Abstract
Background:
Stage III non-small cell lung cancer (NSCLC) being highly heterogeneous requires multimodal therapeutic strategies for optimal management. We present findings on treatment patterns and their associated survival outcomes in patients with stage III NSCLC from the Egypt subset of the KINDLE global real-world study conducted across countries from Asia, Middle East, Africa, and Latin America.
Method:
Retrospective data from the Egypt subset (21 centers) of adult patients diagnosed with stage III NSCLC between January 2013 and December 2017 were analyzed. Descriptive and inferential statistics summarized treatment modalities, progression-free survival (PFS), and overall survival (OS).
Results:
Of 421 patients enrolled (median age: 59.0 years), 77.9% were males, 53.5% had stage IIIA disease, 60.8% had adenocarcinoma, 78.4% had an unresectable disease, and 81.5% had Eastern Cooperative Oncology Group performance status ⩽1. Overall, chemotherapy alone (40.4%) was predominantly used in the initial line, whereas definite radiotherapy was used in only 5.0% of patients. In resectable patients, chemotherapy plus surgery (33.8%), surgery alone (20.6%), or other surgery (20.6%) were the top three modalities used in initial line of treatment. Chemotherapy alone was most preferred (48.8%) in unresectable patients, followed by sequential chemoradiotherapy (CRT) (17.6%) and concurrent CRT (9.3%). The overall median PFS was 10.3 months [95% confidence interval (CI), 9.43–12.02], whereas the median OS was 18.5 months (95% CI, 16.46–21.88). Overall, female gender, adenocarcinoma histology, and radical therapy as surgery or CRT predicted significantly longer OS (all p < 0.05).
Conclusion:
KINDLE-Egypt cohort revealed wide heterogeneities in the treatment patterns of stage III NSCLC. Although deemed resectable, few patients did not undergo surgery, probably due to high smoking rates leading to poor lung function. Lower survival outcomes than other published real-world studies highlight the need for timely approval and availability of novel targeted and immunotherapies to enhance patient outcomes.
Trial registration:
NCT03725475
Keywords
Introduction
Globally, lung cancer (LC) has emerged as a lethal malignancy, with high incidence rates in the Middle East and Africa (MEA) region.1,2 LC is one of the five most common cancers in Egypt with an estimated incidence of 4.9% and 5-year prevalence of 6.95 per 100,000 in 2020. 3 It is reported as the fifth most common cancer in men in Egypt, 4 with an age-standardized mortality rate of 7.2 per 100,000. 3 Tobacco smoking is a significant predictor of LC in Egypt, especially among the males with an age-standardized prevalence of 43.4% [95% confidence interval (CI), 42.2–44.7], which is higher than the global average (32.7%; 95% CI, 32.3–33.0). 5
About 85% of LCs are categorized as non-small cell LC (NSCLC) with adenocarcinoma being the predominant variant of all NSCLC tumors. 6 Nearly one-third of patients with LC are diagnosed with locally advanced (stage III) NSCLC. 7 Staging of NSCLC (TNM: tumor size [T1–T4], lymph node involvement [N0–N3], metastasis [M0-M1c]) during initial diagnosis is a crucial predictor for treatment decisions, prognosis, and survival rates. Majority of patients in Egypt present with either locally advanced or metastatic disease. A 4-year analysis from Egypt (N = 114) revealed that most patients (males: 91.1%, females: 60.9%) presented with T3/T4 staging with N3 lymph node involvement (males: 51.2%, females: 23.4%) and stage IV metastasis (males: 83.7%, females: 73.5%). 8
Due to the wide heterogeneity of stage III NSCLC, optimal management presents a unique challenge. Curative resection is the gold standard treatment recommended for operable and resectable stage III tumors along with adjuvant chemotherapy (CT) and/or radiotherapy (RT). 9 However, for unresectable stage III NSCLC, a multidisciplinary team (MDT) approach involving surgery, RT, and systemic agents form the backbone of treatment.10,11
The presence of epidermal growth factor receptor (EGFR) mutations is proven as a favorable prognostic marker in the treatment of NSCLC. In the MEA region, the prevalence of EGFR mutations in patients with NSCLC ranges from 20.0% to 44.1%.12,13 The National Comprehensive Cancer Network® (NCCN®) now recommends adjuvant-targeted therapy with osimertinib for completely resected (R0) stage IB-IIIA, stage IIIB (T3, N2) NSCLC with EGFR mutations (exon 19 deletion, exon 21 L858R) who received prior adjuvant computed tomography (CT) are ineligible to receive platinum-based CT. 9 Furthermore, adjuvant immunotherapy with programmed death ligand (PD-L1) inhibitor durvalumab after definitive chemoradiotherapy (CRT) was found to be effective in enhancing the progression-free survival (PFS) and overall survival (OS) in patients with unresectable stage III NSCLC, based on the sustained OS and durable PFS 5-year benefit from the PACIFIC trial. 14 In PACIFIC-R, a real-world international study, the efficacy of durvalumab consolidation after CRT was consistent with that observed in the PACIFIC trial. 15 In concordance with the recent guidelines, a consensus statement with experts from the MEA region recommends durvalumab consolidation for patients without disease progression following definitive concurrent chemoradiotherapy (cCRT). 16
The LC mortality rate is high in Egypt, potentially due to increased smoking and tobacco use.2,17 Data on LC treatment patterns, especially for stage III, are scarce. Due to the heterogeneous nature of stage III and IV LC, Egyptian oncologists reported and participated in 44 international clinical trials to investigate newer lines of treatment for LC patients. 18 An international real-world KINDLE study was conducted in developing regions including Asia, MEA, and Latin America for understanding the treatment strategies and their associated clinical outcomes in the pre-immuno-oncology (pre-IO) era in patients with stage III NSCLC. 7 We report the comprehensive treatment practices and associated survival outcomes for the Egyptian subset of the KINDLE study.
Methods
Study design and patient population
The KINDLE-Egypt subset enrolled consecutive adult patients ⩾18 years of age who were diagnosed with de novo locally-advanced stage III NSCLC (as per American Joint Committee on Cancer [AJCC] 7th edition) between 1 January 2013 and 31 December 2017 with at least 9 months of documented follow-up since the index diagnosis. Patients with the initial diagnosis of stage I–II NSCLC who progressed to stage III were excluded. The detailed study design of the overall KINDLE patient population has been presented in the Supplemental Table S1.
Ethics
The independent ethics committees/institutional review boards approved the study protocol (NCT03725475). The study was conducted in accordance with the Declaration of Helsinki, the International Council for Harmonization, good clinical practices, good pharmacoepidemiology practices, and all the applicable legislation on noninterventional studies and/or observational studies. Written informed consent from the patients or next of kin/legal representative was obtained before the study participation. The reporting of this manuscript has been done as per the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist. 19
Data collection and outcomes
The study eligibility criteria and data collection methods have been elaborated in the global manuscript by Jazieh et al. 7 The study outcomes included, demographic and clinical characteristics, treatment patterns, and clinical outcomes (PFS and OS). The PFS was defined as the time from treatment initiation to documented disease progression or death because of any cause, whichever occurred first, and OS was defined as the time from stage III NSCLC diagnosis or time from treatment initiation until death because of any cause. The occurrence and date of disease progression were determined from documentation within the patients’ medical records such as pathology reports, imaging reports, and oncologists’ notes and statements on disease progression. Additional details are presented in Supplemental Table S1.
Statistical analyses
Statistical analyses included descriptive statistics to summarize the patient demographics, disease characteristics, and treatment patterns. The categorical variables were presented as percentages, while the continuous variables were presented as median, minimum, and maximum. The percentage was calculated based on the total number of patients available within each level; unknown and missing data were not included. Survival analysis using the Kaplan–Meier curve was used to present median OS and PFS, along with the two-sided 95% CI. Univariate and multivariate cox proportional hazards models were used to estimate the predictors of survival outcomes while controlling for demographic and clinical covariates. A p-value of <0.05 was considered statistically significant.
Results
Sociodemographic and clinical characteristics
Of the total 421 enrolled patients from 21 centers in Egypt, the median age (range) of patients was 59.0 (24.0–84.0) years and more than three-fourths (77.9%) were men (Table 1). Most patients (64.4%; 257/399) were current or ex-smokers and had an Eastern Cooperative Oncology Group performance status of ⩽1 (81.5%, n = 264/324). At diagnosis (per AJCC, 7th edition), 53.5% (n = 123/230) of patients had stage IIIA disease, and 46.5% (n = 107/230) of patients had stage IIIB disease. At the index date (date of initial diagnosis of stage III NSCLC), 21.6% (n = 69/319) of patients were deemed resectable, and 78.4% (n = 250/319) of them had an unresectable disease. Adenocarcinoma was the most common histologic type (60.8%; 237/390), followed by squamous cell/epidermoid carcinoma (29.0%; 113/390). Most tumors were classified as T3 (41.2%) and T4 (31.3%), with nodal involvement N2 (51.7%) and N3 (11.7%). About 43.4% (173/399) of the population first presented to a pulmonary physician, whereas 18.0% (72/399) presented to a thoracic surgeon; 18.3% (73/399) presented to a primary care physician. CT scans (81.2%; 112/138) followed by positron emission tomography scan (50.0%; 69/138) were predominantly used for diagnosis of NSCLC. Flexible fibreoptic bronchoscopy was performed for 158 (45.4%) patients (Table 1).
Baseline clinico-demographic characteristics of patients with stage III NSCLC in Egypt.

Percentage was calculated based on total number of patients available within each level; unknown and missing data are not included.
Multiple investigations possible.
Current smoker defined as an active smoker; ex-smoker defined as having smoked regularly but stopped ⩾365 days ago; Never smoker defined as never smoked regularly.
The definition for ‘Resectable’ and ‘Unresectable’ was based on the data in electronic Case Record Form as follows:
Resectable: the response to ‘Did the patient undergo curative surgical resection’ is yes.
Unresectable: the response to ‘Did the patient undergo curative surgical resection’ is no and one of the following has to be ticked: ‘considered not correct management’ or ‘medically unfit/comorbid reason’ or other: ‘unresectable Stage III NSCLC’.
AJCC, American Joint Committee on Cancer; BMI, body mass index; CT, computed tomography; EBUS, endobronchial ultrasound; ECOG, Eastern Cooperative Oncology Group; FFB, flexible fibreoptic bronchoscopy; MRI, magnetic resonance imaging; N, number of patients; n, number of patients in the subcategories; NSCLC, non-small cell lung cancer; PET, positron emission tomography.
EGFR testing was conducted in 18.9% (7/37) and 22.4% (34/152) of the resectable and unresectable patients, respectively. Of those tested, four resectable and nine unresectable patients had EGFR mutations. Very few patients underwent PD-L1 testing (resectable: 1/37 and unresectable: 6/152); three unresectable patients were PD-L1 positive (Supplemental Table S2).
The data on both MDT presentation and staging were available for 229 patients (stage IIIA: 123 and stage IIIB: 106); of these, 29.3% (n = 36) and 17.0% (n = 18) had stage IIIA and stage IIIB, respectively. Overall, 25.3% (101/399) of cases were discussed at the MDT. Out of 69 patients with resectable disease, 21 (30.4%) cases were discussed at MDT meetings, whereas in patients with unresectable disease, 76/250 (30.4%) cases were discussed at MDT meetings (Supplemental Table S3).
Treatment patterns
Among resectable patients in the initial line, 89.7% (n = 61) received surgery-based, 2.9% (n = 2) received CRT-based and 7.4% (n = 5) received palliative therapy, whereas among unresectable patients, 34.1% (n = 70) received CRT-based therapy, 61.0% (n = 125) received palliative therapy and 4.9% (n = 10) received surgery-based therapy [Table 2(a)]. Treatment pattern analyses as per resection status revealed that CT+ surgery (33.8%), surgery alone (20.6%) or other surgery (20.6%) were the top three modalities used in resectable patients in initial line treatment. The use of CT as a strategy for downstaging of tumor predominated (48.8%, n = 100) in the unresectable category; the other frequent modalities were sequential CRT (sCRT; n = 36, 17.6%) and cCRT [n = 19, 9.3%; Table 2(b)]. Overall, surgery-based therapy as initial treatment was reported in 76/322 operable patients (23.6%); stage IIIA: 35/113 (31.0%) and stage IIIB: 6/96 (6.3%) (Supplemental Table S4). Different treatment modalities used as per stage are presented in Figure 1 and Supplemental Table S5.
Treatment patterns per resectability.

Other surgery includes any kind of therapy used in combination with surgery (except for the following: surgery alone, surgery + sCRT, surgery + CT) each pattern has <10 patients.
cCRT, concurrent chemoradiotherapy; CT, chemotherapy; N, number of patients; n, number of patients in the subcategories; sCRT, sequential chemoradiotherapy.
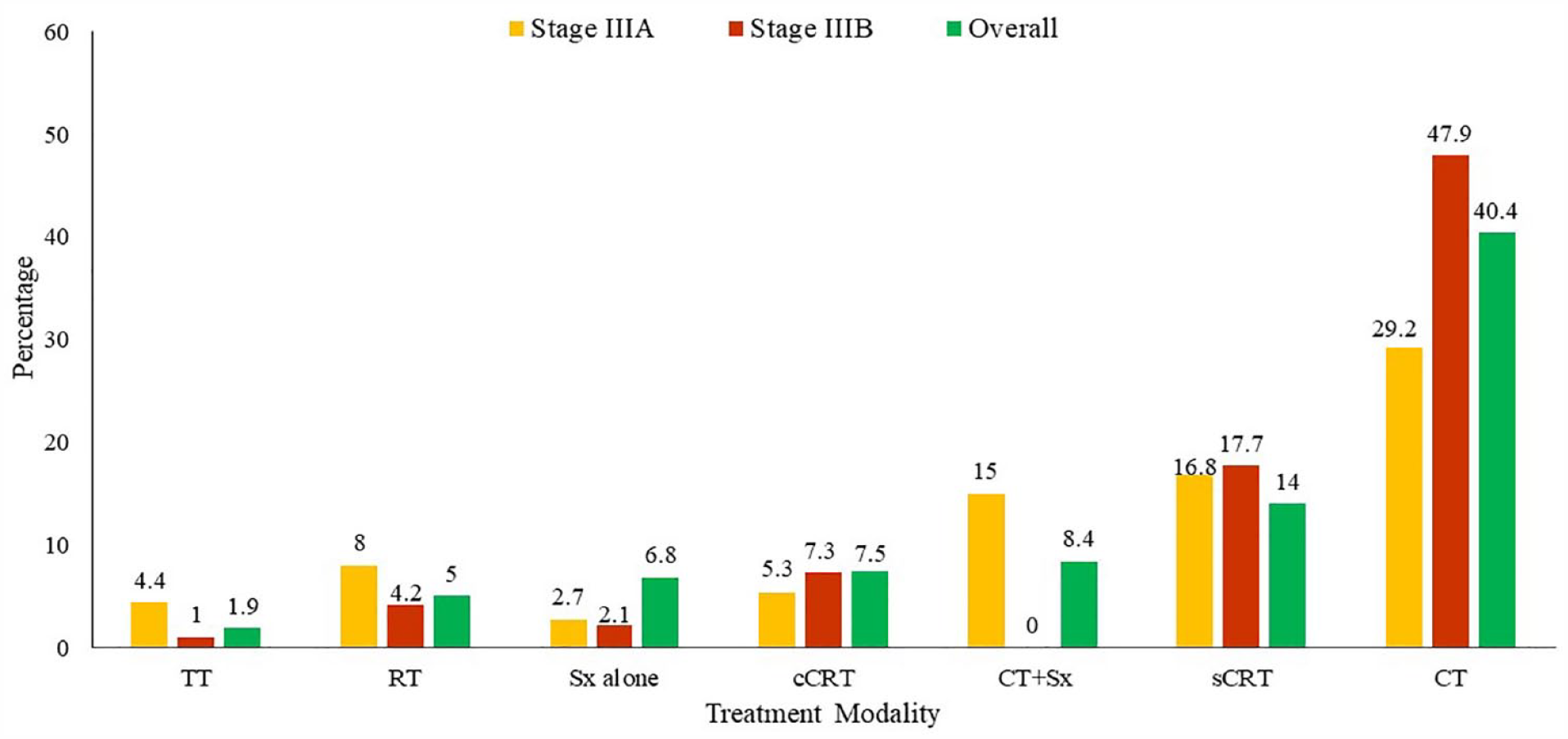
Frequent treatment modalities for stage IIIA and IIIB NSCLC – initial therapy.
Disease control by systemic or local therapy for all patients (resectable: 77.8%, n = 14; unresectable: 71.6%, n = 53) is the main target after disease progression. The predominant second line therapies were CT (resectable: 66.7%, n = 12; unresectable: 40.5%, n = 30), sCRT (resectable: 11.1%, n = 2; unresectable: 18.9%, n = 14) and RT (resectable: 5.6%, n = 1; unresectable: 20.3%, n = 15). In the third line, one patient in the resectable category while three patients in the unresectable category received CT.
Survival outcomes
In the overall Egypt subset, 152/230 (74.5%) patients had PFS events (progressed 122 and died 30) after initial therapy: stage IIIA – 82 PFS events (progressed 66 and died 16) of 123 (73.9%) patients and stage IIIB – 70 PFS events (progressed 56 and died 14) of 107 (75.3%) patients (Supplemental Table S6). After second-line therapy, 51/75 (68.0%) patients had PFS events (progressed 15 and died 36): stage IIIA – 19 PFS events (progressed 4 and died 15) of 36 (52.8%) patients and stage IIIB – 32 PFS events (progressed 11 and died 21) of 39 patients (82.1%); after third-line treatment, 1/4 (25.0%) patients had PFS events (progressed 1 and died 0): stage IIIA – 0/1 (0%) patients had PFS events and stage IIIB – 1/3 (33.3%) patients had PFS events (progressed 1 and died 0). The median PFS of the overall cohort was 10.3 months (95% CI, 9.43–12.02); median PFS being longer in stage IIIA (n = 111, 11.0 months; 95% CI, 9.69–12.42) compared to stage IIIB (n = 93, 9.5 months; 95% CI, 7.62–11.83; Figure 2). The median OS for the overall subset was 18.5 months (95% CI: 16.46–21.88); for stage IIIA (n = 110), it was 23.2 months (95% CI, 18.99–35.32) and for stage IIIB (n = 92), it was 15.8 months (95% CI, 14.42–17.45; Figure 3).

Kaplan–Meier survival curves for progression-free survival by disease stage in KINDLE-Egypt cohort.

Kaplan–Meier survival curves for overall survival by disease stage in KINDLE-Egypt cohort.
The rate of progression events after initial therapy was higher in patients with unresectable disease compared to those with resectable disease [168/250, 84.0% (progressed 123 and died 45) versus 39/69, 57.4% (progressed 33 and died 06); Supplemental Table S6]. The median PFS was longer in resectable (n = 68) patients when compared with unresectable (n = 200) patients [20.8 months (95% CI, 15.34 to 31.61) versus 9.3 months (95% CI, 7.62–9.92)]. In patients with resectable disease (16 out of 69 died), the median OS was 57.1 months [95% CI, 52.57 to Not calculable (NC)], whereas it was 15.8 months (95% CI, 14.42 to 17.35) in unresectable patients (131 out of 250 died; Supplemental Table S6).
Survival outcomes based on initial treatment
In both resectable and unresectable patients, surgery-based therapy rendered the highest median PFS [resectable (n = 61) 20.8 months (95% CI, 15.34–40.94) and unresectable (n = 10) 12.6 months (95% CI, 5.16 to NC)], followed by CRT-based therapy [resectable (n = 2) 18.5 months (95% CI, 5.39–31.61) and unresectable (n = 68) 9.8 months (95% CI, 7.89–11.83); Table 3]. Median PFS was 20.8 months (95% CI, 10.97–NC) for surgery + CT versus sCRT 18.5 months (95% CI 5.39–NC; p = 0.6156) in resectable, and 10.5 months (95% CI, 6.28–15.97) for cCRT versus sCRT 7.1 months (95% CI, 5.36–9.86; p = 0.0246) in unresectable patients [Table 4(a)].
Survival outcomes with broad categories of therapy per resectability.

CI, confidence interval; CRT, chemoradiotherapy; N, Number of patients; n, Number of patients in the subcategories; NC, not calculable; OS, overall survival; PFS, progression-free survival.
Stage and resectability status data captured at the index date was used for determining sub-groups for the sub-group analysis.
Summary of survival outcomes with major treatment strategies per resectability.

Stage and Resectability status data captured at the index date was used for determining sub-groups for the sub-group analysis.
*Other Surgery includes any kind of therapy used in combination with surgery (except for the following: surgery alone, surgery + sCRT, surgery + CT) each pattern has <10 patients.Median OS was 52.6 months (95% CI, 3.02–52.57) for palliative therapy (n = 5) and NC (95% CI, 57.07–NC) for surgery-based therapy (n = 60) in resectable patients, whereas in unresectable patients, median OS was 21.9 months (95% CI, 13.08–NC) and 17.4 months (95% CI, 15.41–21.88) for surgery-based therapy (n = 10) and CRT-based therapy (n = 67), respectively (Table 3). Median OS was 27.8 months for CT (NC for all other treatments) in resectable patients. Improved OS was observed in unresectable patients for cCRT (18.3 months, 95% CI, 14.92–NC) versus CT (14.3 months, 95% CI, 12.29–16.00; p = 0.0415), cCRT (18.3 months, 95% CI, 14.92–NC) versus RT (10.9 months, 95% CI, 2.20–12.02; p = 0.0053), CT (14.3 months, 95% CI, 12.29–16.00) versus RT (10.9 months, 95% CI, 2.20–12.02; p = 0.0115), sCRT (21.3 months, 95% CI, 10.18–22.57) versus RT (10.9 months, 95% CI, 2.20–12.02; p = 0.0291), and RT (10.9 months, 95% CI, 2.20–12.02) versus targeted therapy [16.0 months, 95% CI, 12.81–NC; p = 0.0168; Table 4(b)].
Logistic regression
The univariate and multivariate analyses for clinico-demographic characteristics and treatment regimen-related predictors for PFS and OS are illustrated in Tables 5(a) and (b) and Supplemental Tables S7 and S8 (overall cohort).
Univariate and multivariate analyses for survival outcomes for Stage IIIA and Stage IIIB NSCLC based on clinico-demographic characteristics and treatment regimen.

Includes CT alone, RT alone and targeted therapy.
CI, confidence interval; CRT, chemoradiotherapy; CT, chemotherapy; HR, hazard ratio; OS, overall survival; PFS, progression free survival; RT, radiotherapy.
The bold numbers are marginally significant but cannot change final results, need further investigation.
For the overall cohort of stage III NSCLC, male gender [hazard ratio (HR): 1.439, 95% CI, 1.054–1.963, p = 0.0218], and palliative therapy in first-line (HR: 2.067, 95% CI, 1.594–2.680, p < 0.0001) were associated with a higher risk of progression, whereas surgery in first-line (HR: 0.338, 95% CI, 0.241–0.475, p < 0.0001) was associated with reduced risk of progression in the univariate analysis. In the multivariate analyses, only male gender (HR: 1.393, 95% CI, 1.015–1.912, p = 0.0402) was found to be independently associated with increased risk of progression while surgery as initial therapy (HR: 0.327, 95% CI, 0.162–0.659) was independently associated with reduced risk for progression. Similarly, for OS in the univariate analysis, male gender (HR: 1.516, 95% CI, 1.032–2.226, p = 0.0338), and palliative therapy in the first-line (HR: 2.572, 95% CI, 1.858–3.560, p < 0.0001) were significantly associated with higher risk of death, whereas adenocarcinoma histology (HR: 0.568, 95% CI, 0.416–0.776, p = 0.0004), radical therapy with surgery (HR: 0.253, 95% CI, 0.158–0.406, p < 0.0001), or CRT (HR: 0.678, 95% CI, 0.474–0.971, p = 0.0341) in the first-line were found to be significantly associated with reduced risk of death. In the multivariate analysis, only adenocarcinoma histology was found to be independently associated with reduced risk of death (HR: 0.621, 95% CI, 0.453–0.851, p = 0.0030).
We found a similar trend in patients with stage IIIA disease for risk of progression; however, none of the predictors showed statistical significance in stage IIIB. In the univariate analysis for OS in patients with stage IIIA disease, surgery (HR: 0.347, 95% CI, 0.171–0.706, p = 0.0035) or CRT as initial therapy (HR: 0.369, 95% CI, 0.181–0.751, p = 0.0060) were significantly associated with reduced risk of death, whereas no palliative therapy (HR: 3.411, 95% CI, 1.870–6.221, p < 0.0001) in first-line was significantly associated with increased risk of death. However, in multivariate analysis, only adenocarcinoma histology was independently associated with reduced risk of death (HR: 0.534, 95% CI, 0.288–0.990, p = 0.0464). Among patients with stage IIIB, adenocarcinoma histology was significantly associated with reduced risk of death, both in univariate (HR: 0.537, 95% CI, 0.311–0.930, p = 0.0264) and multivariate analysis (HR: 0.566, 95% CI, 0.324–0.988, p = 0.0451).
Discussion
To the best of our knowledge, the KINDLE-Egypt study is the largest real-world study on locally-advanced stage III NSCLC in Egypt. The study bridges the gaps in knowledge on the treatment patterns and survival outcomes at a regional level in stage III NSCLC through retrospective data collected prior to the approval of durvalumab and EGFR tyrosine kinase inhibitor for stage III NSCLC.
The KINDLE-Egypt subset represented a younger population with a median age of 59 years, as compared to the global KINDLE cohort (63 years) 7 and MEA KINDLE cohort (61 years). 20 The male predominance (male-to-female ratio of 3:1) in the Egyptian cohort could be attributed to high smoking rates among males. Approximately 41% of the patients in the Egyptian cohort were current smokers (current smokers in global cohort: 31.2%, MEA cohort: 37.7%),7,20 and nearly one-fourth were ex-smokers. High smoking rates in the Egyptian population have been widely evaluated and are reported as a prominent risk factor for LC.21,22 A retrospective study in an Egyptian tertiary referral center reported that 71.2% of cases were current or ex-smokers and the most common stage at diagnosis was stage IV (56.4%). The study also reported statistically significant improvements in OS and PFS among the female gender, performance status 1 or 2, never smokers, grade II tumors, lower tumor size, less lymph nodal involvement, and lower stage at diagnosis, among others. 23 Overall, 53.5% of the KINDLE-Egypt population had stage IIIA NSCLC, with adenocarcinomas being the most frequently occurring histologic variant in 60.8% of the patients.
The formulation of a nexus of specialists who can chart judicious pathways involving MDT care can be instrumental for eliminating gaps in care delivery in Egypt. In the KINDLE-Egypt cohort, only one-fourth (25.3%) of the population was treated through an MDT care pathway. Of them, 29.3% had stage IIIA compared to 17.0% for stage IIIB disease. Owing to the widely heterogeneous pathology of stage III NSCLC, integrated management involving a team of thoracic radiologists, interventional radiologists, and thoracic surgeons has been recommended by major guidelines.9,10 Likewise, the MDT approach is the recommended cornerstone for the management of locally-advanced LC in MEA including Egypt. 16 Currently, medical centers like Nasser Institute, National Cancer Institute, Ain Shams University, and Kasr A-Ainy University do provide MDT care in Egypt. A prospective study conducted in Children’s Cancer Hospital Egypt to assess the effects of implementing an MDT approach and standardized protocols for treating pediatric patients with cancer reported improved treatment efficiency and patient care. 24 Hence, MDT implementation should be taken into consideration to improve the quality of healthcare delivery in patients with NSCLC.
Three-fourths (78.4%) of the patients in this Egypt subset had an unresectable disease and are probably unfit for curative resection. In the overall Egypt population, CT alone (40.4%), sCRT (14.0%) and CT plus surgery (8.4%) were the predominant therapies in first-line. Thus, with more than 15 different approaches being used in the first-line, the treatment patterns in Egypt reflect stark diversities in the management of stage III NSCLC due to the heterogeneity of patients’ profiles. On the other hand, cCRT, CT alone, and sCRT were the most frequent treatment modalities in the MEA cohort and the global cohort (cCRT: 32% versus 29%, CT alone: 20% versus 17% and sCRT: 12% versus 10%).7,20 Among resectable patients in the Egypt cohort, the majority (89.7%) underwent surgery-based therapy, in line with the recent NCCN and ESMO guidelines.9,10 Among unresectable patients in Egypt, nearly half (48.8%) received CT alone, followed by 17.6% sCRT; only 9.3% received cCRT in first-line. Though cCRT is recommended as the treatment of choice in unresectable patients, sCRT is a valid alternative in frail patients unable to tolerate concurrent therapy. 9 Tumor down-staging by systemic therapy before surgery or local radiotherapy for initially inoperable patients to improve operability failed in many patients which reflects the lower survival benefits (PFS and OS) in these patients; however, it was successful in a certain group of patients that highlights the unmet need for the addition of novel agents like immunotherapy in this stage of the disease.25,26 The difference in first-line treatment patterns in Egypt compared with the global cohort 7 is primarily due to the low number of operable patients, which is catalyzed by the high number of smoker and ex-smoker patients who tend to have lung fibrosis and as a result become not operable even if the size of the tumor is small. 27 Although deemed resectable, 10% of patients in the Egypt cohort did not undergo surgery, potentially due to high smoking rates leading to poor lung function or spread of chronic obstructive pulmonary disease, which prevents surgery and limits operability and radiation treatment. 28
The majority of LC patients present at an advanced stage in the MEA region. In 2016, a retrospective analysis of lung and pleural cancer patients (N = 804) from Cairo showed that in patients diagnosed with NSCLC (78%), most (70%) had advanced disease (stage IIIB–IV) at the time of presentation. 29 The treatment landscape of the study demonstrated that only 6% of patients underwent curative surgery, whereas 47% of them received CT. 29 The study revealed a palliative pattern of treatment with an overall 88% receiving palliative RT, and only 19% of patients with NSCLC receiving RT to the primary site with radical intent. The study highlighted that marked underutilization of radical RT as a treatment modality may be explained by the preference for systemic CT for patients without access to an RT facility. The study concluded that late presentation in advanced stages leads to poor patient outcomes with many of them not amenable to radical treatment in Egypt. 29 In 2018, Egypt’s Ministry of Health provided new radiation therapy machines with updated technology in all 11 cancer centers and Cairo University to improve access to free radiation therapy.30,31 Over the years, the Egyptian government is taking many efforts to improve cancer prevention, screening, and early detection including the Egypt National Population-based Cancer Registry founded in 2007 and non-governmental organizations coalition to ratify and implement tobacco control policies16,32;albeit, waiting for the difference in clinical outcomes in the near future. Recent recommendations from the United States Preventive Services Task Force suggest annual screening for LC with low-dose CT in adults aged 50–80 years who have a 20-pack-year smoking history and currently smoke or have quit within the past 15 years. 33 Thus, streamlined efforts are needed to encourage screening programs for early diagnosis of NSCLC in Egypt.
In the KINDLE-Egypt subset, nearly three-fourths of the patients progressed after initial therapy; the progression rate was higher in unresectable patients (84.0%) as compared to resectable ones (57.4%). A real-world study conducted in stage III NSCLC reported patients with stage IIIB disease have a worse prognosis compared to stage IIIA disease. 34 However, we found similar progression rates (73.9% versus 75.3%). The median PFS of the Egypt cohort was 10.3 months (95% CI, 9.43 12.02) – marginally lower than the MEA region (11.8 months) 20 and global cohorts (12.5 months). 7 However, the median OS for the Egypt subset was 18.5 months (95% CI, 16.46–21.88), substantially lower than the MEA region (22.9 months) 20 and the global cohort (34.9 months). 7 Despite close monitoring and follow-up of patients at different cancer centers, there is still a high incidence of disease progression in Egypt. Of a total of 122 patients who progressed on the initial therapy, 110 (90.2%) patients received a second-line therapy, predominantly CT alone (47.3%) and RT alone (19.1%). In both resectable and unresectable patients, surgery-based therapy resulted in better PFS (resectable: 20.8 months and unresectable: 12.6 months) compared to CRT-based therapy (resectable: 18.5 months and unresectable: 9.8 months). This could be attributed to unresectable study status assigned at the index date, which might have changed during the course of treatment. Missing data, and selection bias may also have confounded the outcomes. For example, patients with good performance status may have been selected for surgery which might have resulted in better PFS. Hence, these results should be interpreted with caution.
The multivariate analysis of the KINDLE-Egypt study predicted that female gender and surgery as initial therapy was independently associated with better PFS in stage IIIA (p < 0.05), while adenocarcinoma compared to other histological types was independently associated with better OS in both stages IIIA and IIIB (p < 0.05). A retrospective study from Egypt (N = 160) including patients with different histopathological LCs (across all stages) demonstrated the highest survival at 3 years for a combination of surgical, CT, and RT (66.67%), followed by CT (41.57%), and lowest for patients treated by either surgical and CT or surgical and RT approaches (p < 0.000). In the Elzomor et al. study, multivariate analysis showed that positive local signs at presentation, current smoking status, and poorly differentiated tumors were significantly associated with the risk of mortality. The study highlighted that although surgical treatment is the most effective for localized tumors, the majority of patients are diagnosed at a late stage, leading to limited treatment options. 22 In the KINDLE global cohort, in addition to surgery, cCRT alone or triple therapy (surgery plus chemoradiation) as initial treatment were also found to predict better PFS in stage IIIA (p < 0.05), whereas ECOG performance status <2, adenocarcinoma, and surgery or cCRT as initial treatment were independently associated with improved OS in both stage IIIA and IIIB (p < 0.05). 7 However, we did not find the association of CRT-based therapies with the survival outcomes, probably due to the small sample size of the Egypt cohort. Moreover, in contrast with the global cohort where cCRT was the most common treatment modality, in Egypt cohort CT was the most common modality. Robust regional data on treatment patterns, survival outcomes, and their predictors in stage III NSCLC is scarce in Egypt. This study’s results add up to the existing evidence generated through Egypt National Cancer Registry data 35 by presenting the survival outcomes for different treatment modalities in stage III disease. This may help practicing clinicians to improve the quality of care for this group of patients.
The study was conducted in the pre-IO era and before the approval of EGFR tyrosine kinase inhibitors for the early stages of the disease, thus molecular testing rates including EGFR and PD-L1 were low. With the emergence of osimertinib as adjuvant therapy after complete tumor resection (R0) in stage IB-IIIA, stage IIIB (T3, N2) NSCLC with EGFR mutations (exon 19 deletion, exon 21 L858R) and durvalumab in unresectable stage III NSCLC patients across all PD-L1 categories, the treatment landscape has expanded for patients with EGFR/PD-L1 mutations, warranting better biomarker testing to leverage benefit from the novel targeted therapies.9,36 However, in low- and middle-income countries such as Egypt, the disparity in the availability of infrastructure, and economic factors, are substantial barriers to routine biomarker testing and personalized management with targeted therapies. 37 To this end, cost-analysis illustrating the impact of targeted therapies on quality of life, clinical response, outcomes, and pricing, as compared to conventional treatment, can be pivotal. 38 Alongside the economic facets of drug availability, gaps in awareness of the benefits of targeted therapy in a high-risk population with EGFR mutation, and lack of incentives to conduct molecular testing impede the implementation pathway. A claims-based analysis from the United States revealed suboptimal utilization of osimertinib even in locally advanced/metastatic NSCLC with EGFR mutation. 39 Enhancing knowledge among physicians across all tiers of care regarding the importance of targeted therapies for EGFR mutations in stage III NSCLC is imperative. To overcome the gaps, a collaborative approach for developing educational events and promoting research to understand the pitfalls in real-world implementation of personalized therapy should be adopted by medical societies. Fostering the use of targeted therapies through enhanced access to care delivery, and monitoring the implementation in real-world practice, together with strategic MDT approaches to overcome the voids in the uptake of guideline-based management recommendations is needed.
Limitations
The main limitations in the study is that the information and data protection was from patients filing from different cancer centers, which are usually have different systems with limitations of information gathering and different methods of diagnosis, highlighting that the trending staging of localized lung cancer is to PET-CT, if this method was not conclusive, oncologists do the invasive mediastinoscopy.40 Bronchoscopy is usually done for biopsy only not for staging in most of Egyptian cancer centers.
Conclusion
The KINDLE-Egypt cohort provides real-world insights into the treatment patterns and survival outcomes of NSCLC and bridges the data gaps for stage III disease at a regional level. The wide heterogeneities in treatment patterns reflect a varied interplay between clinico-demographic characteristics, risk factors, and economic attributes across the region. This warrants formulation of country-specific guidelines, which can provide equilibrium to the unmet needs of patients while accounting for judicious utilization of available healthcare resources. With substantially lower rates of PFS and OS as compared to the global cohort, the study highlights the need for enhancing patterns of care delivery driven by an MDT approach to increase utilization of CT and adequate management post-relapse. Although the testing rates for EGFR and PD-L1 mutations were low in the current study, the evolution of osimertinib and durvalumab in the arena of early-stage NSCLC presents unique opportunities for the management of patients with curative intent – calling for action to streamline biomarker testing and screening for NSCLC. Timely screening, and increased access to novel targeted therapies, alongside an emphasis on guideline-concordant MDT care can potentially improve survival outcomes in patients with stage III NSCLC in Egypt.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231212182 – Supplemental material for Real-world treatment patterns and clinical outcomes in patients with stage III non-small cell lung cancer: results of KINDLE-Egypt cohort
Supplemental material, sj-docx-1-tam-10.1177_17588359231212182 for Real-world treatment patterns and clinical outcomes in patients with stage III non-small cell lung cancer: results of KINDLE-Egypt cohort by Rasha Aboelhassan, Mohamed Emam Sobeih, Mohamed Alm El-Din, Ramy R. Ghali, Ibtessam Saad El-Din, Ola Khorshid, Mohsen Mokhtar, Ahmed Magdy Rabea, Abdelaziz Belal, Hamdy A. Azim, Mohamed Abdullah, Tamer Elnahas, Hesham Tawfik, Sherif Abdelwahab, Amr Abdelaziz Elsaid, Tarek Hashem, Mohamed Mancy and Heba Farag in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231212182 – Supplemental material for Real-world treatment patterns and clinical outcomes in patients with stage III non-small cell lung cancer: results of KINDLE-Egypt cohort
Supplemental material, sj-docx-2-tam-10.1177_17588359231212182 for Real-world treatment patterns and clinical outcomes in patients with stage III non-small cell lung cancer: results of KINDLE-Egypt cohort by Rasha Aboelhassan, Mohamed Emam Sobeih, Mohamed Alm El-Din, Ramy R. Ghali, Ibtessam Saad El-Din, Ola Khorshid, Mohsen Mokhtar, Ahmed Magdy Rabea, Abdelaziz Belal, Hamdy A. Azim, Mohamed Abdullah, Tamer Elnahas, Hesham Tawfik, Sherif Abdelwahab, Amr Abdelaziz Elsaid, Tarek Hashem, Mohamed Mancy and Heba Farag in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors acknowledge Late Professor Rabab Gaafar and Late Dr. Yasser Abdel Kader for their important contributions to the study. The authors would also like to thank Dr. Sasikala Somara, of Fortrea Scientific Pvt Ltd (formerly Labcorp Scientific Services & Solutions Pvt Ltd) for medical writing support that was funded by AstraZeneca in accordance with Good Publication Practices 2022 guidelines.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
