Abstract
Background:
Early failure of cancer treatment generally indicates a poor prognosis. Here, we aim to develop and validate a pre-treatment nomogram to predict early metachronous metastasis (EMM) in nasopharyngeal carcinoma (NPC).
Methods:
From 2009 to 2015, a total of 9461 patients with NPC (training cohort: n = 7096; validation cohort: n = 2365) were identified from an institutional big-data research platform. EMM was defined as time to metastasis within 2 years after treatment. Early metachronous distant metastasis-free survival (EM-DMFS) was the primary endpoint. A nomogram was established with the significant prognostic factors for EM-DMFS determined by multivariate Cox regression analyses in the training cohort. The Harrell Concordance Index (C-index), area under the receiver operator characteristic curve (AUC), and calibration curves were applied to evaluate this model.
Results:
EMM account for 73.5% of the total metachronous metastasis rate and is associated with poor long-term survival in NPC. The final nomogram, which included six clinical variables, achieved satisfactory discriminative performance and significantly outperformed the traditional tumor–node–metastasis (TNM) classification for predicting EM-DMFS: C-index: 0.721 versus 0.638, p < 0.001; AUC: 0.730 versus 0.644, p < 0.001. The calibration curves showed excellent agreement between the predicted and actual EM-DMFS. The nomogram can stratify patients into three risk groups with distinct EM-DMFS (2-year DMFS: 96.8% versus 90.1% versus 80.3%, p < 0.001). A validation cohort supported the results. The three identified risk groups are correlated with the efficacy of different treatment regimens.
Conclusion:
Our established nomogram can reliably predict EMM in patients with NPC and might aid in formulating risk-adapted treatment decisions and personalized patient follow-up strategies.
Introduction
Nasopharyngeal carcinoma (NPC) is a distinct endemic head and neck malignancy characterized by an unbalanced geographical distribution, wide variety in treatment modalities, and a high distant metastasis rate.1–3 It is rare in non-endemic areas but occurs at relatively high frequency in Southeast Asia and southern China.1,2 Radiotherapy is the primary treatment modality for non-metastatic disease. 3 Distinguished from other head and neck malignancy, NPC is an aggressive disease with a high prevalence of distant metastases.4–8 It is reported that nearly 10%4,5 of patients with NPC present with distant metastases at first diagnosis, termed as synchronous metastatic NPC. In addition, 20–30%6–8 of patients eventually develop distant metastases after chemo-radiotherapy, which is termed metachronous metastatic NPC. The occurrence of distant metastasis, including synchronous/metachronous metastasis, is associated with a poor outcome, the median overall survival (OS) rates for metastatic NPC range from 11 months for those not receiving treatment, to 29.1 months when treated with palliative chemotherapy.9–11
Metachronous metastasis is more prevalent than synchronic metastatic NPC. 12 More importantly, about 70% of the post-treatment failures are ascribed to metachronous metastasis. 13 Time to metachronous metastasis differs between patients. There is no consensus on the optimal cut-off value between early and late metachronous metastasis in NPC. Up to 59% of events occur within the first 2 years after treatment, and the hazard rate of metachronous metastasis displays a sharp peak at 2 years after treatment, which declines after that. 14 In NPC and other cancer types, patients experiencing early treatment failure exhibit poor survival.15–19 Therefore, pre-treatment prediction of the individual risk of early metachronous metastasis (EMM) is central to facilitate early interventions for high-risk patients and the development of individualized follow-up strategies for NPC.
Currently, the International Union against Cancer/American Joint Committee on Cancer (UICC/AJCC) tumor–node–metastasis (TNM) classification, based on tumor size and anatomical tumor invasion information, is the most used prognostic tool for NPC. However, there are large variations in distant metastasis-free survival (DMFS) of NPC patients with equivalent classification receiving similar treatment regimens. Tang et al. demonstrated that the TNM staging system had only 60% accuracy in predicting metachronous metastasis. 20 This finding suggests that solely relying on the TNM classification to predict outcome is inadequate. Many other risk factors might also contribute to metastasis. Previous studies have demonstrated clinical variables such as gender, 21 serum lactate dehydrogenase (LDH),22–24 circulating Epstein–Barr virus (EBV) DNA,25–27 sensitivity C-reactive protein (hs-CRP), 28 tumor infiltrating lymphocytes, 29 CD8 30 and PD-L1 expression31,32 can significantly contribute to the pre-treatment prediction of metastasis. However, in most of these studies, only one or two biomarkers were evaluated without considering others.
Nomograms have been accepted as a reliable prediction tool for the individual numerical probability of a clinical event in cancer by integrating pathological, clinical, and treatment-related variables into a statistical model. 33 Emerging reports have revealed that, for several tumor types, nomograms can achieve more accurate prognoses than the current clinically used TNM classification.34–36 However, to date, no nomograms have been developed for predicting EMM in NPC. Here, based on these premises, we constructed a pre-treatment nomogram to predict EMM in NPC and validated its effectiveness using a different patient cohort, who were treated during the same period as our training cohort.
Materials and methods
Data extraction and study population
The research ethics committee of Sun Yat-sen University Cancer Center (SYSUCC) approved the current retrospective analysis of anonymous data, and the need for individual informed consent was waived (approval number: B2020-267). An NPC-specific database was built to facilitate research into NPC by extracting data from the well-developed big data intelligence platform of SYSUCC. Our intelligence platform supports the automatic extraction, integration, and updating of routine healthcare data. More detailed information about the big data intelligence platform is accessible in our previously published study. 37 A total of 10,126 consecutive patients with newly diagnosed, histologically confirmed, previously untreated non-disseminated NPC diagnosed between April 2009 and December 2015 was identified using the NPC-specific database.
Patients were eligible for inclusion in this study if they met the following criteria: (1) availability of pre-treatment pathological and clinical data; (2) no previous history of malignancy before the NPC diagnosis or other concomitant malignant disease; (3) treated with definitive intensity-modulated radiotherapy (IMRT) with or without chemotherapy; and (4) no serious lung, heart, liver, and kidney diseases. Finally, a total of 9461 eligible patients was identified, who were randomly split into the training (n = 7096) and validation (n = 2365) cohorts at a 3:1 ratio via computer software-generated random numbers.
Baseline evaluation
The routine pre-treatment staging work-up for NPC patients included a complete medical history, biochemistry and hematology profiles, physical examination, fiberoptic nasopharyngoscopy, magnetic resonance imaging of nasopharynx and the whole neck, chest radiography, abdominal sonography, and whole-body bone scans. Each patient was restaged based on the 8th edition of the UICC/AJCC TNM classification. The following potential baseline prognostic factors were collected from the patients’ medical records: age, gender, World Health Organization (WHO) histological type, T classification, N classification, overall stage, cigarette consumption, alcohol consumption, family history of cancer, pre-treatment circulating cell-free (cf) EBV DNA load, hemoglobin (HGB), LDH, albumin (ALB), and CRP. EBV DNA concentrations were detected towards the BamHI-W region of the EBV genome using a real-time quantitative polymerase chain reaction assay, which was described in detail in our previous work. 38
Treatment strategies
Treatment strategies were determined according to standard protocols depending on UICC/AJCC TNM classification and the general condition of the patient. During the study period, institutional guidelines recommended definitive IMRT alone and no chemotherapy for stage I NPC, and concurrent chemo-radiotherapy (CCRT) with or without neoadjuvant/adjuvant chemotherapy (NACT/ACT) for stages II–IVa NPC. The reasons patients had for not undergoing recommended treatment strategies included patient’s refusal, old age, and organ dysfunction. A total of 178 patients that were eligible for chemotherapy underwent IMRT alone due to the above-mentioned conditions. IMRT was administered at one fraction per day, 5 days per week. CCRT consisted of cisplatin, which was administered weekly or on weeks 1, 4, and 7 of IMRT. NACT or ACT consisted of docetaxel with cisplatin, 5-fluorouracil with cisplatin, or cisplatin and 5-fluorouracil with docetaxel every 3 weeks for two to four cycles.
Follow-up and endpoints
After completing definitive treatment, patients returned to the hospital for clinical appointments every 3 months for the first 2 years, and every 6 months thereafter or until death. Follow-up assessment, which included physical examination, assessment of clinical symptoms, and imaging procedures similar to the pre-treatment examinations, was conducted at each follow-up appointment to detect possible treatment failure. Follow-up duration was calculated from the day of diagnosis of NPC to the last visit or death. Clinical suspicion of EMM was confirmed using imaging examination and cytological biopsies. The endpoint of the current study was early metachronous distant metastasis-free survival (EM-DMFS), which was defined as the time from the date of treatment initiation to the first distant metastasis or patient censoring, whichever occurred first. In this study, metastasis within the first 2 years after treatment was defined as EMM, and metastasis more than 2 years after end of treatment was defined as late metachronous metastasis (LMM).
Statistical analysis
The statistical analysis was performed using R software (version 3.4.4) and SPSS version 23.0 (IBM Corp., Armonk, NY, USA). Age was classified into two groups based on the median value, other continuous variables included cf EBV DNA load, HGB, LDH, ALB, and CRP. These variables were converted into categorical variables according to the routine cut-off points in clinical applications as shown in Table 1. The chi-square test or Fisher’s exact test were used to compare the categorical variables of different subgroups. Actuarial survival rates were calculated using the Kaplan–Meier curve and differences between patient subgroups were compared using the log-rank test. Univariate and multivariate Cox proportional hazard regression analyses were performed to quantify the effect of candidate predictors on EM-DMFS. A total of 13 candidate predictors for EMM were included in the univariate Cox proportional hazard regression analysis. Covariates with a univariable p < 0.05 were entered into the multivariable model to validate their significance by using a backward stepwise algorithm. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated using the Cox proportional hazards regression analysis.
Comparison of baseline clinical and treatment characteristic among nasopharyngeal carcinoma patients who experienced early metachronous metastasis and late/non metachronous metastasis.

Statistical comparisons between training cohort and validation cohort were computed using the chi-square test or Fisher’s exact test, a p-value of 0.05 indicates a significant difference.
According to the 8th edition of the AJCC/UICC staging system.
ALB, albumin; CRP, C-reactive protein; cf EBV DNA, circulating cell-free Epstein–Barr virus deoxyribonucleic acid; HGB, hemoglobin; LDH, lactate dehydrogenase; TNM, tumor–node–metastasis; WHO, World Health Organization.
To maximize the predictive power of the model, we constructed a nomogram in the training cohort and validated its predictive power in the validation cohort. Variables considered statistically significant in the multivariate analysis were used to formulate an effective prognostic nomogram in the training cohort using R software (version 3.4.4). The score for each variable was determined by mapping to points on a scale axis ranging from 0 to 100. We calculated the total scores for each patient, by summing the scores of all variables. Finally, we determined the likelihood of EM-DMFS for individual patients. The calibration and discrimination of the nomogram were measured using Harrell’s concordance index (C-index) and calibration curve to assess the predictive accuracy of the model (bootstraps with 1000 resample) in the training and validation cohorts. The calibration ability of the nomogram was assessed graphically using calibration curves, the closer the nomogram-predicted survival curve is to the observation survival curve, the more accurate the prognostic prediction is. The value of the C-index ranges from 0.5 to 1.0, with a value ⩾ 0.7 representing that the constructed nomogram model has a good discriminating ability, and 1.0 indicates perfect predictive ability. Comparisons between nomogram models, TNM classification and other single risk factors were performed with the rcorrp.cens in Hmisc in R software. We further compared the prognostic predictive power of the nomogram and single pre-treatment clinical features, TNM staging system and nomogram using the C-index and the area under the time-dependent receiver operator characteristic (ROC) curve (AUC). All statistical tests were two-sided, with statistical significance set at 0.05.
Results
Clinical and treatment characteristics
Baseline clinical and treatment characteristics of 9461 patients with non-disseminated NPC in the training and the validation cohorts are shown in Table 1 and Supplemental Table 1. Median follow-up duration for the entire cohort, training cohort, and validation cohort was 52.8 months (range: 2.0–111.1 months), 52.7 months (range: 2.6–111.1 months), and 53.3 months (range: 2.0–109.4 months), respectively. The number of events in the entire cohort is summarized in Supplemental Figure 1. Of the 9461 patients analyzed, 9.3% (877/9461) experienced the first metachronous metastasis within the first 2 years after treatment completion (termed EMM), and 3.7% (316/9461) experienced the first metachronous metastasis after more than 2 years after treatment completion (termed LMM). A total of 73.5% (877/1193) metachronous metastases occurred within the first 2 years after treatment completion. For the training and validation cohort, 9.3% (659/7096) and 9.2% (218/7096) patients experienced EMM, and 3.2% (230/2365) and 3.6% (86/2365) patients suffered LMM, respectively.
EMM correlates with poor overall survival in patients with NPC
The 5-year OS for the entire, training, and validation cohorts were 86.2%, 86.3%, and 86.1%, respectively. The median OS time for the EMM and the LMM groups were 31.4 (95% CI: 29.9–33.0) and 63.4 (95% CI: 57.7–69.1) months for the entire cohort; 31.7 (95% CI: 29.9–33.6) and 63.4 (95% CI: 56.7–70.1) months for the training cohort; and 30.1 (95% CI: 25.7–34.6) and 64.3 (95% CI, 51.2–77.3) months for the validation cohort. The 5-year OS for the EMM and LMM groups was 17.7% and 54.5% for the entire cohort (p < 0.001); 18.0% and 54.2% for the training cohort (p < 0.001); 16.8% and 55.2% for the validation cohort (p < 0.001), respectively. Kaplan–Meier plots of overall survival outcome for patients are presented in Figure 1. Overall, patients who experienced EMM had poorer OS.

Kaplan–Meier plots of overall survival outcome for (nasopharyngeal carcinoma) NPC patients in the early metachronous metastasis (EMM) and the late metachronous metastasis (LMM) group. (A) Training cohort, n = 7096; (B) Validation cohort, n = 2365.
Univariate and multivariate analyses of the risk factors for EMM
Univariate analysis results for EM-DMFS in the training and validation cohort are shown in Figure 2. Multivariate analysis revealed that for the training cohort, gender (female versus male, HR = 0.626, 95% CI: 0.514–0.764, p < 0.001), T classification (p < 0.001), N classification (p < 0.001), cf EBV DNA load (<4000 versus ⩾4000 copy/mL, HR = 2.080, 95% CI: 1.742–2.483, p < 0.001), LDH (⩾245 versus <245 U/L, HR = 1.781, 95% CI: 1.445–2.193, p < 0.001) and ALB (⩾40 versus<40 g/L, HR = 0.755, 95% CI: 0.602–0.947, p = 0.015) were independent prognostic factors for EM-DMFS. In the validation cohort, T classification (p = 0.025), N classification (p < 0.001), cf EBV DNA load (<4000 versus ⩾4000 copy/mL, HR = 1.772, 95% CI: 1.312–2.394, p < 0.001), and LDH (⩾245 versus <245 U/L, HR = 1.955, 95% CI: 1.362–2.808, p < 0.001) were found to be independent prognostic factors for EM-DMFS (Table 2).

The forest plot showing univariate association of potential risk factors and early metachronous distant metastasis-free survival in nasopharyngeal carcinoma.
Summary of multivariate Cox proportional hazard regression analysis of independent risk factors for early metachronous metastasis in 9468 nasopharyngeal carcinoma.

ALB, albumin; cf EBV DNA, circulating cell-free Epstein–Barr virus deoxyribonucleic acid; CI, confidence intervals; HGB, hemoglobin; HR, Hazard ratio; LDH, lactate dehydrogenase.
Development and validation of a pre-treatment nomogram for EMM
We further visualized the results of the multivariate analysis of EM-DMFS as a pre-treatment nomogram to predict the EMM in the training cohort (Figure 3A). The variables included in this nomogram were gender, T classification, N classification, cf EBV DNA load, LDH, and ALB. The calibration plots indicated excellent agreement between the nomogram prediction and the actual observed EM-DMFS, both in the training and validation cohorts (Figure 3B). The pre-treatment nomogram-defined scores for each patient are summarized in Figure 3C.
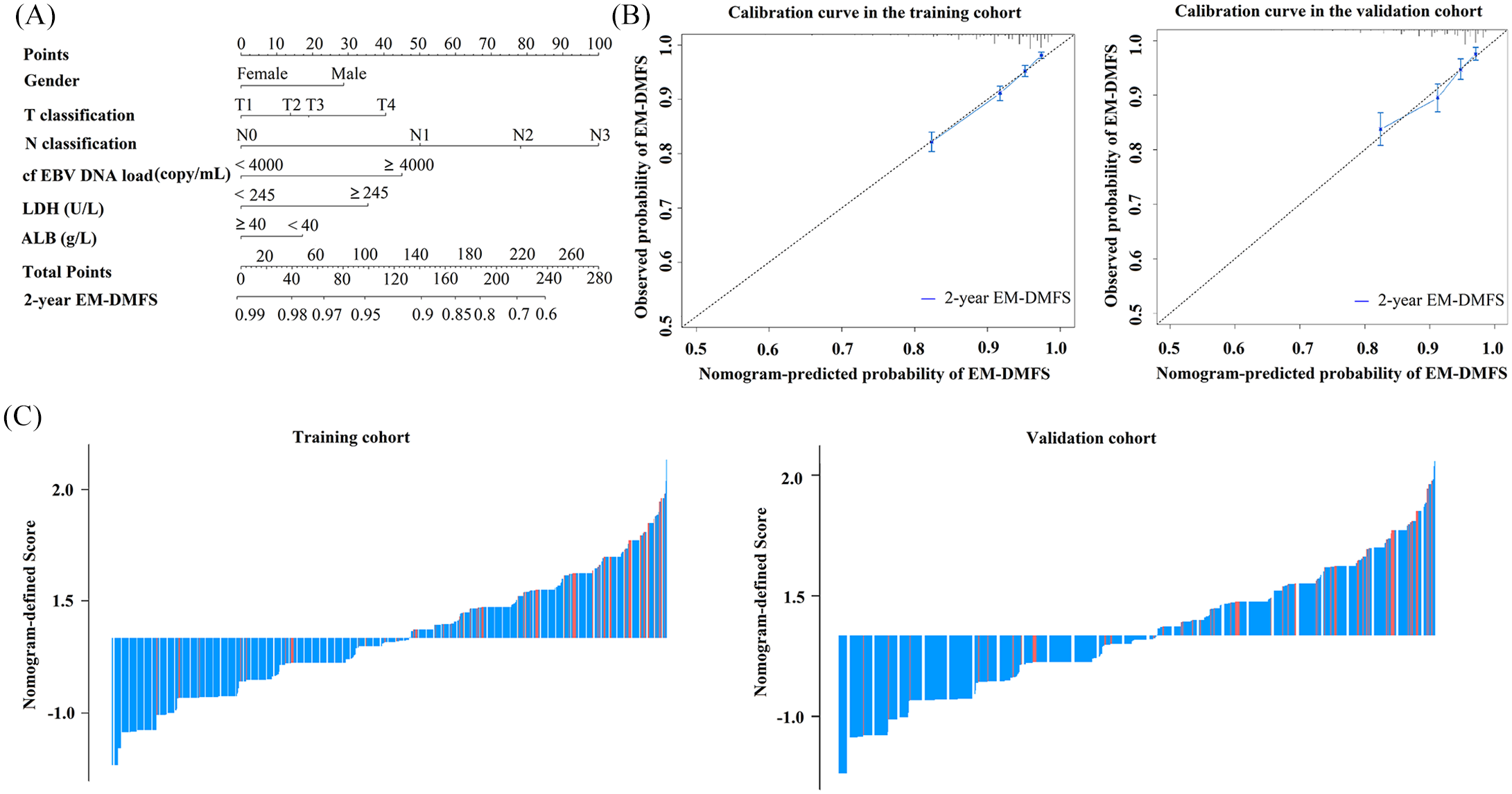
(A) Pre-treatment nomogram to predict early metachronous distant metastasis-free survival (EM-DMFS) in nasopharyngeal carcinoma (NPC) patients. (B) Calibration curves of the nomogram to predict EM-DMFS probability at 2 years in the training (n = 7096) and validation cohort (n = 2365). Nomogram-predicted probability of EM-DMFS is plotted on the x-axis; actual observed probability is plotted on the y-axis. (C) Nomogram-defined score for each NPC patient in the training (n = 7096) and validation cohort (n = 2365).
For the training and validation cohorts, the C-index of the pre-treatment nomogram (0.721, 95% CI: 0.684–0.757; 0.704, 95% CI: 0.637–0.771) was statistically significant and outperformed the 8th TNM classification (0.638, 95% CI: 0.601–0.675, p < 0.001; 0.621, 95% CI: 0.557–0.684, p < 0.001) and single risk factors gender (0.543, 95% CI: 0.513–0.573, p < 0.001; 0.524, 95% CI: 0.468–0.579, p < 0.001), T classification (0.585, 95% CI: 0.545–0.625, p < 0.001; 0.581, 95% CI: 0.515–0.647, p < 0.001), N classification (0.635, 95% CI: 0.615–0.691, p < 0.001; 0.650, 95% CI: 0.582–0.718, p < 0.001), cf EBV DNA load (0.642, 95% CI: 0.607–0.677, p < 0.001; 0.624, 95% CI: 0.561–0.687, p < 0.001), LDH (0.551, 95% CI: 0.522–0.579, p < 0.001; 0.552, 95% CI: 0.503–0.601, p < 0.001), and ALB (0.524, 95% CI: 0.499–0.55, p < 0.001; 0.526, 95% CI: 0.483–0.569, p < 0.001) for EM-DMFS prediction (Table 3).
Summary of the C-index of prognostic models and single risk factors for early metachronous metastasis in nasopharyngeal carcinoma.
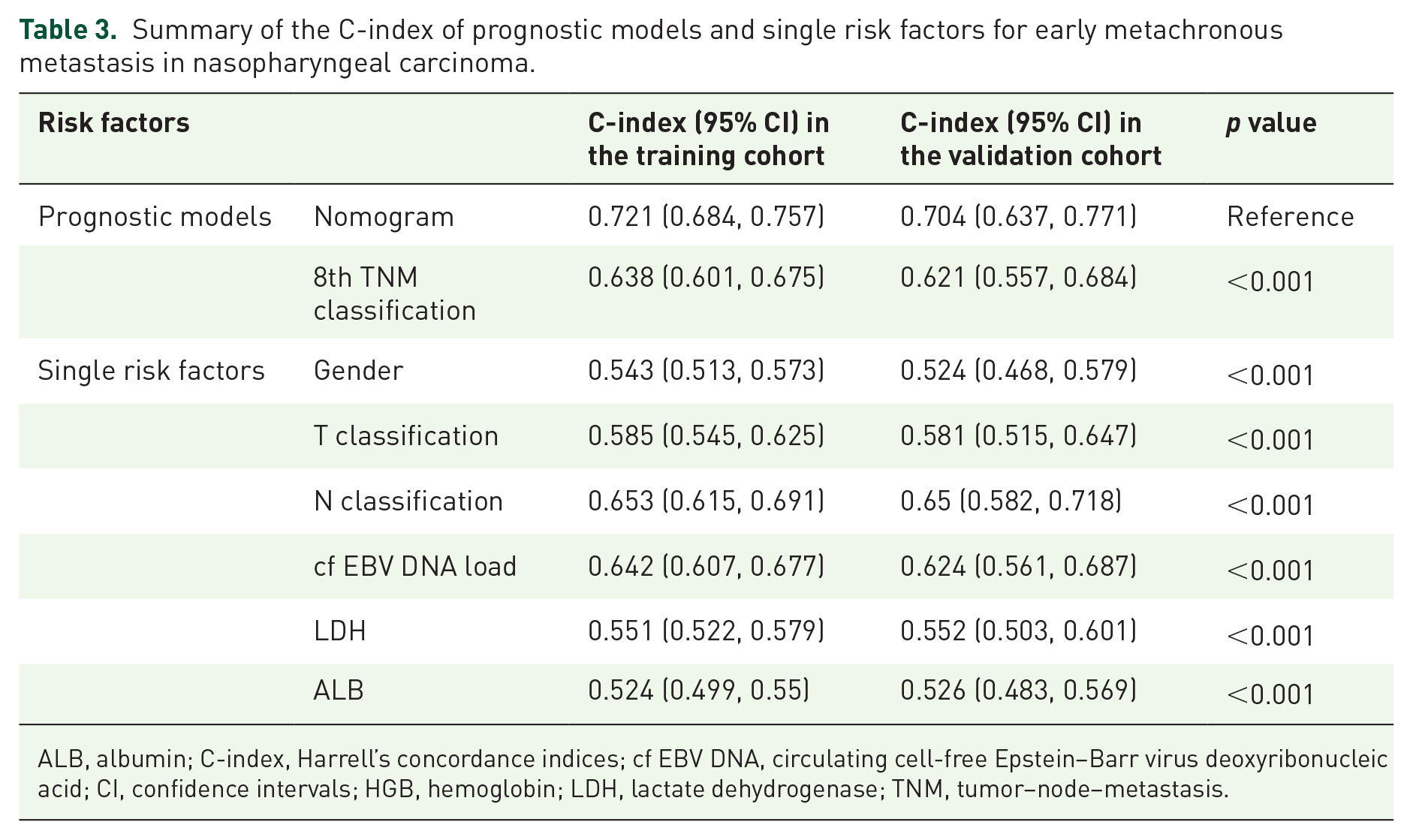
ALB, albumin; C-index, Harrell’s concordance indices; cf EBV DNA, circulating cell-free Epstein–Barr virus deoxyribonucleic acid; CI, confidence intervals; HGB, hemoglobin; LDH, lactate dehydrogenase; TNM, tumor–node–metastasis.
To substantiate further the prediction performance of the pre-treatment nomogram, a ROC curve analysis was performed to evaluate the predictive accuracy of the nomogram (AUCs: 0.730, 0.712; sensitivity: 0.800, 0.684; specificity: 0.544, 0.652), TNM classification (AUCs: 0.644, 0.626; sensitivity: 0.774, 0.913; specificity: 0.337, 0.243), gender (AUCs: 0.546, 0.523; sensitivity: 0.818, 0.775; specificity: 0.274, 0.271), T classification (AUCs: 0.588, 0.584; sensitivity: 0.337, 0.789; specificity: 0.639, 0.332), N classification (AUCs: 0.661, 0.657; sensitivity: 0.574, 0.569; specificity: 0.682, 0.681), cf EBV DNA load (AUCs: 0.649, 0.628; sensitivity: 0.694, 0.656; specificity: 0.601, 0.601), LDH (AUCs: 0.551, 0.554; sensitivity: 0.017, 0.170; specificity: 0.932, 0.938), and ALB (AUCs: 0.525, 0.527; sensitivity: 0.134, 0.124; specificity: 0.916, 0.930) in the training and validation cohorts, respectively. Among these, the pre-treatment nomogram showed the highest accuracy. The AUC of the pre-treatment nomogram was statistically significant and outperformed the 8th TNM classification (p < 0.001), gender (p < 0.001), T classification (p < 0.001), N classification (p < 0.001), cf EBV DNA load (p < 0.001), LDH (p < 0.001), and ALB (p < 0.001) for EM-DMFS prediction (Figure 4).
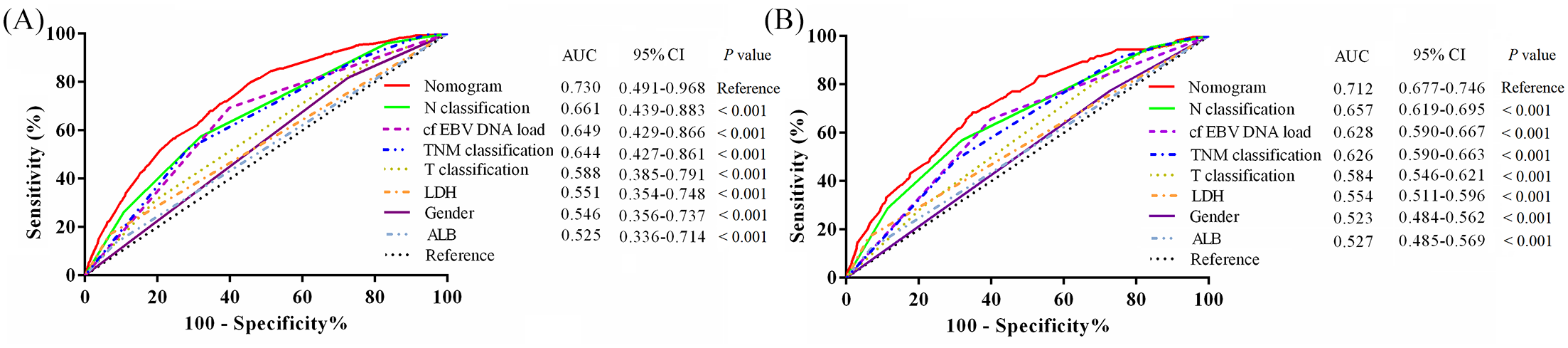
Receiver operating characteristic curves comparing the predictive power of the proposed nomogram, 8th TNM classification and risk factors gender, T classification, N classification, Epstein-Barr virus (EBV)DNA load, lactate dehydrogenase (LDH), and albumin (ALB) for early metachronous distant metastasis-free survival prediction for nasopharyngeal carcinoma (NPC). (A) Training cohort, n = 7096; (B) Validation cohort, n = 2365.
Stratification of the risk of EMM
Using recursive partitioning tree analysis, the optimum cut-off value for the pre-treatment nomogram-defined score for early metachronous DMFS prediction was generated as 1.68 and 2.58 in the training cohort. Patients were categorized as ‘low’, ‘intermediate’, or ‘high’ risk groups in the training and validation cohort. Accordingly, 3636 (51.2%), 2204 (31.1%), and 1256 (17.7%) of patients in the training cohort, and 1217 (51.5%), 733 (31.0%), and 415 (17.5%) patients in the validation cohort were categorized into the low-risk, intermediate-risk and high-risk groups, respectively. The number of events of EMM in each subgroup is summarized in Supplemental Table 2. The subgroup survival analysis showed that the DMFS gradually lowered comparing the low-risk, intermediate-risk and the high-risk group in both the training cohort (2-year EM-DMFS: 96.8% versus 90.1% versus 80.3%, p < 0.001) and validation cohort (2-year EM-DMFS: 96.3% versus 89.6% versus 81.1%, p < 0.001, Figure 5, Supplemental Table 3).
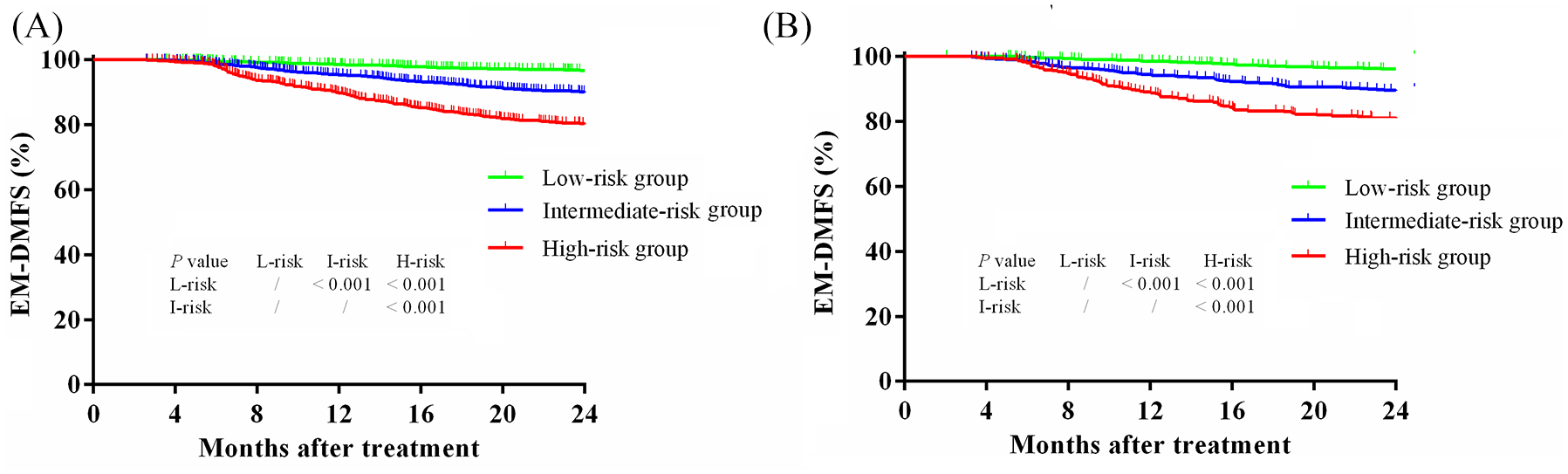
Kaplan–Meier plots of early metachronous distant metastasis-free survival between nomogram-defined high, intermediate, and high-risk groups for nasopharyngeal carcinoma (NPC) patients in the training cohort (A) and validation cohort (B). The log-rank test was used to calculate p-values.
Individualized therapeutic strategies based on nomogram-defined subgroups
We further explored whether the pre-treatment nomogram constructed in this study could guide individualized treatment. We compared the efficacy of different treatment regimens in the nomogram-defined three risk groups. For the low-risk group, patients who received intensive treatments of CCRT (p = 0.444), NACT+IMRT (p = 0.122), NACT+CCRT (p = 0.673) or CCRT+ACT (p = 0.148) did not show significantly better survival in terms of EM-DMFS compared with those who received the IMRT alone (Figure 6A). Therefore, IMRT is recommended for patients in the low-risk group. Within the intermediate-risk group, survival outcomes were comparable between the CCRT, NACT+IMRT, and NACT+CCRT groups, and the above three treatment strategies were significantly superior to IMRT alone (Figure 6B). Compared with CCRT or NACT+IMRT, patients did not benefit from intensive treatment with NACT+CCRT. Therefore, CCRT or NACT+IMRT are recommended for patients in the intermediate-risk group. For the high-risk group, patients were more likely to benefit from therapy intensification with NACT+CCRT than from IMRT alone (p < 0.001), CCRT (p = 0.024), NACT+IMRT (p = 0.018), or CCRT+ACT (p = 0.026) (Figure 6C). Thus, NACT+CCRT is recommended for patients in the high-risk group.

Efficacies of different treatment regimens for nasopharyngeal carcinoma (NPC) patients in nomogram-defined low-risk (A), intermediate-risk (B) and high-risk groups (C). The log-rank test was used to calculate p-values.
Discussion
About 70% of the treatment failures are ascribed to distant metastasis in NPC. 13 Early treatment failure usually indicates a poor prognosis.15–19 TNM classification is not the sole risk factor for EMM in NPC as the EM-DMFS rates of patients who share the same classification are highly variable. Thus, TNM classification is not capable of accurately predicting EMM in patients with NPC pre-treatment. Therefore, establishing prediction models with a more accurate prediction ability is urgently needed.
To the best of our knowledge, this is the first big-data intelligence platform-based analysis to develop a reliable, visualized, and user-friendly prognostic nomogram for EMM prediction in non-metastatic NPC patients before treatment-related decisions are being made. This nomogram was established in the training cohort and validated in the validation cohort. The resulting nomogram achieved excellent discriminative performance, which significantly outperformed the 8th TNM classification for predicting EMM in NPC. Using the nomogram-defined score for individual patients, we successfully classified patients into low, intermediate, and high-risk groups. These proposed risk groups significantly predicted the risk of EMM in NPC.
Previously, a series of studies have proposed nomogram models for facilitating the development of individualized treatment regimens in NPC.39–46 For example, Sun et al. 40 constructed a nomogram for guiding CCRT in stage II NPC. Zhao et al. 41 radiomics proposed a nomogram which may predict the response to NACT and survival in locally advanced NPC. Zhang et al. 44 established a nomogram for locoregionally advanced NPC patients to select individuals who may benefit from additional NACT compared to CCRT alone. To enhance the therapeutic efficacy and increase the proportion of long-term DMFS patients, it is necessary to determine the best candidate treatment for NPC patients. In the current study, we made recommendations for individualized treatment strategies based on the efficacy of different treatment regimens in the three risk groups defined by nomogram.
In addition to enabling clinicians to predict the pre-treatment risk of EMM and formulate individualized treatment regimens for NPC patients, the nomogram may also be useful for facilitating patient counseling and helping to develop individualized follow-up strategies. For example, patients with high EMM risk predicted by the nomogram should be closely monitored after treatment, which allows timely treatment when EMM occurs. This may improve the survival of this patient group.
Our results showed that the majority of metachronous metastasis (73.5%) cases occur within the first 2 years after treatment completion. Patients with EMM have inferior overall survival rates in comparison to patients with LMM. This indicates that characterizing the cohort of patients who are likely to suffer LMM is essential and may assist physicians in formulating more efficient therapeutic strategies.
Our multivariable analyses showed that the variables gender, T classification, N classification, cf EBV DNA load, LDH, and ALB could independently predict EM-DMFS. In line with previous studies, each of these variables was correlated with DMFS prediction in NPC.21–28,47,48 In our nomogram, N classification, cf EBV DNA load, and T classification (C-index in the training cohort, 0.653 versus 0.642 versus 0.585; C-index in the validation cohort, 0.650 versus 0.624 versus 0.581) contributed the most to predicting EM-DMFS of NPC patients. TNM classification is widely accepted as the most important predictor for survival in NPC. Similar to our results, Wu et al. 49 reported that the accuracy of the N classification to predict DMFS is higher than that of T classification (C-index: 0.796 versus 0.673). Accumulating studies report that a high cf EBV DNA load pre-treatment is associated with the prediction of poor DMFS in NPC.24–26 Consistently, our results found that high cf EBV DNA load adversely affected EM-DMFS in NPC patients.
We also confirmed that elevated serum LDH is inversely associated with EM-DMFS. Consistently, many studies have reported elevated LDH levels as a negative prognostic indicator for NPC. High LDH levels have been hypothesized to reflect the hypoxic status of tumor cells.20–22 Tumor hypoxia plays an important role in the development of chemo and radio-resistance, which eventually leads to distant metastasis and tumor progression.50–52 Moreover, ALB is known as a common nutritional index marker and acts as a valuable prognostic indicator in NPC.38,47 Our study confirmed that a decreased level of pre-treatment ALB implies a poor EM-DMFS in NPC. Furthermore, our results indicate that the female sex is a favorable prognostic factor for EM-DMFS in NPC, confirming previous studies. Although the mechanism of gender difference in survival remains unclear, it may be explained by the immune-enhancing properties exerted by female sex steroids, and the adverse effects of testosterone on the immune function of male patients. 20
There were two limitations of the current study that need to be addressed. First, potential bias may exist due to the retrospective nature of this study. Nevertheless, the large sample size may reduce the bias resulting from retrospective data collection. Second, even though the internal validation was conducted to ensure the generalizability of the nomogram, additional validation of the nomogram by external datasets or prospective datasets is still necessary to confirm our results.
In conclusion, we have developed and validated a pre-treatment nomogram as a powerful prognostic tool for predicting EMM in NPC patients. The proposed nomogram can successfully stratify NPC patients into three risk groups with a distinct EM-DMFS and achieved significantly better discrimination performance than the traditional TNM classification. We further made recommendations for individualized treatment strategies based on the efficacy of different treatment regimens in the three risk groups. This nomogram may facilitate clinicians to predict risk, formulate individualized treatment, conduct counseling, and design tailored post-treatment follow-up strategies for NPC patients.
Supplemental Material
sj-pdf-1-tam-10.1177_1758835920978132 – Supplemental material for Development and validation of a prognostic nomogram for the pre-treatment prediction of early metachronous metastasis in endemic nasopharyngeal carcinoma: a big-data intelligence platform-based analysis
Supplemental material, sj-pdf-1-tam-10.1177_1758835920978132 for Development and validation of a prognostic nomogram for the pre-treatment prediction of early metachronous metastasis in endemic nasopharyngeal carcinoma: a big-data intelligence platform-based analysis by Lu-Lu Zhang, Fei Xu, Wen-Ting He, Meng-Yao Huang, Di Song, Yi-Yang Li, Qi-Ling Deng, Yong-Shi Huang, Ting Wang and Jian-Yong Shao in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors sincerely thank Wei Liang (Yidu Cloud Technology Ltd., Beijing, China) for providing technical support in extracting study data from their big data intelligence database platform.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the China Postdoctoral Science Foundation Grant (no. 2019M663305).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
