Abstract
Objective
To assess the efficacy and safety of perioperative melatonin and melatonin agonists in preventing postoperative delirium (POD).
Methods
We conducted a systematic search for randomized controlled trials (RCTs) published through December 2022. The primary outcome was efficacy based on the incidence of POD (POD-I). Secondary outcomes included efficacy and safety according to the length of hospital or intensive care unit stay, in-hospital mortality, and adverse events. Subgroup analyses of POD-I were based on the type and dose of drug (low- and high-dose melatonin, ramelteon), the postoperative period (early or late), and the type of surgery.
Results
In the analysis (16 RCTs, 1981 patients), POD-I was lower in the treatment group than in the control group (risk ratio [RR] = 0.57). POD-I was lower in the high-dose melatonin group than in the control group (RR = 0.41), whereas no benefit was observed in the low-dose melatonin and ramelteon groups. POD-I was lower in the melatonin group in the early postoperative period (RR = 0.35) and in patients undergoing cardiopulmonary surgery (RR = 0.54).
Conclusion
Perioperative melatonin or melatonin agonist treatment suppressed POD without severe adverse events, particularly at higher doses, during the early postoperative period, and after cardiopulmonary surgery.
Introduction
Postoperative delirium (POD) is a common neuropsychiatric complication in patients after surgery that is characterized by altered consciousness and cognitive disturbances, and its incidence rates vary from 10% to 60%.1–3 Elderly patients undergoing surgery are susceptible to POD, resulting in prolonged hospital stays and long-term impaired functional outcomes with significant morbidity and mortality.4,5 The pathophysiology of POD is multifactorial, involving neuroinflammation, altered neurotransmitter levels, sleep deprivation, and physiological changes induced by drugs, surgery, or general anesthesia.1,3,6,7
There has been growing interest in the perioperative use of melatonin (N-acetyl-5-methoxytryptamine) or melatonin agonists because of their antinociceptive, anxiolytic, and sedative effects and their association with neuroinflammatory mechanisms related to POD.8–11 Melatonin, an endogenous neurohormone produced by the pineal gland, plays an important role in regulating the sleep–wake cycle. 12 Some studies demonstrated that low serum melatonin levels are associated with a higher incidence of POD in patients in the intensive care unit (ICU).13,14 Therefore, the administration of melatonin or melatonin agonists could represent a cost-effective measure for preventing POD. However, recent clinical studies reported conflicting results regarding the effects of melatonin or melatonin agonists on POD.15–30
This study examined the efficacy and safety of perioperative treatment with melatonin and melatonin agonists for POD prevention, as measured by the incidence of POD (POD-I), length of hospital stay (LOS-H), length of ICU stay (LOS-ICU), in-hospital mortality, and adverse events. We conducted a systematic review and meta-analysis of randomized controlled trials (RCTs) to evaluate the efficacy and safety of perioperative melatonin or melatonin agonist treatment in the prevention of POD.
Materials and methods
This systematic review and meta-analysis of clinical trials was conducted following the guidelines of the Cochrane Handbook for Systematic Reviews of Interventions 31 and was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses 2020 statement. 32 This review was registered with the International Prospective Register of Systematic Reviews (https://www.crd.york.ac.uk/prospero/; registration no. CRD42021245806). Ethical approval was not required for this study because all analyses were based on previously published RCTs.
Literature search
In accordance with the protocol recommended by the Cochrane Collaboration, we systematically searched the literature for RCTs that evaluated the efficacy and safety of perioperative melatonin or melatonin agonists in preventing POD in patients undergoing anesthesia and surgery. We systematically searched PubMed, Embase, the Cochrane Central Register of Controlled Trials, and other sources to retrieve clinical trials involving adults ( >18 years old) published up to December 2022 with no language restrictions. The search used keywords such as “delirium,” “melatonin,” “melatonin agonist,” “ramelteon,” ”postoperative,” “surgery,” and “randomized,” as outlined in the Supplementary Information (Suppl. 1_Data).
Study selection
We included peer-reviewed RCTs that evaluated the perioperative use of melatonin or melatonin agonists in preventing POD in patients undergoing anesthesia and surgery. Review articles, observational studies, case reports, letters to the editor, commentaries, conference proceedings, laboratory studies, and other nonrelevant studies were excluded. Two authors (Kwak JS and Kim JW) independently assessed the articles to ensure compliance with the inclusion and exclusion criteria. Disagreements were resolved by discussion or consultation with a third independent investigator (Shin HW).
Data extraction and outcome assessment
The primary outcome of this study was the efficacy of the perioperative use of melatonin or melatonin agonists in preventing POD, as measured by POD-I. The secondary outcomes were the efficacy and safety of perioperative melatonin or melatonin agonists, which were assessed using LOS-H, LOS-ICU, in-hospital mortality, and adverse events. Using standardized forms, two authors (You HS and Jang YK) independently extracted the following data: first author’s name, publication year, study country, clinical inclusion criteria, number of patients in the control, melatonin, and melatonin agonist groups, patient age, POD-I, delirium assessment scales used, delirium duration, LOS-H, LOS-ICU, and adverse events. The authors of studies with insufficient or missing data were contacted, and data were extrapolated from figures when necessary to obtain the required information. The control group consisted of patients who received a placebo or no drug, whereas the intervention group (Mel-agonist group) included patients who received melatonin or melatonin agonists during the perioperative period.
Assessment of risk of bias
Two authors (Jang YK and Kim JW) independently evaluated the quality of clinical trials. We used the Cochrane risk-of-bias tool to assess the quality of the included RCTs, considering seven potential sources of bias: random sequence generation, allocation concealment, blinding of participants, blinding of outcome assessment, incomplete outcome data, selective outcome reporting, and others. 31
Statistical analysis
Statistical analyses were performed using RevMan 5.4 (Cochrane Collaboration, London, UK). Mean differences (MDs) with 95% confidence intervals (CIs) were calculated for continuous variables, and risk ratios (RRs) and their corresponding 95% CIs were obtained for dichotomous variables. All reported P-values were two-sided, and P < 0.05 denoted statistical significance. The chi-square test and I2 statistics were used to assess heterogeneity. If heterogeneity was observed (P < 0.05, I2 > 50%), a random-effects model was used; otherwise, a fixed-effects model was used. To evaluate the robustness of the results and the impact of individual studies, we performed a sensitivity analysis by excluding one study at a time to examine its influence on the overall estimate. If significant heterogeneity was observed, we performed a subgroup analysis for POD-I. Funnel plots were used to assess publication bias if more than 10 RCTs were included in the comparison.
Results
Literature search
During the initial electronic search, we identified 34 potentially eligible clinical trials (11 from PubMed, 14 from Embase, 9 from the Cochrane Central Register of Controlled Trials, 8 from other sources, and 14 with duplicate citations; Figure 1). Finally, we identified 16 RCTs that investigated the efficacy and safety of perioperative melatonin or melatonin agonist treatment in patients undergoing anesthesia and surgery. All studies were published between 2010 and 2022.

Preferred Reporting Items for Systematic Reviews and Meta-analyses 2020 flow diagram for this systematic review, which included searches of databases, registers, and other sources. 32
Study characteristics and data
The clinical characteristics of the included RCTs are presented in Table 1. These 16 RCTs collectively investigated the efficacy and safety of perioperative treatment with melatonin or a melatonin agonist to prevent POD in 1981 patients undergoing anesthesia and surgery. Most RCTs were single-center studies, whereas three studies were conducted in multiple hospitals.20,26,28 Melatonin was administered orally in all studies, with eight studies using a dose of 3 mg17,18,20,23–26 and five studies using a dose of 5 or 6 mg.15,16,27,29,30 The other four studies administered ramelteon at a dose of 8 mg.19,21,22,28 The patients included in this systematic review underwent various types of surgeries, including breast, 27 cardiac,17,18,20,23,25 pulmonary, 22 orthopedic,15,16,19,21,26,30 liver, 29 and trauma surgeries. 24 The screening tools used to diagnose POD include the Confusion Assessment Method (CAM),18,21 CAM for the Intensive Care Unit,18,20,22–24 Diagnostic and Statistical Manual of Mental Disorders (DSM)-4, 26 DSM-5, 19 Intensive Care Delirium Screening Checklist, 25 Abbreviated Mental Test,15,16,30 Delirium Observation Screening Scale, 26 and Delirium Rating Scale-Revised-98. 28
Characteristics of the studies included in the meta-analysis.

C, control; M, melatonin; AMT, Abbreviated Mental Test (delirium score < 8); CAM, Confusion Assessment Method; CAM-ICU, Confusion Assessment Method for the Intensive Care Unit; DSM, Diagnostic and Statistical Manual of Mental Disorders; ICDSC, Intensive Care Delirium Screening Checklist (delirium: score ≥ 4); DOSS, Delirium Observation Screening Scale; DRS-R98, Delirium Rating Scale-Revised-98; GI, gastrointestinal; ICU, intensive care unit; PCI, percutaneous transluminal coronary intervention.
Assessment of risk of bias
The results of the risk-of-bias assessment, aimed at determining the quality of the included studies, are summarized in the Supplementary Data (Suppl. 2_Data 1). All studies described the allocation concealment and blinding of participants and personnel. The risk of bias regarding selective reporting was unclear for several RCTs15–17,21,23,25,27,29,30 because no patients were lost during the study period. Some RCTs exhibited a high risk of bias, particularly because of differences in perioperative conditions (e.g., age, opioid use, and operation time) 25 and low POD-I, with missing details regarding the methods used to assess delirium.15,29
Publication bias
Funnel plots were used to assess publication bias. Symmetrical funnel plots indicated the absence of publication bias (Suppl. 2_Data 2).
Efficacy and safety of interventions
POD-I (number of patients)
POD-I was 16.9% in the Mel-agonist group, versus 25.3% in the control group (RR = 0.57; 95% CI = 0.40–0.81; I2 = 61%; P = 0.002; Figures 2–4). We performed a subgroup analysis based on the type or dose of the drug, postoperative follow-up period, and type of surgery. Subgroup analyses for POD-I were based on the type or dose of drug (low-dose melatonin, 3 mg; high-dose melatonin, ≥5 mg; ramelteon, 8 mg), postoperative period (early period, up to 3 days; late period, >7 days), and type of surgery (cardiopulmonary surgery with a high risk of POD, other surgeries). POD-I was significantly lower in the high-dose melatonin group than in the control group (RR = 0.41; 95% CI = 0.24–0.70; I2 = 0%; P = 0.001). However, there were no significant differences in POD-I in the low-dose melatonin (RR = 0.62; I2 = 75%; P = 0.07) and ramelteon groups (RR = 0.55; I2 = 57%; P = 0.25) compared with the control group (Figure 2). In the early postoperative period, the POD-I was significantly lower in the melatonin group than in the control group (RR = 0.35; 95% CI = 0.23–0.54; I2 = 9%; P < 0.001). However, there were no significant differences in POD-I between these groups in the late postoperative period (RR = 0.86; 95% CI = 0.61–1.20; I2 = 46%; P = 0.37; Figure 3). Among patients who underwent cardiac surgery, POD-I was significantly lower in the melatonin group than in the control group (RR = 0.54; 95% CI = 0.33–0.89; I2 = 68%; P = 0.01). However, among patients who underwent other surgeries, no difference in POD-I was identified between these groups (RR = 0.57; 95% CI = 0.31–1.05; I2 = 58%; P = 0.07; Figure 4). In the sensitivity analysis based on surgery type, we excluded data from the study by Abbasi et al. 24 in the other surgery group. Subsequently, a significant preventive effect on POD-I was observed for melatonin (RR = 0.52; 95% CI = 0.27–0.97; I2 = 60%; P = 0.04; Suppl. 2_Data 3). Sensitivity analyses for all other subgroups did not alter the results for POD-I.

Subgroup analyses for POD-I, based on the type and dose of the drug. Forest plot demonstrating POD-I in the control and Mel-agonist groups (melatonin and melatonin agonist). POD-I, incidence of postoperative delirium.

Subgroup analyses for POD-I based on the postoperative period (early or late). Forest plot demonstrating POD-I in the control and Mel-agonist groups (melatonin and melatonin agonist). POD-I, incidence of postoperative delirium.
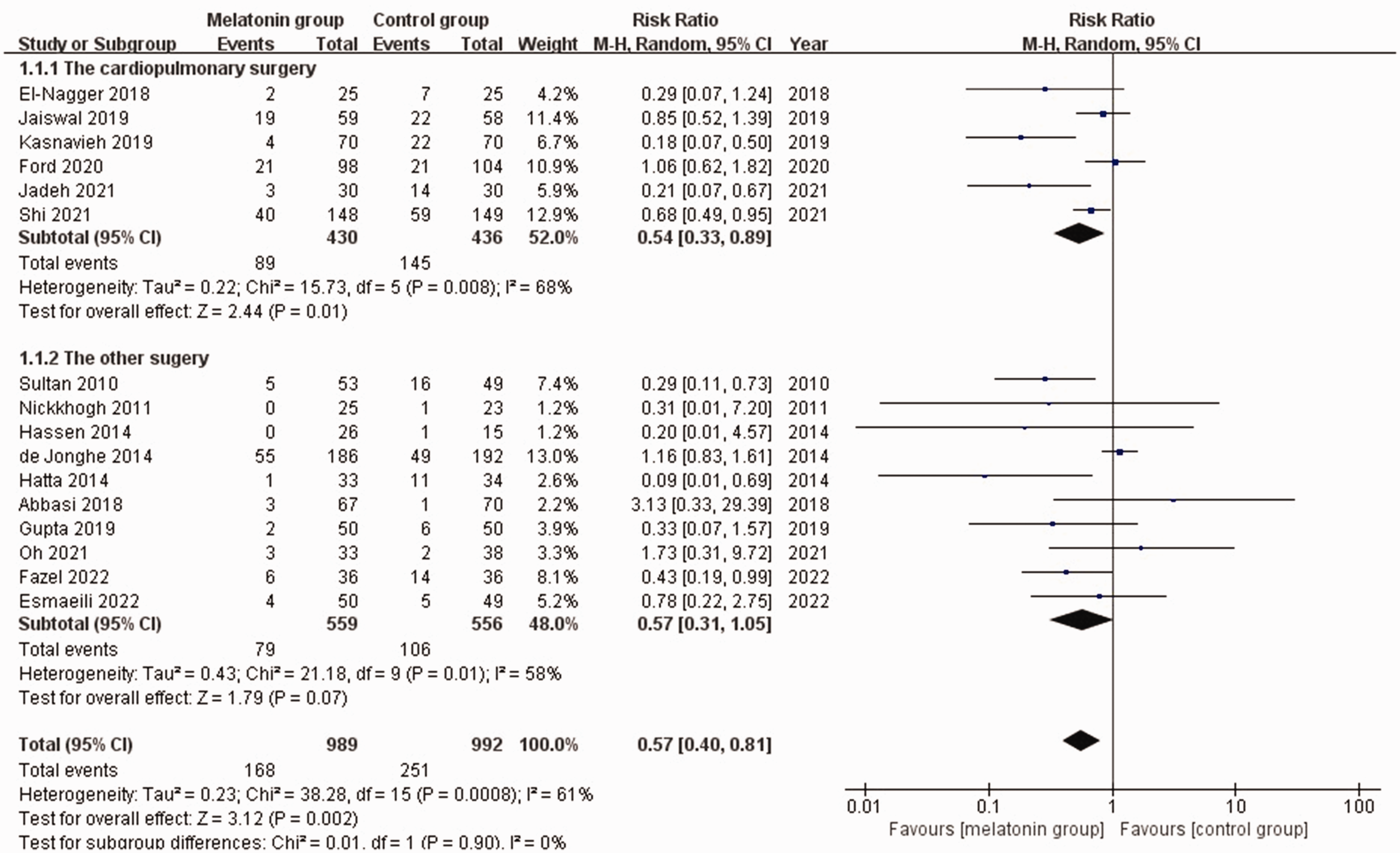
Subgroup analyses for POD-I based on the type of surgery (cardiopulmonary surgery with a high risk of POD or other surgeries). Forest plot demonstrating POD-I in the control and Mel-agonist groups (melatonin and melatonin agonist). POD-I, incidence of postoperative delirium.
LOS-H and LOS-ICU
There were no significant differences in LOS-H18,20,24,26,29 and LOS-ICU17,22,24,29 between the Mel-agonist and control groups. The MD for LOS-H was −0.11 (95% CI = −2.23 to 2.01; I2 = 94%; P = 0.92) with 1062 participants across five studies, and the MD for LOS-ICU was −0.09 (95% CI = −0.60 to 0.42; I2 = 0%; P = 0.74) with 362 participants across four studies. Additionally, the in-hospital mortality rate18,22,24,26 did not differ between the Mel-agonist and control groups (RR = 1.07; 95% CI = 0.68–1.67; I2 = 0%; P = 0.77) with 973 participants across four studies (Suppl. 2_Data 4–6).
Adverse events
No severe adverse events were reported in any of the included studies. The common drug-induced adverse events were dizziness, nausea, hypotension, headache, and gastrointestinal disorders. No differences were reported between the Mel-agonist and control groups regarding the rates of the following adverse events: dizziness27,30 (RR = 1.51, I2 = 31%; P = 0.71; n = 112), nausea19,27,29 (RR = 1.26; I2 = 0%; P = 0.78; n = 160), hypotension29,30 (RR =1.69; I2 = 0%; P = 0.57; n = 119), headache23,27 (RR = 0.81, I2 = 76%; P = 0.81; n = 181), and gastrointestinal disorders 23 (RR = 2.20; P = 0.12; n = 140; Suppl. 2_Data 7–11).
Discussion
We found that the perioperative use of melatonin or melatonin agonists was effective in preventing POD in patients undergoing anesthesia and surgery. Furthermore, our research indicated that the use of melatonin and melatonin agonists during the perioperative period reduces the incidence of POD-I, particularly in the early postoperative phase (within 3 days), when administered at higher doses (≥5 mg), and in cardiopulmonary surgeries. Notably, our study included the largest number of RCTs reviewed to date on this topic.
The incidence of POD increases with age, 23 with a prevalence ranging from 14% to 24% upon hospital admission and potentially increasing to 56% during hospitalization. 23 Therefore, the early detection of POD, identification of risk factors, and implementation of preventive measures are vital for reducing morbidity, mortality, and healthcare costs. Numerous studies have suggested that an imbalance in melatonin levels, resulting from disrupted circadian rhythms and sleep deprivation, can contribute to POD.33,34 Sleep disturbances are particularly common among patients undergoing major surgeries, especially among elderly patients, who already have low baseline serum melatonin levels that are further reduced by surgery. 35 POD is triggered by brain inflammation and dopaminergic dysfunction.26,36 The perioperative use of melatonin can prevent POD by regulating circadian rhythms and exerting anti-inflammatory and antioxidant effects.26,36 Melatonin is believed to scavenge free radicals and minimize brain tissue damage. 34
Melatonin is a US Food and Drug Administration (FDA)-approved dietary supplement. Adult doses range from 1 to 10 mg. Approximately 30 to 60 minutes are required for the drug to take effect, and its duration of action is approximately 5 hours.37,38 The bioavailability of melatonin varies widely (10%–56%), and it is subject to hepatic first-pass metabolism, being influenced by factors such as age, weight, and supplement type. 38 By contrast, ramelteon, another FDA-approved drug, acts as a melatonin receptor agonist with strong affinity for MT1 and MT2 receptors, effectively reducing wakefulness for up to 6 hours.19,39 Moreover, ramelteon has been reported to be 10-fold more potent than melatonin in promoting sleep. Its peak concentration is reached within 0.75 hours. 40 Initially, we anticipated that ramelteon would display significant benefits in preventing POD. However, our review revealed that ramelteon did not have effect on POD-I (Figure 2). Melatonin acts on MT1, MT2, and MT3 receptors in the brain. 38 Conversely, ramelteon is a selective agonist of MT1 and MT2 receptors in the brain. 41 It does not bind to MT3, serotonin receptors, or BDZ receptors in the brain, which are believed to be involved in the pathways leading to delirium.38,42 Ramelteon is a suitable choice for controlling circadian rhythms, but it lacks the anti-inflammatory or antioxidant effects essential for preventing POD. Further research is required to determine the efficacy of ramelteon in preventing POD.
It should be noted that the optimal melatonin dose has yet to be determined. In our review, we found that the efficacy of melatonin in reducing POD-I was particularly notable in the early postoperative period and at higher melatonin doses (≥5 mg). Given its short duration of action (4–8 hours) when administered orally, it might be safer to administer drugs closer to surgery to enhance their effectiveness.1,37 Generally, the incidence of POD is higher in cardiac surgeries (26%–52%) than in other surgeries, 43 possibly because of factors such as hypoperfusion or microemboli during surgery. 44 Our subgroup analysis revealed a lower POD-I in the melatonin group than in the control group. Furthermore, in the sensitivity analysis based on the type of surgery, data from the study by Abbasi et al. 24 were excluded from the analysis, yielding statistically significant results indicating a preventive effect of melatonin on POD-I in other surgeries. The study by Abbasi et al. included patients from various categories, such as medical, surgical, and ICU-admitted trauma patients, which might have influenced the statistical significance of the results in the other surgery analysis.
Orally administered melatonin undergoes extensive first-pass metabolism in the liver and exhibits variable bioavailability in humans. 36 Therefore, a higher melatonin dose than that used in daily clinical practice might be required to prevent POD in older patients undergoing surgery.
Unlike other sleep medications, melatonin does not cause sleep withdrawal, dependence, or tolerance. However, it can lead to mild and transient adverse events, such as headache, dizziness, confusion, vivid dreams, hypotension, abdominal pain, and cramps. 27 In our review, no significant differences in adverse event rates were observed between the melatonin and control groups. In a study by Ng et al., 42 hospitalized patients treated with melatonin exhibited a significantly shorter LOC-ICU.
Three systematic reviews and meta-analyses assessed the effect of perioperative melatonin and melatonin agonist treatment in preventing POD in surgical patients.45–47 However, Wang et al. 48 reported contradictory results. They omitted previously reported RCTs,23–25,27,28,49 including one RCT that examined melatonin precursors, such as tryptophan, 49 and included another publication of a conference proceeding. 50 Han et al. 46 reported that melatonin and ramelteon effectively prevent POD. However, the number of studies included was small, including two cohort studies and one case–control study. Campbell et al. 47 also found that perioperative melatonin use reduced POD-I. However, they included three RCTs,26,29,30 two non-RCTs,41,51 and one conference proceeding. 50 In the three previously mentioned reviews,46–48 methodological heterogeneity might have been present because RCTs and non-RCTs were pooled together in the meta-analysis because of the limited available clinical data.
Our study had multiple limitations. First, the incidence of POD-I was low in some of the included studies, and some studies had small sample sizes, making it challenging to obtain statistically significant results. Second, there were significant differences in patient characteristics, delirium assessment tools, the type of melatonin agonist, the dosage and duration of melatonin agonists, and follow-up periods among the included studies. Additionally, the serum melatonin levels reported in these studies might have been influenced by variations in melatonin regimens, such as manufacturer variability, pharmacokinetic characteristics, and pharmacogenomic heterogeneity. 52
In our study, the perioperative use of melatonin and melatonin agonists decreased POD without severe adverse events, particularly at higher melatonin doses, during the early postoperative period, and in cardiopulmonary surgeries. However, the conclusions of this review should be interpreted with caution in clinical practice because of the high heterogeneity of the included studies. Further studies using standardized diagnostic tools and designs with larger numbers of participants are required to confirm these findings.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241239854 - Supplemental material for Efficacy and safety of perioperative melatonin for postoperative delirium in patients undergoing surgery: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605241239854 for Efficacy and safety of perioperative melatonin for postoperative delirium in patients undergoing surgery: a systematic review and meta-analysis by Hye Won Shin, Ji Su Kwak, Yoon Ji Choi, Jae Woo Kim, Hae Sun You, Hyun Ju Shin and Yoo Kyung Jang in Journal of International Medical Research
Footnotes
Author contributions
Conceptualization: Shin HW, Choi YJ, You HS, and Shin HJ. Data curation: Shin HW, You HS, Jang YK, and Kim JW. Formal analysis: Shin HW and Choi YJ. Investigation: Jan YK, Kim JW, and Shin HW. Methodology: Shin HW, Choi YJ, and Kawk JS. Statistical analysis of the meta-analysis: Shin HW, Shin HJ, and Jang YK. Validation: Kwak JS and Kim JW. Writing the original draft: Shin HW and You HS. Writing, reviewing, and editing: Shin HW.
Data availability statement
All data used in our study are included in this published article and Supplementary Files S1 and S2.
Declaration of conflicting interests
The authors declare that they have no conflicting interests.
Funding
Supplementary file
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
