Abstract
Introduction:
Clinical stage 1 (CS1) nonseminomatous (NS) germ cell tumors involve a 30% probability of relapse upon surveillance. Adjuvant chemotherapy with one course of bleomycin, etoposide, and cisplatin (1xBEP) can reduce this risk to <5%. However, 1xBEP results are based solely on five controlled trials from high-volume centers. We analyzed the outcome in a real-life population.
Patients and Methods:
In a multicentric international study, 423 NS CS1 patients receiving 1xBEP were retrospectively evaluated. Median follow-up was 37 (range, 6–89) months. Primary end points were relapse-free and overall survival evaluated after 5 years. We also looked at associations of relapse with clinico-pathological factors using stratified Kaplan–Meier methods and Cox regression models. Treatment modality and outcome of recurrences were analyzed descriptively.
Results:
The 5-year relapse-free survival rate was 96.2%. Thirteen patients (3.1%; 95% confidence interval, 1.65–5.04%) relapsed after a median time of 13 months, of which 10 were salvaged (77%). Relapses were mostly confined to retroperitoneal nodes. Three patients succumbed, two to disease progression and one to toxicity of chemotherapy. Pathological stage >pT2 was significantly associated with relapse rate.
Conclusion:
The relapse rate of 3.1% found in this population of NS CS1 patients treated with 1xBEP at the routine care level was not inferior to the median rate of 2.3% reported from a meta-analysis of controlled trials. Also, the cure rate of relapses of 77% is consistent with the previously reported rate of 80%. This study clearly shows that the 1xBEP regimen represents a safe treatment for NS CS1 patients.
Keywords
Introduction
Nonseminomatous tumors (NS) of clinical stage 1 (CS1) comprise approximately 20% of all testicular germ cell tumors (GCTs).1,2 If managed with surveillance after orchiectomy, 50% of patients with lymphovascular invasion (LVI) of the primary tumor will progress, 3 while the risk is only 15–20% in those without.4–7 Recurrences can be cured with standard therapy in the vast majority of cases.8–10 Adjuvant chemotherapy with cisplatin-based regimens can safely prevent progression in the majority of cases. 11 Traditionally, this prophylactic chemotherapy consisted of two cycles of bleomycin, etoposide, and cisplatin regimen (BEP) that had evolved from adjunctive chemotherapy for patients with positive lymph nodes found upon primary retroperitoneal lymph node dissection (RPLND). 12 However, as cisplatin-based chemotherapy involves significant early and late toxicity in a dose-dependent manner,13–15 attempts have been made to reduce the dosage of adjuvant chemotherapy. Accordingly, a two-course regimen of BEP with reduced dosage of 360 mg etoposide per cycle (BE360P) became popular in the United Kingdom. 16 Also, two courses of etoposide and cisplatin only (EP) showed promise. 17
In 1992, Oliver suggested application of only one course of BEP (1xBEP) in the adjuvant setting of nonseminoma and demonstrated promising results in a small pilot series. 18 Since 2000, several studies have confirmed Oliver’s hypothesis,11,19–23 and a recent study reported a markedly low long-term toxicity of the regimen. 24 Accordingly, guidelines recommend the 1xBEP regimen as adjuvant treatment for nonseminoma CS1 patients with risk factors.9,25,26
However, evidence for efficacy of the 1xBEP regimen in CS1 nonseminoma rests on only five controlled studies of 22–517 patients primarily from specialized treatment centers or single country populations.19–23 Therefore, more data are needed. Generally, outcome research studies have revealed that favorable treatment results obtained in well-selected patients of properly conducted clinical trials are usually not achieved in patients treated at the routine care level.27–32 Such disparities have also been observed in testicular cancer.33–36 Undertreatment is the most important factor engendering inferior outcomes;37–39 it may be caused by a variety of patient-related factors including age, comorbidities, and compliance, as well as socio-economic factors such as economic situation, insurance status, educational level, and ethnic subtypes.40–45 Finally, inadequate experience of low-volume institutions may compromise outcome.39,46 With regard to the adjuvant 1xBEP regimen, only sporadic data relating to patients treated outside of published studies are available.47,48 Furthermore, a recent collaborative recurrence outcome study suggested inferior survival rates of relapses after 1xBEP. 49 Accordingly, that regimen was strongly cautioned by some leaders in the field. 50 The aim of this study was to analyze outcomes in unselected NS CS1 patients treated with 1xBEP at the routine care level and to compare the results with those of controlled studies.
Patients and methods
A total of 33 institutions from six European countries participated in a retrospective observational study on patients with testicular NS CS1 undergoing 1xBEP from 2004 to 2020. Eligibility criteria included: inguinal orchiectomy, histologic confirmation of NS GCT, age >18 years, absence of metastases, negative tumor markers postoperatively, application of one complete course of BEP with standard dosing within 8 weeks after surgery, and follow-up of ⩾6 months. We registered the following data: age at diagnosis (years), tumor size (mm), presence of ⩾50% embryonal carcinoma (⩾50% EC) in primary tumor (yes/no), length of follow-up (months), and relapse (yes/no). Lymphovascular invasion was rated as absent in the case of local stage pT1 and as present in all stages >pT1. This was done for reasons of simplicity, although the latter stages are principally defined as local extension of the primary tumor into or beyond testicular margins and lymphovascular invasion. 51 Follow-up was performed according to institutional guidelines. Relapse was defined as development of metachronous metastatic disease. Second, testicular tumors were not considered relapses. In relapsing cases, we also registered localization of metastases, treatment modalities, and outcome. Health events occurring subsequent to chemotherapy were not systematically assessed but recorded if reported.
For statistical analysis of quantitative baseline characteristics, measures for location (arithmetic means, medians) and dispersion [standard deviations, range, first and third quartiles (Q1 and Q3)] were provided. For binary and categorical variables, proportions and 95% CIs were calculated. The primary end points relapse-free survival and overall survival were analyzed by means of Kaplan–Meier methods; 5- and 10-year relapse-free and overall survival rates were deduced together with their 95% CIs. To allow for exploratory comparison of the overall relapse rate reported in the meta-analysis of controlled studies 11 with the present results, the crude proportions of relapses, the exact Blyth-Still-Casella 95% CIs, and the unconditional 95% CI on the difference of two binomial proportions and Barnard’s unconditional test for testing differences between rates were provided.
The association of relapse-free survival with clinico-pathological factors was analyzed in an exploratory approach using stratified Kaplan–Meier analyses and log-rank tests. Hazard ratios with 95% CIs were deduced from univariate Cox regression models. In addition, a multivariable Cox-regression model was fitted. Significance was assessed by means of Wald tests. For these analyses, the continuous factors age and tumor size were categorized at respective median values.
All patient-related data were entered into a commercially available database (MS Excel, version 2017) and stored until final evaluation. SAS software (version 9.4) was used for data cleaning and statistical analyses.
Treatment of relapses was analyzed descriptively and compared with reported data.11,49
Ethical approval was provided by Ethikkommission der Ärztekammer Hamburg on 2 March 2020 (PV7288). The need for informed consent of patients was waived by the Ethics committee because only anonymized patients’ data were used during execution of this study. All study activities were conducted according to the Declaration of Helsinki of the World Medical Association as amended by the 64th General Assembly, October 2013.
Results
A total of 423 patients of 455 originally registered were included in the final analysis (study profile, Figure 1). Clinical patient characteristics are listed in Table 1. Relevant comorbidities were noted in 58 patients (14%; details in Table 2).

Study profile.
Patients’ characteristics.
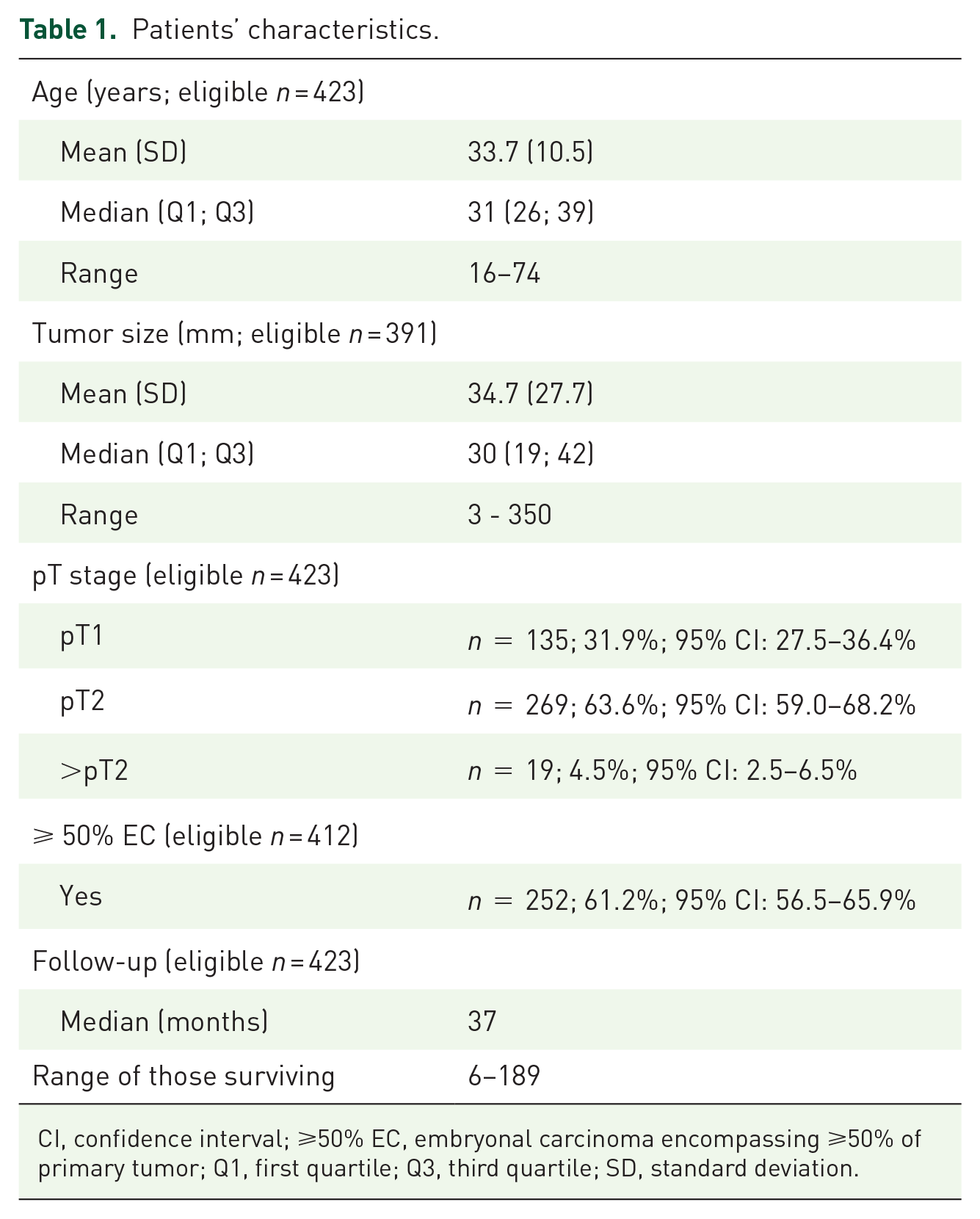
CI, confidence interval; ⩾50% EC, embryonal carcinoma encompassing ⩾50% of primary tumor; Q1, first quartile; Q3, third quartile; SD, standard deviation.
List of comorbidities of the patients included in the study (n, number of patients).

Thirteen patients relapsed within a median time of 14 months (range, 3–64 months) with four recurrences after 36 months or later. The crude relapse rate was 3.1% (exact 95% CI: 1.65–5.04%) compared with 2.3% (95% CI: 1.50–3.38%) in the published meta-analysis; 11 the absolute difference between the two studies amounted to 0.8 percentage points (exact unconditional 95% CI: −0.97% to 3.08%, p = 0.4722). The calculated 5- and 10-year relapse-free survival rates were 96.2% (95% CI: 93.2–97.9%) and 94.2% (95% CI: 87.7–97.3%), respectively (Figure 2). In total, three patients died; the calculated 5- and 10-year overall survival rates were both 99.2% (95% CI: 97.6–99.8%) (Figure 3).

Progression-free survival rate in NS CS1 patients following treatment with 1xBEP.
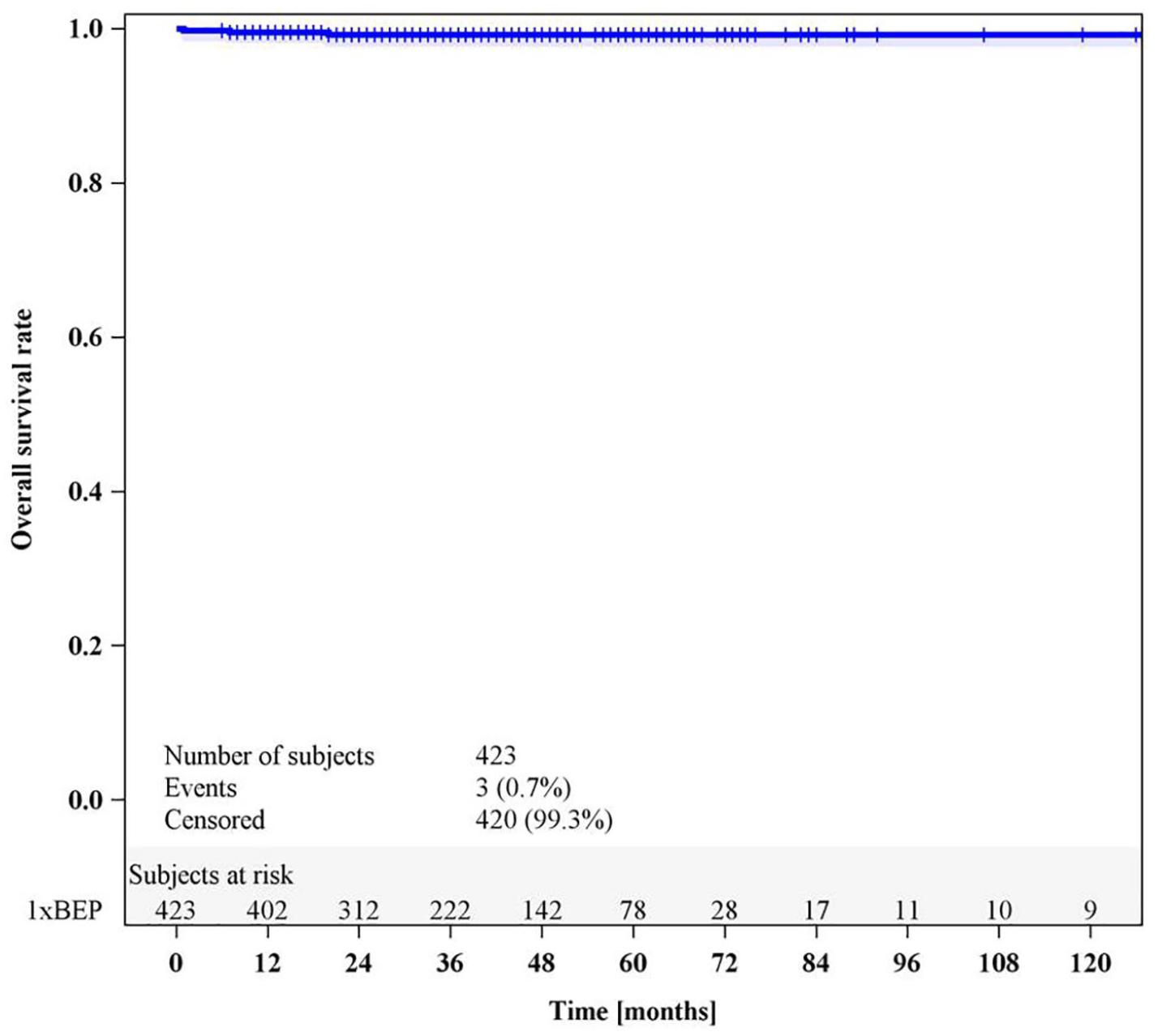
Overall survival rate in NS CS1 patients following treatment with 1xBEP.
Results of the exploratory search for associations of relapses with clinical factors are listed in Table 3. Only local tumor stage >pT2 was significantly associated with relapse rate. This association remained relevant in the multivariable Cox-regression model (Figure 4, Table 4). In this subgroup, 2 (10.5%) of 19 patients relapsed; 11 patients among those with lower local stages relapsed (2.7%; p = 0.0170).

Progression-free survival stratified by local tumor stages (pT stages).
Individual effects of clinico-pathological factors on RFS rates.
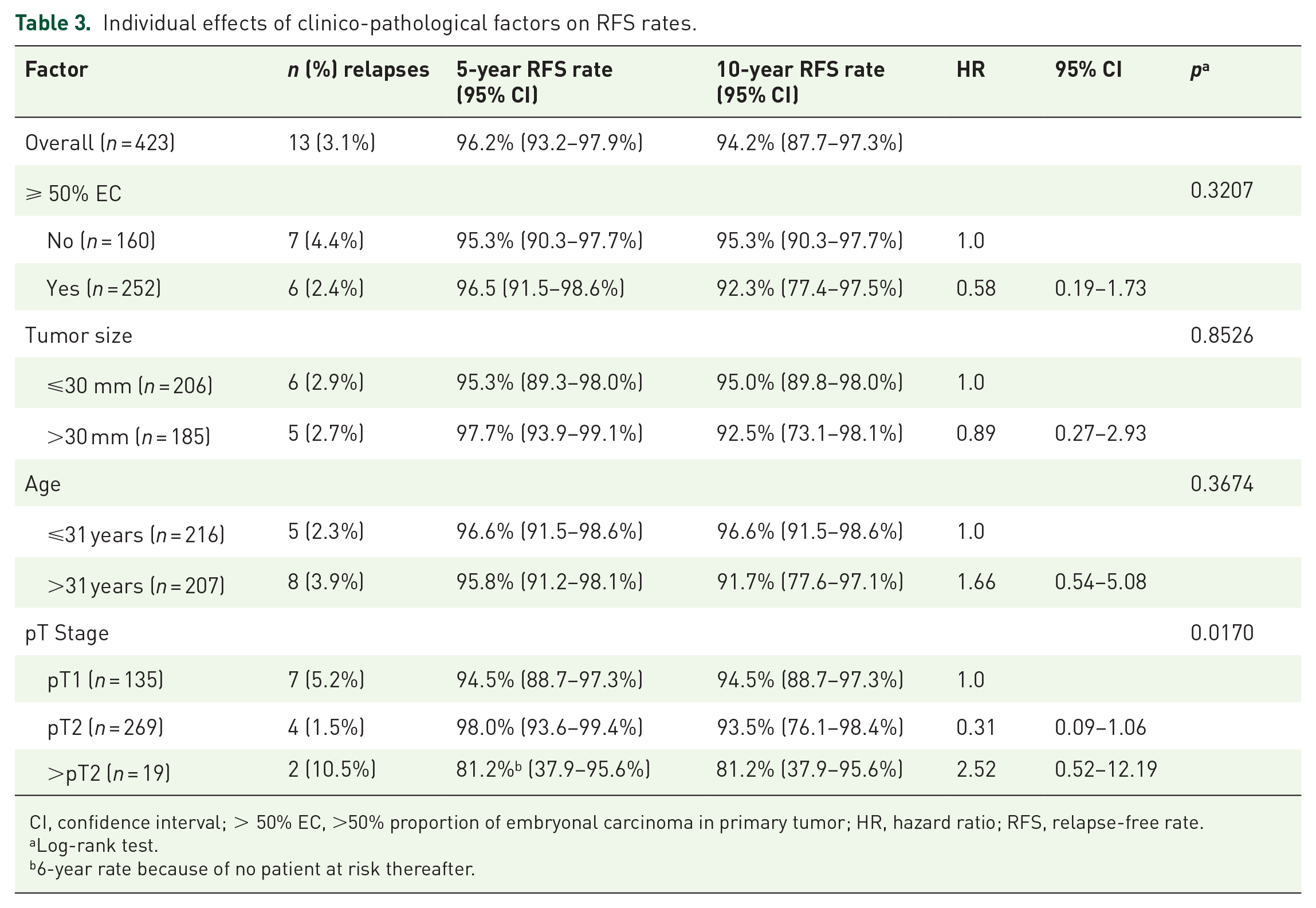
CI, confidence interval; > 50% EC, >50% proportion of embryonal carcinoma in primary tumor; HR, hazard ratio; RFS, relapse-free rate.
Log-rank test.
6-year rate because of no patient at risk thereafter.
Effects of clinico-pathological factors on relapse-free survival rates: multivariable Cox regression models.
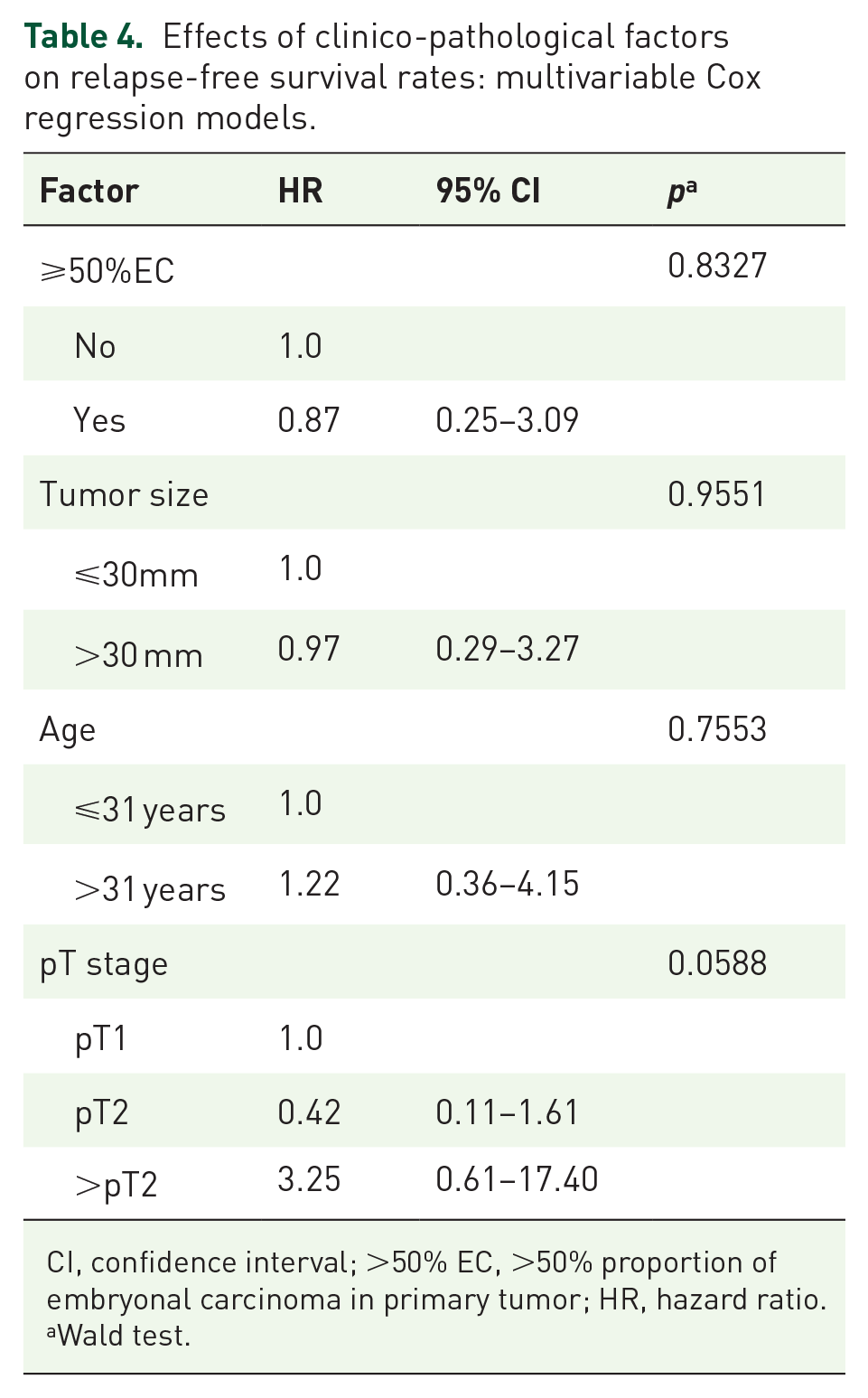
CI, confidence interval; >50% EC, >50% proportion of embryonal carcinoma in primary tumor; HR, hazard ratio.
Wald test.
Relapses were localized in the retroperitoneal nodes in 12 cases, one with additional pelvic node involvement and one with additional pulmonal metastases (Table 5). One patient developed pulmonal metastases only. Treatment consisted of cisplatin-based chemotherapy alone in four patients, while four had chemotherapy and additional surgery (Table 5). Noteworthily, four patients were cured with surgery alone, all of whom had teratoma only, histologically. One patient unfit for chemotherapy received best supportive care. Ten of the relapsing patients were cured (77%), and none experienced a second relapse after a median follow-up time of 24 months (range, 0–44 months).
Clinical details of patients relapsing after one-course BEP.

AWD, alive with disease; BEP, bleomycin, etoposide, and cisplatin; BSC, best supportive care; CR, complete remission; DOD, dead of disease; IGCCCG, International Germ Cell Cancer Collaborative Group; NED, no evidence of disease; PEI cisplatin, etoposide, ifosfamide; RPLND, retroperitoneal lymph node dissection; TIP, paclitaxel, ifosfamide, cisplatin.
We noted three major complications of 1xBEP therapy: one case of neutropenic sepsis, one case of fatal bowel ischemia, and one case of cerebral stroke. Five patients developed contralateral testicular tumors (1.2%).
Discussion
The central result of this retrospective observational study is that NS CS1 patients treated with 1xBEP faced a low overall relapse rate of 3.1%, which is consistent with the previously reported rate of 2.3%. 11 Similarly, 77% of relapsing patients were cured, consistent with the previously reported rate of 80%. 49 Although this study was conducted in unselected patients at the routine care level, the therapeutic outcome was not significantly inferior to that obtained in selected patient populations of controlled trials. 11
Direct comparison of the present results with those of the controlled trials is impractical because there is significant variation among these studies in terms of patient characteristics and methodologies. The reported crude numbers of relapses of the 1xBEP trials range from 1.1% 20 to 4.5% 21 and are in accordance with the frequency of 3.1% of this study. This figure is also close to the 1.8% relapse rate of CS1 patients receiving the traditional two cycle regimen, 11 and it is considerably lower than the 7–11% relapse rate observed in patients receiving primary RPLND.20,52 The calculated 5-year relapse-free survival rate of 96.2% of this study appears to be marginally lower than the 97% 2-year relapse-free rate reported in the UK study, 23 the 99.6% 5-year cancer-specific survival rate reported by Swenoteca, 22 and the 99.41% 2-year relapse-free rate of a German trial. 20 However, the differences are marginal with wide overlap of 95% CIs. More importantly, the figures cannot be directly compared because they relate to methodologically different analytical variables, mainly due to divergent observation times. Overall, the therapeutic outcome observed in this study is likely not inferior to the results of previous trials. Accordingly, the present report, which is the second largest study to date, adds evidence for safety of the 1xBEP regimen and clearly supports its recommendation in guidelines.
The present results are particularly noteworthy because the favorable outcome was achieved in unselected patients in everyday clinical practice. The non-inferiority of the 1xBEP treatment in this population to the results of clinical trials likely relates to the absence of most of the factors that usually precipitate inferior results in routine care patients such as undertreatment, non-compliance, and selection bias.29,39,53 The former two factors are likely negligible because the application of 1xBEP is simple. Selection bias generally contributes to the disparities regarding therapeutic outcome among trial populations and routine care patients.28,29,31,44 However, this kind of bias is low in testicular cancer patients since comorbidities and older age are uncommon in these mostly young individuals. We noted relevant comorbidities in 14% of patients, and 10% were aged ⩾50 years. Although age did not affect relapse-free survival rates statistically in the Cox regression model, it appears noteworthy that 5 of the 13 relapsing patients were aged >50 years, and that all of those not cured were in that age group. This observation is consistent with the recently reported revision of the International Germ Cell Cancer Collaborative Group (IGCCCG) classification system where older age is now considered a poor prognostic factor. 54 It is conceivable that age failed to reach statistical significance in our analysis because of the small numbers of relapses in this study. Conversely, one could speculate that several of the elderly patients of this study and particularly those with comorbidities would not have qualified for inclusion in the controlled trials.
As this study included both patients with and without the risk factor of lymphovascular invasion (i.e. local stages pT1 and >pT1), it is rational to assume that patients with low risk of recurrence might have biased the overall result toward a lower relapse rate. However, local stage >pT1 was present in 67% of our patients, which represents a higher frequency than reported in previous studies.19,20,22 Only the UK-based study exclusively enrolled patients with lymphovascular invasion (pT2) and the relapse rate was 3.0%. 23 In contrast to the Swenoteca study, 22 we observed fewer relapses among patients with the risk factor >pT1 (2.1%) than in those without (5.2%), although the difference was not significant. It thus appears that inclusion of patients without this risk factor did not greatly impact the overall results of our study.
Exploratory evaluation of possible risk factors revealed advanced local stage (> pT2) to be the only parameter significantly associated with recurrence despite adjuvant BEP chemotherapy. Although our finding of local stage >pT2 representing a risk factor for progression will require confirmation in future studies, caregivers of GCT patients are likely best advised to apply particular vigilance to these patients.
The median follow-up length of 37 months in this study is shorter than the intervals of 49–186 months reported in previous trials.19,23 As the number of relapses generally increases with follow-up time, a longer observation time may incur more relapses than shorter intervals. Thus, the low relapse rate found in this study might partly relate to the comparatively short observation time. However, relapses predominantly occur in the first 2 years after completion of treatment; only sporadic cases recur later. 24 Relapses in this study occurred after a median interval of 14 months, which is nearly identical to the interval of 13 months reported by Fischer et al., 49 suggesting that the confounding effect of the comparatively short observation time of our study is likely small. We noted four patients (31%) who recurred later than 36 months after chemotherapy, which is consistent with the rate of 29% reported by Fischer et al. 49 Thus, late relapses will require further attention.
Localization of relapse was confined to abdominal and pelvic nodes in 10 patients; only three (23%) had mediastinal or pulmonal metastases. Accordingly, 12 of the recurrences in this study were classified as IGCCCG good prognosis; only one had intermediate prognosis. These findings correspond to those of the two largest 1xBEP trials. The Swenoteca series reported 2 of their 12 relapses had CS3 disease, yet all were classified as IGCCCG good prognosis. 22 The UK series reported seven relapses, six with good prognosis. 23 A slightly higher incidence of extra-abdominal relapses with 33% CS3 disease was noted in the collaborative recurrence outcome study. 49 Similarly, their proportion of 16% intermediate and poor prognosis cases was higher than that of the present series and of other reports. However, these differences may relate to selection bias since the Fischer study exclusively enrolled patients with ascertained relapses and most of these patients were from tertiary referral centers. 49 Ten of our relapsing patients (77%) were salvaged with standard therapy, which is consistent with the previously reported salvage rate of 80%.11,49 It is worth noting that four patients had teratoma only and were cured with RPLND alone. Thus, surgery should always be considered in NS CS1 patients relapsing after adjuvant chemotherapy. Selection bias must generally be considered when trial results are compared with outcomes achieved at the population level. 29 This is highlighted by two of our failing patients who would likely have been disqualified from inclusion into any chemotherapy study: one 60-year-old patient with a history of Crohn’s disease who was unfit for postchemotherapy RPLND, and one 67-year-old patient with Parkinson’s disease who was unfit for salvage chemotherapy.
Although treatment-related toxicity was not systematically assessed, we noted one cerebral stroke in a patient with no comorbidities and one fatal bowel ischemia in a man with pre-existing arterial hypertension. This rate of major vascular complications (0.5%) corresponds to the previously reported rate of 0.3% and underscores the current experience that major vascular events may occur even during the first cycle of chemotherapy.55,56 The one fatality secondary to bowel ischemia corresponds to a therapy-associated mortality of 0.3%. By contrast, none of the trials reported any treatment-related mortality.
Limitations of this study relate to the retrospective enrollment of patients, which is generally open to selection bias. The comparatively short follow-up interval of 37 months could have contributed to the low rate of relapse particularly in light of the several late relapses. Other limitations relate to the lack of central pathologic review and to non-standardized follow-up. On the other hand, strengths come from the overall large number of cases, the multi-institutional, international patient accrual, and the inclusion of unselected routine care patients.
Overall, 1xBEP chemotherapy can safely prevent relapse in patients with clinical stage 1 nonseminomatous GCTs regardless of the presence of histological risk factors. Only patients with advanced local tumor stage (>pT2) may require particular attention. The regimen proved to be safe in patients at the routine care level.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
