Abstract
Purpose:
To establish a risk classification of de novo metastatic nasopharyngeal carcinoma (mNPC) patients based on 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET-CT) radiomics parameters to identify suitable candidates for locoregional radiotherapy (LRRT).
Methods:
In all, 586 de novo mNPC patients who underwent 18F-FDG PET-CT prior to palliative chemotherapy (PCT) were involved. A Cox regression model was performed to identify prognostic factors for overall survival (OS). Candidate PET-CT parameters were incorporated into the PET-CT parameter score (PPS). Recursive partitioning analysis (RPA) was applied to construct a risk stratification system.
Results:
Multivariate Cox regression analyses revealed that total lesion glycolysis of locoregional lesions (LRL-TLG), the number of bone metastases (BMs), metabolic tumor volume of distant soft tissue metastases (DSTM-MTV), pretreatment Epstein–Barr virus DNA (EBV DNA), and liver involvement were independent prognosticators for OS. The number of BMs, LRL-TLG, and DSTM-MTV were incorporated as the PPS. Eligible patients were divided into three stages by the RPA-risk stratification model: M1a (low risk, PPSlow + no liver involvement), M1b (intermediate risk, PPSlow + liver involvement, PPShigh + low EBV DNA), and M1c (high risk, PPShigh + high EBV DNA). PCT followed by LRRT displayed favorable OS rates compared to PCT alone in M1a patients (p < 0.001). No significant survival difference was observed between PCT plus LRRT and PCT alone in M1b and M1c patients (p > 0.05).
Conclusions:
The PPS-based RPA stratification model could identify suitable candidates for LRRT. Patients with stage M1a disease could benefit from LRRT.
Background
Nasopharyngeal carcinoma (NPC) is an epithelial malignancy endemic in east and southeast Asia with distinct epidemiology, biological characteristics, and treatment patterns.1–3 Compared with other head and neck squamous cell carcinomas, NPC is more predisposed to metastasize to distant sites, with an incidence of approximately 10% in newly diagnosed NPC patients, 4 and distant metastases have a tendency to be more aggressive. Even when adopting systematic chemotherapy, many patients still have uncontrolled locoregional disease, leading to disease progression. The median survival for metastatic NPC (mNPC) patients who receive palliative chemotherapy (PCT) is entirely 10–15 months.5,6 The combination of PCT and local treatment has been shown to be associated with increased survival rates in mNPC patients by previous literature, with a 5-year overall survival (OS) rate of approximately 20%. 7 However, the clinical outcomes of mNPC patients are heterogeneous, and the survival duration ranges from several months to more than 10 years.8,9 Only a subset of patients could benefit from locoregional radiotherapy (LRRT) and local treatment of metastatic lesions.10–12 Growing studies have shown that LRRT is related to increased survival and support strategies incorporating LRRT with PCT for mNPC.13–15 According to the current American Joint Committee on Cancer (AJCC) TNM staging system, mNPC patients are grouped into M1 without detailed subgroups, covering patients from potentially curable to incurable. (Tumor Node Metastasis) It is unclear which patients should be selected to receive LRRT utilizing the TNM staging system. Therefore, risk stratification and screening of mNPC patients for individualized treatment are definitely necessary.
18F-Fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET-CT), a noninvasive imaging modality that can distinguish tumors from normal tissue by detecting enhanced glycolysis, is widely applied in the diagnosis, staging, and prognostic evaluation of tumors.16–18 The semiquantitative metabolic parameters calculated by 18F-FDG PET-CT, such as the standardized uptake value (SUV), total lesion glycolysis (TLG), and volumetric parameter metabolic tumor volume (MTV), which represent intralesional heterogeneity in metabolism, have been reported as prognostic factors correlated with clinical outcomes in various malignancies.19,20 PET-CT radiomics is considered as a promising method for guiding personalized cancer treatment.21,22 Nevertheless, the clinical significance of these structural and textural indices evaluated by PET-CT remains unclear for patients with de novo metastatic NPC (mNPC).
Liver involvement has been demonstrated to contribute to detrimental clinical outcomes, and mNPC patients with liver involvement suffer worse survival than those with bone and lung involvement.8,23 Epstein–Barr virus deoxyribonucleic acid (EBV DNA) is another powerful prognostic factor of NPC, and quantification of plasma EBV DNA has been verified as an effective indicator of tumor burden and widely applied in screening, risk stratification, monitoring, and prognostic prediction of NPC patients.24,25 However, relevant studies regarding 18F-FDG PET-CT parameters, plasma EBV DNA levels, and anatomic factors in de novo mNPC are lacking. Thus, we conducted this retrospective study to evaluate the metabolic parameters on baseline 18F-FDG PET-CT correlated with clinical outcome for de novo mNPC patients and developed a risk stratification model based on 18F-FDG PET-CT parameters to identify suitable candidates for LRRT.
Methods
Study population
From March 2011 to October 2019, a total of 586 primary NPC patients diagnosed with distant metastases at Sun Yat-Sen University Cancer Center (SYSUCC) were enrolled in this study. Eligibility criteria were as follows: (1) histologically confirmed World Health Organization type II–III NPC; (2) newly diagnosed with distant metastases; (3) no previous or synchronous malignant tumors; (4) Karnofsky performance score ⩾80; (5) available baseline clinical and radiologic evaluation data; and (6) receipt of PCT with or without LRRT. Clinical data of eligible patients were reviewed and reclassified. All recruited patients were restaged according to the 8th edition of the AJCC/Union for International Cancer Control staging system. This study was approved by the Institutional Review Board and the Ethics Committee of the SYSUCC (approval number: B-2022-264-01). Flow chart of patient selection is shown in Figure 1.
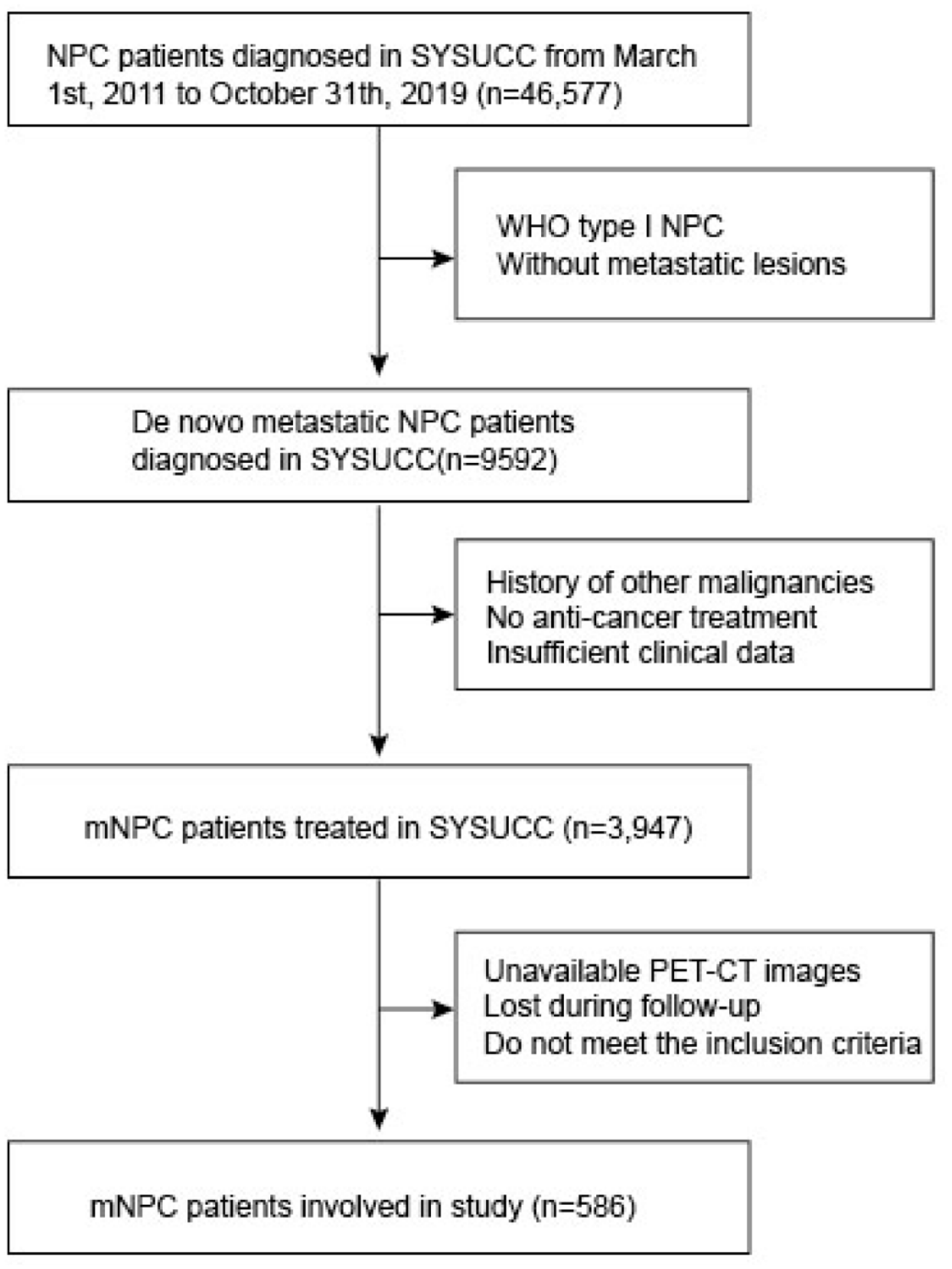
Flow chart of patient selection.
18F-FDG PET-CT procedure
Whole-body 18F-FDG PET-CT scans were performed within 1 week before treatment using integrated PET-CT scanners (Discovery ST, GE Healthcare, Waukesha, WI, USA; or Biograph mCT, Siemens Healthcare, Henkestr, Germany). Patients fasted at least 6 h before the 18F-FDG PET-CT scan. Serum glucose levels were measured prior to tracer injection (target serum glucose ⩽11.1 mmol/l). A single-time-point PET scan (skull to mid-thigh) was performed with two-dimensional (2D) mode in Discovery ST or three-dimensional (3D) mode in Biograph mCT 60–90 min after the 18F-FDG injection (5.55 Mbq/kg for Discovery ST system and 3.7 Mbq/kg for Biograph mCT suytem), with 6–8 bed positions within a scan time of 1.5–3 min for each position. The low-dose CT scan was executed prior to PET scans using the following parameters: automatic tube current modulation, tube voltage 140 kV, collimation 16 × 1.25 mm, slice thickness 3.75 mm for the Discovery ST or tube current 80–200 mAs, voltage 120 kV, collimation 32 × 1.25 mm, and slice thickness 3 mm for the Biograph mCT. Attenuation-corrected PET images and CT images without contrast enhancement were reconstructed using an ordered-subset expectation maximization iterative imaging reconstruction algorithm, with a slice thickness of 3.25 mm (2D) or with 2 mm (3D). Image interpretation was accomplished by two nuclear medicine physicians experienced in PET-CT diagnosis in transaxial, coronal, and sagittal views.
Tumor delineation and quantification
The volumes of interest (VOIs) of PET images were delineated semiautomatically using Syngo.via software. All tumor lesions, including the primary tumor of the nasopharynx, neck lymph nodes, and distant metastases, were delineated in axial, coronal, and sagittal 18F-FDG PET-CT images (Supplemental Figure S1). An SUV of 2.5 was adopted as the threshold for VOI analysis, in accordance with previous studies.26,27 SUVmax was defined by the voxel with the highest activity within the VOI. SUVpeak represented the highest quantitative value of SUV in a 12-mm diameter sphere of VOI. SUVmean was the mean activity in the tumor VOI, while TLG was defined as the SUVmean plus MTV. The number of metastases was acquired from the 18F-FDG PET-CT images, and the PET-CT-derived number of metastases was included in the analyses. In our study, we categorized all lesions and calculated 18F-FDG PET-CT parameters of each patient into two subgroups: locoregional lesions (LRLs, including the primary tumor of the nasopharynx and neck lymph nodes) and distant metastases. Distant metastases were further divided into two groups, bone metastasis (BM) and distant soft tissue metastases (DSTMs, including lung, liver, mediastinal lymph nodes), due to the diversity of metastatic sites. Radiomics indices were evaluated for LRLs and the three largest lesions in each metastatic organ. Judgment of skeletal lesions was based on the metabolic imaging and the morphological information from CT images of the PET-CT examination. Bony lesions with increased SUVmax (>2.5) were localized on CT images and adequately assessed to determine if BM was present. Further tests such as magnetic resonance imaging (MRI) or biopsy were considered if there was any doubt for the diagnosis of BM. BMs in our study were mainly osteolytic or mixed lesions, and a proportion of BMs were small tumors without soft tissue masses ⩾10 mm, which were designates as unmeasurable lesions by RECIST 1.1. 28 Considering TLG and MTV might not be accurate in reflecting the real metabolism of these small BMs and the evaluation indices of BMs in the study should be consistent, we only assessed three indices (SUVmax, SUVpeak, and number of BM) for BMs.
Baseline evaluation and treatment
Pretreatment evaluation of patients included medical history, physical examinations, hematologic and serum biochemical profiles, plasma EBV DNA, nasopharynx histopathology, and 18F-FDG PET-CT and MRI of the nasopharynx and neck. Real-time quantitative polymerase chain reaction was used to measure the level of pretreatment EBV DNA. All the patients in the cohort received PCT. Combined PCT regimens applied in this study included gemcitabine plus cisplatin [GP; cisplatin 20–30 mg/m2 intravenously (IV) on days 1–3 plus gemcitabine 800–1000 mg/m2 IV on days 1 and 8], TP (docetaxel 75 mg/m2 IV on day 1 plus cisplatin 20–25 mg/m2 IV on days 1–3), TPF (docetaxel 60 mg/m2 IV on day 1 plus cisplatin 20–25 mg/m2 IV on days 13 plus 5-fluorouracil 500–800 mg/m2 continuous IV infusion for 24 h on days 1–5), and PF (cisplatin 20–30 mg/m2 IV on days 1–3 plus 5-fluorouracil 800–1000 mg/m2 continuous IV infusion for 24 h on days 1–5). Intensity-modulated radiotherapy was performed in patients who received LRRT. The prescribed doses were 66–72 Gy to gross tumor volume of nasopharynx (GTVnx; the primary NPC tumor and retropharyngeal nodes) and 60–70 Gy to gross tumor volume of lymph node (GTVnd; gross cervical lymph nodes), according to a previous study. 29
Endpoints, follow-up, and tumor response assessment
OS was the primary endpoint of our study, which was defined as the date from diagnosis to the date of death from any cause or to the end of the last follow-up. All patients were followed every 3 months during the first year, every 6 months from the 2nd to 5th years, and annually thereafter. Clinical examination, nasopharyngeal endoscopy, MRI of the nasopharynx and the neck, chest CT and abdominal ultrasonography, and 18F-FDG PET-CT were routinely performed or upon clinical indication of tumor progression. Adverse events (AEs) were evaluated according to Common Terminology Criteria for Adverse Events version 4.0.
Statistical analysis
The optimal threshold analyses of the candidate variables, including EBV DNA, number of metastases, and 18F-FDG PET-CT metabolic parameters (SUVs, TLG and MTV), were conducted by receiver operating characteristic (ROC) curves, and original parameters were converted to dichotomous variables. The chi-square test or Fisher’s exact test was used to compare the categorical data. The survival analyses were performed with the Kaplan–Meier method, and the log-rank test was used to calculate the p values for the intergroup differences. Univariate and multivariate Cox proportional hazards regression models were used to identify significant characteristics for OS and to calculate hazard ratios (HRs) with 95% confidence intervals (CIs). Significant 18F-FDG PET-CT parameters were incorporated into an 18F-FDG PET-CT parameter score (PPS). Each binary variable in the PPS system was assigned a score value; that is, variables with values less than the cutoff value were assigned 1, and those with values greater than the cutoff value were assigned 2. Then, total scores of PPS were calculated. The entire group of patients was divided into two groups according to the cutoff value of the total PPS score. Recursive partitioning analysis (RPA) was used to derive a risk stratification system incorporating PPS, pretreatment EBV DNA, and liver involvement with respect to OS. Inverse probability of treatment weighting (IPTW) was applied to control for differences in baseline characteristics, including age, sex, body mass index (BMI), T stage, N stage, and chemotherapy cycle among the different subgroups, and IPTW-adjusted survival analyses were performed to estimate OS for patients in different subgroups. Statistical significance was defined as p < 0.05 based on two-sided tests. SPSS version 25.0 (IBM Corporation, Chicago, IL, USA) and R 4.0.1 (The R Foundation for Statistical Computing, Vienna, Austria) were used to perform all statistical analyses.
Results
Patient characteristics and OS
A total of 586 mNPC patients (496 males, 84.6% and 90 females, 15.4%) between March 2011 and October 2019 were recruited for this study. The median age of the cohort was 46 years (range, 11–82 years), and all the patients were pathologically diagnosed with nonkeratinizing undifferentiated NPC. With a median follow-up time of 33 months, the 1- and 3-year OS rates of the entire cohort were 76% and 56%, respectively. The most common site of metastasis was bone (288, 49.1%), followed by liver (166, 28.3%) and lung (110, 18.8%). In all, 203 patients (34.6%) exhibited multiple sites of metastases at the time of diagnosis. The median cycle of PCT in the whole cohort was four. In total, 348 patients (59.4%) received LRRT after PCT. The median dose to GTVnx and GTVnd were 70 Gy and 66 Gy, respectively. Detailed clinicopathologic characteristics of patients are listed in Table 1.
Patient demographics and clinical characteristics.

Data are presented as number (percentage) unless stated otherwise.
BM, bone metastases; BMI, body mass index; DSTM, distant soft tissue metastases; EBV, Epstein-Barr virus; GP, gemcitabine and cisplatin; GTVnd, gross tumor volume of lymph node; GTVnx, gross tumor volume of nasopharynx; LRL, loco-regional lesion; LRRT, locoregional radiotherapy; M1a (low risk, PPSlow + no liver involvement); M1b (intermediate risk, PPSlow + liver involvement, PPShigh + low EBV DNA); M1c (high risk, PPShigh + high EBV DNA); MTV, metabolically tumor volume; No., number; PPS, 18F-FDG PET-CT parameter score; SUV, standard uptake value; TLG, total lesion glycolysis.
p Value was calculated with unadjusted χ² test or Fisher’s exact test.
Identification of independent prognostic indicators for OS
18F-FDG PET-CT metabolic parameters of tumors (SUVs, TLG, and MTV) and clinical factors (BMI and EBV DNA) were converted into dichotomous variables using optimal cutoff values obtained by ROC analysis (Table 1). Using univariate COX analyses, LRL-SUVmax, LRL-TLG, and LRL-MTV were significantly correlated with OS (p < 0.05). In terms of BM, higher levels of SUVmax and SUVpeak were related to worse clinical outcomes (p < 0.05). DSTM-SUVmax, DSTM-SUVpeak, DSTM-SUVmean, DSTM-TLG, and DSTM-MTV were significantly associated with OS (p < 0.05). In addition, the 18F-FDG PET-CT derived number of BMs and number of DSTMs were also significantly correlated with OS (p < 0.05). Pre-treatment EBV DNA was identified as a significant predictor of OS (p < 0.001). Patients with liver involvement had significantly worse survivals than those without liver involvement (p < 0.001). However, bone involvement and lung involvement were not significantly correlated with OS (p > 0.05). No significant difference was demonstrated in the OS rate between the patients received GP regimen and non-GP regimens (p = 0.289). However, the group of patients received PCT more than four cycles possessed a better OS than those received less than four cycles (p < 0.001). There was no significant difference of OS between the patients received more than 70 Gy to GTVnx and less than 70 Gy, and similar result was observed in the groups received more than 66 Gy to GTVnd and less than 66 Gy (Table 2). Furthermore, we calculated the number of metastatic organs and number of metastatic lesions, and considered them in the Cox proportional hazards model, as substitutes for number of BMs and number of DSTMs. Multivariable analysis showed that number of metastatic lesions was an independent prognostic factor for OS. The results of univariable analysis and multivariable analysis are presented in Supplemental Table S1.
Univariable and multivariable analyses in the cohort.

BM, bone metastases; BMI, body mass index; CI, confidence interval; DSTM, distant soft tissue metastases; EBV, Epstein-Barr virus; GP, gemcitabine and cisplatin; GTVnd, gross tumor volume of lymph node; GTVnx, gross tumor volume of nasopharynx; HR, hazard ratio; LRL, loco-regional lesion; LRRT, locoregional radiotherapy; MTV, metabolically tumor volume; SUV, standard uptake value; TLG, total lesion glycolysis.
Establishment of PPS
Based on univariate COX analyses, 12 parameters derived from 18F-FDG PET-CT: number of BMs, number of DSTMs, metabolic parameters of LRLs (LRL-SUVmax, LRL-TLG, and LRL-MTV), metabolic parameters of BMs (BM-SUVmax and BM-SUVpeak), metabolic parameters of DSTMs (DSTM-SUVmax, DSTM-SUVpeak, DSTM-SUVmean, DSTM-TLG, and DSTM-MTV) were subjected to multivariate Cox regression analysis. Using backward method, three variables including number of BMs, LRL-TLG, and DMST-MTV remained to be prognostic factors of OS ultimately and were used for the establishment of PPS (Figure 2 and Table 2). Individuals with BMs numbers ⩽3, LRL-TLG ⩽367.9, or DSTM-MTV ⩽18.4 cm3 were assigned the value of 1, while individuals with BMs numbers >3, LRL-TLG >367.9, or DSTM-MTV >18.4 cm3 were assigned the value of 2. The total PPS score for each patient was obtained by adding the three scores. Using the median PPS score of 4 (range 3–6), all the patients in the cohort were categorized into PPShigh (PPS score > 4) and PPSlow (PPS score ⩽ 4) groups. A significant difference in OS was observed between the two groups (p < 0.001, HR: 4.094, 95% CI: 3.042–5.509).

Process of PPS building.
RPA-generated risk stratifications and subcategorization of mNPC
RPA algorithm was performed applying PPS, pre-treatment EBV DNA, and liver involvement. Ultimately, the whole cohort was categorized into three risk groups with divergent survivals: M1a (low risk, PPSlow, and no liver involvement) group, M1b (intermediate risk, PPSlow and liver involvement, PPShigh and EBV <13,050 copies/ml) group, and M1c (high risk, PPShigh and EBV ⩾13,050 copies/ml) group (Figures 3 and 4). The corresponding 3-year OS rates of the three groups were 72%, 47%, and 22%, respectively (p < 0.001). Differences in baseline characteristics including age, sex, T stage, N stage, BMI, and chemotherapy cycle between the PCT alone and PCT plus LRRT subgroups among the three RPA-derived risk groups were controlled using the IPTW approach. IPTW-adjusted Kaplan–Meier curves showed that only the low-risk group (M1a) had OS benefit from LRRT, while no significant statistical differences in patients with and without LRRT were observed in the intermediate-risk (M1b) and high-risk (M1c) groups (Figure 5). Therefore, we believed that low-risk patients (M1a) were suitable candidates for LRRT. The details of all patients and different RPA-derived risk groups are displayed in Table 1 and Figures 3–5.

Process of RPA algorithm of M1 subdivisions.

Kaplan–Meier survival curves for RPA-generated M1 subdivisions.
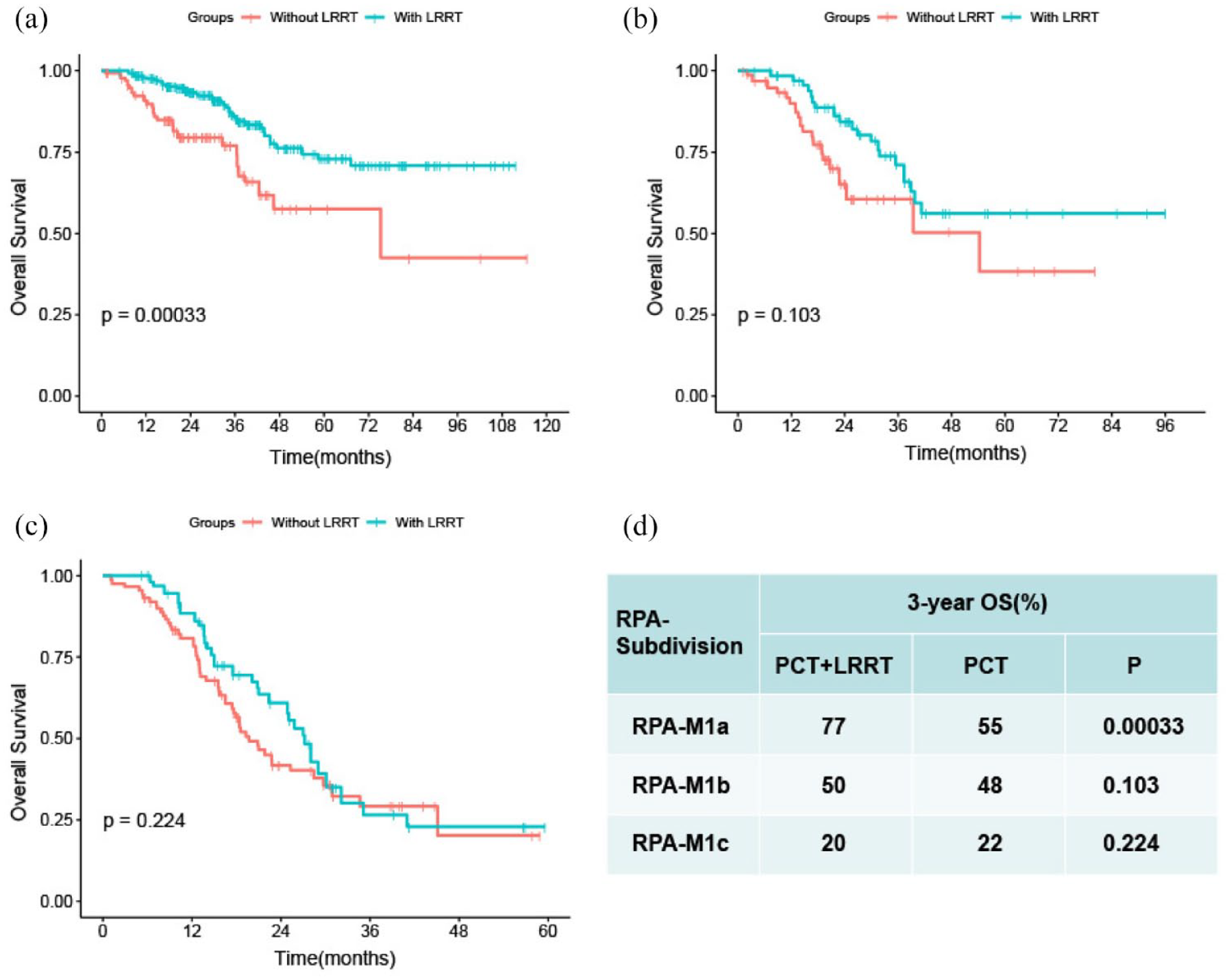
(a–c) IPTW-adjusted Kaplan–Meier analyses of overall survival for de novo metastatic nasopharyngeal carcinoma patients receiving LRRT after PCT versus palliative chemotherapy alone in the RPA-generated M1a (5A), M1b (5B), and M1c (5C) groups; (d): 3-year OS for patients with or without LRRT in each subdivision, accompanied with log-rank p value.
Adverse event
Systematic chemotherapy and LRRT were well tolerated, no treatment-related fatal AEs were observed. Grade 3 to 4 neutropenia was the most common toxic effect, which occurred in 287 (49%) of all patients. Non-hematological acute toxicities including grade 3 nausea/vomiting, mucositis, and dermatitis were observed in occurred in 70 (12%), 123 (21%), and 47 (8%) of all patients, respectively. No significant differences in overall treatment-related AEs were observed between the groups with and without LRRT.
Discussion
To the best of our knowledge, this is the first study to combine 18F-FDG PET-CT parameters with clinical characteristics for prognostic prediction and individualized treatment guidance of de novo mNPC patients. In this study, we evaluated the prognostic value of 18F-FDG PET-CT parameters and established an RPA-generated risk stratification incorporating PET-CT parameters, EBV DNA and liver involvement. We found that LRL-TLG, number of BMs, and DMST-MTV, which might have different roles in evaluating diverse lesions, were useful predictors for OS in mNPC patients. In addition, liver involvement and pretreatment EBV DNA were remarkable predictors and may play different roles in mNPC subgroups. Low-risk (M1a) patients could benefit from LRRT, whereas LRRT was of no value for moderate-risk (M1b) and high-risk (M1c) patients.
Platinum-containing PCT has been the mainstay therapeutic modality for initial mNPC patients, with response rates of 40–65% and a median OS of 10–15 months. 4 The recently established regimen of GP increased the OS of mNPC to 29 months. 30 Retrospective studies have demonstrated survival benefits with the addition of LRRT to PCT in mNPC patients.12–15 This conclusion was also strongly supported by a recently published phase III randomized clinical trial. According to the trial, LRRT combined with chemotherapy significantly improved OS in chemotherapy-sensitive mNPC patients. The 2-year OS of patients who received PCT and LRRT was improved to 76.4%, compared with 54.5% in the chemotherapy-alone group. 31 Nevertheless, a subset of, rather than all, mNPC patients could benefit from LRRT. Systemic chemotherapy incorporating LRRT is recommended for selected patients with distant metastases with a small tumor burden or at limited sites according to the contemporary National Comprehensive Cancer Network guidelines. However, there is no consensus currently regarding which patients should be selected to receive LRRT after PCT. To identify individuals who could benefit from LRRT, risk stratification and subdivisions based on biomarkers and anatomic features have been executed for mNPC patients. Zou et al. subgrouped mNPC patients according to anatomic features, including metastatic number and location, and found that LRRT combined with chemotherapy could improve the OS of patients in the oligometastases and multiple metastases without liver involvement groups, while patients with liver involvement did not benefit from LRRT. 13 Investigators of another study reported that they stratified mNPC patients by EBV DNA at diagnosis of metastasis (mEBV DNA) and radiological features, with the conclusion that patients in the low-risk group (low mEBV + oligo lesion and low mEBV + multiple lesions) may benefit from curative local treatment and patients in the high-risk group (high mEBV + no liver involvement and high mEBV + liver involvement) are recommended to undergo intensive systemic treatment. 15 With different study population and inclusion criteria, our study showed that patients with high pretreatment EBV DNA levels and liver involvement had significantly worse survival than those with low EBV DNA levels and without liver involvement, in line with the studies above. Both univariate and multivariate analyses showed that EBV DNA and liver involvement were powerful predictors for the survival of mNPC patients, and subsequently, we incorporated EBV DNA and liver involvement into our model. However, the metabolic characteristics of tumors were not included in these studies.
The prognostic value of 18F-FDG-PET-CT in tumors has been demonstrated by many previous studies.16,17,19,20 Growing evidence shows that PET-derived metabolic parameters, including SUVmax, TLG, and MTV, are promising indicators of therapeutic evaluation and prognostic estimation for NPC patients.17,32 Qi et al. explored the feasibility of 18F-FDG-PET-CT parameters in predicting the treatment response of locoregionally advanced NPC patients who received neoadjuvant chemotherapy followed by concurrent chemoradiotherapy (CCRT) and found that parameters of FDG PET were strongly correlated with tumor regression, indicating that 18F-FDG-PET-CT has the potential to monitor and predict tumor response. 33 Chen et al. 34 utilized 18F-FDG PET-derived parameters and EBV DNA to evaluate the early response of NPC patients to CCRT. Their results revealed that the combination of TLG and EBV DNA is useful in predicting clinical outcomes and enables satisfactory stratification of NPC patients with distinct survival rates. Similar results were observed in Ma’s study, which showed that SUVmax incorporating EBV DNA is useful in predicting the survival and response to chemotherapy in advanced or recurrent NPC patients. 35 In terms of mNPC, SUVmax, TLG, and EBV DNA were also demonstrated to be independent predictors of clinical outcomes. 36 However, studies of risk stratification based on 18F-FDG-PET-CT parameters to identify suitable candidates for LRRT in mNPC patients are rare. Therefore, we conducted this study aiming to develop a tool for guiding individualized therapy in mNPC patients. Using 18F-FDG PET-CT parameter-based PPS, together with liver involvement and EBV DNA, we developed a risk stratification of mNPC patients and explored the value of LRRT in patients with different risk groups. We found that only patients in the M1a group could benefit from LRRT; therefore, these patients are considered suitable candidates for LRRT. However, the survival of M1b and M1c patients was not improved by LRRT; beyond that, radiation-related toxicity was increased, and LRTT was not recommended for these patients, while systemic treatment involving targeted therapy and immunotherapy, or clinical trial was suggested for these patients.
In our study, PPS was shown to be the most essential impact factor for OS in all patients, followed by liver involvement (HR: 2.363, 95% CI: 1.752–3.186) and EBV DNA (HR: 2.163, 95% CI: 1.564–2.991). Patients in the PPShigh subgroup had significantly worse survival than those in the PPSlow subgroup (3-year OS 58% versus 24%). However, when trying to further stratify the groups of PPSlow without liver involvement and PPSlow with liver involvement by the level of pretreatment EBV DNA, we failed to find significant discriminations of survival among these subgroups. Similar results were observed in the PPShighEBVlow and PPShighEBVhigh subgroups. We hypothesized that in the early stage of metastases, with approximately the same tumor load, the site of metastases is the most essential factor of survival, whereas EBV DNA performed well in reflecting differences in tumor burden, leading to discrimination in survival in the PPShigh group, as metastases of multiple numbers or locations are commonly observed in this group. To establish the PPS system, we subdivided distant metastases of NPC into BMs and non-BMs (DSTM), and we found that the most influential parameters of distant metastases were DSTM-MTV and the number of BMs using multivariate Cox regression analysis. For local regional lesions, TLG was a significant predictor of clinical outcomes, in accordance with Chang’s study. 37 However, in terms of DSTMs, MTV, rather than TLG, was the unique prognostic factor for OS in our multivariate analysis among the PET-CT parameters of DSTMs. We hypothesize that MTV might be more suitable than TLG to evaluate the prognosis of mNPC patients with DSTMs. Similar results were observed in Chan’s study, which evaluated the primary and distant metastatic tumors of NPC and demonstrated that MTV is an independent risk factor in mNPC patients. 38 This might suggest that TLG is a good metabolism predictor for the evaluation of LRLs, while MTV performed better than TLG in assessing DSTMs. Nevertheless, further validations from other centers are needed.
Our study had several limitations. First, the primary limitation is its single-center nature and retrospective design, and no external validation was performed. Second, although we acquired the TLG and MTV metrics of BMs with soft tissue masses ⩾ 10 mm, we did not assess these two indices for small BMs without soft tissue masses <10 mm. However, the prognostic value of TLG and MTV for BMs has been evaluated in other studies. This is indeed a defect of our research. Third, in our study, we used two PET-CT scanners of different models to obtain patients’ images and data; in addition, due to many uncontrollable factors of clinical work in real world, PET scans were performed within a period of time(60–90 min) after the injection of 18F-FDG instead of at a strict time point. All the above may influence the FDG uptake and leads to errors between patients’ metabolic parameters. Thus, the cutoff values of metabolic parameters in our study need further validation in other centers. Fourth, posttreatment EBV DNA and tumor response proved useful indicators in predicting clinical outcomes, which were also confirmed by our study and might improve our model; these indicators were not included in our study due to its deficiency in some patients.
Conclusions
In conclusion, our findings suggest that certain PET-CT-derived texture parameters are independent predictors of outcomes in de novo mNPC patients. We established an RPA-generated subdivisions of mNPC patients based on PET-CT parameters, which could perform accurately in risk stratification and identification of suitable candidates for LRRT. However, more studies are needed to validate this novel M1 stage subdivisions.
Supplemental Material
sj-doc-2-tam-10.1177_17588359221118785 – Supplemental material for M1 stage subdivisions based on 18F-FDG PET-CT parameters to identify locoregional radiotherapy for metastatic nasopharyngeal carcinoma
Supplemental material, sj-doc-2-tam-10.1177_17588359221118785 for M1 stage subdivisions based on 18F-FDG PET-CT parameters to identify locoregional radiotherapy for metastatic nasopharyngeal carcinoma by Hui-Zhi Qiu, Xu Zhang, Sai-Lan Liu, Xue-Song Sun, Yi-Wen Mo, Huan-Xin Lin, Zi-Jian Lu, Jia Guo, Lin-Quan Tang, Hai-Qiang Mai, Li-Ting Liu and Ling Guo in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-1-tam-10.1177_17588359221118785 – Supplemental material for M1 stage subdivisions based on 18F-FDG PET-CT parameters to identify locoregional radiotherapy for metastatic nasopharyngeal carcinoma
Supplemental material, sj-jpg-1-tam-10.1177_17588359221118785 for M1 stage subdivisions based on 18F-FDG PET-CT parameters to identify locoregional radiotherapy for metastatic nasopharyngeal carcinoma by Hui-Zhi Qiu, Xu Zhang, Sai-Lan Liu, Xue-Song Sun, Yi-Wen Mo, Huan-Xin Lin, Zi-Jian Lu, Jia Guo, Lin-Quan Tang, Hai-Qiang Mai, Li-Ting Liu and Ling Guo in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
