Abstract
Background:
The paradigm of adjuvant treatment for patients in pathological stages II and III of non-small-cell lung cancer has changed toward the adjuvant combination of chemotherapy and immune checkpoint inhibitors, neoadjuvant chemoimmunotherapy, or ultimately periadjuvant chemoimmunotherapy. The introduction of a new standard requires significant changes in preoperative assessment. A real description of its current state in Poland is required, which can form the basis for systemic changes.
Objectives:
The aim of our study was to analyze the current standards for preoperative assessment in Poland.
Design:
Observational study.
Methods:
A comprehensive survey was conducted across seven thoracic surgery centers in Poland, focusing on preoperative pathological examination techniques, imaging methodologies, multidisciplinary team consultations, and the interval between imaging and surgical resection.
Results:
The survey analyzed data from 459 patients who were operated between January 2024 and October 2024. The most common pathological diagnostic methods were intraoperative frozen section, and preoperative core biopsy. Almost all patients in stages pIA-IIIB had contrast-enhanced computerized tomography (CT) of the chest, positron emission tomography (PET/CT), and bronchoscopy before surgery. In Poland, brain imaging is not a standard part of the preoperative assessment, and brain scans are obtained only exceptionally (pooled median 0% of patients in stages IA–IIIB). The median time between the CT and surgery was 61 days (range 30–90 days), and between PET/CT and surgery 35 days (range 10–66 days). The median time between the surgery and pathological report was 17 days (range 5–30 days). Multidisciplinary tumor boards are organized in the postoperative period for all patients comparing to pooled median 50% in preoperative period. In the vast majority of cases, the expression status of Programmed Death Ligand-1 and next-generation sequencing were carried out only in postoperative samples.
Conclusion:
Current compliance with perioperative assessment standards in Poland is inadequate. The consequence of this situation may be low availability of perioperative treatment protocols.
Plain language summary
The standard of treatment for patients with operable stage non-small cell lung cancer has changed with the advent of the possibility of complementing surgery with immunotherapy or targeted drugs. Such treatment allows for significant improvements in treatment outcomes. The introduction of this new standard requires significant changes to the treatment qualification process and the tests performed as part of the patient’s preoperative assessment. A true description of its current state is needed, which can form the basis for systemic changes. The aim of this study was to analyse the current standards of preoperative assessment in Poland. A multidisciplinary survey was conducted in 7 thoracic surgery centres in Poland. Topics included questions about methods of preoperative pathological examination, imaging procedures, multidisciplinary team discussion and the period between imaging and resection. The survey analysed data from 459 patients operated on between January 2024 and October 2024. The most common pathological diagnostic methods were intraoperative frozen slices and preoperative core biopsy. Almost all patients in stages pIA-IIIB had a contrast-enhanced chest CT scan, PET/CT and bronchoscopy before surgery. However brain imaging was performed in an inadequate proportion of patients. Multidisciplinary tumor boards were most commonly organized in the postoperative period. In the vast majority of cases, the expression status of PD-L1 and next-generation sequencing were carried out only in postoperative samples. The current adherence to the standards for perioperative evaluation in Poland is insufficient. The consequence of the defined situation can result in the low accessibility of perioperative treatment protocols.
Keywords
Introduction
Surgical resection followed by adjuvant chemotherapy for pathological stages II and III was the most effective method of treatment of patients with early-stage non-small cell lung cancer (NSCLC) until 2021.1–4 In general, 5-year survival of the patients did not exceed 60% in stage II and 36% in stage III. 5 This was related to the incremental effect of platinum-based adjuvant chemotherapy in patients with NSCLC 1 ; however, low adherence to the complete surgical protocol and four cycles of adjuvant chemotherapy was reported.6,7 The paradigm of adjuvant chemotherapy has changed to the adjuvant combination of chemotherapy and immune checkpoint inhibitors,8,9 neoadjuvant chemoimmunotherapy, 10 or periadjuvant chemoimmunotherapy.11–13 Targeted therapies have also completed the altered horizon of treatments in early NSCLC.14,15
Patients with stage III NSCLC benefit the most from perioperative treatment, but at the same time, most patients diagnosed with stage III lung cancer are unresectable at the time of diagnosis, and chemoradiotherapy was established as the standard of care for unresectable stage III NSCLC after the publication of the PACIFIC trial.16–18
The debate regarding the appropriate definitions of resectability and radical resection continues both in Poland and globally. Among Polish thoracic oncology specialists, the definition of resectability provided by the International Association for the Study of Lung Cancer (IASLC) is the most widely accepted and has also been recommended in the latest multidisciplinary consensus of Polish experts. 17 This new classification provides a more accurate categorization of patients for resectability, but the decision on treatment remains a matter of multidisciplinary team (MDT) consensus and should be made individually, especially in view of the resectability consensus proposed above.17,19
Until now, protocols have focused on surgical treatment of stages I–II NSCLC, and decisions regarding adjuvant systemic treatment were only made in the postoperative period. Recent advances enabling neoadjuvant chemoimmunotherapy demand a broader preoperative evaluation. A truly multidisciplinary discussion must consider a comprehensive evaluation of the patient and tumor biomarker status. Concurrent chemoradiotherapy (cCRT) should also be considered as an option for patients with locally advanced disease. The PACIFIC, CheckMate-816, and Keynote-671 studies all documented a significant improvement of disease-free and overall survival, and this should be emphasized. However, proper patient selection is essential for optimal treatment in each case. 16
In Poland, approximately 22,000 new cases of lung cancer are diagnosed annually, with about 4000 patients undergoing surgical resection, almost exclusively performed by board-certified thoracic surgeons. The national network is structured around dedicated Lung Cancer Units, primarily based in academic centers, with at least 1 unit in each of the 16 regions. Smaller centers contribute minimally and are typically not integrated into multidisciplinary care pathways. 20 In Poland, NSCLC diagnosis procedures and treatment are fully reimbursed by the National Health Care Fund, ensuring that patients do not incur out-of-pocket costs. As of October 2025, many therapeutic options are covered by reimbursement, as listed below. For locally advanced stage III patients, the most common approach is concurrent platinum-based chemoradiotherapy followed by consolidative durvalumab or osimertinib for EGFR-positive patients. In adjuvant settings, atezolizumab has been reimbursed since January 2024 for patients with Programmed Death Ligand-1 (PD-L1) expression ⩾50%, pembrolizumab since April 2025 for patients with PD-L1 expression <50% (since April 2025), and for patients with anaplastic lymphoma kinase (ALK) or epidermal growth factor receptor (EGFR) positive NSLCC, alectinib (since April 2025) and osimertinib (since January 2025), respectively. Treatment involving preoperative immunochemotherapy was reimbursed in July 2024—first as a regimen based on the results of the CheckMate-816 study, and a few months later as a regimen with pembrolizumab based on the Keynote-671 study. The expanding scope of multidisciplinary management protocols for stages II–III NSCLC requires substantial adaptation of previous existing clinical pathways in Poland. Patients in these stages should be systematically discussed in MDT meetings, to ensure optimal qualification for neoadjuvant or peri-adjuvant treatment strategies. Recent evidence confirms that structured organization of MDT contributes to improved overall survival in patients with NSCLC. 21 Polish national guidelines integrate recommendations from IASLC and the European Society of Medical Oncology (ESMO). 17 However, the implementation of these guidelines into everyday clinical practice seems incomplete. The underlying reasons for this suboptimal adoption are not fully understood. Introducing innovative standards into well-established and convenient routines often meets resistance due to institutional inertia, limited resource allocation, and cognitive comfort with existing workflows. As a result, even evidence-based recommendations can require sustained leadership engagement and structured change-management strategies to achieve effective integration into clinical practice.
The purpose of our study was to analyze the current standards of clinical practice for preoperative evaluation across different centers in Poland. Our aim was to identify any gaps and areas for improvement in order to optimize the process of assessing and qualifying patients for perioperative treatment. To the best of our knowledge, no similar multicenter data have been published to date.
Materials and methods
Between the October 1, 2024, and the November 15, 2024, thoracic surgeons from comprehensive cancer centers of thoracic surgery in Poland were asked to complete an originally developed multiarea survey (Supplemental File 1). The centers were chosen arbitrarily by the study team in order to adequately balance different regions of Poland. All responding centers were multispecialty teaching hospitals in Poland, selected to ensure representativeness in terms of institutional profile and geographical distribution. Being a multispecialty teaching hospital that constituted an inclusion criterion for participation in the study. The selection process was not based on nationwide or fully random sampling. The questions covered preoperative pathological examination, preoperative imaging procedures, data related to MDT, and the interval between imaging and resection.
The study data collected from the centers did not allow the identification of individual medical records of patients at any point in the analysis. Data were sent to a central database developed for this study. After the data were sent to the leading center, information about the sources of the statistical data was removed. Descriptive statistics were applied to summarize the data. The results were presented in tables and illustrated using bar charts. The reporting of this study conforms to the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement (Supplemental File 2.)
Results
In total, the survey analyzed data from 459 patients across 6 centers in Poland: Thoracic Surgery Department, Medical University of Gdansk, Department of Thoracic Surgery, Poznan University of Medical Sciences, Poznan, Department of Thoracic Surgery, Medical University of Lublin and Department of Thoracic Surgery, Holy Cross Oncological Centre, Kielce, Department of Thoracic Surgery and Tumours, Oncology Centre, Bydgoszcz, Department of Thoracic Surgery, National Research Institute of Chest Diseases, Warsaw, Clinic of Thoracic Surgery, General and Oncological Surgery, USK Nr 2, Lodz, Thoracic Surgery Department, The Center for Pulmonary Diseases, Olsztyn. Thoracic surgeons employed in the centers provided retrospective data from registries and electronic medical records, ranging from 35 to 100 patients per center, operated between January 1, 2024, and October 21, 2024. The data were accepted as submitted from the centers with no further validation. Centers were queried on the methods used to obtain pathological diagnoses during the perioperative period. The data are presented in Table 1. Individual patient data were not aggregated at any point in the study. The most common method of acquiring pathological diagnoses in stage II was a core biopsy (stage IIA median 33.3%, range 0.0%–100.0%). There is a trend toward more patients undergoing operations with preoperative diagnoses obtained via core biopsy, although discrepancies between centers remain significant. Most of the patients in stage I have their pathological diagnosis obtained on the basis of an intraoperative frozen section examination (median 36.4% in stage IA, range 10.0%–88.9%). When all operated patients are taken as a whole including the most commonly represented pathological stage I—the most common methods of reaching the pathological diagnosis in early-stage patients were intraoperative frozen section and preoperative core biopsy. Endobronchial navigation and fine needle biopsy are the least common methods of determining the pathological diagnosis. The data are presented in Figure 1.
Methods of obtaining the pathological diagnosis in the perioperative period.
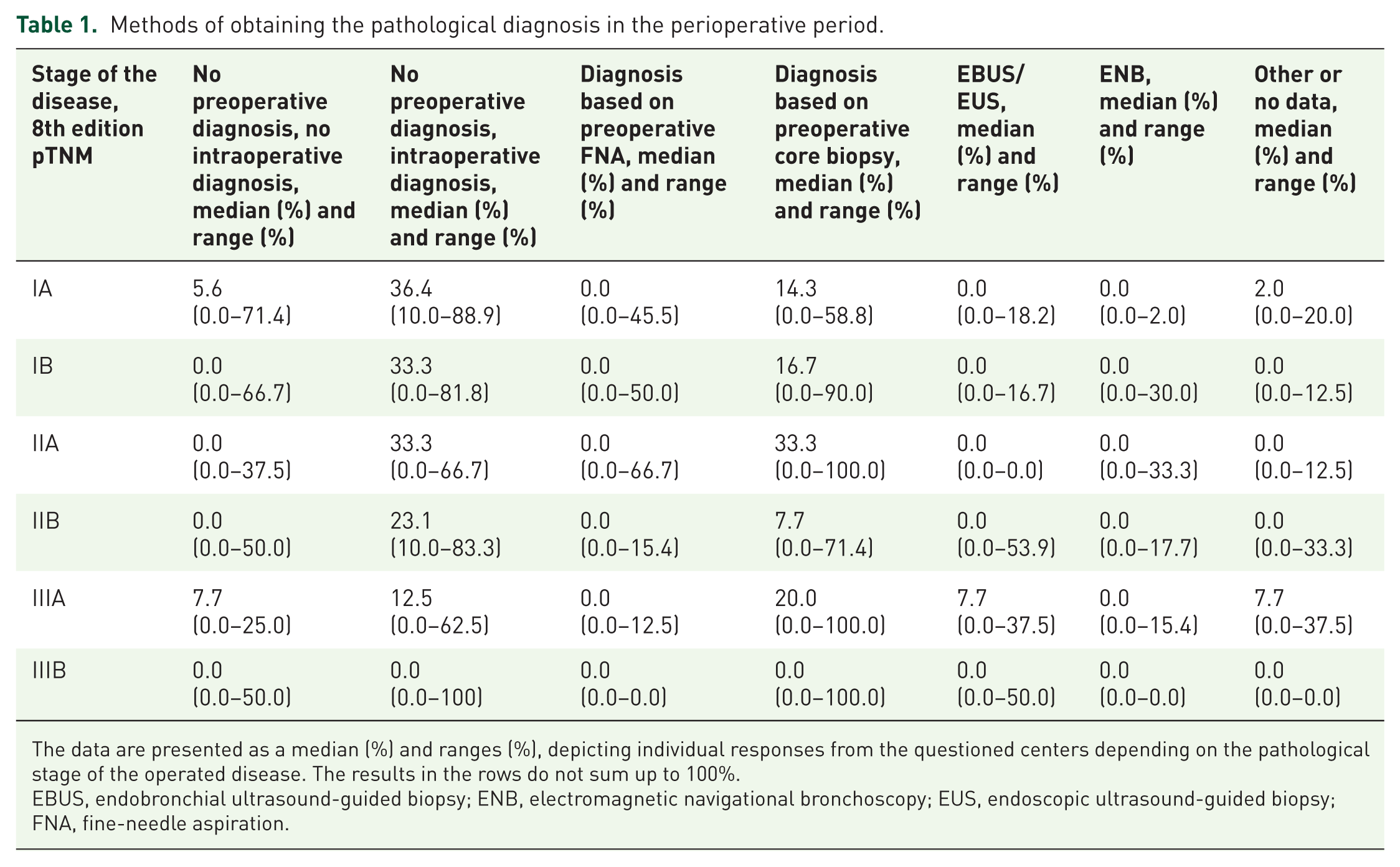
The data are presented as a median (%) and ranges (%), depicting individual responses from the questioned centers depending on the pathological stage of the operated disease. The results in the rows do not sum up to 100%.
EBUS, endobronchial ultrasound-guided biopsy; ENB, electromagnetic navigational bronchoscopy; EUS, endoscopic ultrasound-guided biopsy; FNA, fine-needle aspiration.

Methods of perioperative pathological diagnosis were questioned in an aggregated group of all patients reported by the centers.
Almost all patients in stages pIA–IIIB had chest computerized tomography (CT) of the chest with intravenous contrast, positron emission tomography (PET/CT), and bronchoscopy performed in the preoperative period. Despite its importance, PET/CT is not universally performed in all patients in advanced stages. This study cannot provide the reader with exact numbers but indicates that there are centers in Poland, where one-third of patients with stage IIIA do not undergo preoperative PET/CT. However, only a negligible percentage of patients underwent brain imaging, regardless of the stage of the disease (pooled median 0% of patients in stages IA–IIIB; Table 2). The median time between CT and surgery was 61 days (range 30–90 days), and between PET/CT and surgery was 35 days (range 10–66 days). The median time between surgery and the pathological report was 17 days (range 5–30 days).
Imaging and invasive procedures performed preoperatively.
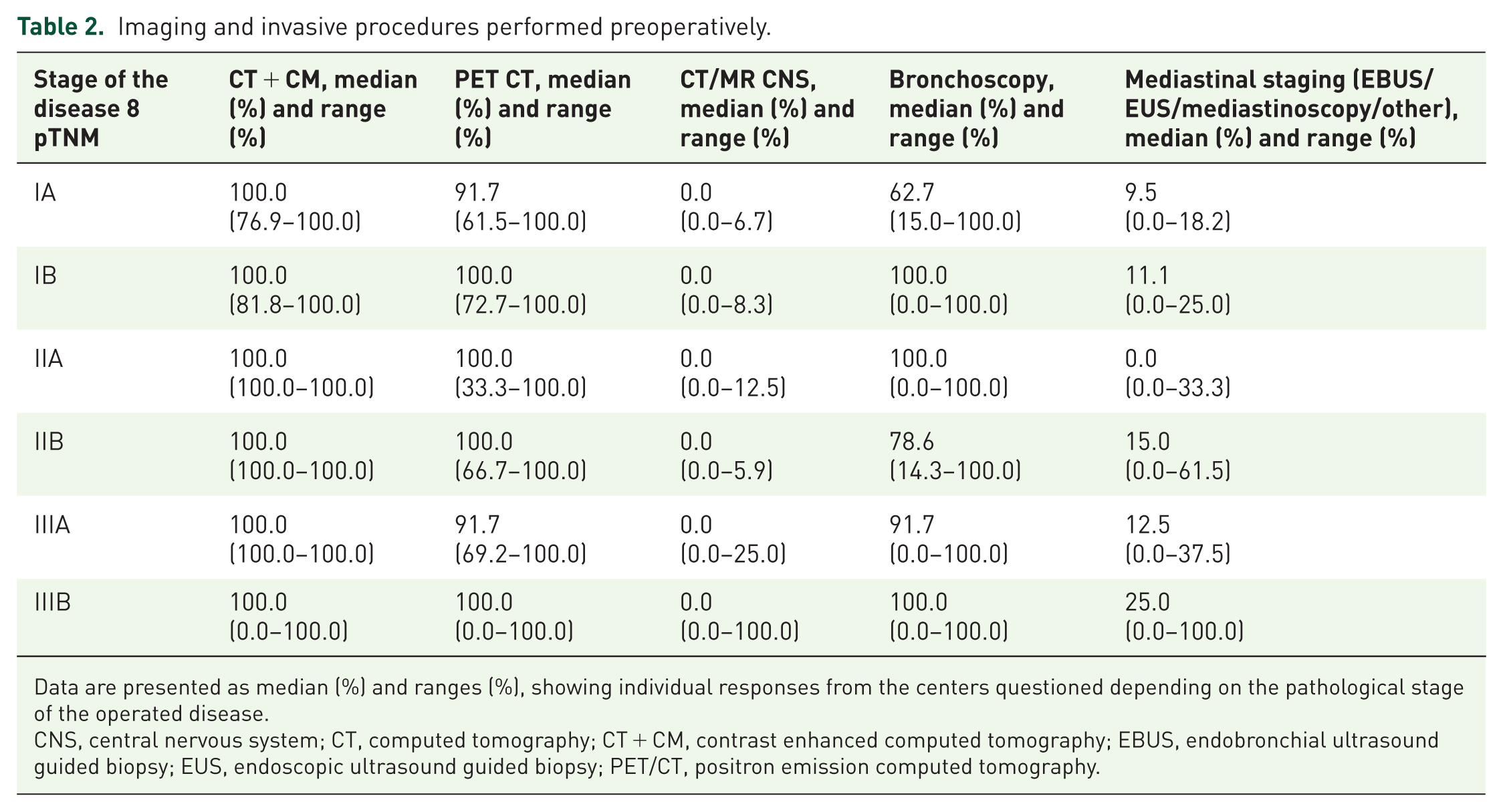
Data are presented as median (%) and ranges (%), showing individual responses from the centers questioned depending on the pathological stage of the operated disease.
CNS, central nervous system; CT, computed tomography; CT + CM, contrast enhanced computed tomography; EBUS, endobronchial ultrasound guided biopsy; EUS, endoscopic ultrasound guided biopsy; PET/CT, positron emission computed tomography.
MDT meetings in Poland are predominantly convened postoperatively, with discussions focused primarily on resected specimens. Preoperative MDTs are rarely conducted in stage IA disease, accounting for approximately 5% of cases, and their frequency increases with advancing stage. Nevertheless, the majority of patients with stage III disease do not have access to preoperative MDT consultations. Overall, MDT discussions are held preoperatively in about 50% of cases. The distribution of patients who underwent preoperative MDT evaluation is illustrated in Figure 2. Surgeons make therapeutic decisions largely independently, without routinely consulting MDT meetings prior to surgery. However, once the complete pathological report is available, nearly all cases are subsequently discussed during postoperative MDT consultations.

The proportion of patients who had multidisciplinary team discussions depended on the pathological stage of the disease. The whiskers represent the range of the responses depending on the center.
In all centers that responded to the questionnaire, radiation oncologists and medical oncologists as well as thoracic surgeons are present during MDT. Patients were included in the decision-making process in 21% of centers (Figure 3). MDTs are organized 1–2 times a week with a median of 18 patients (range 9–50) discussed during a single meeting. In contrast to the attendance list during MDTs, radiation oncologists are employed in only 43% of the centers (Figure 4). Radiation oncologists, geriatricians, and social workers are employed in 43%, 14%, and 71% of hospitals organizing lung cancer treatment, respectively, which limits access to their expertise when formulating comprehensive management plans. This structural gap is particularly relevant for patients with stage III disease, in whom radiotherapy often represents the therapeutic backbone and must always be considered as an alternative to surgery. The lack of in-house geriatric and social care specialists may also reduce the likelihood of delivering adequately tailored treatment for high-risk or elderly patients with age-related limitations.

Attendance list during the multidisciplinary tumor board. The percentages represent the proportion of centers where each individual participant participates in the multidisciplinary tumor board.

Medical specialists employed at the center, which organizes a multidisciplinary tumor board.
The centers were asked about the proportion of patients who undergo next-generation sequencing (NGS) assessment and analysis of PD-L1 expression on the operative samples or small samples harvested preoperatively in all patients with NSCLC. In most cases, the diagnosis was conducted using postoperative samples to assess both PD-L1 expression and NGS. Postoperative evaluation of PD-L1 expression was more frequently performed than NGS, with a higher percentage of cases observed in advanced stages compared to stage I. The data are presented in Figures 5 and 6.
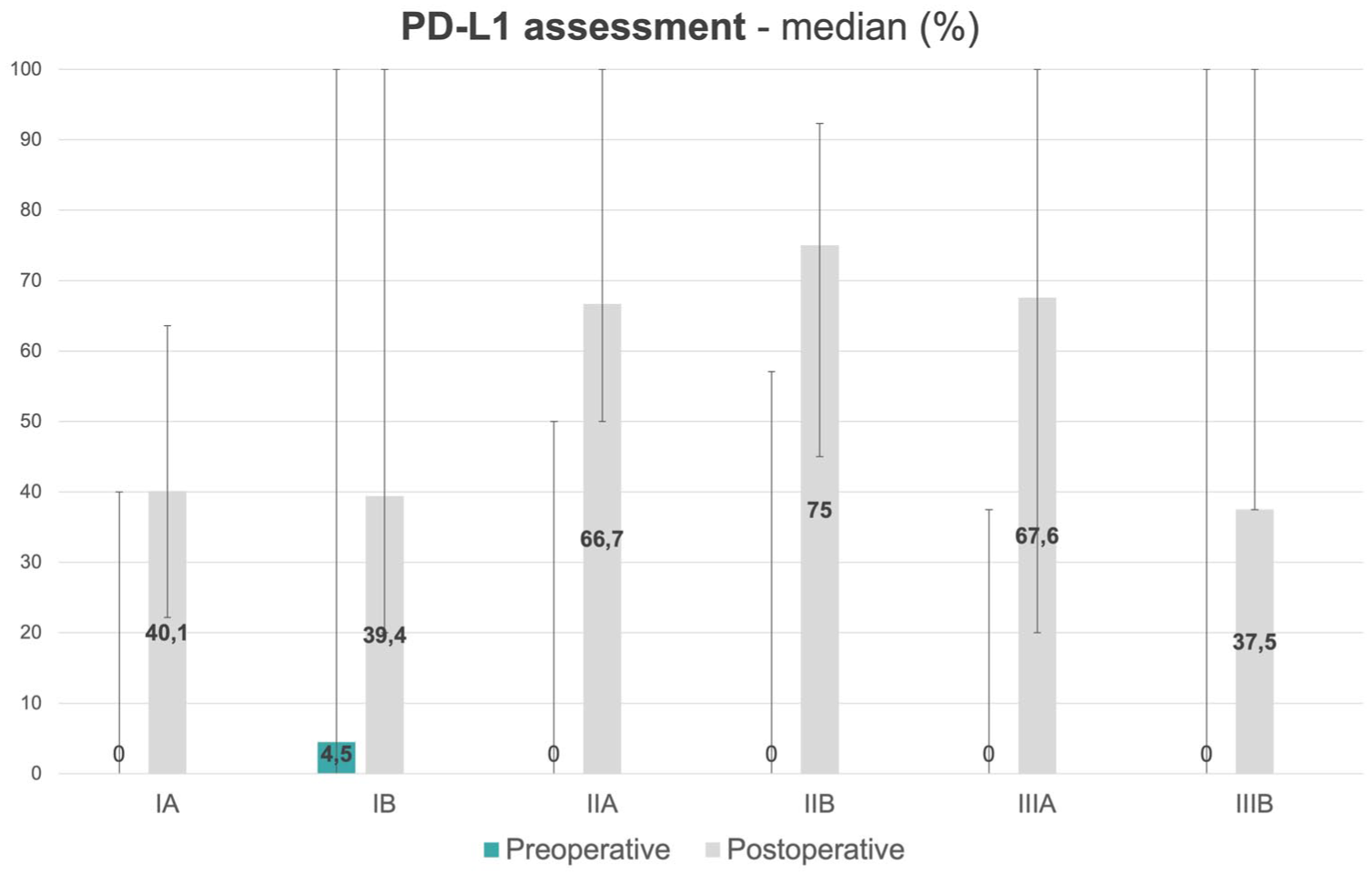
The proportion of patients who had the evaluation of PD-L1 expression in the preoperative or postoperative period, depending on the clinical stage of the disease. The whiskers represent the range of the responses depending on the center.

The proportion of patients who had NGS in the preoperative or postoperative period depending on the clinical stage of the disease. The whiskers represent the range of responses depending on the center.
Discussion
In 2014, the ESMO guidelines stated that histological diagnosis in patients with early-stage lung cancer before surgery is desirable, but may not be possible in certain groups of patients due to objective difficulties and that tumor malignancy can be determined by PET/CT. 22 Now, in 2025, it has become mandatory to assess not only tumor histology but also biomarkers, including predictive factors for immunotherapy and targeted therapies. Implementing new and detailed diagnostic guidelines will require close collaboration between thoracic surgeons and other specialists, the use of modern diagnostic tools, and, most importantly, education to improve the skills and knowledge of perioperative management.
The purpose of this study was to analyze the current standards of clinical practice for preoperative evaluation across different centers in Poland. Our aim was to identify any gaps and areas for improvement in order to optimize the process of assessing and qualifying patients for perioperative treatment. It is important to note that the wide implementation of preoperative and perioperative chemoimmunotherapy requires complex and comprehensive cooperation between different medical specialties. This article may contribute to the development of new standards for the preoperative assessment of NSCLC patients in Poland. Treatment based on preoperative immunochemotherapy according to the CheckMate-816 regimen was reimbursed in July 2024, marking the beginning of an evolution in thinking about the treatment and diagnosis of patients eligible for surgical treatment.
Diagnostics—Objectives and challenges
The clinical staging of a patient eligible for surgical treatment for stages II and III NSCLC should be based on a CT scan of the chest and upper abdomen, PET/CT, and brain imaging. This approach provides the most accurate description of the extent of disease. In our cohort, almost all patients underwent PET/CT, while brain imaging was not performed in a significant number of patients. The prolonged interval between diagnostic imaging (CT and PET/CT) and surgical intervention represents a critical limiting factor for effective treatment delivery in NSCLC. In our cohort, the median time from CT to surgery reached 61 days, which substantially exceeds the 30-day interval recommended by ESMO guidelines. Reducing this delay is expected to improve treatment outcomes by minimizing disease progression during the waiting period and ensuring timely access to definitive therapy. An expert assessment of the initial CT scan, followed by a coordinated decision to simultaneously order complementary studies such as PET/CT, endoscopic diagnostics, and brain imaging, could reduce the diagnostic bottleneck; however, systemic constraints within healthcare delivery remain a major limitation to minimizing delays before the initiation of radical treatment. In Poland, access to magnetic resonance imaging (MRI) and CT imaging of the brain is reimbursed for oncological patients regardless of their stage and diagnosis. However, the lower number of MRI scanners compared to CT scanners often results in a preference for CT brain imaging due to its higher availability. The availability of MRI scanners in Poland is about three times lower than the Organisation for Economic Co-operation and Development average (0.6 vs 1.8 per 100,000 inhabitants). In contrast, the number of CT scanners is relatively similar, at around 2 per 100,000 inhabitants. 23 Similar data have been published showing a discrepancy between over 85% of patients staged with PET/CT and only 10%–23% of patients who undergo brain MRI (by clinical stage). 24 Current ESMO guidelines exactly indicate the need for brain MRI in stages II–III NSCLC prior to the treatment. 16 The low percentage of patients undergoing brain imaging (e.g., up to 5.9% in stage IIB, up to 25.0% in stage IIIA) may reflect poor adherence to clinical practice guidelines as well as limited access to MRI, and we believe that efforts to change this situation are worthwhile—it is important to highlight that approximately 10% of individuals diagnosed with NSCLC will develop brain metastases. Early detection of these metastases is crucial, as timely intervention with local treatments such as stereotactic radiosurgery, whole brain radiotherapy, and surgical resection can improve clinical outcomes. A complete assessment of the clinical stage prior to surgery, including brain imaging, is necessary for optimal planning of the management strategy.
The main purpose of the pathological evaluation of biopsy or cytological samples is to provide an accurate diagnosis and preserve material for molecular studies. Although formalin-fixed paraffin-embedded tissues remain the preferable material, most laboratories accept other specimen types. In particular, testing on cell blocks is highly recommended if it is the only available material or the best material. Material acquired using minimally invasive techniques, including endobronchial ultrasound-guided biopsies, may be insufficient to perform histological, biomarker, and molecular diagnostics. In such cases, histological protocols dedicated to small biopsy examinations may help to conserve the material. The use of circulating tumor DNA acquired by liquid biopsy has been proposed for initial molecular studies. However, such an approach cannot be treated as a substitute for tissue material and is not recommended for patients with early NSCLC. 25 The minimal analytical threshold and lower detection limit should be ensured to fully utilize NGS-based methods. Specifically, for DNA and RNA sequencing methods, the percentage of cancer cell nuclei in the sample plays a greater role than the number of cancer cells; nevertheless, the NGS-based test usually requires at least 1000–5000 cells, with 20% constituting cancer cells. As such, the preservation of the biological material used for histological and immunohistological tests is critical. 26
Similarly to locally advanced and metastatic NSCLC, PD-L1 testing in early lung cancer is no longer optional for completely resected tumors. Data from phase III trials show that neoadjuvant immune checkpoint inhibitors plus chemotherapy increase the complete pathological response (pCR) rate compared with chemotherapy alone in stages IB–IIIA NSCLC, leading to a shift in clinical practice. 27 There are four commercially available FDA-approved PD-L1 IHC staining protocols, each using different monoclonal antibodies (SP142, SP 263, 22C3, or 28-8) and their scoring system. Since the assays are fully interchangeable, it may be necessary to adjust the assay to the drug for which it was developed. 28 Furthermore, there is a considerable discrepancy between the results of the PD-L1 evaluation in biopsies and postresection samples. This results from heterogeneous patterns of PD-L1 expression in tumors (including focal or aggravated expression) and leads to underestimation of PD-L1 expression. While lower positivity thresholds appear to decrease the error margin, more studies are needed. 29
In the cohort of patients analyzed, a proportion of patients—12%–33% of patients depending on clinical stage—were not diagnosed before surgical treatment. In our opinion, this is a high percentage and is particularly relevant in stage III patients, who benefit most from preoperative immunochemotherapy. One reason for this may be the belief that the short time between initial imaging and surgery correlates with better surgical results and that it is, therefore, a “waste of time” to undergo pathological diagnostics and wait for results.30,31 However, it should be emphasized that previously published data on patients before the era of perioperative immunochemotherapy and the timeframe should be interpreted differently now. At the same time, it is worth noting that authors from other countries also outline that for patients with early NSCLC, surgical removal is still considered a diagnostic and therapeutic procedure to avoid extending the time to obtain a final diagnosis. 32
In our cohort, almost no patients had PD-L1 expression evaluated preoperatively. This is correlated with the relatively low proportion of patients who had preoperative samples taken. Given the postoperative eligibility of patients with PD-L1 expression ⩾1%, this is clearly an area for improvement. Furthermore, based on experience from biomarker testing in metastatic disease, we know that in many centers, molecular abnormalities and PD-L1 expression are tested sequentially, significantly extending the start of treatment.
Although NGS testing in Poland is reimbursed, there are organizational disparities between nonacademic and academic centers. This issue is primarily due to limited access to reference pathological diagnostics in smaller centers and insufficient funding for PD-L1 expression testing. Academic centers, with higher productivity, offer more comprehensive biomarker tests. Similar to other European countries, Poland is transitioning from polymerase chain reaction sequential testing and immunohistochemistry screening to NGS testing, including broad gene panels. Liquid biopsy is not commonly used in early-stage NSCLC patients. The ongoing implementation process involves adapting appropriate testing and reporting standards. Variations in turnaround time between centers, low clinician awareness, and inadequate tissue availability and management contribute to access disparities, not only in Poland but also in other European countries.33,34 There is limited literature on what percentage of patients in other countries have their biomarker status assessed as part of preoperative management. Agelaki et al. presented a retrospective analysis of 303 patients collected from 16 Greek centers. Of this group, 75% underwent surgical resection, 20% of whom received neoadjuvant treatment. A total of 46% of patients underwent biomarker testing: 33% for PD-L1 (70% PD-L1 ⩾1%), 31% for EGFR (16% positive), 28% for ALK (2% positive), 16% for BRAF (
In the authors’ opinion, one of the main challenges in organizing early-stage lung cancer care in 2025 is the inadequate adherence of healthcare professionals to established diagnostic and treatment guidelines; moreover, despite full reimbursement, the availability of essential diagnostic and molecular procedures remains limited, a constraint frequently observed in publicly funded healthcare systems.
Systemic treatment
In Poland, neoadjuvant nivolumab was reimbursed in July 2024, perioperative pembrolizumab in April 2025, while adjuvant atezolizumab and osimertinib were previously available. The ongoing controversies include the necessity of postoperative immunotherapy after pCR, the optimal duration of perioperative therapy, treatment-related risks, and the best strategy for patients with stage III disease (N2a, N2b, IIIB), issues that remain unresolved. The biological rationale for neoadjuvant immunotherapy is well established,8–12 as chemoimmunotherapy administered in the presence of the primary tumor and metastatic nodes enhances antitumor immunity, translating into higher pCR rates, longer event-free survival, and in CheckMate-816 and Keynote-671 improved overall survival.10,12 For patients without actionable mutations, immunotherapy constitutes the standard, while targeted agents have become pivotal in adjuvant care. The ADAURA and ALINA trials14,15 demonstrated a substantial benefit in EGFR- and ALK-positive resected NSCLC, underscoring the growing importance of molecular profiling in early-stage disease. A synthesis of these pivotal perioperative trials is provided in Table 3.
Key clinical trials in early-stage non-small cell lung cancer.

DFS, disease-free survival; EFS, event-free survival; HR, hazard ratio; N/A, non-available; OS, overall survival; pCR, pathological complete response; PD-L1, Programmed Death Ligand-1.
Taking into account data on perioperative systemic treatment, it is essential for thoracic surgeons to be actively involved in the treatment plan at every stage of it. Early involvement allows surgeons to coordinate scope of diagnostic procedures, surgical timing with systemic therapy, and assess resectability after neoadjuvant treatment. Close collaboration within the MDT is essential for adapting perioperative strategies, ultimately improving the overall efficacy of treatment, and improving patient outcomes. In the analyzed cohort, biomarker diagnostics were performed in almost all patients only after surgery. This approach prevents qualification for preoperative treatment and should definitely be changed. It is also essential to remodel the reimbursement model for biomarker diagnostics. Currently, many healthcare systems, including Poland’s, do not provide sufficient funding for molecular diagnostics, which is sometimes limited to patients with advanced disease. 38
MDT meetings provide an optimal model for diagnostic and therapeutic decision-making for patients eligible for surgical treatment. MDT-directed patients benefit more from treatment, both in terms of time to recurrence and overall survival. They also have shorter waiting times for bronchoscopy and histopathological diagnosis.21,39,40 We believe that in the perioperative treatment era, it has become much more significant.
In the cohort, we analyzed, the majority of patients were only discussed by the MDT after surgical treatment. This approach is justified by traditionally limited options for neoadjuvant therapies in early-stage NSCLC. However, low awareness and a lack of multidisciplinary discussion during the preoperative diagnostic phase restrict the availability of perioperative and preoperative systemic therapies. We think that this is an important area for change. We believe that MDT should be organized at key points in a patient’s diagnostic and therapeutic pathway. First, at the time of initial eligibility for treatment, then after the imaging, histology, and biomarker results. For patients eligible for presurgical treatment, also after its completion to confirm the indication for surgical treatment. The next step should be a multidisciplinary evaluation of the patient after receiving the results of the postoperative histopathological examination. It is important to highlight that the surgeon’s evaluation of resectability during the initial phase of the treatment planning is critical. The surgeon’s absence from the MDT may lead to misclassification, thereby increasing the risk that the patient being ultimately considered ineligible for surgery. 41
Our study has some limitations. First, the aggregated data do not independently illustrate the situation in the whole of Poland—the centers were chosen for questioning in a nonsystematic way (based on a total number of patients treated every year) and the number of cases reported differed. The selected centers represented various regions of Poland, and all distributed questionnaires were eventually retrieved; however, the collected responses provide an overview of the current situation in Poland rather than precise numerical data. We are convinced that the wide range of data obtained in this study should not be overlooked and, as such, ought to be taken into account and critically assessed when making strategic decisions about the future development of oncology in Poland.
However, the character of the data that was the subject of this study was practically impossible to obtain in another way. Another limitation relates to the dynamic publication landscape and the processes of drug registration and reimbursement policies. It leads to changes in the approach of clinicians, including thoracic surgeons. According to the authors, the results present an indicative overview of the situation in Poland. It should be emphasized that if activities aimed at changing the current situation are to be implemented, it will be difficult to analyze the data over time and compare future results with those currently collected. However, such data can be compared on the basis of medical registries. It seems that existing medical records should be profoundly reorganized to better meet current needs.
Identifying standardization deficiencies requires understanding where and why there are gaps in the adherence to recommended assessment protocols. Variations in practices among different institutions may result from a lack of uniform guidelines or differences in the interpretation of existing rules. It should be noted that even scientific societies in different countries vary in their approach and definitions of resectability. 42 Lack of staff training can lead to the unintentional omission of key assessment steps, affecting the quality and reliability of the results. Additionally, poor implementation of guidelines, due to lack of organizational support or infrastructure, can also contribute to standardization deficits. 43 The reasons for the identified differences between the study centers were challenging to interpret. A trend of improved adherence to standards was observed in multispecialty centers, where broader discussions during well-organized MDTs often included radiotherapists. This may be due to a competitive environment for patients, particularly in stage III NSCLC.
The simplest solution is education. It should be conducted with a focus on immediate and long-term outcomes. Immediate effects can be achieved through training policies targeted at involved specialists. Another obvious path for education is to incorporate new treatment methods into resident training programs. It should be emphasized that, in light of funding for NGS testing, PET/CT, and, above all, therapies based on the CheckMate-816, ADAURA, Keynote-671, Keynote-091, and IMpower010 regimens, it is essential to make these approaches as widely available to patients as possible. In the authors’ opinion, advances in the treatment of stages II and III lung cancer must lead to organizational changes, which constitute the second recommendation. There is no doubt that new treatment methods require a complex approach, which can only be effectively implemented in multidisciplinary high-volume diagnostic and treatment centers. This study contributes to the field of education by emphasizing the importance of understanding the current state to implement systemic changes effectively. Identifying specific areas for improvement is crucial for development. Our research highlights the need for significant efforts to ensure that patients with NSCLC are diagnosed in a manner that enables them to benefit from new treatment protocols. Future research, possibly using more complex questionnaires or qualitative studies, may provide insights into the evolving landscape in Poland, influenced by the findings of this article. In addition, the collection of prospective data through a national lung cancer registry, periodic review of the quality of organized MDTs, and the exchange of expert experience should improves the quality of the diagnostic process and affect patient outcomes. The proposed centralization of services for early-stage NSCLC to regional, high-volume centers of excellence should be carefully balanced with the logistical and economic constraints faced by patients and their families. Regular auditing and quality control, although demanding and challenging to perform continuously, can enhance adherence to standards. A universal digital protocol for documenting patients’ diagnostic and therapeutic milestones, including specific timepoints, could serve as a valuable tool for clinicians and provide informative data for quality measurement purposes.
Conclusion
The data collected indicate that the current adherence to the standards of perioperative assessment in Poland is low. The current state of the perioperative evaluation allows for the proper qualification of patients to treatment protocols based on initial surgery. A high proportion of patients with PET/CT performed leads to adequate preoperative staging of patients in clinical stages I and II. A relatively high proportion of patients undergo an evaluation of PD-L1 expression and NGS of the operative specimens. The Achilles heel in Polish thoracic surgery centers is preoperative assessment, including (1) a low proportion of patients with brain imaging in stage III, (2) a low proportion of patients operated with preoperative diagnosis in stages II and III, (3) a low proportion of patients with molecular analysis performed preoperatively, and (4) a low proportion of patients discussed during MTD preoperatively. The consequence of the defined state results in the low accessibility of perioperative treatment protocols. In conclusion, it is worth reiterating that preoperative immunochemotherapy significantly improves the patient’s prognosis by prolonging DFS and OS, and should be currently the standard of care.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251413971 – Supplemental material for State of the art of perioperative assessment in early non-small-cell lung cancer in Poland in the emerging era of perioperative protocols
Supplemental material, sj-docx-1-tam-10.1177_17588359251413971 for State of the art of perioperative assessment in early non-small-cell lung cancer in Poland in the emerging era of perioperative protocols by Tomasz Marjanski, Magdalena Knetki-Wroblewska, Lukasz Kuncman, Izabela Chmielewska, Lukasz Szylberg, Julia Niedzielska, Cezary Piwkowski, Pawel Rybojad, Maciej Dancewicz, Dariusz Dziedzic, Joanna Lipinska, Grzegorz Liteplo, Piotr Skrzypczak and Maciej Krzakowski in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
