Abstract
Appendiceal cancer is rare and encompasses a diverse group of tumours ranging from low-grade appendiceal mucinous neoplasms to high-grade adenocarcinomas. Appendiceal cancers often spread to the peritoneal cavity causing extensive mucinous dissemination and peritoneal metastases. Prognosis varies with histological subtype. Cytoreductive surgery and heated intraperitoneal chemotherapy is well-established as the most effective treatment achieving long-term survival in some patients. Chemotherapy regimens used to treat appendiceal cancer are extrapolated from the colorectal cancer setting, but disease biology differs and outcomes are inferior. The role of chemotherapy in the treatment of appendiceal cancer remains poorly defined. There is an urgent need to develop novel tailored treatment strategies in the perioperative and unresectable setting. This review aims to evaluate the literature for patients who received intraperitoneal and systemic chemotherapy for appendiceal cancers.
Introduction
Appendiceal cancer is rare with an incidence of 1–2 per million and is listed by the National Organisation for Rare Diseases.1–3 Appendiceal epithelial neoplasms encompass a diverse group of tumours ranging from low-grade appendiceal mucinous neoplasms (LAMNs) to high-grade adenocarcinomas.4,5 The most common pattern of spread for appendiceal cancers is the peritoneal cavity causing extensive mucinous dissemination and peritoneal metastases. 4
Cytoreductive surgery and heated intraperitoneal chemotherapy (CRS-HIPEC) are the most effective treatments, but prognosis varies with histological subtype. Five-year survival ranges from 96% for low-grade disease to 23% with high-grade disease.6–12 Although low-grade disease confers a better 5-year survival, long term it still recurs in up to 30% of patients, many of them will die from the disease. 13
It is imperative in all disease settings to define therapeutic strategies that can meaningfully improve outcomes. The role of perioperative chemotherapy in the multidisciplinary setting is a dilemma. Chemotherapy regimens are extrapolated from the colorectal cancer (CRC) literature. Although some data are within CRC-directed studies, there has been increasingly homogenous data published specifically for appendiceal cancer populations over recent years.14,15 Furthermore, distinctions have emerged between appendiceal and CRC with regard to the biological understanding of peritoneal disease and subsequent outcomes.16–21
This review aims to evaluate the literature for patients who received chemotherapy for appendiceal cancers with peritoneal disease, with previous reviews focused primarily on surgical outcomes or were written prior to the publication of more recent studies using the updated 2019 WHO classification with limited discussion regarding the specific role of systemic chemotherapy.22–24 We have excluded goblet-cell adenocarcinomas as treatment paradigms are generally similar to CRC, and the subgroup with neuroendocrine features is beyond the scope of this review. We have discussed evidence for the specific contribution of HIPEC in addition to CRS, the role of perioperative systemic chemotherapy and the outcomes of chemotherapy in the unresectable setting.
Although there is a need for prospective randomised trials paired with translational studies to improve biomarker and therapeutic advances, we demonstrated that despite significant limitations and challenges that exist in interpreting this literature, an evidence-based and rational therapeutic approach can still be employed. We emphasised that treatment decisions should be made by a multidisciplinary team at centralised, high-volume treatment centres.
Classification of appendiceal cancers
Over many years, the nomenclature of appendiceal cancer has been adapted to homogenise a complex and inconsistent histopathological classification.5,25–27 A further challenge is the discordance between the primary and peritoneal tumour grade.7,28,29 It is now convention to describe the primary and peritoneal disease separately.
The term ‘pseudomyxoma peritonei’ or ‘PMP’ describes the clinical syndrome of abdominal mucinous disease that can arise from both benign and malignant conditions. 4 It is a broad term that fails to capture the heterogeneous biology of the underlying histopathological diagnosis and associated tumour biology. 28
The classification systems developed sequentially by Ronnett et al., 26 Misdraji et al. 28 and Bradley et al. 30 are summarised in Table 1. These proposed classifications systems are used inconsistently,31,32 causing confusion for clinicians and making it impossible to compare clinical outcomes. 4 The Peritoneal Surface Oncology Group International (PSOGI) 2016 classification was achieved by international consensus in a modified Delphi process at the world congress in Berlin, 2012. 27 This nomenclature is summarised in Table 2 and classifies peritoneal disease as low or high grade.
Historical classification of appendiceal epithelial neoplasms.

DPAM, disseminated peritoneal adenomucinosis; LAMN, low-grade appendiceal mucinous neoplasm; PMCA, peritoneal mucinous carcinomatosis; PMCA-I/D, PMCA with intermediate or discordant features.
Peritoneal Surface Oncology Group International (PSOGI) 2016 classification of appendiceal epithelial neoplasms. 27

HAMN, high-grade appendiceal mucinous neoplasm; LAMN, low-grade appendiceal mucinous neoplasm.
The current gold standard World Health Organisation (WHO) 2019 fifth edition refines this further (Table 3), combining the PSOGI nomenclature and including the tiered-grading used in the American Joint Committee on Cancer of G1, well-differentiated; G2, moderately differentiated and G3, poorly differentiated.24,33,34
2019 WHO classification of appendiceal epithelial neoplasms. 33
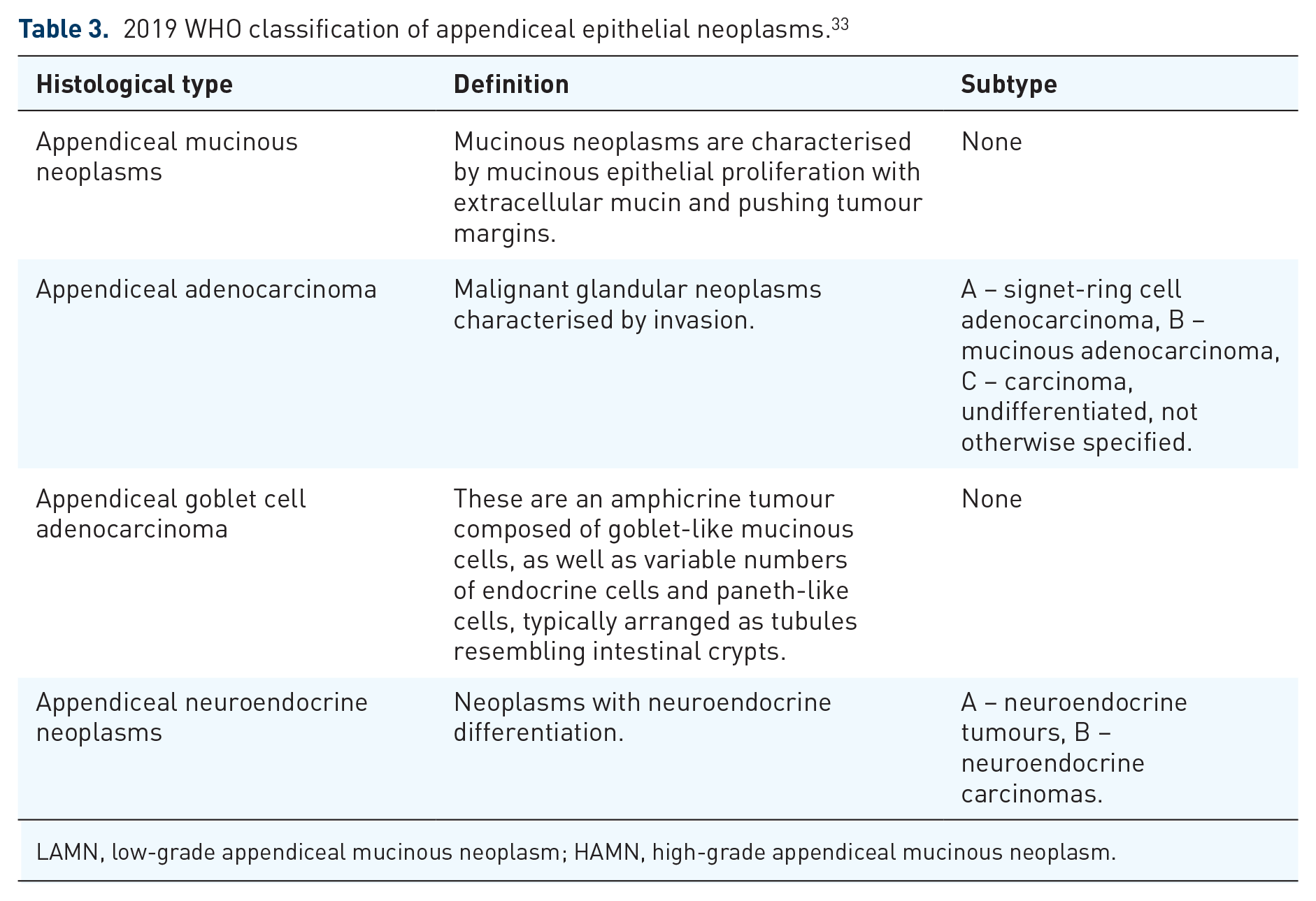
LAMN, low-grade appendiceal mucinous neoplasm; HAMN, high-grade appendiceal mucinous neoplasm.
We reconcile the heterogeneous classification used throughout the literature by describing the primary and peritoneal disease in both contemporary and historic nomenclature (i.e. that originally employed by the author) for clarity of data interpretation. We will also use the terms low-grade disease to refer to primary appendiceal mucinous neoplasms (AMNs) and high-grade disease to refer to primary appendiceal adenocarcinomas.
Methods
Key clinical databases (PubMed/MEDLINE, Scopus, Science Direct, OVID and Google Scholar) were extensively searched through 9 September 2021 with the search terms: appendix/appendiceal, cancer/carcinoma/neoplasm/malignancy and chemotherapy (systemic/intravenous, intraperitoneal/HIPEC, neo/adjuvant, peri/pre/postoperative).
Results
A total of 65 articles were found to be relevant for this review: 33 evaluating the role of HIPEC (Table 4) and 42 evaluating systemic chemotherapy (Table 5), each including 10 articles that reported on both.6,8–10,12,14–16,21,26,35–91 Table 6 is a summary of the characteristics of the literature included in this review.
Literature review of outcomes of HIPEC in addition to CRS for appendiceal cancer with peritoneal disease.

AMN, appendiceal mucinous neoplasm; CC, cytoreductive score; CRS, cytoreductive surgery; CT, chemotherapy; Cyc, cyclophosphamide; DFS, disease-free survival; DPAM, disseminated peritoneal adenomucinosis; EPIC, early postoperative intraperitoneal chemotherapy; 5FU, 5-fluorouracil; HG, high grade; HIPEC, heated intraperitoneal chemotherapy; IP, intraperitoneal; Iri, irinotecan; LAMN, low-grade appendiceal mucinous neoplasm; LG, low grade; LV, leucovorin; M, median; MD, moderately differentiated; MMC, mitomycin-C; NR, not reached; OS, overall survival; Ox, oxaliplatin; PCI, peritoneal cancer index; PD, poorly differentiated; PFS, progression-free survival; PMCA, peritoneal mucinous carcinomatosis; PMCA-I/D, PMCA with intermediate or discordant features; PMP, pseudomyxoma peritonei; adenoca, adenocarcinoma; QoL, quality of life; WD, well-differentiated; y, year.
Literature review of systemic chemotherapy for appendiceal cancer with peritoneal disease.
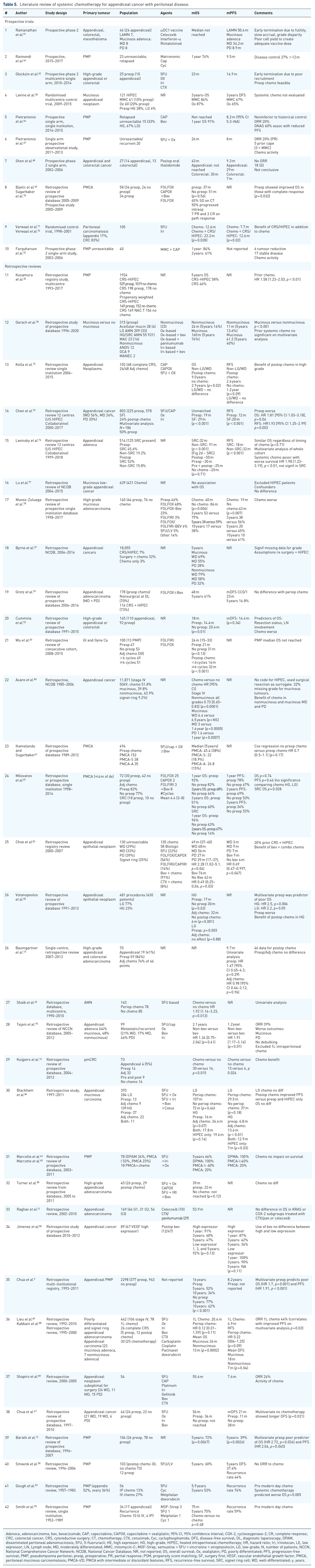
Adenoca, adenocarcinoma; bev, bevacizumab; CAP, capecitabine; CAPOX, capecitabine + oxaliplatin; 95% CI, 95% confidence interval; COX-2, cyclooxygenase-2; CR, complete response; CRC, colorectal cancer; CRS, cytoreductive surgery; CT, chemotherapy; CTX, cetuximab; Cyc, cyclophosphamide; DFS, disease-free survival; DL, diagnostic laparoscopy; DPAM, disseminated peritoneal adenomucinosis; 5FU, 5-fluoruracil; HE, high expressor; HG, high-grade; HIPEC, heated intraperitoneal chemotherapy; HR, hazard ratio; Iri, Irinotecan; LE, low expressor; LN, Lymph node; MD, moderately differentiated; MMC, mitomycin-C; MOF-Strep, semustine + 5FU + vincristine + streptozotocin; LG, low-grade; N, number of patients; NCCN, National Comprehensive Cancer Network; NCDB, National Cancer Database; NR, not reported; OS, overall survival; Ox, oxaliplatin; PD, poorly differentiated; PFS, progression-free survival; PMP, pseudomyxoma peritonei; preop, preoperative; PR, partial response; PSM, propensity score matching; SF, surgery first; VEGF, vascular endothelial growth factor; PMCA, peritoneal mucinous carcinomatosis; PMCA-I/D, PMCA with intermediate or discordant features; RFS, recurrence-free survival; SRC, signet ring cell; WD, well-differentiated; y, years.
Characteristics of literature review for chemotherapy in appendiceal cancer.

Studies that are updates of previous literature are only counted once i.e. the most recent.
10 studies included assessment of both HIPEC and systemic chemotherapy but are counted in each category for the purpose of analysis.
Other HIPEC agent: lobaplatin and docetaxel, doxorubicin/MMC/5FU, melphalan, cisplatin ± MMC, cisplatin ± doxorubicin, oxaliplatin + irinotecan, cyclophosphamide.
Other systemic chemotherapy agents: cyclophosphamide, thalidomide, panitimumab, cetuximab, gefitinib, celecoxib, carboplatin, paclitaxel, melphalan, MOF-strep (semustine, 5FU, vincristine, streptozotocin), doxorubicin, αDC1 vaccine, interferon-α, rintatolimod.
Intravenous 5FU at the time of intraperitoneal oxaliplatin is not considered separately, this is grouped as HIPEC or intraperitoneal chemotherapy.
Numbers are just appendix cancer patients where this is known.
cap, capecitabine; DFS, disease-free survival; DPAM, disseminated peritoneal adenomucinosis; EPIC, early postoperative intraperitoneal chemotherapy; 5FU, 5-fluorouracil; HG, high grade; HIPEC, heated intraperitoneal chemotherapy; LG, low grade; MCP-H, high-grade mucinous carcinomatosis peritonei; MCP-L, low-grade mucinous carcinomatosis peritonei; MMC, mitomycin-C; OS, overall survival; PFS, progression-free survival; PMP, pseudomyxoma peritonei; LAMN, low-grade appendiceal mucinous neoplasm; adenoca, adenocarcinoma; PMCA, peritoneal mucinous carcinomatosis; PMCA-I/D, PMCA with intermediate or discordant features.
Intraperitoneal chemotherapy
Cytoreductive surgery and HIPEC
The combination of CRS-HIPEC is the standard of care for appendiceal cancer with peritoneal disease.63,94 These therapies evolved together, with neither alone demonstrating success. 95 The concept of radical debulking followed by intraperitoneal chemotherapy was first described in 1969 and was followed by case series showing further benefit.6,8,11,12,58,59,96 The CRS or peritonectomy technique (surgical intention is for no residual macroscopic disease) was pioneered by Mr Sugarbaker et al.95,97 with the goal of complete cytoreduction. Compared to debulking procedures alone (surgical intention is limited removal of macroscopic disease), CRS-HIPEC has been shown to improve 5-year survival rates from about 50% to 76 to 96%.6,8,10–12,46,48,50,56,58,95,98 One randomised study by Verwaal et al.71,72 has evaluated CRS-HIPEC versus systemic chemotherapy, but only a small proportion of appendiceal cancers were included (17%) and the specific contribution of HIPEC to the improved outcomes is unclear.
It is difficult to determine the role of complete cytoreduction with or without HIPEC and disentangling the specific contributions of each therapy is the purpose of this review. Furthermore, the definitions of complete and incomplete cytoreduction vary in the literature, therefore we have adopted complete cytoreduction to include cytoreductive score (CC) 0 and 1 (no or less than 0.25 cm residual disease), and incomplete to include CC 2 and 3 (more than 0.25 cm residual disease). However, we acknowledge that further stratification may refine selection of patients for potential treatment options.
Mechanism of action and rationale of intraperitoneal chemotherapy
The aim of intraperitoneal chemotherapy after cytoreduction is to sterilise the peritoneal cavity of occult tumour cells. Intraperitoneal delivery of the chemotherapy intends to improve drug exposure to the peritoneal surface, which is limited with systemic administration. 75 Active drug concentrates in tissue to a few millimetres; therefore, resection to a minimal volume is required for definitive treatment.12,61,99,100 Further studies confirm immediate chemotherapy is needed so that tumour cells do not seed in surgical adhesions.61,101 Factors associated with altered HIPEC clearance include the extent of resection, contracted peritoneal space and completeness of cytoreduction.102,103
Evidence for benefit of HIPEC in addition to CRS
The specific question of whether HIPEC contributes to improved outcomes compared to CRS alone has not been directly addressed. However, there is reasonable evidence in the literature of case series to suggest independent benefit in both appendiceal adenocarcinoma and mucinous neoplasms.35,37,39,42,47,52,55,57
Shaib et al. 42 attempted to answer this by evaluating their institutional registry data from multiple centres including one which did not use HIPEC. Of the 163 patients included, 65 had complete cytoreduction with most (78%) receiving HIPEC. A clear improvement in overall survival (OS) was seen in patients who received HIPEC even after adjustment for HIPEC-treating centre and extent of surgical resection [hazard ratio (HR) 0.42, 95% confidence interval (CI) 0.24–0.73, p = 0.002].
Another study inadvertently resulted in a control group (no HIPEC) as HIPEC was introduced midway through the review period. 52 This enabled a comparison between 60 patients (23 appendiceal cancer) treated with CRS with or without HIPEC and demonstrated an improved survival in those patients who received HIPEC. This study is limited by a mixed tumour population and so the specific contribution of the role of HIPEC for appendiceal cancer is less clear.
Most single-centre retrospective studies lack a comparable control arm. For example, an Indian study evaluating their experience of 33 cases of appendiceal and CRC peritoneal metastases reported improved 4-year OS for CRS-HIPEC compared to CRS alone. 39 However, the CRS alone group represented the poorer prognostic group where HIPEC delivery was abandoned due to high burden disease, which would not achieve complete cytoreduction.
Large database analyses, such as that of the National Cancer Database (NCDB), can only bluntly evaluate the role of HIPEC.37,77,80 Although these analyses suggest a survival benefit of the addition of HIPEC to surgery, the comparison of CRS-HIPEC compared to surgery alone is flawed as the surgery alone group likely represents patients who undergo debulking procedures rather than complete cytoreduction, which is known to be suboptimal for disease control.9,95
A recent retrospective study evaluating outcomes of CRS-HIPEC compared to CRS-alone is the best evidence to date of the efficacy of HIPEC. This study evaluated 1924 patients with PMP from more than 20 centres over 24 years. 35 The addition of HIPEC to CRS in PMP was associated with a 35% reduction in the risk of death (HR 0.65, 95% CI 0.50–0.83). This was a statistically robust cohort study with propensity matched analysis and inverse probability treatment weighting. Subgroup analysis further confirmed the benefit of HIPEC in both low and high-grade disease and those with complete and incomplete cytoreduction. However, there were several limitations of this study. The reasons the CRS-alone group did not receive HIPEC were not available. 104 Additionally, the registry itself was set up for prospective data collection in 2010, yet the period captured was from 1993 requiring a long period of retrospective data collection with incomplete data in 45% of the patients in the registry.
The only randomised trial evaluating HIPEC compared mitomycin-C (MMC) to oxaliplatin in 121 patients with appendiceal cancer with peritoneal disease, but given outcomes are similar in each arm the ultimate benefit of HIPEC in addition to CRS is unable to be evaluated. 38
A systematic review and meta-analysis compiled from single case reports and case series demonstrated most studies had 5-year OS similar to expected. 23 Therefore, mortality was minimally affected by the different treatment regimens. This analysis is flawed by the quality of the studies evaluated making the assessment of the role of HIPEC compared to no HIPEC challenging.
While all studies investigating the addition of HIPEC to CRS in appendiceal cancer are flawed and results are mixed, no data suggests worse survival outcomes with HIPEC. On the whole, the data available suggests a probable benefit for HIPEC supporting its continued use.
Evidence for HIPEC after debulking surgery
Historically, most patients who underwent CRS or debulking received concomitant intraperitoneal chemotherapy. However, the poor outcomes of patients who undergo debulking despite also receiving HIPEC questions its utility. The addition of HIPEC to debulking surgery appears not to improve OS in one Swedish study comparing 110 patients who had CRS-HIPEC compared to 40 patients who had debulking and HIPEC. 94 However, recent literature contradicts this suggesting the even those patients with incomplete cytoreduction benefit.35,42
The challenge in this setting is that a proportion of patients who have a surgical intention of debulking (anticipated macroscopic residual disease) can still achieve surgical outcomes of no residual macroscopic disease. This occurred in 25% of the debulking group in the study by Andreasson et al. 94 It is this subgroup of patients that may still benefit from provision of HIPEC.
Therefore, the literature currently supports current practice of abandoning HIPEC in the setting of gross macroscopic residual disease and continuing its use in the setting of achieving complete cytoreduction (regardless of the initial surgical intention).
Hyperthermia
Hyperthermic infusion of chemotherapy was first clinically demonstrated after cytoreduction in 1980. 105 Hyperthermia alone may confer a therapeutic benefit by inducing heat stress in tumour cells, denaturing proteins and impairing DNA repair mechanisms, especially important after a cytotoxic insult.106,107 Heat improves delivery of the chemotherapy into tissues and has a synergistic effect with particular cytotoxic agents.108–113
The optimal temperature was established initially by in vitro studies showing 42.5°C was more effective than 39°C. 114 Elias et al. 61 evaluated HIPEC at 43°C, the maximal tolerable temperature in animals, but found higher morbidity and mortality compared to other published literature.
Mitomycin-C HIPEC
MMC is the most ubiquitous of agents used for HIPEC with most of the evidence for the combination of CRS-HIPEC historically using this agent.6,8,16,35,36,38,41,42,46,47,49–52,54–57,63
In the randomised control trial that compared MMC- to oxaliplatin-HIPEC, no significant difference was found in survival rates consistent with prior data.38,54 In a follow-up study, patients reported improved physical and functional well-being from oxaliplatin-HIPEC.115,116
The recent PSOGI registry analysis did not show survival benefit in the group receiving MMC-HIPEC compared to no HIPEC (HR 0.93, 95% CI 0.65–1.34) noting that this was not the most common agent in this cohort and has flaws of retrospective analysis. 35
Other clinical considerations in choice of HIPEC agent include prior receipt and intended sequencing of perioperative chemotherapy. This has not been clearly evaluated in the literature, and current clinical practice at most institutions favour initial MMC-HIPEC to facilitate initial oxaliplatin-based systemic chemotherapy to reduce the dose-limiting risk of neurotoxicity.115,117
Oxaliplatin HIPEC
The use of oxaliplatin-HIPEC has been controversial in the CRC setting. 118 In appendiceal cancer, some studies show similar outcomes compared to MMC-HIPEC.38,119,120 In the recent PSOGI registry study, oxaliplatin-HIPEC was shown to reduce the risk of death compared to no HIPEC (HR 0.42, 95% CI 0.19–0.93). 35 However, only two patients in the HIPEC group are evaluable at 5-years compared to 47 in the no HIPEC group, due to significant attrition in this group or short follow-up. 104 Oxaliplatin-HIPEC has also been shown to have survival benefit when compared to irinotecan-HIPEC. 43
Cisplatin and mitomycin HIPEC
This regimen was shown to be an efficacious regimen in the recent PSOGI registry study with HR 0.57 (95% CI 0.42–0.78). 35 This regimen was originally developed for treatment of gastric cancer peritoneal metastases based on preclinical data showing the synergistic effect of MMC enhancing intracellular accumulation of platinum adducts.121,122 The benefits shown in this study are consistent with the effect demonstrated in ovarian cancer. 123 Further randomised study is needed to compare the benefit of this regimen to that of MMC- and oxaliplatin-HIPEC.
Other HIPEC regimens
Data concerning other HIPEC regimens with comparable safety profiles, but no meaningful improvement in efficacy are summarised in Table 4.
Perioperative systemic chemotherapy
The literature evaluating the role of perioperative chemotherapy is conflicting with some studies showing benefit and others suggesting worse outcomes in patients with appendiceal cancer (Table 5). We use the terms pre- and postoperative chemotherapy, as the terms neoadjuvant and adjuvant used in this literature as descriptors imply a ‘curative/definitive’ treatment intent, which is not always applicable. The term perioperative is used when both pre- and postoperative chemotherapy is evaluated, but does not imply that patients have received both pre- and postoperative chemotherapy.
The decision-making process regarding selection of patients for upfront surgery is beyond the scope of this review but involves an assessment of the patients’ histological subtype, rate of disease progression, burden of disease and fitness for surgery. Multidisciplinary team assessment at an expert peritoneal tumour service is essential. Ultimately this decision is based on the likelihood of the CRS-HIPEC procedure to result in a complete cytoreduction with no or minimal residual disease. This is an important concept to bear in mind when reading this review, as patients selected for upfront ‘preoperative’ chemotherapy usually have disease or patient-related factors that suggest complete cytoreduction may not be achieved. This creates bias by selecting patients with poorer prognoses.
Studies suggesting benefit of perioperative chemotherapy
There have been two prospective trials in the perioperative setting. The COMBATAC trial was a prospective phase 2 single-arm study, which reported on 25 patients of whom 10 had appendiceal cancer.65,124 Patients received 3 months of pre- and postoperative chemotherapy using FOLFOX or FOLFIRI with cetuximab. The median progression-free survival (PFS) was 14.9 months, which met the target threshold and is comparable to the literature.14,69,125 Although this study supports the role of systemic chemotherapy, there is no control arm and no subgroup analysis of the appendix cancer cohort so further conclusions about the role of systemic chemotherapy is limited.
The other prospective trial of perioperative chemotherapy assessed the role of postoperative thalidomide following CRS-HIPEC in 27 participants of whom 14 had appendiceal cancer. 68 The median PFS was 9.3 months and failed to meet the prespecified threshold. Given the limited role of anti-angiogenic agents in the unresectable setting, it is unlikely that this treatment will be pursued further in the perioperative setting.66,82,86
Asare et al. 80 interrogated the NCDB and reported on 5049 patients with stage IV appendiceal adenocarcinoma. In the nonmucinous group, there was an improvement in survival with chemotherapy compared to no chemotherapy (HR 0.73, 95% CI 0.65–0.83). In the mucinous group, there appeared to be no benefit from chemotherapy (HR 0.95, 95% CI 0.86–1.04). This effect is driven by the lack of benefit in the well-differentiated group (median OS 6.4 years versus 6.5 years, p > 0.05), but with survival improvement in the moderate and poorly differentiated groups (3.0 year versus 1.6 year, p = 0.0005; 1.6 year versus 1.0 year, p = 0.0007). 80 A flaw with this study is that HIPEC may have been included as systemic chemotherapy, especially as the 5-fluorouracil (5FU) bolus given with oxaliplatin-HIPEC is administered intravenously. Furthermore, the NCDB has no specific code for CRS-HIPEC, which amplifies the potential inaccuracies of this analysis.
Studies suggesting no benefit of perioperative chemotherapy
The subsequent study of the NCDB by Lu et al. 77 focused on the role of chemotherapy in 639 patients with stage IV well-differentiated mucinous appendiceal adenocarcinomas. The majority (90%) of this patient population underwent a surgical resection. Patients who had chemotherapy had improved 5-year OS (61% versus 53%), but after multivariate analysis including surgical resection, there was no association of benefit with chemotherapy (HR 1.1, 95% CI 0.82–1.4). This study further supports the concept that well-differentiated mucinous adenocarcinomas remain relatively chemoresistant despite the increasing use of combination chemotherapy regimens.80,93
Further evidence of the lack of benefit of perioperative systemic chemotherapy is demonstrated in a retrospective study of 393 (72% low-grade) patients from two high-volume centres. 84 Very few had perioperative chemotherapy (13 pre- and 9 postoperative) making conclusions difficult, but lends support for a lack of benefit in low-grade disease (median PFS 30 months versus 37 months, p = 0.18; median OS 109 months versus 72 months p = 0.46). Low-grade appendiceal tumours are thought to be more resistant to conventional cytotoxic agents. 126 Although it should be noted with prolonged survival in low-grade disease, any absolute benefit of perioperative chemotherapy will need to be of sufficient magnitude to provide a clinically meaningful benefit.9,38
Studies that have not been conclusive about the role of perioperative chemotherapy have been those with mixed tumour populations.14,15 For example, one retrospective review included high-grade (moderate or poorly differentiated) peritoneal carcinomatosis from 19 (41%) appendiceal cancer patients in a cohort of CRC. The perioperative chemotherapy status was not known for all of the cohort, and not described specifically for the appendix compared to the colorectal primary subgroups. The median PFS of 9.5 months seems slightly optimistic for this high-grade cohort where expected PFS is around 6 months for both colorectal and appendiceal cancers with peritoneal metastases. Furthermore, this study is likely underpowered for follow-up as median follow-up is shorter than median PFS (with no CI described and many early censored cases).
A study demonstrating survival benefits in OS and PFS regardless of timing of systemic chemotherapy only had a small proportion of appendix origin tumours (5%) compared to the remaining cohort of CRC. 14 Therefore, the true benefit of perioperative chemotherapy for the appendix cancer cohort is difficult to extrapolate.
Studies suggesting worse outcomes with perioperative chemotherapy
Some studies, albeit with significant limitations, suggest that perioperative chemotherapy could in fact cause harm.35,83,92 Thus, assessing which patient groups may be at increased risk of harm from a proposed treatment is essential.
High-grade disease is expected to derive potential benefit from perioperative chemotherapy due to biological aggressiveness and is recommended before and/or after CRS at multiple institutions, regardless of data suggesting otherwise.83,126 However, the literature does not necessarily support this. For example, Cummins et al. 92 evaluated 165 patients, 110 with high-grade appendiceal cancer, which compared outcomes to 55 CRC with peritoneal metastases. Most of the cohort received perioperative chemotherapy. Preoperative chemotherapy was associated with poorer OS (14 months versus 20 months, p = 0.01) and postoperative chemotherapy with improved OS outcomes (5 months versus 35 months, p < 0.0001).
Furthermore, Levinsky et al. 76 evaluated prognostic factors of a subgroup of 125 patients with appendiceal adenocarcinomas with signet ring cells from the same study population described by Chen et al. 75 They have not described the proportion of signet rings cells within the tumour specimen. so it is unclear if these meet the current classification for signet ring adenocarcinoma (more than 50% signet ring cells). Multivariate analysis of the entire cohort suggested worse OS in those receiving systemic chemotherapy (HR 1.98, 95% CI 1.23–3.19, p < 0.01).
This propensity for high-grade ACs to have worse OS after perioperative chemotherapy could be explained by selection bias, given both these studies are retrospective. However, in the second study described by Levinsky et al., 76 the majority of the non-signet ring cell subgroup included well-differentiated adenocarcinomas. These well-differentiated adenocarcinomas are conversely, less chemosensitive, which could also account for worse outcomes with systemic chemotherapy. This argument is strengthened by the study by Shaib et al. who in their multicentre study demonstrated worse survival outcomes in patients with LAMN who received perioperative chemotherapy compared to those who did not (HR 1.92, 95% CI 1.14–3.23, p = 0.013). 42 This demonstrates the selection bias that although lower-grade and well-differentiated tumours may have more indolent biology, at the time clinicians select patients for systemic chemotherapy, the burden of the disease is such that it is more imminently life-threatening. Thereby, if systemic agents do not achieve response, these patients will have worse overall outcomes.
Chemotherapy regimens
Further limitations affecting retrospective studies evaluating systemic chemotherapy is the lack of detail regarding the systemic agents used which is either not recorded in the large databases or difficult to attain from centralised records when delivered in the community.15,77,80 Studies that provide a more detailed insight into treatment regimens confirm that perioperative chemotherapy choice was predominantly fluoropyrimidine and oxaliplatin especially in high-grade disease.40,41,65,74,75,78,79,81,84,85,93
Perioperative chemotherapy in the presence of signet ring cells
Signet ring cells, present in the tumour in any proportion, have been shown to predict for poor survival outcomes.41,76,127–129 Despite small patient numbers, Milovanov et al. 81 showed a large difference in OS from their cohort of 28 patients with signet ring cells out of 70 patients with peritoneal mucinous carcinomatosis (PMCA) (high-grade peritoneal disease) with 1-year OS with preoperative chemotherapy 94% versus 43% in those without chemotherapy (p = 0.028). However, there was no difference seen when a similar analysis was done in a larger cohort of high-grade histology, suggesting that this result may occur from confounding, or chance. In this study, subgroup analysis of those with signet ring cells found a nonsignificant shorter median survival of 25 months in those who received preoperative chemotherapy compared to 39 months for those who did not receive chemotherapy (p = 0.18). 78
One study specifically looked at prognostic variables in 514 patients with appendiceal adenocarcinoma of whom 125 (24%) had signet ring cells. 76 Multivariate analysis of the entire cohort suggested worse OS in those receiving systemic chemotherapy (HR 1.98, 95% CI 1.23–3.19, p < 0.01). However, after multivariate analysis of the signet ring cell subgroup, there was no statistical difference between those who received perioperative chemotherapy compared to those who did not (HR 1.69, 95% CI 0.50–5.68, p = 0.4). 76
Another study of 142 patients with poorly differentiated or signet ring cell appendiceal adenocarcinoma of whom 19 (13%) had signet ring adenocarcinoma and 9 (6%) well or moderately differentiated with focal signet ring cells revealed a 44% response rate in 78 patients who received first-line chemotherapy. 87 One-fifth (20%) of this cohort proceeded to CRS with or without HIPEC. In the group of patients who had complete cytoreduction, 5 had preoperative and 12 had postoperative chemotherapy, but with the assessment of outcomes challenging due to small numbers (recurrence-free survival (RFS), HR 0.22, 95% CI 0.04–1.25; OS HR 0.12, 95% CI 0.01–1.59).
Outcomes for poorly differentiated and signet ring cell appendiceal adenocarcinomas appear to be more comparable to that of CRC than low-grade disease.87,92 This provides some support for extrapolation of treatment regimens for this histological subtype.
Duration of perioperative chemotherapy
The optimal duration of perioperative chemotherapy is not clearly defined. This is most challenging to assess in the preoperative setting, given the conflicting efficacy results.
The approach in most studies is 3 months of chemotherapy followed by clinical and/or surgical reassessment and then a decision for another 3 months or to proceed to surgery. Following surgery, a further 3 to 6 months of postoperative chemotherapy is often considered.70,85,126 For patients with borderline disease, 6 months of treatment might be optimal, if there is an initial response to ensure maintenance of the response. However, for clearly resectable disease upfront, 3 months may be preferable to reduce the risk that chemoresistant disease may progress to be unresectable. 70
One study evaluated the duration of postoperative chemotherapy and demonstrated an OS benefit for six or more cycles of chemotherapy compared to less than six cycles. 40 Unfortunately, this retrospective study was a heterogeneous mix of different cancer types including CRC and gynaecological malignancy and is confounded by the likelihood that patients with prolonged survival will receive more treatment.
Preoperative chemotherapy
Timing of perioperative chemotherapy is important and forms an important discussion point in multidisciplinary meetings. Reasons to consider preoperative or neoadjuvant chemotherapy are summarised in Table 7. Unfortunately, the current literature does not clearly guide the specific role of preoperative chemotherapy. Therefore, recommendation for preoperative chemotherapy remains a case-by-case discussion with individualised treatment decisions.
Summary of reasons to consider preoperative or postoperative chemotherapy in patients with appendiceal cancer with peritoneal disease.

A number of studies suggest that preoperative chemotherapy is associated with worse survival outcomes.12,13,35,58,75,78,83,92 Often the intent is for systemic control of disease, potential downstaging and for observation of disease biology.73,75,85 Therefore, even those who remain or become candidates for CRS-HIPEC will likely have additional disease-related factors that portend a poor prognosis, confounding outcomes.
A large multicentre cohort of 803 patients with appendiceal peritoneal metastases of whom 225 (28%) had preoperative chemotherapy. 75 Following propensity scored matching and multivariable analysis, preoperative chemotherapy was associated with worse outcomes (RFS HR 1.93, 95% CI 1.25–2.99; OS HR 1.81, 95% CI 1.02–3.118).
Another large retrospective multicentre registry study also confirmed that preoperative systemic chemotherapy independently predicted poor survival in a cohort of patients with appendiceal peritoneal disease (30% high-grade). 9 After multivariate analysis, chemotherapy was not associated with detriment in the low-grade group, but predicted poor OS in the high-grade group (HR 1.75, 95% CI 1.2–2.6, p = 0.005). In both of these studies, unmeasured variables leading to selection bias must be considered in addition to a true detriment of administering preoperative chemotherapy, potentially from delays to definitive surgery.
Worse outcomes are not confined to the high-grade group where they might be expected. A recent registry study demonstrated that in a cohort of patients with low- and high-grade PMP receiving CRS, prior systemic chemotherapy was significantly associated with increased risk of death (HR 1.58, 95% CI 1.23–2.03, p < 0.001). 35 This persisted after propensity matching and sensitivity analyses to control for selection bias.
A study by Votanopoulos et al. 83 in 2015 evaluated 481 patients with both low- and high-grade disease. Preoperative chemotherapy predicted for worse OS in both low-grade (HR 2.2, p = 0.05) and high-grade tumours (median OS 17 months versus 32 months, p = 0.02; HR 2.5, p = 0.006) on multivariate analysis.
Baratti et al. 12 evaluated 104 patients with PMP (78 low-grade DPAM, 26 high-grade PMCA). Five-year OS of 72% is consistent with the predominant histology of LAMN compared to adenocarcinoma. Previous chemotherapy was associated with worse OS (HR 2.72, p = 0.033) and PFS after multivariate analysis (HR 2.04, p = 0.045). This is consistent with chemoresistance in the low-grade subgroup.
There are a number of different explanations why preoperative chemotherapy predicts for worse survival outcomes. The role of selection bias was interrogated in the recent PSOGI registry study. 35 Analysis of 1571 excluded patients (192 preoperative chemotherapy, but 958 missing data) revealed similar balance of prognostic factors, except for increased early postoperative intraperitoneal chemotherapy (EPIC) use in the excluded group, but improved rates of 10-year OS and lower severe morbidity suggest the presence of unmeasured confounders. A number of studies also comment that the decision for preoperative chemotherapy sometimes occurs externally to the centralised referral institution.83,84 This results in suboptimal selection of patients for preoperative chemotherapy with functional deterioration in addition to potential delay in definitive CRS. 84 Another theory postulated is the potential for selection pressure of the chemotherapy on chemoresistant clones.9,81
Despite some literature suggesting worse outcomes with preoperative chemotherapy, there are some intriguing aspects that emerge suggesting further biological insights. A prospective consecutive cohort of 34 patients with high-grade PMCA were treated with preoperative 5FU + oxaliplatin.69,70 Most patients (65%) had the full treatment course of 6 months. Although there was no improvement in OS in patients who received preoperative chemotherapy compared to those who did not (median OS 51 months versus 37 months, p = 0.56), there were 10 (29%) patients who had a histological response. These patients did not reach median OS compared to 29.5 months in those who did not achieve histological response (p = 0.032). There were no clinical variables identified that could help predict histological response, but this is compelling data and contrasts to data in the unresectable setting, which suggests mucinous tumours may have poorer outcomes with systemic chemotherapy. 93
Further data points to potential short-term benefits of preoperative chemotherapy in appropriately selected patients including evaluation of 45 high-grade mucinous adenocarcinoma patients. 85 There was no difference in OS based on their primary analysis, but calculation of OS from date of initial therapeutic intervention showed a nonsignificant trend to worse OS in those who had preoperative chemotherapy consistent with other literature suggesting this association.9,12,35,75,78,83,92 However, in this study, there was a high response rate to chemotherapy of 58%, and no patient experienced disease progression. This is consistent with other data also suggesting high rates of stable disease following preoperative chemotherapy. 84 This raises the possibility that ongoing postoperative chemotherapy in those demonstrated to have initial response and tolerability may be warranted in this high-risk group to maintain suppression of disease beyond CRS.
These studies outline the challenges in the literature assessing accurate response to chemotherapy. Mucinous peritoneal disease can be notoriously hard to visualise. Intraoperative assessment of response was shown to be discordant with radiographic assessment, particularly concerning is the increased rate of true progression shown intraoperatively (50%) compared to only 20% by imaging. 70 Other literature evaluating chemotherapy by different response criteria have attempted to compensate for this issue with some investigators recommending a ‘modified peritoneal RECIST criteria’.64,73,89,131
Decision-making around preoperative chemotherapy for this rare and specialised cancer should be reserved for expert multidisciplinary meeting and early referral to these services essential. It also emphasises the importance that reference centres continue to audit outcomes and improve data retention to minimise the loss that comes when patients are treated at external locations. 15
Postoperative chemotherapy
In contrast to the literature evaluating preoperative chemotherapy, outcomes after postoperative chemotherapy appear to be more favourable.83,84 This likely reflects refined patient selection of a fit population, pathological characteristics, post CRS-HIPEC and also the lack of control arm, as the comparator is those who receive preoperative chemotherapy, which overall show worse outcomes. Principles that favour postoperative chemotherapy are summarised in Table 7.
The theme that emerges from the literature is a trend for benefit of postoperative systemic chemotherapy in high-grade tumours.69,74,84 Kolla et al. 74 evaluated the role of postoperative chemotherapy in a retrospective cohort of 103 appendiceal cancer patients. There was a benefit of chemotherapy for non-low-grade tumours with a median OS 9 years compared to 3 years for the low-grade group (p = 0.02) in those who had complete cytoreduction. However, the analysis grouped chemoresistant LAMNs with well-differentiated adenocarcinomas, so any signal for benefit in the adenocarcinoma group will be lost.
In another retrospective review of 430 patients with mostly low-grade disease, postoperative chemotherapy showed no survival benefit for low-grade tumours on univariate analysis (p = 0.88) but conferred a significant benefit for high-grade tumours (median OS 32 months versus 6 months, p < 0.001). 83
Further evidence for a benefit of postoperative chemotherapy in high-grade tumours is shown by a retrospective study by Blackham et al. 84 Of 109 patients with high-grade tumours, those who received postoperative chemotherapy had prolonged median PFS compared to preoperative chemotherapy and HIPEC alone (14 months versus 7 months versus 7 months, p < 0.001). A similar trend to improved OS was not statistically significant (36 months versus 16 months versus 20 months, p = 0.07).
Although systemic chemotherapy was associated with worse survival outcomes in a particularly poor prognostic cohort of appendiceal adenocarcinoma with signet ring cells, this study demonstrated no statistical difference between the timing of pre-, post- or perioperative treatment (p = 0.71). 76
The timing of postoperative chemotherapy also allows selection of high-risk patients based on pathological factors such as positive lymph nodes. Lymph node status is not routinely assessed as part of the patients’ diagnostic work-up prior to CRS; however, there is literature demonstrating lymph node involvement portends worse prognosis and in high-grade disease may select patients who benefit from systemic chemotherapy.
Lymph node involvement may counter-intuitively be a later phase in appendix cancer progression as peritoneal disease can occur directly due to transcoelomic spread without spread to lymph nodes as an intermediary. 4 Baumgartner et al. 15 raised the question of focusing chemotherapy strategies on the lymph node positive group, which was a strong predictor of OS and had short PFS. Kuijpers et al. 14 also suggested the benefit of systemic chemotherapy in lymph node positive disease in those with peritoneal carcinomatosis, but there were questions raised regarding the influence of surgical complications. This was also a predominant CRC population making conclusions difficult for the appendix cancer subgroup.
Cummins et al. 92 concluded that all high-grade appendiceal tumours with positive lymph nodes should have systemic chemotherapy. However, both positive lymph nodes and chemotherapy were associated with poorer survival, and their data do not interrogate if there is any difference in lymph node positive patients who received chemotherapy compared to those who did not. Another study evaluated positive lymph node status as a predictor of improved survival from perioperative chemotherapy but revealed no advantage. 84 While Votanopoulos et al. 83 commented on their institutional approach to lymph node positive patients, they did not explore in their data the proportion and outcomes of chemotherapy in this group. It is also worth noting that lymph node status as a possible selection tool is likely limited to higher-grade disease as a recent study after propensity matching did not demonstrate positive lymph nodes to be prognostic for OS in patients with low-grade disease. 35
Patients who have incomplete cytoreduction (CC 2/3) are a specific subgroup worthy of discussion. Firstly, they have a known volume of residual disease and so could bear similarity to patients who have not had any form of CRS. Therefore, chemotherapy decisions should be based on evidence from the unresectable studies discussed in detail below. This fits with a trend to improved PFS being demonstrated in this patient subgroup without an OS benefit. 84
The discrepancy in outcomes between pre- and postoperative chemotherapy is consistent with the ultimate need to improve selection of patients for these therapies. The literature clearly defines the contrast between those patients who achieve favourable outcomes compared to those who do not with existing treatments. Lymph node involvement in high-grade cases may be a strategy to help select patients who might benefit from systemic chemotherapy, but more evidence is needed to support this hypothesis. While the search for improved therapeutic strategies is important, delineating predictive biomarkers is also critical.
Systemic anticancer agents for unresectable disease
There is limited literature demonstrating the activity of systemic chemotherapy in the unresectable setting, with few prospective trials.21,64,66,67,73,82,86,89 These are summarised in Table 5.
The rare nature and centralised speciality care pathways for treating this disease in most countries leads to some degree of referral bias in the literature. Firstly, more severe cases are likely to be referred for treatment at speciality centres, leading a bias towards poorer outcomes. Conversely, patients with advanced disease whom local physicians pre-empt a pathway of best supportive care may never refer their patient for specialty centre management. Fitter, motivated patients are more likely to be seen at specialised centres, which would lean a possible bias to improved outcomes in patients that are reported from these centres. Most of the literature arises from centralised referral centres, so meaningful chemotherapy data is often missed from patients treated in the community.
5-Fluorouracil or capecitabine
The chemotherapy backbone in appendiceal cancer is 5FU or capecitabine as in CRC. In a single institutional retrospective analysis of chemotherapy agents, 30% of the participants were treated with single-agent 5FU or capecitabine. 89 It is likely that single-agent treatment was given in the advanced setting to those less fit for combination chemotherapy.
The benefit of capecitabine in combination with cyclophosphamide and mitomycin has been demonstrated by two studies.64,73 These were both small cohorts, but demonstrated activity of these agents with disease control rate of 27% in the first by Raimondi et al. and a clinical benefit rate of 38% in the second by Farquharson et al. The Raimondi study of low-grade disease included those with progressing disease on consecutive scans, whereas the Farquharson study of both low and high-grade PMP did not require this. The Farquharson study demonstrated 1-year OS of 84% and 2-year OS of 61%. 73 Interestingly, two patients originally deemed unresectable achieved CRS following treatment with this regimen. This study attempted to overcome the challenges of measuring radiological responses by systematic application of disease volume assessment, discrete deposit measurement and compressive effects on intraperitoneal organs assessed by experienced radiologists.
Oxaliplatin-based combinations
Oxaliplatin and fluoropyrimidine chemotherapy regimens have been the mainstay of CRC chemotherapy for many decades.132–136 Its use has been extrapolated to the treatment of settings of appendiceal cancer and is often the preferential first-line regimen.87,89,93
The best evidence for this doublet treatment is from a single-arm prospective study by Pietrantonio et al. 67 This study evaluated survival outcomes in 20 consecutive patients with unresectable or recurrent PMP (low- and high-grade) treated with FOLFOX-4. This was a high-burden disease group and 45% patients achieved stable disease. This compares to a 24% partial response rate in a retrospective study by Shapiro et al., 89 which did not use RECIST and defined partial response as any degree of response. Median PFS was 8 months and median OS 26 months in the Pietrantonio et al. 67 study. This contrasts to longer median OS of 56 months likely driven by a mostly well-differentiated tumour population in the retrospective analysis by Shapiro et al. 89 The poorer survival in this prospective study is not inconsistent with a high-burden disease population, some of whom have high-grade disease. Two of the six initially unresectable patients underwent laparotomy with one achieving complete CRS.
Tejani et al. 93 demonstrated activity and similar survival outcomes (response rate 39%, median PFS 1.2 years and median OS 2.1 years) in a select group of 112 appendiceal adenocarcinomas from the National Comprehensive Cancer Network (NCCN) database. The majority of this cohort (71%) received combination chemotherapy of 5FU or capecitabine and oxaliplatin. Worse survival outcomes were shown in those with mucinous and poorly differentiated tumours. Whether this is due to chemotherapy being less efficacious for these subgroups or poorer disease biology is difficult to tell without a comparator arm. 80
The impact of systemic chemotherapy on the survival outcomes in both these studies is unclear, but would appear to not overly influence the natural trajectory of high-burden unresectable disease. The modest response rate suggests some degree of treatment activity and confirms oxaliplatin-based treatments as having the best evidence in this setting.
Irinotecan-based combinations
There are small numbers of appendiceal cancer patients treated with irinotecan-based combinations.82,89 No further details are provided in these studies on the outcomes of this subgroup due to low patient numbers. In the retrospective analysis by Lieu et al., 87 there was a trend towards improved PFS in patients who had the first-line irinotecan (1.0 year versus 0.5 year, p = 0.07). In the control arm of the Verwaal et al.71,72 randomised control trial, a small number of patients were given single-agent irinotecan as the second-line chemotherapy agent. No further detail is available about these patients to draw any conclusions.
Biologic therapy
Anti-angiogenic agents
Vascular endothelial growth factor (VEGF) expression has been shown to be associated with poor OS in appendiceal cancer.137–139 Studies that have evaluated the role of anti-VEGF therapy with bevacizumab are conflicting, confounded by the chemotherapy regimen.66,82,86,87,93 The most recent was a prospective phase 2 study that evaluated 15 patients with PMP (mostly low-grade) who relapsed after prior CRS-HIPEC and received capecitabine and bevacizumab. 66 Median PFS was 8.2 months (95% CI 5.3–not assessable) which met the pre-specified non-inferiority threshold of 5 months, and median OS was not reached, with 1-year OS 91%. Three of the 15 patients had a partial response (20%).
Choe et al. 82 evaluated the role of biological therapy through analysis of 130 of 353 patients with appendiceal cancers. Most patients (91%) received bevacizumab in addition to combination chemotherapy, so that the comparison is to those who received single-agent chemotherapy. Median PFS was improved with bevacizumab compared to no bevacizumab (9 months versus 4 months, HR 0.69, 95% CI 0.470–0.995). Median OS was improved by 34 months for patients receiving additional bevacizumab, but this finding could be confounded by the use of combination chemotherapy.
Jimenez et al. 86 evaluated differential VEGFR-2 gene expression in a cohort of 59 of 89 patients with peritoneal carcinomatosis from appendiceal cancer. Twelve of the 47 high expressors received adjuvant bevacizumab. There was no statistical difference between these groups, yet there was a trend to better outcomes in VEGFR-2 low expressors, noting that this comparison is underpowered.
Conversely, a subgroup analysis of 112 patients with appendiceal adenocarcinoma (51% received bevacizumab) suggested worse PFS in those who received bevacizumab (HR 1.91, 95% CI 1.17–3.14, p = 0.01) and no advantage to OS. 93 Most patients who received bevacizumab had combination chemotherapy, likely reflecting selection of more aggressive disease biology; however, this study does not support the use of bevacizumab in treatment of advanced appendiceal adenocarcinoma.
There are no studies that provide strong evidence for the benefit of the addition of anti-angiogenic agents to systemic chemotherapy agents in the unresectable setting.
Molecular-directed therapy
EGFR-inhibitors
In the study by Shapiro et al., 89 11 (20%) of the patients studied received biologic therapy alone or in combination with chemotherapy. Five patients were treated with gefitinib alone, which is interesting as this is not an extrapolated CRC regimen.
Another study by Choe et al. 82 also included a small subgroup of patients treated with anti-EGFR monoclonal antibodies in addition to systemic chemotherapy. In this group, OS outcomes were worse, (18 months versus 20 months; HR 3.83 95% CI 1.04–14.14). The authors have not described RAS testing and the study period included the time prior to knowledge of the lack of benefit from EGFR-antibodies in RAS-mutant CRC. 140
In the COMBATAC trial, perioperative systemic chemotherapy (48% 5FU + oxaliplatin; 48% 5FU + irinotecan) and cetuximab was given to KRAS wild-type appendiceal cancer patients in a cohort of CRC.65,124 No additional conclusions for the role of cetuximab could be made from this study as the appendiceal cancer subgroup was not evaluated independently.
There was a trend to prolonged OS in 20 of 49 KRAS wild type patients who received cetuximab or panitumumab. However, in another study of 149 of 600 patients who had molecular testing, there was no statistical difference compared to those who did not receive an EGFR inhibitor (median OS 68.4 m versus 51.7 m, p = 0.83). 21
Cox inhibitors
In one study evaluating molecular markers, 30% of the patients found to have cyclooxygenase-2 (COX-2) expressing tumours received selective COX-2 inhibition with celecoxib. 21 Median OS was not statistically different between those receiving celecoxib compared to those who did not (57.6 months versus 55.7 months, p = 0.84).
Immunotherapy
In the current era of immunotherapeutics, appendiceal cancers are considered ‘cold’ tumours as they lack the ability to initiate an effective immune response, and few are MMR-deficient or high tumour mutation burden.20,141,142
Given the recency in advances for the role of immunotherapy in MSI-high cancers, there is sparing literature on appendiceal cancer patients receiving this treatment. Lu et al. 77 reported in their cohort of stage IV well-differentiated mucinous adenocarcinomas that 5% received immunotherapy.
A novel approach of a dendritic vaccine (αDC1) as an adjuvant treatment in combination with immunomodulators celecoxib, interferon-α and rintatolimod was evaluated in a phase 2 study including 24 patients with appendiceal cancer following CRS-HIPEC (7 LAMN, 16 mucinous adenocarcinoma). 91 This study was hampered by technical issues with difficulty isolating adequate cells to achieve the target dose and ultimately the trial was stopped prematurely for futility, slow accrual and grade disparity in the appendiceal cancer group with PFS for low- and high-grade tumours 50.4 and 8.9 months respectively.
Conclusion and future directions
This review reconciles the evidence for the role of intraperitoneal and systemic chemotherapy for the treatment of appendiceal cancer with peritoneal disease. Challenges to reaching definite conclusions include retrospective study designs and broad study populations due to disease rarity and inconsistent use of tumour nomenclature due to changing classification systems. Heterogeneous chemotherapy regimens, inadequate chemotherapy data due to the centralised nature of the surgical service with local delivery of chemotherapy and difficulty accurately measuring radiological treatment responses further complicate interpretation.
It is clear from the literature that the addition of HIPEC to complete cytoreduction has survival benefits with a more limited role of HIPEC in the setting of incomplete cytoreduction.
There is contradictory evidence as to the benefit of perioperative chemotherapy in the setting of appendiceal cancer, especially of any additional benefit to that of complete CRS-HIPEC. Studies of preoperative chemotherapy generally appear to be associated with worse survival outcomes, although this group will be most influenced by selection bias. Studies of postoperative chemotherapy generally show some degree of benefit, especially in high-grade disease with presence of signet ring cells and lymph node involvement as possible selection tools. Based on evidence from this review there is a minimal role of perioperative systemic chemotherapy in addition to complete CRS-HIPEC for low-grade mucinous peritoneal disease (from AMNs) and that use of systemic agents should be reserved for use within clinical trials.
Systemic chemotherapy agents demonstrate some activity in the treatment of unresectable disease, but it remains unclear the optimal way to refine selection of patients who can benefit. This review also highlights the necessity of thorough collection of information about systemic agents and ensuring accuracy of clinical outcomes. Furthermore, there are a number of unstudied treatment strategies such as maintenance chemotherapy, intermittent-dosing or histology-tailored therapy that should be the focus of future prospective study.
Ultimately, further studies of the same agents are unlikely to yield more meaningful or convincing information. There is an urgent need for novel treatment agents and strategies. Preclinical and translational research models that interrogate the biological nature of this rare and unpredictable malignancy are needed to help postulate rational therapeutic development. We propose a translational medicine platform where we can interrogate the true biology of each individual patients’ tumour and microenvironment. By establishing robust preclinical models and evaluating a multi-omic profile using cutting edge technology such as single-cell RNA sequencing and spatial transcriptomic analysis of these tumours and their microenvironment, our research group is attempting to rationally identify targets and pathways of novel and repurposed therapeutic strategies. In the meantime, individualised treatment decisions should be made in the setting of a multidisciplinary discussion at a high-volume appendiceal cancer treatment centre and international collaboration is vital for the design of feasible prospective studies that can evaluate these clinical dilemmas more definitively.
