Abstract
Background:
Patients with non-small cell lung cancer (NSCLC) presenting with mesenchymal–epithelial transition (
Methods:
We performed a retrospective, international, multicenter efficacy and safety analysis in patients with NSCLC treated with capmatinib in an early access program between March 2019 and December 2021.
Results:
Data from 81 patients with advanced
Conclusion:
In patients with
Introduction
During the last decade, remarkable progress has been made in the personalized treatment of non-small cell lung cancer (NSCLC), which is the most frequent type of lung malignancy, accounting for 84% of all lung cancer diagnoses.1,2
The c-mesenchymal–epithelial transition proto-oncogene – known as
In the last two decades, MET-targeting small-molecule kinase inhibitors, conventional therapeutic monoclonal antibodies, and antibody-based biotherapeutics led mainly to disappointing outcomes in preclinical and clinical trials.15,16 Different MET-targeted therapies are currently under investigation, including monoclonal antibodies against MET or its ligand [hepatocyte growth factor (HGF)],
17
and small-molecule MET inhibitors. The role and efficacy of immune-checkpoint inhibitors in treatment of patients with
Capmatinib is a potent and highly selective small molecule MET inhibitor, which has shown substantial and clinically meaningful antitumor activity in cancers presenting with various types of MET activation.8,19–21 Moreover, capmatinib is known to cross the blood–brain barrier and demonstrates intracranial efficacy in patients with NSCLC harboring
Real-life data of
Methods
Study design
This is a retrospective, non-interventional, multicenter real-world analysis called Real-world Experience with Capmatinib (RECAP), which aims to evaluate the efficacy and safety of
The primary endpoint of this retrospective data analysis was the ORR – proportion of patients with complete response (CR) and partial response (PR) defined according to RECIST v1.1 criteria. 23 The secondary endpoints were the following: (i) evaluation of treatment-related adverse events (TRAEs) determined by the treating physician; (ii) disease control rate (DCR) defined as the proportion of patients with CR, PR, and stable disease (SD); (iii) intracranial ORR (icORR); (iv) median duration of response (mDoR) assessed as the time between the initial response to therapy and subsequent disease progression or death due to any cause; (v) mPFS measured as the time from the first dose of capmatinib to the first progression event [progressive disease (PD) or death if no PD documented until then, irrespective of cause of death]; and (vi) median overall survival (mOS) defined as the time between date of diagnosis of advanced stage lung cancer and death.
Study population and treatment
Oncological and pneumological centers specialized in the treatment of lung cancer from seven different countries (Austria, France, Israel, The Netherlands, Slovenia, Sweden, and Switzerland) contributed to this dataset. Data from all eligible patients treated by physicians who participated in a capmatinib EAP were included. For inclusion, the following criteria had to be met: histologically confirmed NSCLC with locally advanced or metastatic disease, age ⩾18 years, confirmed
Next-generation sequencing (NGS)-based genomic profiling, Sanger sequencing, or polymerase chain reaction (PCR) from tissue and/or liquid biopsy were used for the identification of
Capmatinib was taken orally (standard dose of 400 mg twice daily). Reduced starting dose, dose reductions, and re-escalations were decided at treating physicians’ discretion. Capmatinib treatment was continued until disease progression, lack of clinical benefit, unacceptable toxicity, patient’s withdrawal of consent, or the treating physicians’ decision.
Data collection
In 2019, capmatinib became available through an EAP for the treatment of patients with advanced NSCLC harboring a
Clinical characteristics and treatment data were extracted from medical records, anonymized by the treating physicians and transferred for statistical analysis. Data included information about patients’ demographics and clinical characteristics [country, gender, date of birth, ethnicity, smoking and Eastern Cooperative Oncology Group (ECOG) performance status, disease stage, previous treatments, histology,
Efficacy and safety assessments
According to clinical practice at each institution, a CT scan of the chest and abdomen performed every 6–12 weeks was used to evaluate tumor response and progression per RECIST v1.1. Additional brain CT and/or MRI assessment were done according to institutional standard of care.
In the overall population, as well as for each subgroup (treatment-naïve and pretreated patients), the following efficacy parameters were analyzed: ORR, DCR, duration of response (DoR), progression-free survival (PFS), and overall survival (OS). Tumor response (maximum change in tumor size) was compared through pretreatment lesion measurements performed at baseline and post-treatment (at least one imaging evaluation). For calculation of intracranial response, only patients with untreated or progressing brain lesions were included. Measurable brain lesion was defined as ⩾5 mm at baseline.
Adverse events (AEs) were graded as per Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. Treatment relation of an AE to capmatinib was assessed by the treating physicians. Safety monitoring was performed at baseline, at every subsequent evaluation visit, or as clinically indicated. A documentation of each dose modification or interruption, as well as treatment discontinuations, related to TRAEs was done.
Ethics approval and informed consent
The study protocol was approved by the ethics committee of the city of Vienna, Austria (EK-21-239-1121). Informed consent was obtained in accordance with local legislation in the respective countries at each study side. According to Austrian laws, informed consent for each patient was not necessary for this retrospective analysis. The study was conducted according to the principles of the Declaration of Helsinki.
Statistical analysis
PFS, OS, and DoR have been analyzed using the Kaplan–Meier method and derived related 95% confidence intervals (CI). DoR was calculated for all patients who achieved CR or PR; if a patient died, irrespective of cause of death, without PD beforehand, then the date of death has been used as end date. Concerning the PFS, patients without any documented progression and who are alive at the time of data cut-off have been censored at time of data cut-off or last contact. Patients who initiated subsequent anticancer therapy in the absence of documented PD (e.g., discontinued treatment due to an AE) have been censored at time of treatment discontinuation. Data for patients who were lost to follow-up or alive have been censored for the OS at the date of last contact. Confidence interval for proportions, such as ORR and DCR, has been calculated using the exact Clopper–Pearson method. For a comparison of subgroups defined by previous lines of systemic anticancer therapy, a log-rank test with a level of significance of 5% (chi-square
All statistical analyses have been conducted using SPSS software (v.27.0, IBM SPSS Statistics) and RStudio v.1.4.1106. Tables and figures have been created by using SPSS v.27.0 (IBM SPSS Statistics), Microsoft Excel 2019, and RStudio v.1.4.1106.
Results
Patients
We included 81 patients with locally advanced or metastatic
Demographics and characteristics of patients prior to capmatinib administration.

Data cut-off date: November 8, 2021.
Percentage may not be 100 because of rounding.
As defined by the National Lung Screening Trial. 24
ECOG performance status, with higher numbers indicating worse daily living capability.
One patient was treated in stage IIIa but with palliative intend and one patient was down-staged after capmatinib therapy.
Previous regimens defined as at least one dose of chemotherapy and/or immunotherapy or one dose of TKI; one patient received a combination of capmatinib and pembrolizumab; one patient received first osimertinib during 2.5 months before capmatinib was additionally administered; two patients received tepotinib (c-MET inhibitor) but had to stop the therapy because of adverse events and received then capmatinib; one patient received APL 101 (c-MET inhibitor).
Five patients received chemo- and anti-VEGF therapies.
One patient received immuno- and anti-TIGIT therapy. One patient received immunotherapy and lenvatinib.
TKIs administered include crizotinib (12 patients), cabozantinib (3 patients), tepotinib (2 patients), afatinib (1 patient), gefitinib (1 patient), and osimertinib (1 patient). Two patients received more than one prior TKI.
As already defined, most common clones used for PD-L1 testing were SP263 and 22C3. 17
Five patients were tested negative for
Anti-PD-1, anti-cell death protein 1; anti-TIGIT, T-cell immunoreceptor with immunoglobulin and immunoreceptor tyrosine-based inhibitory motif domain; anti-VEGF, vascular endothelial growth factor; ECOG, Eastern Cooperative Oncology Group; NA, not applicable; NOS, not otherwise specified; NSCLC, non-small cell lung cancer; PCR, polymerase chain reaction; PD-L1, programmed death-ligand 1; EGFR, epidermal grow factor receptor; GCN, gene copy number; py, pack years; TKIs, tyrosine kinase inhibitors.
At the time when capmatinib treatment started, most patients (86%) had stage IV disease, and the most frequent site of metastasis was bones (36%), lung (35%), pleura (31%), and/or brain (27%). In total, 40% of patients presented with only a single-site metastatic lesion. Pretreated patients had received a median number of one therapeutic agent prior to capmatinib (range, 1–5). Prior regimens included platinum-based therapies (70%), anti-PD-1 (anti-cell death protein 1) or PD-L1 treatments (61%), and tyrosine kinase inhibitors (TKIs) (41%).
The majority of patients (78%) presented with an adenocarcinoma. The primary testing method of
Response
The efficacy results are presented in Table 2. The overall ORR was 58% (95% CI, 47–69), with two patients (3%) having CR and 45 patients (56%) having PR. Non-responders included 12 patients (15%) showing PD and 18 patients (22%) SD. Four patients (5%) had no measurable target lesion. Both CRs were observed among pretreated patients, but the proportion of PRs was higher in the treatment-naïve group (68%
Efficacy of capmatinib in

Data cut-off date: November 8, 2021. ORR, PFS assessed according to RECIST v1.1 for patients with measurable disease.
Percentage may not equal to 100 because of rounding.
ORR was including complete or partial response.
DCR was including complete response, partial response, or stable disease.
PFS was calculated from start of therapy to progression or death independent of reason of death. Patients who have no documented progression and are alive at the time of data cut-off have been censored at time of data cut-off or last contact. Patients who initiate subsequent anticancer therapy in the absence of documented PD (e.g., discontinued treatment due to adverse events) have been censored at time of treatment discontinuation.
OS was calculated from start of capmatinib treatment to date of death independent of cause. Patients who are alive or lost to follow-up have been censored at last date known alive.
Only includes patients with measurable brain lesions ⩾5 mm and recent follow-up MRI or CT, who did not have prior intervention of brain metastasis, or prior intervention but progression of brain lesions before capmatinib start.
CI, confidence interval; CR, complete response; CT, computed tomography; DCR, disease control rate; (ic)DCR, (intracranial) disease control rate; (ic)ORR, (intracranial) ORR; MRI: magnetic resonance imaging; NR, not reached; ORR, objective response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; TRAEs, treatment-related adverse events.
Maximum change in tumor size related to baseline of 75 patients (retrospective measurement of lesions was not possible for two patients) is shown in Figure 1(a).

Best response to capmatinib. Waterfall plots of maximum change in tumor size measured according to RECIST v1.1 in all target lesions between baseline and follow-up imaging in pretreated and treatment-naïve patients in the overall population (a) and in patients with baseline intracranial target lesions (b). Both growth (+20%) and shrinkage (−30%) of tumor size are indicated by the dashed lines.
The median DoR was still immature. The DoR for individual patients are presented in Supplemental Figure S1. At a median follow-up of 9.5 months, 70% (33 of 47) of the responses were ongoing.
Figure 2(a) shows the response to capmatinib according to the starting dose, PD-L1 expression, and TP53 status. A slightly higher response was observed among patients having received a full starting dose compared with reduced starting dose (ORR, 61%
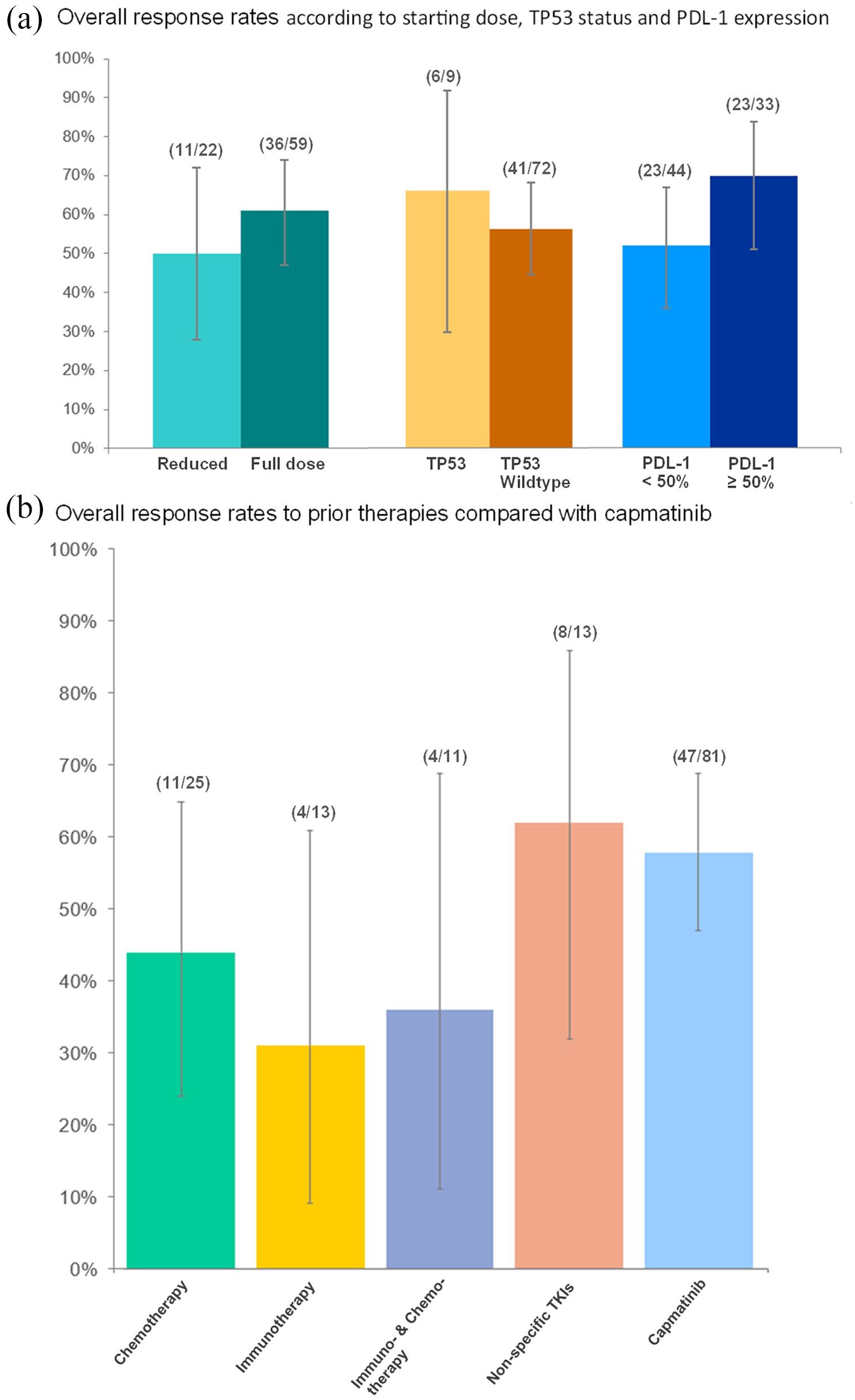
Comparison of response rates. (a) Response according to starting dose and molecular characteristics. (b) Response to prior therapies compared with capmatinib.
Figure 2(b) and Supplemental Table S1 show the ORR to prior therapies of pretreated patients compared to capmatinib. ORR to capmatinib and non-specific TKIs (crizotinib and cabozantinib) (ORR, 62%) were higher compared with chemotherapy (44%), chemoimmunotherapy (36%), or immunotherapy (31%). Higher DCR were observed for capmatinib (81%
Progression-free survival and overall survival
After a median follow-up of 10.7 months, the mPFS was 9.5 months (95% CI, 4.7–14.3) in the overall population, 10.6 months (95% CI, 5.5–15.7) in the treatment-naïve, and 9.1 months (95% CI, 3.1–15.1) in the pretreated patients (Table 2, Figure 3(a) and (b)). At data cut-off, 43% of treatment-naïve patients and 59% of the previously treated patients had disease progression, which was mainly systemic (81%
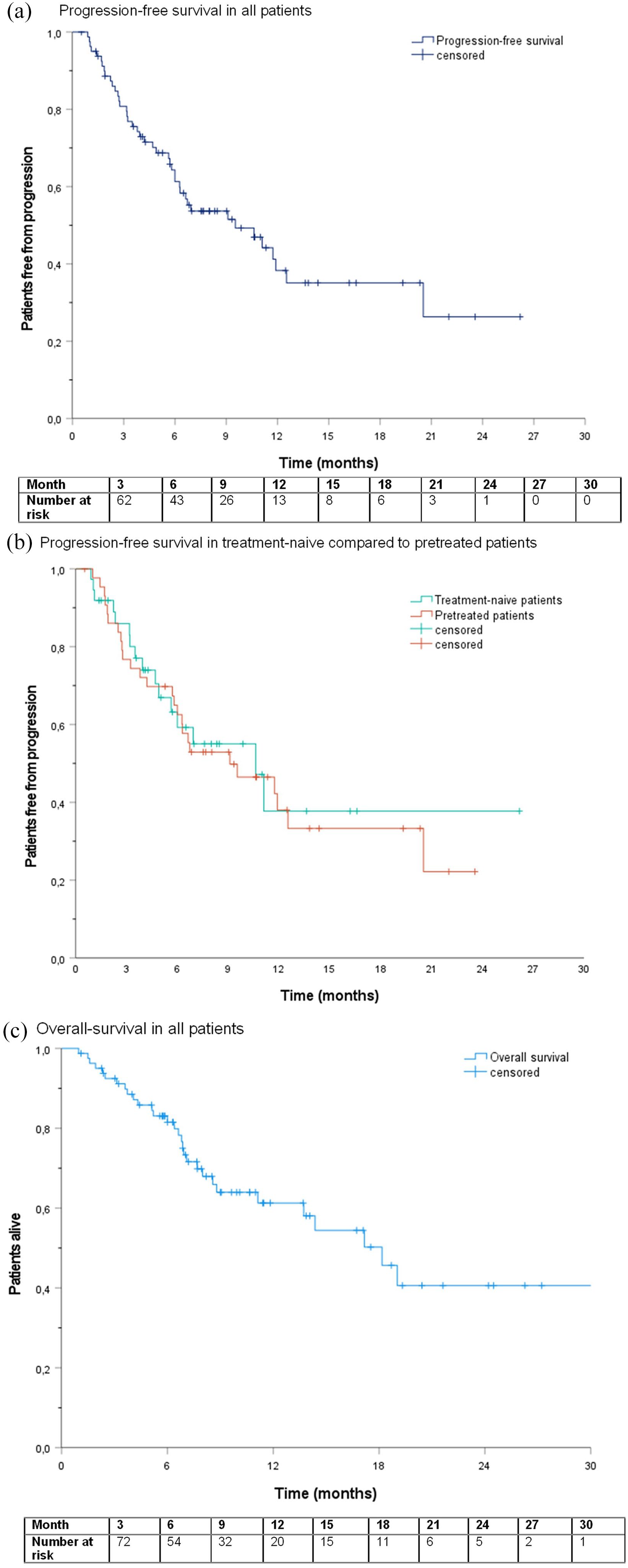
Progression-free survival (PFS) and overall survival (OS). Kaplan–Meier plots of median PFS in the overall population (a) and in previously treated and treatment-naïve patients (b), as well as of the median OS in the overall population (c).
The mOS was 18.2 months (95% CI, 13.2–23.1) after a median follow-up of 11.0 months (Figure 3(c)). In pretreated patients, the mOS reached 17.2 months (95% CI, 6.7–27.7), whereas the mOS was not reached in the treatment-naïve group (Table 2).
Intracranial outcome
At baseline 22 patients (27%) had confirmed brain lesions, 55% of them being asymptomatic (Table 2). The overall mPFS in these patients was 9.1 months (95% CI, 4.0–14.2), with 38% of patients showing intracranial progression. The progression rate was similar in both analyzed groups (58%
Safety
The TRAEs that occurred at any grade are presented in Table 3. Overall, 61 patients (75%) experienced TRAEs, although most of them of low severity (grade ⩽ 2) (Figure 4). Peripheral edema was the most common TRAE (
Treatment-related adverse events (TRAEs) that occurred at any grade in patients treated with capmatinib (

Data cut-off date: November 8, 2021.
This analysis included any patient who received at least one dose of capmatinib; TRAEs were graded as per Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, as determined by the treating physician.
Percentage may not equal to 100 because of rounding.
In three patients, dose reduction occurred because of two simultaneously TRAEs; in two other patients, dose reduction was due to three TRAEs at once.
In two patients, treatment interruption occurred because of two simultaneously TRAEs.
Liver enzymes are related to aspartate aminotransferase (AST) and alanine aminotransferase (ALT), bilirubin, and gamma-glutamyl transferase (GGT).

Treatment-related adverse events (TRAEs). Data cut-off date: November 8, 2021; TRAEs that occurred at any grade in at least 2% of treated patients. The analysis included all patients who received at least one dose of capmatinib. Relatedness of any adverse event to the treatment was assessed by the treating physician. TRAEs were graded as per Common Terminology Criteria for Adverse Events (CTCAE, version 5.0) as determined by the treating physician. Percentage may not equal to 100 because of rounding; liver enzymes were including aspartate aminotransferase (AST), alanine aminotransferase (ALT), bilirubin, and gamma-glutamyl transferase (GGT).
For most patients (
Discussion
Because of a significant unmet medical need for the treatment of patients with advanced NSCLC harboring
The RECAP analysis evaluated capmatinib under real-world conditions, and, so far, represents the largest published retrospective data set on capmatinib for the treatment of
As expected, the real-world RECAP patient population was less selected and therefore, patients presented with less favorable baseline conditions compared to the ones recruited in the GEOMETRY Mono-1 trial. In our population, 31% of patients had an ECOG performance status ⩾2 (
Both the ORR and DCR in our patient population were comparable with the results from the GEOMETRY Mono-1 trial. In contrast, a higher rate of previously treated patients responded in our study (50%
In the RECAP population, an encouraging intracranial activity of capmatinib was observed. In patients with measurable brain lesions according to protocol, the icORR reached 46% (including 18% with a CR) and a icDCR of 91%. In addition, mPFS in patients with intracranial disease was similar to overall population in RECAP. These results are comparable with the results of the phase II trial with an icORR of 54% and a icDCR of 92%.
No new safety signals were reported in the RECAP study. Capmatinib showed a manageable safety profile and low discontinuation rates, with mainly low-grade and reversible TRAEs. However, dose reductions and treatment interruptions were frequently necessary emphasizing the importance of regular monitoring of patients during capmatinib therapy. TRAEs grade 3/4 were reported in 23% of patients in the RECAP cohort, compared to 53% of patients with
When analyzing the prior therapies administered to patients in RECAP, capmatinib achieved a higher response rate than chemotherapy or immunotherapy, either as monotherapy or combined. This finding must be interpreted with caution due to the small sample size. In RECAP, 40% of patients had a high PD-L1 expression (TPS ⩾ 50%); which is in line with previous published report.
27
However, in
Additionally,
The challenge for pathologists is to timely identify
This retrospective analysis carries several inevitable limitations, such as selection bias, reporting bias, and information bias. Moreover, given the small sample size of some subgroups, only descriptive efficacy outcomes have been presented. Additionally, inherent limitations to clinical routine practice in each participating center – especially in terms of MET testing methods, intervals of radiographic assessments, and national- or hospital-based treatment guidelines – should be considered. With those limitations in mind, our real-world results were however in line with previously reported phase II clinical data. 8
In the case of rare disease, data from patients treated in real-world settings are essential to assess treatment efficacy and safety in non-selected patient populations presenting with comorbidities and poor performance status. To date, several trials are testing selective MET inhibitors – as monotherapy or in combinations – in NSCLC.11,31 Anticipating the emergence of acquired resistance against current MET inhibitors, co-alterations that might be involved in treatment escape mechanisms have been already identified32,33; however, the frequency and the type of resistance may change with broader use of more potent and specific MET-TKIs or MET molecular antibodies. 11
Supplemental Material
sj-docx-1-tam-10.1177_17588359221103206 – Supplemental material for Real-world experience with capmatinib in MET exon 14-mutated non-small cell lung cancer (RECAP): a retrospective analysis from an early access program
Supplemental material, sj-docx-1-tam-10.1177_17588359221103206 for Real-world experience with capmatinib in MET exon 14-mutated non-small cell lung cancer (RECAP): a retrospective analysis from an early access program by Oliver Illini, Hannah Fabikan, Aurélie Swalduz, Anders Vikström, Dagmar Krenbek, Michael Schumacher, Elizabeth Dudnik, Michael Studnicka, Ronny Öhman, Robert Wurm, Luciano Wannesson, Nir Peled, Waleed Kian, Jair Bar, Sameh Daher, Alfredo Addeo, Ofer Rotem, Georg Pall, Alona Zer, Akram Saad, Tanja Cufer, Hadas Gantz Sorotsky, Sayed M. S. Hashemi, Katja Mohorcic, Ronen Stoff, Yulia Rovitsky, Shoshana Keren-Rosenberg, Thomas Winder, Christoph Weinlinger, Arschang Valipour and Maximilian J. Hochmair in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Medical writing assistance was provided by Dr. Florence Boulmé.
Author contribution(s)
Compliance with ethical standards
The study protocol was approved by the ethics committee of the city of Vienna, Austria (EK-21-239-1121). According to Austrian laws, informed consent for each patient was not necessary for this retrospective analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This academic collaboration was supported by the Karl Landsteiner Institute for Lung Research and Pulmonary Oncology.
Conflict of interest statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
