therapy were reviewed. The median follow-up duration was 11.4 months, with 94% patients completed six or more cycles of chemotherapy with docetaxel. The efficacy results indicated that median time to CRPC and time to PSA nadir were 19.5 and 7 months, respectively. PSA response (>50% drop in PSA level from baseline) was achieved in all patients and the median maximal PSA response was 99.6%. 23
Metastatic CRPC
Abiraterone acetate plus prednisone
One RCT 26 and four real-world studies29–33 contributed evidence for AAP in patients with mCRPC. In the RCT, Ye et al. 26 investigated the efficacy and safety of AAP in 313 chemotherapy-naïve, asymptomatic or mildly symptomatic mCRPC patients from China, Malaysia, Thailand, and Russia, and patients were randomized in equal proportion to AAP or matching placebos. At the median follow-up of 3.9 months, median time to PSA progression was not reached in AAP group versus 3.8 months in placebo, attaining 58% reduction in PSA progression risk (HR 0.42; 95% CI 0.27–0.65; p < 0.0001). AAP treated patients had higher confirmed PSA response rate and were 5-times more likely to achieve radiographic response than placebo treated patients. Median OS was not reached. 26
Four real-world studies, one each from Japan (113 patients), 29 Singapore (200 patients), 30 Hong Kong (110 patients),32,33 and Malaysia and Thailand (93 patients), 31 provided evidence on the effectiveness of AAP. The majority of patients in each study (53–82%) were chemotherapy naïve. The results indicated that in three studies,30–32 median OS and PFS were numerically greater among chemotherapy-naïve patients compared with post-chemotherapy patients. In a study comparing AAP and docetaxel as first-line treatment in chemo-naïve mCRPC patients, AAP resulted in improved PFS and comparable OS as compared to docetaxel. 33 A study by Lim et al. also showed that patients from Malaysia had a relatively lower median OS (17.8 versus 27.0 months) and PFS (10.4 versus 14.0 months) than patients from Thailand, although these differences were not statistically significant. 31
Enzalutamide
One RCT (PREVAIL) 25 and two real-world studies29,34 provided data for enzalutamide in mCRPC patients. In the PREVAIL trial, 25 the efficacy and safety of enzalutamide was evaluated versus placebo in 148 East Asian (Japan, Korea, and Singapore) chemotherapy-naïve men with mCRPC. Results showed that treatment effect of enzalutamide compared with placebo was consistent with that for overall population as demonstrated by the HRs (95% CI) of 0.59 (0.29–1.23) for OS, 0.38 (0.10–1.44) for rPFS, and 0.32 (0.20–0.50) for time to PSA progression. 25 In the real-world study from Hong Kong reviewing the clinical records of patients retrospectively, 117 patients had received enzalutamide as first line (chemo-naïve or no AAP; n = 34, or 29.1%), second line (post-docetaxel or AAP; n = 57, 48.7%), and third or fourth line (previously received ⩾2 of docetaxel, AAP, cabazitaxel, and/or radium-233; n = 26, or 19.3%). 34 The overall PSA response rate was 43.6%, with varying PSA rates by lines of treatment (first/second/third or fourth line: 73.5/35.1/19.2%). OS and PFS were significantly associated with the line of treatment in the univariate survival analysis (first/second/third or fourth line: OS, not reached/15.8/7.4 months; PFS, 7.1/3.9/2.2 months; both p = 0.0002) but not in the multivariate analysis. 34 The real-world study from Japan showed that PSA response rate and PSA PFS were significantly improved in patients treated with enzalutamide versus those treated with AA. 29
Radium-223
Two non-RCTs (single-arm, interventional studies) provided evidence on efficacy of radium-223 in patients with symptomatic CRPC and bone metastases.27,28 There were 226 patients in the study from Asian countries (Singapore, China, Taiwan, and South Korea) 27 and 49 patients in the study from Japan 28 . In both studies patients received a median of six injections of radium-223, with 51–57% completing all six scheduled injections. In the Asian study, the median OS was 14.0 months (95% CI 11.2–17.4); median time to PSA progression was 3.6 months (95% CI 3.1–3.7); and median symptomatic skeletal-related event (SSE)-free survival was 26.0 months (95% CI 12.6–not reached). 27 The study from Japan study showed 1-year OS and SSE-free rate of 78 and 89%, respectively. Kaplan–Meier curve showed median OS of 12.5 months. 28
Docetaxel
Evidence for docetaxel was available from two real-world studies.36,37 A prospective, observational study from China included 403 patients with mCRPC who received ⩾1 dose of docetaxel following failure of hormonal therapy (disease progression with serum testosterone <50 ng/dL; first line, 42%; second line, 31%; third line, 13%). The mean (SD) number of docetaxel cycles was 4.4 (2.9). The median OS was 22.4 months (95% CI 20.4–25.8) and PSA response rate was 71%. 36 A study from Singapore compared the standard dose regimen of docetaxel 75 mg/m2 every 3 weeks (n = 11) with two attenuated regimens (docetaxel 60 mg/m2 every 3 weeks (n = 38); docetaxel 20–35 mg/m2 weekly (n = 40)) in patients with mCRPC. Results showed comparable median OS between 75 and 60 mg/m2 every 3 weeks regimens of docetaxel (18.0 versus 16.9 months; p = 0.05). However, the median OS in docetaxel 60 mg/m2 every 3 weeks was significantly longer than in the weekly regimen (16.9 versus 10.6 months; p = 0.01). 37
Cabazitaxel
A post-marketing surveillance study from Japan evaluated the real-world effectiveness of cabazitaxel in 660 patients with mCRPC previously treated with a docetaxel-containing regimen. The median OS was 319 day (95% CI 293–361) and time to treatment failure was 116 days (95% CI 108–135). The PSA response rates for decrease of ⩾30 or ⩾50% from baseline was 28 and 18%, respectively, in patients with baseline PSA of ⩾5 ng/mL. 38
Lutetium-177 prostate-specific membrane antigen
Four real-world studies provided data for the effectiveness of 177Lu-PSMA, including one from Singapore (20 patients) 39 and three from India (10–40 patients)40–42. In the study from Singapore, median four cycles per patient were administered, and the median OS and PFS was 13.1 (95% CI 8.6–15.0) and 5.9 (95% CI 2.8–7.4) months, respectively. 39 In two Indian studies, patients were given only one cycle of 117Lu-PSMA, and on the PSA response evaluation, 10–23% of patients had PR, 59–60% SD, and 18–30% PD.40,42 In another Indian study with 40 mCRPC patients, three cycles were administered on average. The median OS and PFS was 12 and 7 months, respectively. Based on PSA response assessment, 43% of patients had CR, 5% PR, 5% SD, and 48% PD. 41
Pembrolizumab
In a retrospective review study from China with previously untreated mCRPC patients harboring PD-L1 staining, 100 patients received pembrolizumab plus enzalutamide whereas 106 received pembrolizumab alone. The study indicated that median OS was 25.1 months for patients receiving pembrolizumab plus enzalutamide versus 18.3 months for patients receiving pembrolizumab alone (HR 0.56; 95% CI 0.39–0.80; p = 0.001). A marked distinction was also observed in the median PFS (6.1 months for pembrolizumab plus enzalutamide versus 4.9 months for pembrolizumab alone; HR 0.55; 95% CI 0.41–0.75; p = 0.001). 43
Evidence on safety and tolerability
Table 2 provides the summary of safety results reported in included studies. In both RCTs of apalutamide, SPARTAN in patients with nmCRPC 18 and TITAN in mCSPC, 20 no new safety signals were identified in the Japanese subpopulation. The safety profile of apalutamide with ADT was comparable with that of the global population, except for skin rash that was higher in the apalutamide group in Japanese subpopulation (50% versus 27% 20 ). Darolutamide was reported to be well tolerated in Japanese patients in the ARAMIS study. 17 However, due to small number of patients in the Japanese subgroup, it was not possible to conclude with certainty whether differences in the safety profile exist between Japanese and overall ARAMIS populations. Patients randomized to darolutamide had numerically higher treatment emergent adverse events (TEAEs) compared with placebo: overall TEAEs, 86% versus 64%; serious TEAEs, 32% versus 9%; and treatment discontinuations due to TEAEs, 8% versus 6%. 17 In both RCTs of enzalutamide, ARCHES in patients with mCSPC 19 and PREVAIL in mCRPC, 25 the safety profile of enzalutamide was tolerable in Japanese and East Asian patients and were generally consistent with that observed in the overall study population. Nevertheless, nasopharyngitis (25% versus 4%), hypertension (20% versus 8%) and abnormal hepatic function (14% versus 1%) were the frequently reported TEAEs in enzalutamide plus ADT group in Japanese patients versus the overall population. 19 Data from real-world studies indicated fatigue (32–64%), hypertension (23%), and appetite loss (19%) as the common events with enzalutamide.29,34
Safety outcomes reported in RCTs, non-RCTs, and real-world studies of advanced/metastatic PC included in this review.

N is the total number of patients in the study; n is the number of patients on a particular treatment.
AA, abiraterone acetate; ADT, androgen deprivation therapy; AE, adverse event; ALT, alanine transaminase; APA, apalutamide; AST, aspartate aminotransferase; CBT, cabazitaxel; CN, China; DAR, darolutamide; DOC, docetaxel; EA, East Asia; ENZA, enzalutamide; HK, Hong Kong; JP, Japan; 177Lu-PSMA, lutetium-177 prostate-specific membrane antigen; mCRPC, metastatic castration-resistant prostate cancer; mCSPC, metastatic castration-sensitive prostate cancer; nmCRPC, non-metastatic castration-resistant prostate cancer; MY, Malaysia; P, prednisone; PBO, placebo; PC, prostate cancer; PEMB, pembrolizumab; Q3W, every 3 weeks; RCT, randomized controlled trial; RTI, respiratory tract infection; RU, Russia; SAE, serous AE; SG, Singapore; TH, Thailand; WBC, white blood cell.
In Japanese subpopulation in LATITUDE study, AAP demonstrated favorable safety outcomes and the safety profile was similar to the overall population. Of note, TEAEs that were particularly higher among Japanese population versus global population included hypertension (51% versus 38%), hypokalemia (43% versus 24%), hepatotoxicity (37% versus 25%), and osteoporosis including osteoporosis-related fractures (23% versus 7%). 21 Safety profile in real-world studies appeared to be in line with RCTs, with hypertension, hypokalemia, fatigue, hepatotoxicity, peripheral edema being the frequent events with AAP.
Radium-223 was reported to be well tolerated in both studies. Grade ⩾3 TEAEs were reported in 39–46% of patients in both studies, with anemia being the most common (approximately 15%), followed by decreased lymphocyte count (14%), anorexia (10%), and bone pain (10%). Serious TEAEs occurred in 24–29% of patients. In one study, 7 (3%) patients had a TEAE leading to death; none were considered to be related to radium-223.27,28
The safety/toxicities observed for docetaxel in real-world studies23,24,36,37 was consistent with those known/reported in the overall population in RCTs. The most common grade 3/4 TEAEs included febrile neutropenia (13%), neutropenia (41%), granulocytopenia, leukopenia (5% each), and anemia (3%).23,36 Also, the grade 3/4 neutropenia was found to be lower with 60 mg/m2 compared with 75 mg/m2 every 3 weeks (18% versus 36%). 37 Evidence indicates that 27% to 34% of mCSPC patients and 16% of mCRPC patients were administered primary granulocyte colony stimulating factor (GCSF) prophylaxis by the treating oncologist to alleviate the risk of docetaxel-related febrile neutropenia.23,24
In the single post-marketing surveillance study of cabazitaxel, the median dose and the median dose per cycle was 20 mg/m2. The initial dose of cabazitaxel was 25 mg/m2 in only 30% of patients. The frequent adverse drug reactions (ADRs; any grade) reported with cabazitaxel were neutropenia (49%), febrile neutropenia (18%), and anemia (15%). Most ADRs occurred in cycle 1. Of note, 34% of 660 patients started cabazitaxel without prophylactic GCSF. Neutropenia and febrile neutropenia were significantly less frequent in patients who received prophylactic GCSF. 38
In four studies included for 177Lu-PSMA, the most common TEAE was hematologic toxicity, which occurred in up to 20% of patients. Grade 3/4 toxicities were grade 3 anemia (15%), grade 3 thrombocytopenia (5%), and grade 3 neutropenia (5%). Grade 4 toxicities were generally not reported in patients from India. Grade 1 xerostomia was observed in two patients (5%) after 177Lu-PSMA therapy with mean xerostomia inventory score of 23 in these patients.39–42
TEAEs frequently observed with pembrolizumab included immune-related events (15%), hypothyroidism (8%), hyperthyroidism (5%), and fracture (4%). Immune-related events (21%) and hypothyroidism (11%) were numerically greater when pembrolizumab was given with enzalutamide. The statistically significant differences (pembrolizumab plus enzalutamide versus pembrolizumab alone) were also observed for fatigue (7% versus 1%; p = 0.025) and musculoskeletal events (9% versus 1%; p = 0.007), but these events tended to be manageable. 43
Quality of studies
All included studies were assessed for quality assessment. There was low risk of bias in the RCTs in most of the assessed domains (randomization, intervention effect, missing outcome, and selection of reported results) based on Cochrane risk of bias tool. The quality score for real-world studies ranged from five to six stars, with majority of studies having five stars on the Newcastle-Ottawa Scale. A higher number of stars indicates a better quality of study (Supplementary Tables S5 and S6).
Discussion
This SLR provides comprehensive evidence on the efficacy, effectiveness and safety of therapies used in the treatment of advanced or metastatic PC in the Asian countries. The literature searches conducted allowed collection and assessment of evidence from 24 studies of various treatments (e.g., apalutamide, darolutamide, enzalutamide, AAP, radium-223, docetaxel, cabazitaxel, 177Lu-PSMA, pembrolizumab) and study designs (e.g., RCTs, non-RCTs, prospective, and retrospective observational studies) from different Asian countries. The overall evidence from this SLR suggests that Asian data on efficacy and safety of treatments from pivotal trials and real-world studies confirmed similar trend in the outcomes consistent with the findings from overall pivotal trials. The evidence from this SLR is anticipated to help physicians to enable better treatment decisions in clinical practice in Asian patients with advanced or metastatic PC.
Two studies in Japanese subpopulation (i.e., ARAMIS with darolutamide 17 and SPARTAN with apalutamide 18 ) reported evidence for patients with nmCRPC. The median MFS, OS, and PFS were not reached in ARAMIS and SPARTAN studies. The authors of ARAMIS study concluded that efficacy outcomes favored darolutamide given with ADT in patients with nmCRPC, supporting the clinical benefit of darolutamide in this patient population. Darolutamide was reported to be well tolerated; however, due to the small sample size, it was not possible to draw a firm conclusion on the safety profile between Japanese and overall ARAMIS populations. 17 Treatment with apalutamide given in combination with ADT also demonstrated favorable efficacy outcomes with comparable benefit-risk profile to the global population with nmCRPC who are at high-risk of developing metastases. 18 Overall, the evidence showed that next-generation androgen receptor inhibitors with comparable mechanism of action (apalutamide and darolutamide) remarkably prolonged the MFS in Asian patients with nmCRPC.
Three studies in Japanese subpopulation, ARCHES (enzalutamide), 19 TITAN (apalutamide), 20 and LATITUDE (AAP), 21 and one real-world study (docetaxel) in Chinese population23,24 reported evidence for patients with mCSPC, including patients with mHSPC19,24 and mHNPC21,23. Median OS and rPFS were not reached in all three studies. Enzalutamide plus ADT demonstrated clinical benefit with a tolerable safety profile in Japanese men with mHSPC, which was consistent with the overall population. 19 Treatment with apalutamide plus ADT exhibited favorable efficacy compared with ADT alone, and these findings are comparable to those in the overall population. The authors of TITAN study concluded that apalutamide plus ADT can be considered as one of the therapeutic options for a broad spectrum of mCSPC regardless of prior treatment and disease extent in Japanese patients. 20 In LATITUDE study, the addition of AAP to ADT demonstrated favorable efficacy and safety outcomes in patients with newly diagnosed, high-risk mCSPC. Survival benefits observed in the Japanese subgroup were consistent with the overall population. 21 The evidence from these studies demonstrated that next-generation androgen receptor inhibitors (apalutamide and enzalutamide) and androgen biosynthesis inhibitor (AAP) significantly delayed the time to development of radiographic disease. The real-world evidence for docetaxel suggests that chemohormonal therapy is efficacious in Chinese patients with mCSPC and comparable to the pivotal study, while the chemotherapy-related hematologic toxicities are more frequent in Chinese patients.23,24
The number of studies included for patients with mCRPC was more than the number of studies for nmCRPC or mCSPC (two RCTs, two non-RCTs, and 15 real-world studies). Treatment effects and safety of enzalutamide in East Asian patients were generally consistent with those observed in the overall study population from PREVAIL. 25 A clinical trial confirmed favorable benefit-to-risk ratio of AAP in chemotherapy-naïve men with mCRPC, consistent with global study, thus supporting use of AA in this patient population. 26 Two studies supported the use of standard radium-223 regimen for the treatment of Asian patients with CRPC and bone metastases, with well-tolerated safety profile.27,28 Evidence from real-world studies revealed that in patients with mCRPC, combination therapy of docetaxel plus prednisone, cabazitaxel with prednisone, AAP, enzalutamide provided remarkable OS and PFS benefits.23,24,30,31,34,36–38 No new safety signals were identified in Asian patients, and safety profiles appeared to be tolerable, in line with previous studies. Of note, the low dose regimen of docetaxel (60 mg/m2) has been shown to exhibit similar efficacy versus standard regimen (75 mg/m2) and an acceptable tolerability profile. 37 However, it should be interpreted cautiously as this study was an observational, non-randomized study. A considerable proportion of patients on chemotherapy with docetaxel in real-world have been reported to administer primary GCSF prophylaxis to alleviate the chemotherapy-related hematologic toxicities.23,24 Nevertheless, in contrast, a majority of patients started cabazitaxel chemotherapy with prophylactic use of GCSF. 38 Evidence suggested 177Lu-PSMA to be well-tolerated and able to produce disease control with good symptomatic and biochemical responses in the context of heavily pre-treated mCRPC with progressive disease, with low toxicity profile.39–42 Although data is limited, among selected populations of men with previously untreated mCRPC harboring PD-L1 staining, pembrolizumab added to enzalutamide treatment may significantly increase the survival benefits compared with pembrolizumab alone regardless of tumor mutation status. The safety profile for pembrolizumab in combination with enzalutamide was also reported to be manageable. 43
Of note, we did not find any study of PARP inhibitors to be eligible to be included in this SLR. However, in the PROfound study of olaparib in men with mCRPC, a subgroup analysis by region showed that HR of olaparib for progression or death in patients from Asia was 0.57 (95% CI 0.34–0.98) compared with enzalutamide or AA, and the HR in the overall population was 0.34 (95% CI 0.25–0.47). 44 Although this data provides some evidence on efficacy of PARP inhibitor in Asian patients, there is a need for more data or more studies evaluating PARP inhibitors in Asian population so that definitive conclusions can be made about their use in clinical practice.
It is important to mention other Asian studies and pivotal global trials that reported data for the treatments of interest for this SLR but were not included; either they were published beyond the cut-off period or not providing separate data for Asian patients. De Giorgi et al. very recently reported the results of enzalutamide in patients with nmCRPC by subgroup based on region (Asia versus North America) from PROSPER trial which showed that enzalutamide plus ADT reduced the risk of death independent of region, meaning that OS benefits of enzalutamide in Asian patients were comparable to that in patients from North America. 45 STAMPEDE trial demonstrated significantly higher rates of OS and treatment failure-free survival in locally advanced/metastatic PC patients treated with AAP plus ADT versus ADT alone. 46 Evidence from a clinical study in mCRPC patients from the US and Singapore also indicated that low-dose AAP (250 mg; with low-fat meal) was noninferior to standard dose AAP (1000 mg fasting) in terms of median PFS and PSA response rate. 47 The OS benefits of chemohormonal therapy (ADT plus docetaxel) than ADT alone were shown in CHAARTED trial in patients with mHSPC, with more clinical benefit observed for high-volume disease patients versus low-volume disease.48,49 A clinical study of docetaxel in Chinese patients with mCRPC demonstrated that docetaxel plus prednisone significantly prolonged the OS compared with mitoxantone plus prednisone and improved the PSA response rate and pain. 50 A real-world study showed that docetaxel-based chemotherapy is tolerable and efficacious in Chinese mCRPC patients and suggested that maximizing exposure to docetaxel-based chemotherapy followed by novel therapies would have a favorable survival impact on mCRPC patients. 51 Real-world data suggest that docetaxel is not as well tolerated in the Asian patients compared to the predominantly Caucasian patient profile in pivotal trials.23,51 AFFIRM trial demonstrated enzalutamide to significantly prolong OS among patients with mCRPC after chemotherapy (prior docetaxel exposure). 52 Very recently, PEACE-1 investigators have shown that addition of AAP to standard therapy with docetaxel plus ADT significantly improved the OS in patients with de novo mCSPC. The survival benefit observed in PEACE-1 trial with triple systemic therapy adds to the advances recently made for patients with metastatic hormone/castration-sensitive PC. 53 In ARASENS trial involving patients with mHSPC, the OS was significantly longer with triple therapy with darolutamide, ADT and docetaxel than with placebo plus ADT and docetaxel (HR 0.68; 95% CI 0.57–0.80). Approximately 35% of patients in ARASENS were Asian, in whom the survival benefit of triple therapy with darolutamide was not significant versus placebo (HR 0.838; 95% CI 0.617–1.137). 54 Overall, all these data further strengthen the evidence for therapies in Asian patients with advanced/metastatic PC to enable decision making by the clinicians. The safety profile is consistent with the drug experience in terms of types of AEs. However, the rates of some AEs could be higher in Asian patients warranting further research on drug dosing and systemic exposure, etc., specially in Asian patients. Given that higher incidence of rash observed with apalutamide in Asian patients and increased toxicities with standard dose docetaxel chemotherapy, these treatment options should be considered carefully in the context of triple therapy for patients with mCSPC.
This SLR has a few limitations. Firstly, we included studies that were published in English. This may be considered a source of bias although most scientific articles are published in English. Some of the studies were observational studies which generally lack methodological rigor to make comparisons. 55 Nevertheless, they provide valuable insights on treatment practices and patient characteristics among patients in the real-world settings and are considered to form a bridge from the results of RCTs to routine clinical practice. 55 Two RCTs (PEACE-1 54 and ARASENS 55 ) reporting efficacy of triple systemic therapies in patients with mCSPC were not included as these were published after the data collection period for this SLR (May 2021); however, their efficacies have been discussed briefly above. Finally, the global RCTs that reported results for Asian population have limited number of Asian patients (5–10% of the total trial population) and the results should be interpreted with caution. Nonetheless, it is noteworthy to mention that some recent trials such as ARASENS had up to 35% of patients from Asia. 55 Thus, if this trend of including more Asian patients to have more diverse population in pivotal trials continues, it will increase the generalizability of findings to the diverse populations and their applicability in clinical practice. Despite these, we still feel that more studies with patients specifically from Asia are needed.
Conclusions
This SLR collating data from 24 studies on patients with advanced/metastatic PC, including nmCRPC, mCSPC, and mCRPC, indicated that evidence from Asian patients is relatively more for AAP, enzalutamide, and 177Lu-PSMA than other advanced PC therapies. Although studies in Asian patients are limited, current analysis of Asian data from pivotal trials and real-world studies confirmed a similar trend in outcomes consistent with the findings from pivotal trials. The evidence is reassuring and may increase the confidence of Asian clinicians in applying clinical trial data related to PC therapies on their patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221131525 – Supplemental material for Efficacy and safety of therapies for advanced prostate cancer in Asia: Evidence from a systematic literature review
Supplemental material, sj-docx-1-tam-10.1177_17588359221131525 for Efficacy and safety of therapies for advanced prostate cancer in Asia: Evidence from a systematic literature review by Marniza Saad, Rainy Umbas, Edmund Chiong and Ravindran Kanesvaran in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
