Abstract
Background:
Immune checkpoint inhibitors (ICIs) combined with chemotherapy, with or without angiogenesis inhibitors, have been investigated as the first-line treatment for extensive-stage small-cell lung cancer (ES-SCLC). However, it remains unclear which treatment modalities are the most effective and safest, as comparative studies evaluating these treatment options are limited.
Objectives:
This article aims to compare the relative efficacy and safety of ICIs + Chemo + angiogenesis inhibitors versus ICIs + Chemo as the first-line treatment for ES-SCLC.
Design:
A network meta-analysis was conducted to systematically compare the efficacy and safety data obtained from various clinical trials.
Data source and methods:
This study presents a systematic review and Bayesian network meta-analysis of data sourced from PubMed, Cochrane Library, EMBASE, ClinicalTrials.gov, and major international conferences up to August 31, 2024. Furthermore, this study analyzed both published works and gray literature on randomized clinical trials (RCTs).
Results:
A comprehensive analysis was conducted on 10 phase III RCTs comprising 2672 untreated ES-SCLC patients treated with two PD-L1 inhibitor combinations with angiogenesis inhibitors (ICIs + Chemo + angiogenesis) and eight PD-1/PD-L1 inhibitor combinations (ICIs + Chemo). Patients treated with ICIs + Chemo + angiogenesis inhibitors had higher progression-free survival (PFS) (hazard ratio (HR) = 0.56, 95% CI: 0.47–0.66) and overall response rate (ORR) (OR = 1.64, 95% CI: 1.17–2.31) compare to those who were treated with ICIs + Chemo. However, no significant difference was observed in the overall survival (OS; HR = 0.97, 95% CI: 0.79–1.19) and Grade ⩾ 3 adverse events (OR = 1.28, 95% CI: 0.81–2.04) between these patients. The subgroup analyses revealed that the addition of angiogenesis inhibitors improved the OS in patients under 65 years. Moreover, PFS was improved in all subgroups except the central nervous system metastasis.
Conclusion:
This study revealed that first-line immunochemotherapy combined with angiogenesis inhibitors improves PFS and ORR in ES-SCLC patients; however, it did not affect OS. Therefore, it was inferred that patients under the age of 65 can gain survival benefits from the addition of anti-angiogenic therapy.
Trial registration:
INPLASY (INPLASY2023110061).
Plain language summary
Extensive-stage small-cell lung cancer (ES-SCLC) is an aggressive neuroendocrine tumor. Immune checkpoint inhibitors (ICIs) combined with chemotherapy, with or without angiogenesis inhibitors, have been investigated as the first-line treatment for ES-SCLC. However, it remains unclear which treatment modalities is the most effective and safest, as comparative studies evaluating these treatment options are limited. This study compared the differences between the treatment modalities through a network meta-analysis and revealed that first-line immunochemotherapy combined with angiogenesis inhibitors improves PFS and ORR in ES-SCLC patients; however, it did not affect OS. Therefore, it was inferred that patients under the age of 65 can gain survival benefits from the addition of anti-angiogenic therapy. There was no difference in the safety.
Keywords
Introduction
Small-cell lung cancer (SCLC) is an aggressive neuroendocrine malignancy characterized by high vascularization and metastasis rate. Furthermore, it accounts for 15% of all lung cancers, with a 5-year survival rate of less than 7%. 1 Due to its high metastasis rate (60%–70%), most patients indicate extrathoracic cancer cell proliferation at the time of diagnosis, which limits surgical resection and hinders treatment. 2 Currently, the etiology and the pathogenesis of SCLC remain unclear. Studies have indicated that a history of heavy smoking and the inactivation of tumor suppressor genes, including TP53 and RB1, are associated with SCLC. 3 Furthermore, in the past three decades, the median overall survival (OS) of platinum-based chemotherapy has only reached 10 months. 4
Immunotherapy is an emerging approach that has been widely used in clinical practice. Several phase III clinical trials have revealed that co-treatment with first-line chemotherapy plus PD-1 or PD-L1 inhibitors, such as atezolizumab, durvalumab, and serplulimab, can prolong overall survival (OS) to 12.3–15.8 months.5–7 Moreover, due to the improved therapeutic response and acceptable safety profile, the FDA and NMPA have approved the above drugs in combination with chemotherapy as a first-line treatment for extensive-stage SCLC (ES-SCLC). However, first-line immunochemotherapy only improves the OS of ES-SCLC patients by 2–5 months. Because of the inevitable drug resistance and the constraints of subsequent treatment lines, improvement of the efficacy of first-line treatment and innovative therapeutic models are urgently required for SCLC patients.
Antiangiogenic therapy is another promising avenue for cancer treatment. Angiogenesis is modulated by various pro-angiogenic and anti-angiogenic factors and is critical for tumor recurrence, progression, invasion, and metastasis. 8 The inhibitors of vascular endothelial growth factor (VEGF) are of three types, including monoclonal antibodies, small molecule tyrosine kinase inhibitors (TKIs), and multitarget vascular disrupting agents. 9 A small-molecule and a multitarget TKI, Anlotinib, has indicated an increased OS of 2.4 months when applied as a later-line therapy for ES-SCLC patients compared to placebo.10,11 Therefore, NMPA has recommended anlotinib as a third-line or subsequent-line treatment option for ES-SCLC patients. Furthermore, the phase III clinical trial GOIRC-AIFA FARM6PMFJM indicated that compared to traditional chemotherapy, bevacizumab in combination with chemotherapy increased PFS by 1 month and the efficacy of antiangiogenic therapy in ES-SCLC. 12
According to preclinical research, immune checkpoint inhibitors (ICIs) and angiogenesis inhibitors may work better together by facilitating the reciprocal control of vascular normalization and immunostimulatory reprogramming. 13 A phase III clinical trial of benmelstobart (PD-L1 inhibitor) with anlotinib + chemotherapy enhanced the median OS and PFS to nearly 19 and 7 months, respectively, compared to chemotherapy alone. 14 However, since in these trials, standard chemotherapy was employed as the control group, and the adverse reactions were caused by drug superposition, the obtained results are controversial. Furthermore, the research on direct comparisons between immunochemotherapy and immunochemotherapy in conjunction with angiogenesis inhibitors is scarce; therefore, it is necessary to define the most effective combined strategy for future head-to-head clinical trials. Network meta-analysis (NMA) can quantitatively compare different intervention strategies and rank the effectiveness of certain outcome indicators to select the optimal treatment plan.
The present investigation assessed the efficacy and safety of all currently available first-line immunotherapy combinations for ES-SCLC patients. This systematic review and meta-analysis were performed by adjusting indirect comparisons grounded in a Bayesian framework and subgroup analyses of OS and PFS. These investigations are designed to identify the optimal immunotherapy combinations.
Materials and methods
Data sources and search strategy
A systematic search of electronic medical databases was performed on February 3, 2024, and updated on 31 August, 2024. The relevant literature was screened from the electronic databases, including PubMed, Cochrane Library, Embase, and ClinicalTrials.gov. Furthermore, the latest literature was obtained by reviewing online proceedings from various annual conferences. The search keywords comprised “immunotherapy, PD-1, PD-L1, Toripalimab, Atezolizumab, Durvalumab, Pembrolizumab, Adebrelimab, Serplulimab, Tislelizumab, Nivolumab, Benmelstobart, antiangiogenic, Anlotinib, bevacizumab.” The detailed overview of the search strategy is presented in Table S1. All the data were searched, screened, and extracted by two researchers (ZLJ and LYW) independently. A third researcher (WL) was approached in case of any disagreements. A systematic review was conducted according to the 2020 preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines 15 (Supplemental File 1). This study was registered on the International Platform of Registered Systematic Review and Meta-Analysis Protocols (INPLASY) on November 15, 2023, and was last updated on the same date (registration number INPLASY2023110061).
Selection criteria
The inclusion criteria were as follows: (1) randomized clinical trials (RCTs) that included ES-SCLC patients with diagnosis confirmed either histologically or cytologically, (2) RCTs that employed combinations of PD-1/PD-L1 inhibitors, either used with or without angiogenesis inhibitors, as the first-line treatment, (3) RCTs that compared combinations of PD-1/PD-L1 inhibitors, with or without angiogenesis inhibitors, against other treatment approaches for ES-SCLC, and (4) phase II and III studies that reported at least one of these clinical outcomes: (a) OS: specified as the duration from randomization until the incidence of death by any cause, (b) PFS: defined as the period from randomization until either disease progression or death by any cause, (c) ORR: the percentage of patients who achieved an objective response, and (d) adverse events (AEs): of any grade or grade ⩾ 3 was classified according to the National Cancer Institute Common Terminology Criteria for Adverse Events.
The exclusion criteria were as follows: (1) RCTs with overlapping patient populations, (2) RCTs presenting unclear clinical outcomes, (3) RTCs lacking PD-1/PD-L1 treatments, (4) studies that employed a single-arm design, (5) maintenance therapies, and (6) duplicate records.
Data extraction and quality assessment
Key clinical features, including trial name, publication sources, year of publication, sample size, age and sex distribution, smoking status, and the Eastern Cooperative Oncology Group Performance Status (ECOG PS) score, were obtained from the selected studies. The primary endpoints of this study were OS and PFS, for which hazard ratios (HRs) and 95% confidence intervals (95% CIs) were carefully extracted. Whereas the secondary endpoints included ORR and the incidence of Grade ⩾ 3AEs. This study evaluated the potential bias risks in the selected trials via the Cochrane Risk of Bias Tool. Moreover, quality assessment consists of selection bias, performance bias, detection bias, attrition bias, reporting bias, and other biases. The included studies were divided into low risk of bias (−), high risk of bias (+), and unclear bias (?) groups. The quality assessment was carried out by two independent reviewers (ZLJ and LYW), and any disagreements were resolved after reaching a consensus among ZLJ, LYW, and WL.
Statistical analysis
All the statistical assessments were carried out via the “netmeta” package in R Studio to derive HRs for time-to-event data and ORs for dichotomous outcomes. Furthermore, OS and PFS were represented as HRs, along with 95% CIs. Moreover, ORR and Grade ⩾ 3 AEs are presented as ORs with 95% CIs. To evaluate heterogeneity between the studies, the inconsistency test (I-squared, I2) was carried out. In case the I2 value exceeded 50%, indicating a significant risk of heterogeneity, a random-effects model was used for the meta-analysis; otherwise, a fixed-effects model was applied. 16
The standard Bayesian model was employed, which considers inconsistency and heterogeneity among the included studies. The effect size’s posterior distribution was computed using Gibbs sampling, following the Markov Chain Monte Carlo methodology, with a non-informative prior distribution applied. In total, 50,000 iterations were performed, and the first 10,000 iterations were designated as a burn-in sample (with a thinning interval of 1) to reduce the impact of the initial value. Effect sizes were articulated as HRs and ORs, along with their respective 95% CIs. Moreover, the SUCRA values were assessed to rank the safety and efficacy outcomes. To achieve a convergent diagnosis across all comparisons, the Brooks–Gelman–Rubin (BGR) diagnostic method was implemented. To ensure transitivity, trials adhering to strict patient allocation criteria were identified and included, and conditions for evaluated treatments were optimized. 17 In this study, the network structure of the NMA did not form any closed loops, making it impossible to directly assess inconsistency. Model fit was investigated by assessing the posterior deviance information criterion (DIC). The goodness-of-fit between the consistency model and the inconsistency model was compared. Differences between DIC from different models below 5 (Table S2), indicating that the consistency model provided a good fit to the data. Furthermore, sensitivity analyses were carried out by excluding studies with relatively low literature quality or high risk to verify whether these studies would affect the final results and conclusions. Finally, the consistency model was chosen because of stable results under different assumptions and without significant heterogeneity or inconsistency.
Results
Systematic review and characteristics of the included studies
During the preliminary literature search, 632 studies were identified from the database, and 5 extra online entries were sourced from meeting records. After removing duplicates and irrelevant articles via abstract screening, 45 studies were considered suitable for full-text review. Finally, 11 studies met the eligibility requirements (Figure 1) and included 4815 patients who underwent the following 12 treatments: adebrelimab + chemotherapy (adeb-chemo), atezolizumab + chemotherapy (atez-chemo), durvalumab + chemotherapy (durv-chemo), nivolumab + chemotherapy (nivo-chemo), pembrolizumab + chemotherapy (pemb-chemo), serplulimab + chemotherapy (serp-chemo), tislelizumab + chemotherapy (tisl-chemo), toripalimab + chemotherapy (tori-chemo), chemotherapy (chemo), tiragolumab with atezolizumab and chemotherapy (tira-atez-chemo), benmelstobart with anlotinib + chemotherapy (benm-anlo-chemo), atezolizumab with bevacizumab + chemotherapy (atez-beva-chemo). Comprehensive details of all the included studies are presented in Table 1. Each study’s full outcome reports were obtained, and all the studies adhered to the principle of random allocation and exhibited a low risk of bias. The evaluation of the risk of bias is illustrated in Figure S2, and the network diagrams are shown in Figures 2 and 3 and Figure S1.

Literature search and selection. The study process followed the PRISMA guidelines.
Baseline characteristics of studies included in the network meta-analysis.


Network diagrams of comparisons on different treatments.

Network map of three treatment arms of ICIs + Chemo + angiogenesis inhibitors, ICIs + Chemo, and Chemotherapy.
The chemo and tira-atez-chemo treatments functioned solely as transitivity nodes for indirect comparisons; therefore, they were not included in the subsequent analyses.
Comparisons of OS, PFS, and ORR
The OS analysis (Figure 3) revealed that ICIs + Chemo + angiogenesis inhibitors (HR = 0.97, 95% CI: 0.79–1.19) did not substantially improve OS when compared to ICIs + Chemo. Furthermore, the most effective first-line treatment regimen for ES-SCLC patients was also assessed. There were no significant differences in OS across the different treatment regimens for ES-SCLC (Figure 4). However, according to the Bayesian ranking profiles (Figure 5), benm-anlo-chemo had the highest probability (41%) of enhancing OS (Table S3).

HRs, ORs, and their 95% confidence intervals from network meta-analysis of different first-line therapeutic regimens in ES-SCLC patients. (a) HRs and 95% CI for overall survival (upper triangle in yellow) and progression-free survival (lower triangle in green). The hazard ratio < 1.00 provides better survival benefits. (b) ORs and 95% CI for objective response rate (upper triangle in yellow) and grade ⩾ 3 adverse events (lower triangle in green).

Bayesian ranking profiles of comparable treatments on efficacy and safety for ES-SCLC patients.
The PFS analysis (Figure 3) showed that ICIs + Chemo + angiogenesis inhibitors (HR = 0.56, 95% CI: 0.47–0.66) substantially improved PFS compared to ICIs + Chemo. Furthermore, benm-anlo-chemo surpassed all other regimens (Figure 4). As per Bayesian ranking profiles (Figure 5), benm-anlo-chemo had the highest probability (98%) of achieving improved PFS (Table S3). Among the combination of immunotherapy and chemotherapy, serp-chemo significantly outperformed adeb-chemo, tori-chemo, nivo-chemo, atez-chemo, pemb-chemo, and durv-chemo. Moreover, atez-beva-chemo significantly increased PFS relative to atez-chemo (HR = 0.67, 95% CI: 0.52–0.86), pemb-chemo (HR = 0.64, 95% CI: 0.44–0.95), and durv-chemo (HR = 0.64, 95% CI: 0.44–0.95).
The analysis of endpoints of ORR (Figure 3) showed that compared with ICIs + Chemo, ICIs + Chemo + angiogenesis inhibitors (OR = 1.64, 95% CI: 1.17–2.31) significantly increased ORR. Furthermore, this study conducted the ORR analyses for nine studies (Figure 4), which showed that benm-anlo-chemo (OR = 2.59, 95% CI: 1.47–4.63) improved the patient’s ORR compared with atez-chemo. However, there was no difference in ORR between atez-beva-chemo and other regimens. Based on the Bayesian ranking profiles (Figure 5), benm-anlo-chemo indicated the highest probability (63%) of achieving ORR (Table S3).
Safety and toxicity
The findings indicated no significant difference in the incidence of Grade ⩾ 3 AEs (Figure 3) between ICIs + Chemo + angiogenesis inhibitors and ICIs + Chemo (OR = 1.28, 95% CI: 0.81–2.04). Furthermore, Grade ⩾ 3 AEs analysis revealed that benm-anlo-chemo had a higher risk of Grade ⩾ 3 AEs than durv-chemo and tisl-chemo (OR = 2.36, 95% CI: 1.01–5.60) (Figure 4). Moreover, per the Bayesian ranking profiles (Figure 5), nivo-chemo had the highest probability (50%) of ranking first as the most toxic treatment strategy. Included clinical trials reported the following treatment-related AEs of any grade associated with immunotherapy combinations: neutropenia, white blood cell count, platelet count, anemia, nausea, vomiting, and constipation. Moreover, the addition of angiogenesis inhibitors produced some unique AEs, including hypertension, proteinuria, and hematuria (Table S4). In the BEAT-SC study, the incidence of hypertension was 31.3%, proteinuria was 24%, and palmar-plantar erythrodysesthesia syndrome was 11.4%. This study also compared specific AEs, such as neutropenia, anemia, nausea, vomiting, and constipation, which indicated no significant difference between ICIs + Chemo + angiogenesis inhibitors and ICIs + Chemo. The incidence of all grade AEs, serious AEs, and those resulting in treatment interruption and death was not significantly increased with the addition of angiogenesis inhibitors (Figure 6).

The forest plot of adverse events for ICIs + Chemo + Angiogenesis inhibitors versus ICIs + Chemo.
Subgroup analysis
The subgroup analysis was carried out using five dimensions: gender, age, performance status, brain metastasis status, and liver metastasis status (Figure 7). The data revealed an imbalance in OS benefits among patients of different ages. Furthermore, ICIs + Chemo + angiogenesis inhibitors (HR = 0.42, 95% CI: 0.29–0.61) yield better OS in patients under 65 years old than ICIs + Chemotherapy. In patients with brain and liver metastases, no difference in OS was observed between these treatment options. For PFS, the addition of angiogenesis inhibitors (HR = 0.66, 95% CI: 0.41–1.07) may not benefit patients with CNS metastasis.
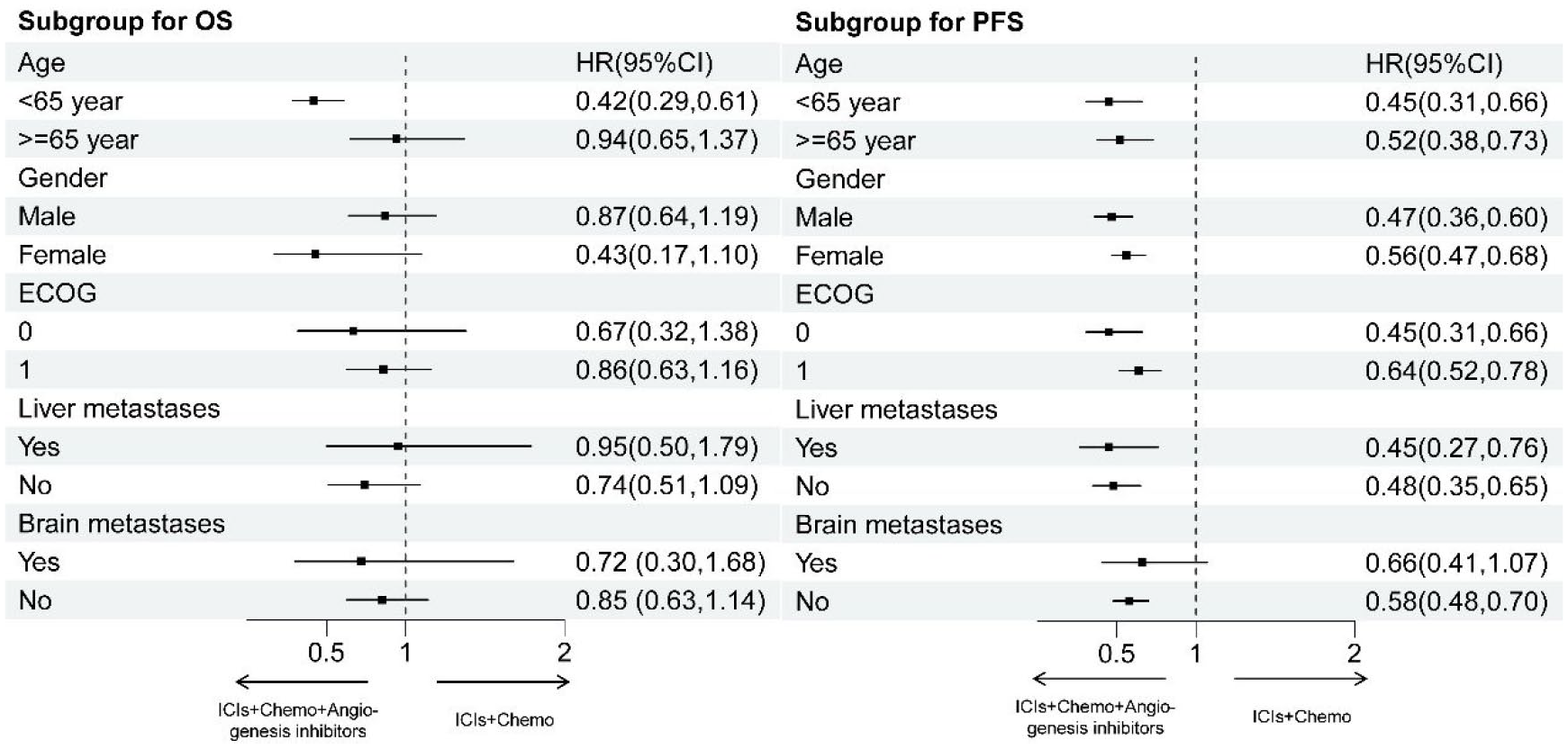
Subgroup analyses for OS and PFS.
Discussion
The treatment of SCLC has always been a challenge, and platinum-based therapy has historically served as the first-line treatment for a long time; however, recently, novel treatment strategies have emerged. The development of immunotherapy has accelerated since the advent of atezolizumab and durvalumab.5,6 Serplulimab is the first PD-1 drug with positive results as the first-line regimen for ES-SCLC treatment. Furthermore, in the Chinese population, adebrelimab has indicated excellent therapeutic outcomes. 7 Compared to the 2% previously attained with platinum-based therapy, a new trial on atezolizumab revealed a 5-year OS rate of 12% in patients with ES-SCLC, indicating a considerable increase in treatment options. 18 To further enhance the therapeutic effect, angiogenesis inhibitors have been added to the first-line treatment. Currently, only a few studies directly compare the efficacy and toxicity of immunochemotherapy combined with angiogenesis inhibitors and immunochemotherapy. This study revealed that immunochemotherapy combined with angiogenesis inhibitors can improve PFS and ORR in untreated ES-SCLC patients. However, these benefits have not been observed for OS.
To the best of our knowledge, this article aims to compare the relative efficacy and safety of ICIs + Chemo + angiogenesis inhibitors versus ICIs + Chemo as the first-line treatment for ES-SCLC. The results of the present study are consistent with Ying Liu’s conclusion, which suggests that immunochemotherapy combined with angiogenesis inhibitors primarily benefits PFS and does not provide a significant advantage in OS compared to PD-1 or PD-L1 combined with chemotherapy. 19 The ETER701 and BEAT-SC trials incorporated anti-angiogenic drugs into the co-treatment of immunotherapy and chemotherapy, indicating an innovative approach. 14 Benmelstobart is a novel PD-L1 inhibitor that enhances the immune system’s attack on tumors by inhibiting the binding of PD-L1 to PD-1, thus alleviating the immune suppression of tumor cells on T cells. Furthermore, anlotinib (multi-target TKI) and bevacizumab (monoclonal antibodies) have been found to inhibit tumor angiogenesis by targeting the vascular endothelial growth factor (VEGF) receptor, thereby disrupting the tumor’s blood supply. 10 Moreover, it has been observed that targeting VEGF can induce an immune response in cancer models by normalizing tumor vasculature, increasing T-cell infiltration, promoting dendritic cell maturation, and inhibiting the activity of immunosuppressive cells.20–22 Although preclinical studies have revealed potential benefits, due to the short OS of ES-SCLC, the inability to improve OS will impede the development of immunotherapy combined with angiogenesis inhibitors and chemotherapy.
The subgroup data analysis revealed that, in terms of OS, only patients under the age of 65 showed divergent outcomes from the general population. This discrepancy may be attributed to the toxicity tolerance in this demographic. Therefore, more effective treatment strategies should be pursued to further increase benefits.
For PFS, the subgroup population was largely comprised of the overall population, while patients with brain metastases did not benefit from the addition of angiogenesis inhibitors. In this study, brain metastasis patients from each study made up 1.3%–22.8% of the total, and only four literature reports on subgroup analysis of brain metastases were analyzed, which may have increased the overall uncertainty of the obtained results. The treatment of CNS metastasis patients is relatively complex, and the efficacy of chemotherapy, immunotherapy, and angiogenesis inhibitors is limited. Furthermore, traditional radiotherapy still plays an important role. Subgroup analysis of the Ideate-lung01 trial suggested that B7-H3 antibody–drug conjugate therapy offers specific advantages for targeting brain lesions. 23 Hopefully, in the future, more new drugs will be developed for ES-SCLC patients with brain metastasis.
Liver metastasis is a common site of extra-thoracic metastasis, with an incidence rate of 35% 24 and a poor prognosis. The liver is characterized as an immunosuppressive organ, and although immune therapies have shown some advantages over traditional chemotherapy, these benefits remain limited. 25 The low immunogenicity and reduced infiltration of CD8+ T cells may contribute to this trend. Moreover, incorporating angiogenesis inhibitors has indicated improvements in only PFS and not OS. These observed benefits in PFS may be related to their potential to ameliorate the immunosuppressive environment, which improves therapeutic outcomes. 26
The spectrum of AEs between ICIs + Chemo + angiogenesis inhibitors and ICIs + Chemo is relatively similar. Here, several representative AEs were analyzed, which revealed no significant differences between the two types of treatments. Moreover, the addition of angiogenesis inhibitors did not increase the incidence of serious AEs and death, indicating that the adverse reactions of ICIs + Chemo + angiogenesis inhibitors can be controlled. However, angiogenesis inhibitors may result in some unique AEs, such as 31.3% and 24% of hypertension and proteinuria of any grade were observed in the benm-anlo-chemo, respectively, necessitating vigilance from clinical practitioners. It is, therefore, crucial to understand the toxicity profiles of first-line treatments for informed clinical decision-making, particularly in combination therapies. This knowledge enables physicians to customize the most suitable individualized treatment plans, especially for elderly patients.
Limitations
There are certain limitations in this study. First, although a comprehensive search strategy was established, the potential for publication bias exists due to the exclusion of unpublished literature. Second, the small number of studies meeting our inclusion criteria may have increased uncertainty in the results, particularly as some had limited sample sizes. Third, patients were not stratified by factors such as race; therefore, studies like CAPSTONE-1, RATIONALE-312, and EXTENTORCH exclusively included participants from China, which could potentially affect the perceived treatment benefits. It should be considered that the effectiveness of immunotherapy and chemotherapy may differ between Asian and Western populations. Therefore, future research should investigate the relative therapeutic effects associated with these clinical characteristics.
Conclusion
In summary, this study indicated that immunochemotherapy combined with angiogenesis inhibitors has certain advantages in PFS and ORR in untreated ES-SCLC patients. However, these benefits were not translated into improved OS, and only patients under 65 years old may potentially benefit from this co-treatment strategy. Moreover, the addition of angiogenesis inhibitors did not significantly increase toxicity. In the future, further randomized studies are required to confirm these findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251348310 – Supplemental material for Efficacy and safety of first-line PD-1/PD-L1 inhibitors combined with or without anti-angiogenesis therapy for extensive-stage small-cell lung cancer: a network meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251348310 for Efficacy and safety of first-line PD-1/PD-L1 inhibitors combined with or without anti-angiogenesis therapy for extensive-stage small-cell lung cancer: a network meta-analysis by Linjing Zhou, Yuwei Li, Le Wang, Kaiyan Chen, Shichao Zhou, Yunfei Chen, Jing Sun, Yunfeng Tong and Yun Fan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
