Abstract
Objectives
We conducted a systematic review and meta-analysis to evaluate the clinical effectiveness and safety of riluzole to treat neurodegenerative dyskinesia in patients, using the Cochrane collaboration guidelines.
Methods
We searched databases including Medline, Embase, and Cochrane without any language filters. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement was used as a guideline, and the study protocol was registered in PROSPERO (CRD42022354627).
Results
Eleven studies involving 1376 patients were included. There was a significant overall effect of riluzole on changes in motor function scores. However, the level of heterogeneity was I2 = 74%. In the subgroup analyses, there were no significant effects of riluzole on motor scores in hereditary ataxia, Parkinson’s disease, or Huntington’s disease. In the sensitivity analysis, there were no significant effects of riluzole on motor function scores. Furthermore, there were no significant differences in adverse events between the riluzole and placebo groups.
Conclusions
Although riluzole may not have significant efficacy for improving motor function in neurodegenerative dyskinesia syndromes compared with placebo, it seems to have an acceptable safety profile. Moreover, it may be effective for hereditary ataxia syndromes, although there was a relatively small effect size and limited quality of evidence.
Introduction
As a manifestation of an underlying neurodegenerative disease or secondary to a widespread neurological or systemic disease, neurodegenerative movement disorders can occur as primary (idiopathic) or genetic disorders with neuronal loss or glial cell proliferation. 1 Clinical manifestations include excessive abnormal involuntary movements or hypokinesia (i.e., motor bradykinesia). 2 Movement disorders include Parkinson’s disease (PD), parkinsonism-plus syndromes (e.g., progressive supranuclear palsy and multiple system atrophy), Huntington’s disease (HD), hereditary ataxia, and Tourette syndrome. 3 However, non-motor symptoms are also common in the course of PD, HD, and hereditary ataxia. Neurodegenerative diseases are relatively rare and have a lack of available treatments, with no complete cure currently available. 4 Although neurodegenerative diseases are a key research topic in the field of neuroscience and many investigations into a variety of interventions have been conducted, there has been relatively little success. For example, studies have indicated that coenzyme Q10, idebenone, and mitoquinone are not promising for the prevention and delay of disease progression in neurodegenerative movement disorders. 5 Current clinical treatments are therefore mostly focused on the relief of specific symptoms, such as hypokinesia, hyperkinesia, or ataxia.
Levodopa has traditionally been the drug of choice for both PD and complications of spinocerebellar ataxia; however, halting disease progression and late-onset hyperkinesia remain important problems.6,7 Riluzole is a benzothiazole compound with anti-glutamatergic effects. It is currently the only drug approved by the US Food and Drug Administration for treating amyotrophic lateral sclerosis. Recently, riluzole has also been reported to interfere with growth signaling pathways, calcium homeostasis, glutathione synthesis, reactive oxygen species production, DNA integrity, and autophagy and apoptosis pathways. 8 Moreover, neuroprotective effects of riluzole have been demonstrated in experimental models of PD, spinocerebellar ataxia, and HD.9–11 However, clinical trials into the efficacy of riluzole in neurodegenerative movement disorders have provided non-conclusive results. For example, a few randomized controlled trials (RCTs) with relatively small sample sizes have reported good efficacy of riluzole for improving motor function in patients with PD, HD, and hereditary ataxia.12–14 By contrast, a recent high-quality RCT reported a lack of efficacy of riluzole in hereditary ataxia compared with placebo. 15 This lack of clear evidence for its efficacy is one of the major reasons why riluzole is not used to treat neurodegenerative movement disorders in many countries.
In the present study, we thus aimed to systematically evaluate the motor function effects of riluzole in neurodegenerative movement disorders, to assess its efficacy and safety.
Methods
The present study was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 16 The protocol of this study was registered in PROSPERO (CRD42022354627).
Literature search
Literature searches were performed across PubMed, Embase, Cochrane Library, Web of Science, and China National Knowledge Infrastructure databases using the following phrases: (“riluzole” OR “2-amino-6-trifluoromethoxybenzothiazole” OR “Rilutek” OR “RP54274” OR “PK26124”) AND (“dyskinesia” OR “movement disorders” OR “ataxia*” OR “spinocerebellar ataxia” OR “spinocerebellar degeneration” OR “spinocerebellar atrophy” OR “SCA” OR “Machado–Joseph disease” OR “ataxia telangiectasia” OR “Parkinson’s disease” OR “Parkinson” OR “parkinsonism” OR “multiple system atrophy” OR “progressive supranuclear palsy” OR “corticobasal degeneration” OR “Huntington’s disease” OR “tic disorders”). No language or regional limitations were imposed.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) studies on PD, HD, hereditary ataxia, or tic disorders; (2) study design was an RCT comparing the efficacy of riluzole with a placebo; and (3) studies had the full text and complete data available.
The exclusion criteria were as follows: (1) studies that were not RCTs or were low-quality RCTs; (2) studies that were not performed on humans; (3) studies that were published as letters, editorials, expert opinions, reviews, or case reports; and (4) duplicate publications.
Data extraction and quantitative synthesis
All relevant literature was reviewed by all four reviewers (MQQ, RW, HLF, and YNS). A consensus was reached between MQQ, RW, and HLF regarding the included studies. The search deadline was 20 December 2023. We also contacted the relevant corresponding authors to confirm the extracted data or to request missing data.
The primary observation was changes in motor function scores in patients with PD, progressive supranuclear palsy, HD, hereditary ataxia, and Tourette syndrome. The motor function scores of dyskinesia syndromes were measured using the Unified PD Rating Scale (UPDRS) motor, Unified HD Rating Scale (UHDRS) motor, Short Motor Disability Scale (SDMS) Index score, International Cooperative Ataxia Rating Scale (ICARS), Scale for the Assessment and Rating of Ataxia (SARA), and Yale Global Tic Severity Scale (YGTSS) motor. In addition, the “Revised Cochrane risk-of-bias tool for randomized trials” was used to assess the methodological quality of all included RCTs. 17
Statistical analysis
The meta-analysis was performed using Review Manager 5.3 (The Cochrane Collaboration, Copenhagen, Denmark). Heterogeneity was tested using Cochran’s Q test and I2. p < 0.05 or I2 ≥ 75% indicated significant heterogeneity, and a random-effects model was adopted; otherwise, a fixed-effects model was used. For continuous data, we calculated the mean differences with 95% confidence intervals (CIs) or used standardized mean differences (SMDs) if studies did not use the same scales to measure outcomes. For dichotomous data, we calculated the risk ratios with 95% CIs. If a trial (or group within a trial) reported no adverse events or dropouts, we calculated the risk differences instead of the odds ratios.
Funnel plots were inspected for potential asymmetry to check for publication bias, and a sensitivity analysis was performed if necessary. We also performed subgroup analyses based on the included conditions.
Results
Search results
Of the 236 records that were identified, 61 were excluded as duplicates and a further 94 were excluded as irrelevant. After a full-text review of the remaining 81 studies, we excluded 16 reviews or conferences, 21 data mismatches, nine duplicate cases, and 24 other types of studies (e.g., case reports). Finally, we included 11 studies12–15,18–24 involving 1376 participants (Figure 1).

PRISMA flowchart of study selection and reasons for exclusion. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Characteristics of included studies
Of the 11 studies, 10 were RCTs and 1 was a random crossover trial. All studies were written in English; half of the studies were conducted in Europe and the other half in America. Changes in motor scores after treatment with riluzole versus placebo were recorded and compared. Patients with a diagnosis of PD were scored using the UPDRS13,24 motor portion, and patients with HD were evaluated using the UHDRS21–23 motor part and SDMS 14 Index score. Patients with ataxia were assessed using ICARS 19 and SARA12,15 scores. Tic disorders were scored using the YGTSS 17 motor score. The characteristics of the included studies are shown in Table 1.
Summary of the included studies.

HD, Huntington’s disease; MSA, multiple system atrophy; PD, Parkinson’s disease; PSP, progressive supranuclear palsy; RCT, randomized controlled trial.
Risk of bias in included studies
For the RCTs, the risk-of-bias tool was used; all 10 RCTs were referred to as “double-blind” and described the method of generating a random sequence. Furthermore, all trials were reported “randomly.” Nine trials were fully reported in the results, with the primary outcome listed in the published protocol. In the 10 RCTs, we did not identify any other potential sources of bias. A detailed assessment is shown in Figure 2.

Risk of bias items (presented as percentages across all included trials). ICARS, International Cooperative Ataxia Rating Scale; SARA, Scale for the Assessment and Rating of Ataxia; SMDS, Short Motor Disability Scale; UHDRS, Unified Huntington’s Disease Rating Scale; UPDRS, Unified Parkinson’s Disease Rating Scale; YGTSS, Yale Global Tic Severity Scale.
Efficacy outcomes
We included 11 studies involving 1376 patients in which motor function in dyskinesia syndromes were compared between treatment with riluzole and placebo. The change in motor scores was significantly different between the two groups (SMD −0.34, 95% CI −0.64 to −0.04, p = 0.03; level of heterogeneity χ2 = 38.02, df = 10, p < 0.0001, I2 = 74%) (Figure 3).

Changes in motor scores after treatment with riluzole versus placebo. CI, confidence interval; SD, standard deviation.
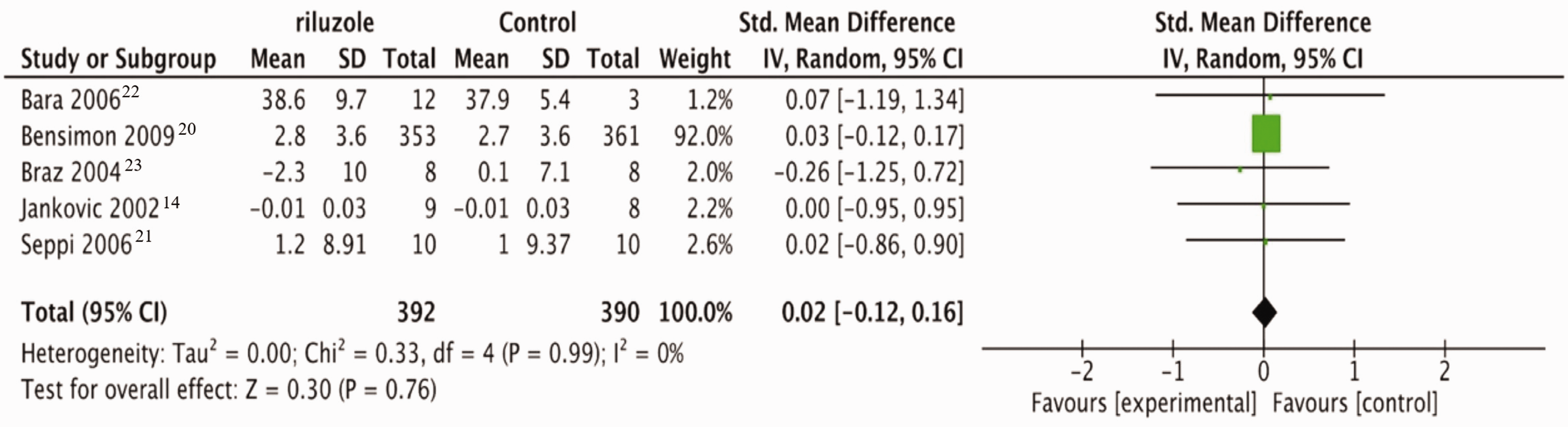
We then performed subgroup analyses on the included conditions: PD, HD, and hereditary ataxia. For PD and its complications (i.e., progressive supranuclear palsy and multiple system atrophy), two RCTs comprising 734 patients with multiple system atrophy or progressive supranuclear palsy and three RCTs comprising 48 patients with PD were synthesized (three RCTs were assessed using UPDRS motor, one using SMDS, and one using the Movement Time Index score). The change in motor scores did not significantly differ between the riluzole and placebo groups (SMD 0.02, 95% CI −0.12 to 0.16; level of heterogeneity χ2 = 0.33, df = 4, I2 = 0%) (Figure 4).

Changes in motor scores in Parkinson’s disease after treatment with riluzole versus placebo. CI, confidence interval; SD, standard deviation.
For HD, two RCTs comprising 442 patients were included (both were assessed using the UHDRS). The change in motor scores did not significantly differ between the riluzole and placebo groups (SMD −0.26, 95% CI −0.81 to 0.30; level of heterogeneity χ2 = 3.94, df = 1, I2 = 75%) (Figure 5).

Changes in motor scores in Huntington’s disease after treatment with riluzole versus placebo. CI, confidence interval; SD, standard deviation.
For hereditary ataxia, three RCTs comprising 137 patients were included (two RCTs were assessed using SARA and one was evaluated using ICARS). Although there was a relatively large effect size compared with the other subgroup analyses, the change in motor scores did not significantly differ between the riluzole and placebo groups (SMD −0.91, 95% CI −1.95 to 0.12; level of heterogeneity χ2 = 15.91, df = 2, p = 0.0004, I2 = 87%) (Figure 6).

Changes in motor scores in hereditary ataxia after treatment with riluzole versus placebo. CI, confidence interval; SD, standard deviation.
Adverse events
Adverse effects were reported in 10 of the 11 reports. However, most studies reported no differences in the incidence of adverse events between the riluzole and placebo groups. The results of the random effects model (I2 = 33%) indicated no significant differences between the riluzole and placebo groups (odds ratio 1.26, 95% CI 0.81 to 1.94) (Figure 7).
Numbers of participants with adverse events. CI, confidence interval.
Publication bias and sensitivity analysis
The funnel plot in Figure 8 shows that all 11 RCTs were predominantly distributed in the upper part. It is thus reasonable to infer that the included studies were free of publication bias and other relevant biases.

Funnel plot. SMD, standardized mean difference.
In the sensitivity analysis, after removing the paper by Ristori et al., 19 the change in motor scores did not significantly differ between the riluzole and placebo groups (SMD −0.19, 95% CI −0.42 to 0.04; level of heterogeneity χ2 = 18.97, df = 9, p = 0.03, I2 = 53%) (Figure 9).

Sensitivity analyses for treatment with riluzole versus placebo after removing Ristori et al. 19 CI, confidence interval; SD, standard deviation.
Discussion
The present study comprised a systematic and up-to-date overview of recent randomized, double-blind, placebo-controlled studies on the efficacy of riluzole for treating neurodegenerative movement disorders.
At present, the exact mechanism of riluzole remains unknown. Earlier studies have demonstrated that riluzole may have a neuroprotective effect by increasing the synaptic uptake of glutamate while decreasing its release and preventing excitotoxicity; it also inhibits voltage-gated calcium channels to enhance potassium channels, thereby modulating other excitatory neurotransmitters. 11 Notably, in a mouse model of spinocerebellar ataxia type 3, treatment with riluzole for 10 months reportedly leads to decreased motor performance. 11 By contrast, the neuroprotective effects of riluzole have been observed in experimental models of PD, multiple system atrophy, and HD.9,10,25 However, although preclinical studies have supported the use of riluzole, the clinical results of the present meta-analysis do not support the use of riluzole for movement disorders as a whole.
In the included studies, Coarelli et al. 15 reported no differences between riluzole and placebo in patients with spinocerebellar ataxia type 2; this finding was different from the studies of Romano et al. 12 and Ristori et al., 19 which included ataxia syndromes. In general, the results of the present analysis indicate that riluzole does not differ from placebo in improving motor function in the included conditions. Although the SMD of the overall analysis favored riluzole in a significant manner, the meta-analytical effect size seemed to be almost completely driven by two studies (Romano et al. 12 and Ristori et al. 19 ) that included ataxia syndromes; for all other conditions, no significant differences were observed in our subgroup analyses. It is also worth noting that in the studies by Romano et al. 12 and Ristori et al., 19 the cohorts were very heterogeneous; they included hereditary and non-hereditary patients and even pre-ataxic carriers (for which SARA progression is not linear, as it is for ataxic stages). 26 After excluding the study by Ristori et al. 19 in our sensitivity analysis, there was a substantial decrease in overall heterogeneity. Furthermore, for the overall analysis, 74% of the inconsistency was present, whereas for the subgroup analyses, the I2 was 0% for PD, progressive supranuclear palsy, and multiple system atrophy, and was 87% for ataxia syndromes and 75% for HD. Even if we hypothetically accepted the generalization of the results of the overall analysis to all included motor disorders, without acknowledging the evident differences in effects between them, the SMD effect size was limited; an SMD between 0.2 and 0.4 is generally considered a small effect, and the 95% CI spanned from a moderate to a null effect.
There are some limitations to our study. The UPDRS motor, UHDRS motor, ICARS, and SARA are the most commonly used scales for assessing motor function in PD, HD, and hereditary ataxia, and higher scores imply more severe movement disorders. However, differences in data between the scales may also lead to differences in the results of the analyses and individual studies, meaning that the heterogeneity of the findings in the present study was moderate. More homogeneous studies should be included in future investigations.
Given that there are currently no effective treatments for motor function in neurodegenerative movement disorders, the application of riluzole offers new possibilities for the treatment of PD, ataxia syndromes, Tourette syndrome, and HD. Alongside the promising results of this drug in animal studies and clinical trials, it is hoped that more—and better quality—RCT studies of riluzole for neurodegenerative movement disorders will be conducted, to further elucidate the associations between the two and any related mechanisms. A network meta-analysis may also be applied in the future to compare multiple factors, to better determine the efficacy of riluzole.
Conclusion
Compared with placebo, riluzole may not have significant efficacy for treating motor function in dyskinesia syndromes of central nervous system disorders. However, it may be effective and safe for hereditary ataxia syndromes, although there was a relatively small effect size and a limited quality of evidence.
Supplemental Material
sj-xlsx-1-imr-10.1177_03000605241276489 - Supplemental material for Efficacy and safety of riluzole for treating motor function in rare dyskinesia syndromes: a systematic review with meta-analysis
Supplemental material, sj-xlsx-1-imr-10.1177_03000605241276489 for Efficacy and safety of riluzole for treating motor function in rare dyskinesia syndromes: a systematic review with meta-analysis by Rui Wang, Hongli Fang, Yenan Shen and Mengqiu Qiu in Journal of International Medical Research
Footnotes
Author contributions
Conception and design of the study: RW and HLF. Literature search; study selection and assessment; data extraction and analyses: MQQ, YNS, and RW. Drafting the manuscript; figures and tables: RW, HLF, and MQQ.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by a grant from the Zhejiang Provincial Health Department New Technology Project Foundation of China (No. 2021PY060).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
