Abstract
Patients presenting with synchronous or metachronous colorectal cancer liver metastases (CLM) should be evaluated for multimodal management with curative intent. Preoperative systemic chemotherapy shows beneficial impact on adjuvant progression-free survival and also borderline on overall survival, without significantly increasing initially R0 resectable patients postoperative complication rates. Postoperative chemotherapy recommended based on the perioperative trial experience for those patients achieving at least stable disease during preoperative chemotherapy, or based on the adjuvant trials for patients receiving upfront resection. ‘Borderline’ resectable CLM, preoperative chemotherapy plays an important role in both in achievement of a resectable status and improvement of prognosis. Recent 4 drug combinations demonstrated response rates up to 80% even for advanced disease and are thus promising regimens for further evaluation in patients with resectable or unresectable liver-limited (+/- lung) disease.
Background
Colorectal cancer (CRC) is the most frequently diagnosed cancer in Europe and one of the leading causes of cancer death worldwide [Ferlay et al. 2010; Jemal et al. 2011]. One in four patients with CRC presents with metastatic disease at the time of diagnosis and a further quarter of patients will develop metastases after curative resection, of whom 20–30% present with liver metastases only [Mantke et al. 2012; Van der Pool et al. 2012]. Liver or lung metastases without further sites of disease represent a unique and favourable biology of advanced CRC as metastases can be restricted to these visceral organs for a long time or even forever, making the resection or elimination of these metastases in addition to chemotherapy an important and meaningful tool to enable cure or at least long-term survival.
Upfront stratification of patients with metastatic CRC (mCRC) into clinical groups according to clinical presentation and treatment aim by a multidisciplinary team is important for tailoring the treatment approach especially for either initially or potentially resectable liver ± lung metastases [Schmoll and Sargent, 2007; Van Cutsem et al. 2010].
Recent retrospective series have demonstrated profound improvement in the outcome of patients with CRC over the past 20 years due to advances in chemotherapy and a dramatic increase in patients undergoing liver resection for either primarily or secondarily resectable colorectal liver metastases (CLM), resulting in an overall survival (OS) comparable with that of stage III [Union for International Cancer Control (UICC)] colon cancer [Kopetz et al. 2009; Morris et al. 2010]. However, with the majority of patients relapsing after liver resection, either intrahepatic (about 70%), extrahepatic (about 50%) or both, further research in terms of patient selection as well as improved treatment modalities and intensity is urgently warranted [de Jong et al. 2009].
Stratification of patients with CLM
Patients with hepatic metastases should be stratified in upfront ‘clearly R0’ resectable CLM and not R0, but potentially resectable CLM after downsizing with (neoadjuvant) chemotherapy. Although the status of the primary tumour in cases of synchronous disease needs to be taken into account, especially in cases of rectal cancer, a patient’s prognosis depends on the extent and resectability of CLM. The treatment approach differs in regard of resectability. Initially resectable patients receive perioperative systemic treatment with fluoropyrimidine and oxaliplatin for up to 6 months to improve prognosis, similar to adjuvant treatment of stage III colon cancer. For not R0 resectable patients the most active induction chemotherapy, for as long as necessary and as short as possible, is required to ensure achievement of secondary R0 resectability.
Prognostic factors for liver resection of CLM
Already in the late 1980s, risk factors for poor outcome after resection of clearly R0 resectable CLM were identified, e.g. presence of positive portal lymph nodes, extrahepatic metastases, four or more hepatic metastases or primary tumour stage Duke’s C [Hughes et al. 1986]. The scoring system described by Fong and colleagues includes five risk factors: node-positive primary, disease-free interval <12 months, >1 lesion, size >5 cm, and carcinoembryonic antigen (CEA) >200 ng/m [Fong et al. 1999]. Patients with five risk factors had a 5-year OS rate (5-yOSR) of 14% with a median of 22 months compared with a 5-yOSR of 44% and a median of 51 months in the presence of one risk factor. The Fong score, however, is only validated for upfront resectable CLM and does not include preoperative chemotherapy, which limits the use of this score for the current treatment situation.
Recently, the international liver metastases survey, including data for about 4500 patients with preoperatively treated CLM, confirmed the poor prognosis of patients with lymph node positive primary, abnormal CEA levels and number of CLM, although the cutoff value was three in the current analyses. Furthermore, more than one line and six cycles of preoperative treatment, incomplete macroscopic resection (R2), concomitant extrahepatic metastases and progression during preoperative chemotherapy were associated with poor prognoses [Adam and Barroso, 2011].
Besides clinicopathological variables, molecular markers might gain importance for prognostic information on patients selected for surgery. The worse prognosis of BRAF-mutated mCRC has been shown in several trials with a progression-free survival (PFS) of about 3–6 months and an OS of up to 15 months, although patient numbers are limited by the low frequency [Bokemeyer et al. 2012; Price et al. 2011; Tol et al. 2009a]. Recent analysis indicated that upfront intensive four-drug treatment, FOLFOXIRI (irinotecan, oxaliplatin, fluorouracil and folinate) and bevacizumab, for BRAF-mutant mCRC might result in more favourable PFS (11.8 months) and OS (23.8 months), comparable with the overall mCRC population [Salvatore and Loupakis, 2012]. However, the significant and independent worse prognostic value of KRAS (58 out of 202 patients) and BRAF mutation (6 out of 292 patients) was recently demonstrated in patients with resection of CLM (hazard ratio [HR] 2.13, 95% confidence interval [CI] 1.25–3.65 for KRAS mut and HR 6.245, p < 0.003 for BRAF mut) [Karagkounis and Torbenson, 2012; Teng et al. 2012].
Determination of resectability
Criteria for resectability are not well defined. Several approaches have been published but with as yet no consensus [Nordlinger et al. 2009; Poston et al. 2008; Yamaguchi et al. 2008]. Concordance in determination of resectability could be achieved in only 64.5% of cases in the CELIM trial with a board of surgeons blinded for point-in-time of imaging (baseline or after treatment), patients’ clinical outcome and the vote of the other participants, whereas critical disagreement (resectable versus nonresectable) was reported in 6.8% [Folprecht et al. 2010]. In the EORTC 40983 trial, patients with up to four potentially resectable liver metastases and no evidence of extrahepatic disease were included [Nordlinger et al. 2008].
Recent clinical trials include patients depending on resectability determined by a surgeon with hepatic surgery expertise [ClinicalTrials.gov identifier: e.g. NCT01540435, NCT01266187]. In general, limitations for liver surgery are remnant liver of less than 30%, unfavourable location, comorbidity excluding major surgery, aggressive tumour biology and/or the presence of extrahepatic disease. Although number (more than four) and size (>5 cm) have an impact on prognosis [Yamaguchi et al. 2008], patients could still be deemed resectable and achieve long-term survival with these characteristics. Furthermore, positive margins (R1 resection) seem to be less important for outcome after response to preoperative chemotherapy [Tanaka et al. 2011]. Localized (hepatoduodenal ligament, retropancreatic area) rather than distant (celiac, para-aortic) lymph node metastases does not preclude from long-term survival (5-yOSR: 25–30%) after resection of CLM and lymph nodes [Adam et al. 2008; de Haas et al. 2008; Pulitano et al. 2012]. With regard to evolving surgical techniques (e.g. in situ split enabling potential complete and macroscopically curative resections of CLM even in case of disseminated and bilobar disease), technical resectability might often be given, although not every resection might be biologically meaningful.
Systemic treatment of upfront clearly R0 resectable CLM
The role of perioperative treatment in resectable CLM, especially the point in time (preoperative versus postoperative), mode (systemic versus local) and intensity (monotherapy versus combination) is still a matter of debate. Although postoperative therapy after resection of CLM has become an accepted standard of care in many parts of the world, data on the benefit are limited. There are only a few randomized trials comparing preoperative and/or postoperative intravenous chemotherapy or hepatic artery infusion with or without intravenous chemotherapy to surgery alone. Moreover, in adjuvant systemic chemotherapy none of the conducted clinical trials achieved the recruitment goal, leading to solely premature data.
Current guidelines such as European Society for Medical Oncology (ESMO) consensus guidelines recommend upfront surgery for a single liver metastasis with a diameter of up to 2 cm and good prognosis, whereas for larger single or multiple, clearly R0 resectable CLM preoperative chemotherapy should be administered [Schmoll et al. 2012].
Perioperative systemic treatment
The EORTC 40983 trial accrued 364 patients to be randomized between surgery alone and chemotherapy with 5-fluorouracil (5FU) and oxaliplatin (FOLFOX4 regimen), administered 3 months preoperatively and postoperatively [Nordlinger et al. 2008]. Patients had to be technically resectable, as stated by computed tomography (CT) scan, with a maximum of 4 CLM and no prior treatment with chemotherapy. Patients’ characteristics were favourable, with more than 50% of patients bearing only one CLM and about 80% with up to two, respectively. A significant increase in PFS rate at 3 years from 33.2% to 42.4% was shown in the group of patients (n = 171) receiving chemotherapy and resection of CLM (HR 0.73, 95% CI 0.55–0.97). In the randomized population (‘intent-to-treat analyses’), the difference was not statistically significant due to ineligibility of 6% of patients (HR 0.79, 95% CI 0.62–1.02), but still favouring the chemotherapy arm (Table 1). Recent updated OS analyses (secondary endpoint) revealed a small albeit nonsignificant benefit in 5-yOSR of 4.1% in all eligible patients, corresponding to a median OS prolongation of 9 months (55 versus 64 months) [Nordlinger and Sorbye, 2012]. However, the trial was not powered to detect an OS benefit.
Results from EORTC 40983 trial [Nordlinger et al. 2008].

CT, chemotherapy; HR, hazard ratio; OSR, overall survival rate; PFSR, progression-free survival rate.
No unusual toxicities occurred in the chemotherapy arm. Nearly 80% of patients completed the preoperative part. After resection, 76% of patients were receiving postoperative treatment with 52% of patients (n = 80) completing the six scheduled postoperative cycles.
During preoperative chemotherapy main grade 3/4 toxicities were nausea and vomiting 4%, diarrhoea 8%, stomatitis 7%, sensory neuropathy 2% and neutropenia 18% (febrile 2%). Postoperative reversible complications were significantly higher (25 versus 16%, p = 0.04) in the chemotherapy group. Particularly intra-abdominal infections, biliary fistulas with an output of more than 100 ml per day for more than 10 days and hepatic failure with a bilirubin-elevation grade 3/4 for more than 3 days occurred more often in the perioperative group. There was no impact on postoperative death rate (1% in both arms). During postoperative chemotherapy, toxicities were similar with a higher rate of grade 3 sensory neuropathy (10%) and other neurological toxicities (12%) due to the higher cumulative dose of oxaliplatin.
The results of the EORTC 40983 trial consequently led to the EORTC 40051 ‘BOS’ study with FOLFOX and cetuximab ± bevacizumab, which was stopped after disadvantageous results for the combination of epidermal growth factor receptor (EGFR) and vascular endothelial growth factor (VEGF) antibodies in CAIRO2 and PACCE [Hecht et al. 2009; Tol et al. 2009b].
Postoperative systemic treatment
Already the standard of care in patients with stage III disease (and some stage II), data for adjuvant treatment after resection of stage IV disease are still limited (Table 2). Two randomized phase III trials have compared adjuvant systemic chemotherapy with 5FU/folinic acid (FA) after resection of CLM to surgery alone, but both closed prematurely due to slow accrual. A small but significant improvement in recurrence free survival rate (RFSR) could be shown in the FFCD trial at the time of closure, with a 5-year RFSR of 26.7 versus 33.5% (p = 0.028), favouring the group with adjuvant treatment [Portier et al. 2006]. Although the 5-yOSR increased from 41.1% to 51.1%, this difference was not significant (p = 0.13). The ENG (EORTC/NCI-CTG/GIVIO) trial, whose results are still not fully published, displayed a nonsignificant trend towards a prolonged RFS with a median of 39 versus 20 months (p = 0.35) and an increased OS with a median of 53 versus 43 months (p = 0.39) [Langer et al. 2002]. A combined analysis of both trials (n = 278) showed a nonsignificant prolongation of median disease-free survival (DFS) from 18.8 to 27.9 months (p = 0.058) and OS from 47.3 to 62.2 months (p = 0.095) [Mitry et al. 2008]. However, both trials were using a noncontemporary 5FU bolus regimen, which is more toxic and possibly not active enough to reveal a significant increase in this limited number of patients. Postoperative treatment with 5FU plus irinotecan in a prematurely stopped, randomized phase III trial revealed no additional benefit over 5FU alone in terms of OS (3-year survival rate of 73 versus 72%) [Ychou et al. 2009a]. Regarding median DFS, there was a nonsignificant increase of 3 months (21.6 versus 24.7 months, HR 0.90, 95% CI 0.67–1.2, p = 0.47). However, with overall 306 treated patients the trial might have been insufficiently powered to demonstrate a significant DFS benefit. Moreover, the lack of a clear benefit might also be related to the relatively poor efficacy, as shown in the adjuvant treatment of stage III colorectal disease with 5FU/FA plus irinotecan (FOLFIRI) [Saltz et al. 2007; Van Cutsem et al. 2009; Ychou et al. 2009b]. Recently results of the again prematurely closed Dutch HEPATICA trial were presented, demonstrating a DFS benefit after 2 years of 18% (70% versus 52%) in favour of 12 months bevacizumab with 6 months chemotherapy (capecitabine and oxaliplatin) compared with 6 months chemotherapy alone after R0 resection of CLM in 79 patients [Voest and Snoeren, 2011]. Further follow up might reveal whether this benefit is only transient, as demonstrated for bevacizumab containing adjuvant treatment after resection of stage II or III colon cancer [Allegra et al. 2011; de Gramont, Van Cutsem, et al., 2012]. It is questionable, however, whether further results regarding adjuvant chemotherapy will be available as further postoperative trials (ADHOC, NSABP C-09 and HEPATICA) were closed due to insufficient accrual.
Results for adjuvant treatment after resection of stage IV disease.

beva, bevacizumab; CAPOX, capecitabine and oxaliplatin; CI, confidence interval; FA, folinic acid; FOLFIRI, FA, 5FU and irinotecan; 5FU, 5-fluorouracil; HR, hazard ratio; LV5FU, leucovorin and 5FU; nr, not reported; OS, overall survival; OSR, overall survival rate; p value, significance level; R0, number of R0 resections.
Ongoing trials in R0 resectable CLM
Several randomized trials are currently ongoing evaluating intensified perioperative regimens, for example, CRUK06/031 (FOLFOX ± cetuximab) and EORTC BOS-2 (FOLFOX alone versus FOLFOX plus EGFR antibody or FOLFOX plus bevacizumab), both in KRAS wild-type patients, as well as intensified perioperative versus postoperative approaches such as the German PANTER (FOLFOX plus cetuximab 3 months preoperative and postoperative versus 6 months postoperative) in KRAS wild-type, the Australian ATTACHE (FOLFOX/XELOX/FOLFIRI plus bevacizumab 3 months preoperative and postoperative versus 6 months postoperative) or PERIMAX (FOLFOXIRI plus bevacizumab 3 months preoperative and postoperative versus FOLFOX 6 months postoperative).
Induction treatment in not R0, but potentially resectable, CLM after chemotherapy-induced downsizing
The group of patients with potentially resectable metastases warrants a highly active, neoadjuvant or induction chemotherapy that aims to achieve resectability, with tumour shrinkage as the most important goal. Regarding the reported Response Evaluation Criteria in Solid Tumors (RECIST) overall response rates (ORR) displayed in clinical trials in the first-line treatment for mCRC, a three-drug combination containing EGFR-targeting antibody in KRAS wild-type patients or bevacizumab or a three-drug chemotherapy regimen (FOLFOXIRI) should be chosen [Bokemeyer et al. 2011; Douillard and Siena, 2011; Falcone et al. 2007; Hurwitz et al. 2004; Maughan et al. 2011; Saltz et al. 2008; van Cutsem et al. 2011] (Table 3). These trials included all patients with mCRC, and the benefit for “liver only” disease cannot be assessed properly.
Results following induction treatment in not R0, but potentially resectable, CLM after chemotherapy-induced downsizing.
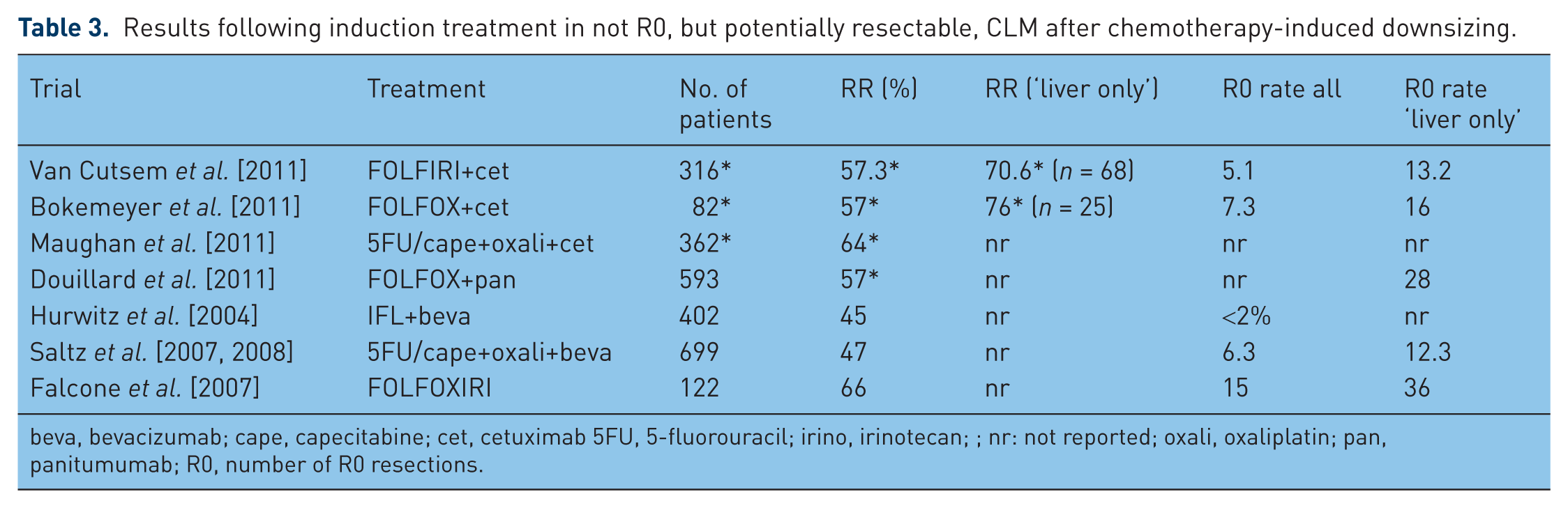
beva, bevacizumab; cape, capecitabine; cet, cetuximab 5FU, 5-fluorouracil; irino, irinotecan; ; nr: not reported; oxali, oxaliplatin; pan, panitumumab; R0, number of R0 resections.
Recent data from single arm phase II trials with different 5FU/FA, irinotecan and oxaliplatin schedules in combination with either bevacizumab or EGFR antibodies consistently reported a high ORR of about 80% [Assenat et al. 2011; Bruera et al. 2010; Garufi et al. 2010; Lonardi and Fornaro, 2012; Masi et al. 2010]. In particular, the four-drug regimens with EGFR antibodies were compromised by grade 3/4 diarrhoea, leading to dose reduction of chemotherapy with maintained efficacy. In one trial with FOLFOXIRI plus bevacizumab, the ORR of 80% in ‘liver only’ patients resulted in a favourable R0 resection rate of 26% (n = 15) [Masi et al. 2010].
Several trials including mCRC patients without extrahepatic disease have shown ORR in the range of 71 to 78% with current three-drug regimens (5FU, irinotecan, and oxaliplatin (FOLFIRINOX), capecitabine plus oxaliplatin (CAPOX) plus bevacizumab, FOLFOX/FOLFIRI plus cetuximab [Folprecht and Gruenberger, 2010; Gruenberger et al. 2008; Wong et al. 2011; Ychou et al. 2008]. Although the above-mentioned limitations about determination of resectability need to be taken into account, the CELIM and BOXER trial reported rates of secondary resectability of 28 and 40%, respectively [Folprecht et al. 2010; Wong et al. 2011]. Recently, survival analysis of the CELIM trial demonstrated a significant PFS benefit of 15.4 versus 8.9 months (HR 2.07, p = 0.001) and OS improvement of 46.7 versus 27.3 months (HR 2.34, p = 0.002) in favour of R0 resected patients (n = 36) compared with those with no or R1/2 resection (n = 70); this further strengthens the approach of aiming at and performing secondary resection if feasible [Folprecht and Gruenberger, 2012].
If resectability cannot be achieved by systemic chemotherapy alone, combination with hepatic artery infusion might be a possible approach as recently demonstrated in a small group of 61 treated patients with a resection rate of 44% [Kemeny and Fong, 2012].
Ongoing trials are evaluating different 5FU/FA, irinotecan and oxaliplatin regimens in patients with liver-limited disease with either panitumumab or cetuximab in KRAS wild-type or bevacizumab (e.g. Austrian MO18725, CELIM 2, PRODIGE 14, VOLFI).
Controversies about neoadjuvant/induction treatment
Although tumour shrinkage is important in order to achieve technical resectability and RECIST response rates are likely to correlate well with the percentage of resected or ‘converted’ patients, patient prognosis may be determined even more by the effect of treatment on tumor tissue and -biology than by the achievement of resectability alone. Accordingly, good correlations of pathohistological response to preoperative chemotherapy, which seemed to be increased with bevacizumab-containing combinations, were reported with OS from different cohorts of patients after resection of CLM [Blazer et al. 2008; Klinger et al. 2010; Rubbia-Brandt et al. 2007]. These have to be kept in mind in case of cross-trial comparison for response rates.
One of the major drawbacks in neoadjuvant treatment is the change into irresectability in cases of progression during first-line induction chemotherapy. In the EORTC 40983 trial, 12 (7%) patients experienced progressive disease during preoperative chemotherapy leading to irresectability in 8 patients, half of whom presented with new lesions [Nordlinger et al. 2008]. However, the switch to irresectability reveals aggressive tumour biology and predicts a worse outcome even with resection, or defines inadequate chemotherapy with a 2drug regimen only, or both.
Another issue is the complete radiological response during neoadjuvant treatment, as complete clinical response does not mean complete pathological response [Benoist et al. 2006; Gaujoux et al. 2011]. Therefore patients should be closely monitored during preoperative chemotherapy and ideally resected before complete remission. If an anatomical resection can be performed, complete response is not a major problem because resection will be based on initial sites of liver metastases. In cases of complete response on CT and no option for anatomical resection, different imaging methods might be used [magnetic resonance imaging (MRI), positron emission tomography (PET) scan, contrast-enhanced ultrasound] or resection might be delayed until relapse occurs.
Chemotherapy can result in severe damage of liver tissue. Whereas oxaliplatin-based combinations are associated with increased risk of vascular lesions, irinotecan-containing regimens have been associated with increased risk of steatohepatitis, especially in obese patients with a body mass index (BMI) over 25 kg/m2 [Aloia et al. 2006; Benoist and Nordlinger, 2009; Vauthey et al. 2006]. The clinical impact of chemotherapy-associated histological changes seems to favour oxaliplatin in the neoadjuvant setting. Increased risk of perioperative bleeding, postoperative complication rate and poorer functional reserve was found to be associated with vascular lesions. Although influence on morbidity is still controversial, mortality was not affected [Nakano et al. 2008; Vauthey et al. 2006]. In the EORTC 40983 trial, the perioperative treatment resulted in a similar postoperative death rate to surgery alone, with a significantly higher rate of reversible postoperative complications (p = 0.04). Higher risk of complications and an increased 90-day mortality [1.6% versus 14.7%, odds ratio (OR) 10.5] was reported for irinotecan-associated steatohepatitis [Vauthey et al. 2006]. The addition of bevacizumab to neoadjuvant oxaliplatin-based treatment is feasible, does not limit the ability of liver regeneration, and even seems to protect the liver from oxaliplatin-induced liver toxicity [Gruenberger et al. 2008; Ribero et al. 2007].
Duration of preoperative treatment seem to have a major impact on perioperative morbidity and prognosis. Several retrospective analyses suggested an increased postoperative morbidity (19% versus 54% p = 0.047 for </≥6 cycles, higher rate of liver insufficiency (4% versus 11%, p = 0.035) for </≥9 cycles and higher rate of reoperation (0% versus 11%, p = 0.02) and longer hospitalization (11 versus 15 days, p = 0.02) for ≤/>12 cycles [Aloia et al. 2006; Karoui et al. 2006; Kishi et al. 2010]. Moreover, the recent liver metastases survey showed a DFS detriment for patients with more than six cycles of preoperative chemotherapy [Adam and Barroso, 2011]. Duration of preoperative chemotherapy should therefore be limited to 3 months in case of initially R0 resectable disease (followed by 3 months postoperative treatment) or as short as possible in case of initially unresectable disease. Although liver regeneration and postoperative complication rates do not seem to be affected by preoperative bevacizumab treatment, the interval to surgery is a critical factor with regard to wound-healing complications and bleeding [Erinjeri et al. 2011; Kesmodel et al. 2008; Wicherts et al. 2011]. According to the summary of product characteristics (SmPC), the minimum interval between bevacizumab administration and operation should be 4 weeks; thus the last preoperative chemotherapy might be applied without bevacizumab.
Besides treatment-naïve patients with synchronous liver metastases, a large group of patients with metachronous disease will have received adjuvant chemotherapy. Whereas adjuvant single-agent fluoropyrimidines do not influence choice of treatment, the interval between oxaliplatin-based adjuvant chemotherapy and hepatic recurrence is important for selection of neoadjuvant or induction chemotherapy. In cases of recurrence within 6 months after completion of adjuvant oxaliplatin, irinotecan-based chemotherapy might be more appropriate even in clearly R0 resectable liver metastases.
Conclusion
The prognosis of patients with CLM depends on their access to an optimal multimodal treatment. Systemic perioperative chemotherapy administered preoperatively and/or postoperatively has been shown to reduce recurrence and prolong survival in initially clearly R0 resectable disease. Besides the subgroup of patients with a single small CLM, for whom upfront resection should be performed, perioperative chemotherapy for 3 months preoperatively and 3 months postoperatively should be administered.
For not R0 but potentially resectable CLM after chemotherapy, an aggressive treatment approach is broadly accepted with at least chemotherapy triplets or chemotherapy doublets in combination with EGFR or VEGF antibodies. This approach might possibly also be useful for patients with R0 resectable CLM, as DFS is still poor after curative resection. Current trials reflect ongoing discussions about the optimal treatment approach for CLM comparing either perioperative versus postoperative, or two versus three or even four drug regimens in resectable and three versus four drug regimens in potentially resectable liver-limited disease.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
AS has received honoraria from Roche and Merck Serono. HJS has performed a consultant or advisory role at Roche, received an honorarium from Roche, and research support from Roche and Merck Serono.
