Abstract
Background and purpose:
Chemotherapy-induced neutropenia and neutrophil-to-lymphocyte ratio (NLR) are potentially useful prognostic markers in patients with metastatic castration-resistant prostate cancer (mCRPC). This post hoc analysis investigated whether these markers can be utilized for dose considerations and evaluated the prognostic impact of leukocyte subtypes.
Patients and methods:
PROSELICA assessed the non-inferiority of cabazitaxel 20 mg/m2 (C20; n = 598) versus 25 mg/m2 (C25; n = 602) for overall survival (OS) in patients with mCRPC previously treated with docetaxel. The association of grade ⩾ 3 neutropenia, NLR, baseline neutrophilia and lymphopenia with OS, progression-free survival (PFS), and prostate-specific antigen response rate (PSArr) was investigated by an unplanned uni- and multivariate analyses.
Results:
PROSELICA confirmed the negative prognostic value of increased baseline NLR [⩾3, hazard ratio (HR) 1.40; p < 0.0001], but did not identify a subgroup of patients benefiting more from C20 or C25. In this post hoc analysis, patients who developed grade ⩾3 neutropenia (n = 673) had a significantly improved OS [∆OS = 2.7 months, HR = 0.78 (95% CI 0.68–0.89)] with the greatest advantage observed in patients with baseline neutrophilia [n = 85; 5.3 months, 0.60 (0.42–0.84)]. After adjustment for the Halabi criteria, neutropenia grade ⩾ 3 was the only biomarker that remained significantly associated with OS [ (HR 0.86 (0.75–0.98)], PFS [HR 0.78 (0.68–0.88)], and PSArr [odds ratio (OR) 1.82 (1.37–2.41)] while neutrophilia showed the strongest association with OS [1.53 (1.29–1.81)].
Conclusions:
Grade ⩾ 3 neutropenia was the only leukocyte-based biomarker associated with all key outcome parameters in mCRPC patients receiving cabazitaxel and might be able to overcome the negative prognostic effect of baseline neutrophilia.
NCT number:
NCT01308580
Introduction
Prostate cancer is a leading cause of male morbidity and mortality, with over 300,000 men worldwide dying from the disease in 2018. 1 Patients with metastatic castration-resistant prostate cancer (mCRPC) may receive first-line treatment with docetaxel or novel androgen receptor-targeted agents, such as abiraterone and enzalutamide,2,3 with darolutamide, apalutamide, and enzalutamide approved for non-metastatic CRPC.4,5 Following progression on first-line therapies, patients may receive an alternative androgen receptor-targeted agent, cabazitaxel or radium-223 and sipuleucel-T.6 –10 Although some patients may respond to treatment, many will experience progressive disease. Consequently, there is a great need to identify biomarkers to maximize the benefit from the different treatments available.11 –13
Inflammation has a vital role in initiating tumor growth through targeted tissue damage and has gained more recognition as a potential prognostic marker of treatment response.14 –17 Neutrophils comprise 50–70% of all leukocytes in the human immune cell population, with numerous studies describing the critical pathways exploited by tumors to disturb normal immune homeostasis. 18
Myeloid-derived suppressor cells (MDSCs) are a heterogeneous population of cells, morphologically and phenotypically similar to neutrophils and monocytes, which are generated during diseases, including cancer. 19 Multikinase inhibitors have minimal antitumor activity, but in combination with immune checkpoint blockade are synergistic and inhibit PI3K signaling to modulate MDSC-regulating cytokines. 20 MDSC depletion may be a mechanism to overcome immune resistance in mCRPC. 21 Elevated levels of circulating neutrophils are often observed in various tumor entities. 18 Tumor-induced neutrophils suppress cytotoxic CD8 + T cell activation leading to accelerated metastatic spread in a breast cancer mouse model. 15 Furthermore, in patients with breast cancer, an elevated level of neutrophils can predict worsened metastasis-specific survival, with elevated pretreatment neutrophil-to-lymphocyte ratio (NLR) leading to poorer disease-specific survival.15,22,23
Several studies have identified treatment-associated neutropenia, and pre- and post-treatment NLR as cost-effective prognostic biomarkers in patients with mCRPC. 24 In prostate cancer, high baseline NLR is associated with poor overall survival (OS) and progression-free survival (PFS), regardless of treatment received. 25 Pre-treatment NLR is a useful prognostic tool in patients with metastatic solid tumors, irrespective of primary tumor site, surgical intervention, chemotherapy line, age, gender, and performance status. 26 In prostate cancer, several large studies and even meta-analysis have been conducted, which cover the entire disease spectrum, including early non-metastatic as well as asymptomatic, mildly, and heavily symptomatic mCRPC.27 –30 Interestingly, the NLR cutoffs seem to increase with symptom and disease burden.
Other studies have suggested that the change in NLR following treatment may have a better prognostic value31,32 or included additional inflammatory markers.33,34 In the TROPIC trial, patients with a high baseline NLR that remained high following treatment had worse disease outcomes compared with patients who had a high NLR at baseline that decreased following treatment. 32 Furthermore, post hoc analyses of the TROPIC data, patients receiving cabazitaxel who developed grade ⩾ 3 neutropenia had prolonged OS and PFS, suggesting that depletion of re-programmed, pro-tumorigenic neutrophils could improve the prognosis of mCRPC patients. 35
The primary objective of this study was to confirm the prognostic role of neutropenia and NLR in patients who received cabazitaxel in the PROSELICA trial, a phase III trial that demonstrated cabazitaxel 20 mg/m2 (C20) maintained 50% of the survival benefit of 25 mg/m2 (C25) in patients with mCRPC who had received prior docetaxel treatment. 36 Since it is unclear whether the poor prognosis associated with a high NLR is driven by elevated neutrophil or diminished lymphocyte counts, we integrated neutrophilia and lymphopenia into the analysis. In PROSELICA, grade ⩾ 3 neutropenia occurred more frequently in patients receiving C25 versus C20 (73% vs 42%, respectively). 36 This has opened the unique opportunity to explore whether neutropenia can predict improved outcomes independently of the dose applied.
Methods
Study design and population
PROSELICA (NCT01308580) was a randomized, phase III trial that assessed the non-inferiority of C20 (n = 598) versus C25 (n = 602) in terms of OS in patients with mCRPC who had previously received docetaxel treatment. 36 Patients received either cabazitaxel 20 or 25 mg/m2 every 3 weeks. Granulocyte colony-stimulating factor was allowed in a curative or prophylactic intent, however, prophylactic use was not allowed during cycle 1 to assess hematological toxicity. Key inclusion criteria were diagnosis of mCRPC, disease progression on or after docetaxel, and Eastern Cooperative Oncology Group performance status (ECOG PS) 0–2. Patients were stratified according to ECOG PS (0–1 vs 2), measurable disease (yes vs no), and region. The patient demographics and complete study protocol can be found in the primary study report. 36 The distribution of baseline hematological parameters [absolute neutrophil count (ANC), absolute lymphocyte count (ALC), and NLR] is displayed in Supplementary Table 2.
Efficacy analysis
OS and PFS were analyzed using Kaplan–Meier estimates and Cox proportional hazard models. p- values were determined by log-rank tests. OS was defined as time from randomization to death due to any cause. In absence of confirmation of death, survival time was censored at the last date the patient was known to be alive or study cutoff. 36 PFS was defined as the time from randomization to the first occurrence of any of the following events: tumor progression per Response Evaluation Criteria in Solid Tumors (RECIST 1.1), prostate-specific antigen (PSA) progression, pain progression, or death due to any cause. 36 PSA response rate (PSArr), defined as the proportion of patients with a ⩾50% PSA decline from baseline, was analyzed in the eligible population using Kaplan–Meier estimates with chi-square tests and odds ratios.
Statistical analysis
Differences in OS, PFS, and PSArr were determined by uni- and multivariate analyses with grade ⩾ 3 neutropenia occurrence for the intention-to-treat (ITT) population and for patients with baseline neutrophilia (ANC > 7000 G/l). For multivariate analysis, following parameters were used for adjustment: treatment group, ECOG PS, current use of opioid analgesic, liver or lung metastases, lactate dehydrogenase, albumin, alkaline phosphatase, hemoglobin, and PSA as summarized in Supplementary Table 1. NLR cutoffs were defined as NLR < 3 versus ⩾ 3, < 5 versus ⩾ 5 based on previous publications.32,35,37 To estimate the continuous prognostic value log-transformed NLR was included into the analysis. Efficacy analysis differed to data previously published as the analysis presented here included all patients and was not restricted to patients who had received ⩾ 3 cabazitaxel cycles. 38
Results
Effect of baseline NLR status on efficacy outcomes and prognostic values of leukocyte subtypes
This post hoc analysis of the PROSELICA study confirmed the negative prognostic impact of a high NLR on OS independently of the cutoff applied but was unable to identify any subpopulation benefiting from either dose (Tables 1 and 2). Median PFS was similar regardless of baseline NLR level and dose. Patients who received the C25 dose had consistently improved response rates compared with C20, regardless of NLR baseline status. Other parameters of NLR [ANC (⩾ 7 × 109/L vs < 7 × 109/L) and lymphocytes (< 1 × 109/L vs ⩾ 1 × 109/L)] were associated with OS, including after adjustment for Halabi criteria (ECOG PS, liver or lung metastases, lactate dehydrogenase, opioid use, hemoglobin, aspartate transaminase, and alkaline phosphatase). 39 Neutrophilia ANC ⩾ 7x109/L showed the strongest association with OS supporting our hypothesis that neutrophils are driving the poor prognosis in mCRPC patients.
Efficacy outcomes in patients stratified by baseline NLR status in the PROSELICA study.

C20, cabazitaxel 20 mg/m2; C25, cabazitaxel 25 mg/m2; CI, confidence interval; HR, hazard ratio; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio; OS, overall survival; PFS, progression-free survival; PSA, prostate-specific antigen.
Uni- and multivariate analyses of OS, PFS, and PSA response to evaluate different parameters of NLR (ITT population).

ANC, absolute neutrophil count; CI, confidence interval; HR, hazard ratio; ITT, intention-to-treat; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio; OS, overall survival; PFS, progression-free survival; PSA, prostate-specific antigen.
Univariate logistic regression model with treatment group and factor. Adjusted analysis: multivariate logistic regression model adjusted by treatment group, Eastern Cooperative Oncology Group performance status, current use of opioid analgesic (Y/N), liver or lung metastases (Y/N), lactate dehydrogenase >1 × upper limit of normal (Y/N), albumin, alkaline phosphatase, hemoglobin, and PSA.
Neutrophilia is defined as present for baseline ANC ⩾ 7 × 109/L (yes) and < 7 × 109/L (no). Lymphopenia is defined as present for baseline ALC < 1 × 109/L (yes) and ⩾1 × 109/L (no). The median NLR in the PROSELICA trial was 3.38.
Effect of grade ⩾ 3 neutropenia on efficacy outcomes
In the PROSELICA ITT population, development of grade ⩾ 3 neutropenia was associated with improved efficacy outcomes (Figures 1 and 2, Supplementary Figure 1 and Table 2). OS [15.1 vs 12.4 months; HR (95% CI) = 0.78 (0.68–0.89), p = 0.0002], PFS [3.7 vs 2.8 months; HR (95% CI) = 0.74 (0.65–0.84), p < 0.0001], and PSArr [44.1% vs 25.5%; OR (95% CI) = 2.06 (1.57–2.71), p < 0.0001] were all significantly improved for patients who developed grade ⩾ 3 neutropenia compared with those who did not. This trend was seen for both cabazitaxel doses.
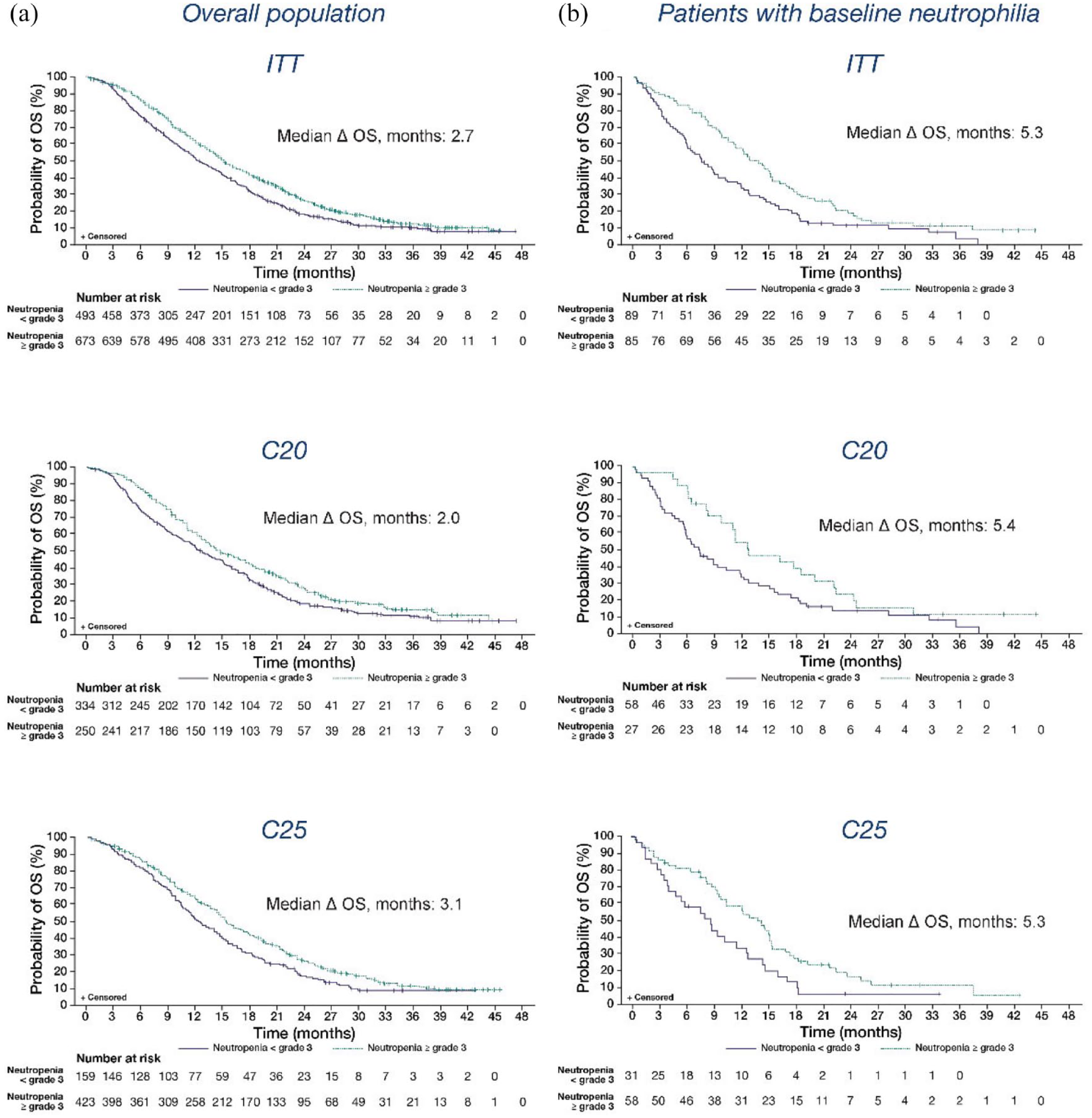
Kaplan–Meier curves for OS for (a) the overall population and (b) patients with baseline neutrophilia grouped by development of grade ⩾ 3 neutropenia during treatment.

Impact of grade ⩾ 3 neutropenia on patient outcomes in the overall study population and in patients with baseline neutrophilia.
When multivariate analysis factoring in Halabi criteria 39 was performed, the ITT and C20 populations still had significant improvements in efficacy associated with development of grade ⩾ 3 neutropenia, but not for C25.
Effect of grade ⩾ 3 neutropenia on efficacy outcomes in patients with baseline neutrophilia
Development of grade ⩾ 3 neutropenia was associated with improved survival outcomes in the poor-risk subgroup of patients with neutrophilia (ANC > 7000 G/l) at baseline (n = 174) (Figures 1–3, Supplementary Figure 1). In patients with baseline neutrophilia, grade ⩾ 3 neutropenia was associated with improved OS [12.8 vs 7.5 months; HR (95% CI) = 0.60 (0.42–0.84), p = 0.0030], PFS [4.3 vs 2.1 months; HR (95% CI) = 0.59 (0.43–0.83), p = 0.0019], and PSArr [43.8% vs 16.9%; OR (95% CI) = 2.77 (1.27–6.05), p = 0.0103] compared with neutropenia grade < 3. This trend was also seen in patients with baseline neutrophilia receiving C20 with versus without grade ⩾ 3 neutropenia for OS [12.7 vs 7.3 months; HR (95% CI) = 0.60 (0.37–0.98), p = 0.0428] and PFS [5.0 vs 1.8 months; HR (95% CI) = 0.46 (0.28–0.76), p = 0.0023]. Development of grade ⩾ 3 neutropenia was also associated with improved OS for patients with baseline neutrophilia who received C25 [13.8 vs 8.5 months; HR (95% CI) = 0.59 (0.36–0.94), p = 0.0265]. Patients with baseline neutrophilia receiving C20 who developed grade ⩾ 3 neutropenia had numerical improvements in PSArr compared with patients who did not develop grade ⩾ 3 neutropenia, and those who received C25 had numerical improvements in PFS and PSArr. Furthermore, waterfall plots for percentage PSA change (Figure 3) indicate a clear relationship between the depth of response and the degree of neutrophil decline in this poor-risk subpopulation. Patients with grade ⩾ 3 neutropenia show the deepest responses and cluster at the right side of the waterfall plots, whereas most patients with a rapid PSA progression as their best response develop no grade ⩾ 3 neutropenia and cluster at the left side.

Waterfall plot of maximum percent change in PSA in patients with baseline neutrophilia for (a) the ITT population, (b) patients who received C20, and (c) patients who received C25. Patients with ⩾3 neutropenia show the deepest responses. In contrast, many patients without ⩾3 neutropenia show no PSA response or progressive disease as their best response to therapy.
When multivariate analysis factoring in Halabi criteria was performed, the ITT and C20 populations still had significant improvements in PFS associated with development of grade ⩾ 3 neutropenia, but not for C25. OS and PSArr were not significant for the ITT or individual doses, which can be partially explained by the limited number of patients (approximately 15% of the overall population).
Linear regression analysis
A linear regression analysis was performed to compare the maximum percentage change in PSA from baseline with the nadir ANC (Figure 4). Although there was relatively high variability in the relationship between PSA response (⩾ 50% decline from baseline) and nadir ANC value, a cluster of patients who had a PSA response and a low nadir ANC value of ⩽ 2 could be identified (red).

Linear regression analysis of nadir ANC with percentage change in PSA from baseline in the PROSELICA ITT population (n = 1134; R2 = 0.0487).
Discussion
There is growing evidence that immune response and especially an immunosuppressive tumor microenvironment may impact treatment outcome in mCRPC and previous studies have suggested a prognostic role for high baseline NLR with regards to survival.16,25,40 In contrast, development of neutropenia has been associated with improved response to taxanes.32,35,41 This post hoc analysis of the PROSELICA trial supports these two hypotheses and was able to provide evidence that patients who developed grade ⩾ 3 neutropenia during treatment had improved survival and response to cabazitaxel (p < 0.001 for PFS, OS, and PSA response) in the largest cohort tested to date (n = 1200). Improved outcomes were observed in both treatment arms (C20 and C25), suggesting that there is a positive and dose-independent association of grade ⩾ 3 neutropenia with PSArr, PFS, and OS. Of note, neutropenia ⩾ 3 was the only parameter that was associated with the primary and key secondary endpoints. Our study confirmed the negative predictive impact of a high baseline NLR on OS for all cutoffs applied (NLR < /⩾ 3, p < 0.001). We consider it a strength of this analysis that we have explored the predictive value of different leukocyte subtypes. Baseline lymphopenia and neutrophilia were both associated with an increased risk of death, but neutrophilia showed the strongest association with OS. Compared with all other biomarkers, the HR for neutrophilia further increased after adjustment for the Halabi criteria (HR 1.53; p < 0.001).Therefore, neutrophilia identifies a subset of patients with a very poor prognosis and can be considered as an independent predictor of a markedly reduced survival probability. Furthermore, among patients with baseline neutrophilia, those who developed grade ⩾ 3 neutropenia during treatment had improved survival and response to cabazitaxel compared with those who did not develop grade ⩾ 3 neutropenia, suggesting that the depletion of neutrophils can overcome the negative prognostic impact of neutrophilia and subsequently leading to the largest absolute survival gain of 5.3 months in this subpopulation (twice as much as in the ITT population with 2.7 months). Taken together, these results are consistent with previous studies, including analyses of the phase III TAX327, VENICE (docetaxel), and TROPIC (cabazitaxel) trials.32,35,40,41
Compared with earlier studies,42,43 including the retrospective study of the TROPIC phase III trial where the onset of grade ⩾ 3 neutropenia was discussed as a surrogate for optimal dose exposure, 35 the current analysis has established an instantaneous and novel role of increased neutrophils as drivers of poor prognosis mCRPC, which is independent of the parameters of the current standard prognostic model. 39 We believe the negative prognostic impact of increased neutrophils before treatment initiation and the reversion of this effect by the induction of neutropenia may emphasize the importance of neutrophils in mCRPC biology, 16 although an important confounder is the increased drug exposure associated with pronounced neutropenia that may also convey more direct antitumor activity. Our results highlight that neutropenia, which is depleting neutrophils as one form of MDSCs, should not be exclusively considered an adverse effect, but as a mode of action with direct immunological implications. 15
One of the limitations of this study is its retrospective nature. Despite the pro-tumorigenic effect of neutrophils there are alternative genetic and non-genetic resistance mechanisms.12,44 Currently, there is no reliable and clinically validated method to identify patients with an intrinsic cabazitaxel resistance. This has compromised the linear regression analysis. In the C25 arm, the overall neutropenia rate was 88.9% (73.3%, grade 3/4; 15.3%, grade 1/2; and 11.1%, no neutropenia). Therefore, the reference group is small and we cannot exclude that milder forms of neutropenia have an impact on cabazitaxel efficacy. This impacts the statistical analysis, since grade ⩾ 3 neutropenia is mainly compared with milder forms of neutropenia and it also explains why some effects are more pronounced with C20, where the size of the analyzed groups was more balanced. With 33.4% in the C20 arm versus 11.1% in the C25 arm, the reference group included a significantly greater proportion of patients without any neutropenia. Although, grade ⩾ 3 is a clinical useful cutoff for neutropenia, the optimal range of neutrophil decline remains to be defined and might be influenced by factors like the duration of neutropenia, the distribution of T and B cells or rare subtypes of granulocytes like eosinophils and basophils.
Nevertheless, the importance of the NLR has also been observed in other cancer types including non-small cell lung cancer (NSCLC). Comparable survival outcomes have been noted in patients with advanced NSCLC when neutropenia has occurred during chemotherapy. 45 High NLR has also been shown to counteract elevated programmed death ligand 1 (PD-L1) expression, which is associated with a good prognosis in NSCLC treated with immune checkpoint inhibitors (ICIs). 46 Together with our results, these studies suggest that neutropenia should be further explored as a concept to optimize the dose exposure to cytotoxic drugs, including novel combination regimen, to positively influence the tumor microenvironment, and to adjust the treatment to the individual needs of the patients. Such a concept could represent a way to personalize mCRPC treatment along with next-generation sequencing, which is increasingly used in mCRPC. 12
In conclusion, the data presented here suggest that the development of grade ⩾ 3 neutropenia may be associated with improved outcomes in patients with mCRPC receiving cabazitaxel and may be able to overcome the negative prognostic effect of baseline neutrophilia, a novel prognostic marker in mCRPC. We hypothesize this may be related to the clearance of myeloid cells fueling tumor growth. These observations suggest that patients started on a lower dose of cabazitaxel might benefit from a dose escalation. Clinical trials are needed to prospectively evaluate whether dose escalation from 20 to 25 mg/m2 is warranted in patients without grade ⩾ 3 neutropenia and might represent a possible way to personalize mCRPC treatment in the future.
Footnotes
Acknowledgements
The authors want to thank Amber Wood of MediTech Media for their editorial and medical writing support.
Ethics approval and consent to participate
This is a post hoc analysis of the PROSELICA phase-III trial. The protocol, including all amendments, was reviewed and approved by independent ethics committees and institutional review boards. The protocol complied with recommendations of the 18th World Health Congress (Helsinki, Finland, 1964) and all applicable amendments and with the laws, regulations, and guidelines of the countries where the study was conducted. All patients provided written informed consent before participation. Ethics approval for this post hoc analysis was waived since only anonymized data were used.
Consent for publication
All patients provided written informed consent in the original study.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: research and analysis were supported by Sanofi. The authors were responsible for all content and editorial decisions and received no honoraria for development of this manuscript. Editorial support was provided by Amber Wood of MediTech Media, funded by Sanofi.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AM has provided a consulting, advisory or speaker role for Amgen, Astellas, Boehringer Ingelheim, Celgene, Janssen, Gerresheimer, Glaxo-Smith-Kline, Merck, MSD, Novartis, Roche, Sanofi, Servier, Takeda, and Vifor, has received research funding from Bayer (personal) and Merck & Cie (institutional), has intellectual property interests relating to Merck (not related to this report), has been paid to provide expert testimony for Sanofi, and has reported travel/accommodation expenses paid for by Amgen, Astellas, Boehringer Ingelheim, Janssen, Merck, Roche, Sanofi, and Servier. RdW has provided a consulting or advisory role for Sanofi, Merck Sharp & Dohme, Roche/Genetech, Janssen, Bayer and Clovis Oncology. RdW has also received honoraria and/or research funding from Sanofi and Bayer. SO has provided a consulting or advisory role for AstraZeneca, Sanofi, Roche/Genetech, Janssen, Bayer and Astellas, Pfizer, Merck Sharp & Dohme, Novartis, Bristol Myers Squib. SO has also received honoraria and/or research funding from Sanofi, AstraZeneca, Bayer, Pfizer, Janssen, and Bristol Myers Squib. OS has provided a consulting role for Advanced Accelerator Applications (AAA), Astellas, AstraZeneca, Bayer, Blue Earth Diagnostics, Inc., Bavarian Nordic, Bristol Myers Squibb, Clarity Pharmaceuticals, Clovis, Constellation, Dendreon, EMD Serono, Fusion, Isotopen Technologien Meunchen, Janssen, Merck, Moyvant, Myriad, Noria Therapeutics, Inc., Novartis, Noxopharm, Progenics, POINT Biopharma, Pfizer, Sanofi, Tenebio, Telix, Theragnostics. OS has received grant/research support from Advanced Accelerator Applications, Amgen, AstraZeneca, Bayer, Constellation, Endocyte, Invitae, Janssen, Lantheus, Merck, Progenics, and Tenebio. FS-L has provided a consulting or advisory role for Bayer, BMS, Janssen, MSD, AstraZeneca and Sanofi. FS-L has also reported travel/other expenses from BMS, Sanofi, and Roche. ZS was an employee of Sanofi. MF and AO are current/former Sanofi employees. AO owns stocks in Sanofi. ME is on the board of directors for VERU Inc. JSdB has provided a consulting or advisory role for AstraZeneca, Sanofi, Roche, Astellas Pharma, Bayer, Pfizer, Merck Sharp & Dohme, Merck Serono, Boehringer Ingelheim, Sierra Oncology, Menarini Silicon Biostystems, Celgene, Taiho Pharmaceuticals, Daiichi Sankyo, Janssen, Genmab, GSK, Orion Pharma GmbH, Eisai, and BioXCel therapeutics, and received travel/accommodation/expenses from AstraZeneca, Astellas Pharma, GSK, Orion Pharma GmbH, Sanofi, Genmab, Taiho Pharmaceuticals, Qiagen, and Vertex. JSdB is also associated with patents/royalties/other IP for abiraterone, PARP inhibitors, IL-23 targeting in prostate cancer, CHK1 inhibitor. JSdB has also received honoraria and/or research funding from AstraZeneca, Sanofi, Astellas Pharma, Pfizer, Roche/Genentech, Janssen, Menarini Silicon Biosystems, Daiichi Sankyo, Sierra Oncology, Taiho Pharmaceuticals, Merck Serono, Astex Pharmaceuticals, Merck Sharp & Dohme, Orion Pharma GmbH, CellCentric, Celgene, Bayer, MedImmune, Medivation, and BioExcel.
Availability of data and materials
The data sets generated and/or analyzed during the current study are not publicly available (registrational trial). Any request should be submitted to the data provider.
Supplemental material
Supplemental material for this article is available online.
