Abstract
Testicular Leydig cell tumors are rare neoplasms of the testes. These tumors are generally benign but malignancy and metastatic potential have been described. Here, we present a case of Leydig cell tumor in a 49-year-old male, incidentally discovered as a testicular mass. The patient had no significant previous medical history. Ultrasonography revealed a hypoechoic, well-defined, vascularized lesion measuring 7 × 7 × 4 mm adjacent to the tunica albuginea. The patient underwent testis sparing surgery, employing a modified approach including intraoperative ultrasound-guided localization, en-bloc wedge resection of the lesion with surrounding tunica albuginea and seminiferous tubules, and gubernaculum sparing surgery. Postoperatively, the patient had an uneventful recovery and was discharged on the same day. Histopathological examination confirmed the diagnosis of Leydig cell tumor, with no high-risk pathological features observed. Regular follow-up intervals were scheduled to monitor for potential recurrence, emphasizing the importance of vigilant postoperative surveillance in cases of testis-sparing surgery for Leydig cell tumors.
Introduction
Testicular cancer is one of the rarest forms of male cancer, representing 1%–3%. Leydig cell tumors (LCT) of the testicles are uncommon neoplasms arising from the interstitial cells of Leydig, constituting 1%–3% of all testicular tumors. 1 The clinical presentation of LCT can vary widely, ranging from an asymptomatic mass to diverse clinical manifestations. including virilization, gynecomastia, and changes in libido. LCT tumors are typically benign, with malignancy being rare. Malignant LCTs are characterized by metastasis to lymph nodes and other visceral organs. 2 The recommended standard treatment for testicular masses is radical inguinal orchiectomy, with testicular biopsy not advised. Definitive tumor classification is based on histopathological examination. Literature reports have documented cases of testis-sparing surgery (TSS) for LCT, yielding satisfactory clinical outcomes.2,3 The current indications adopted by the American Urological Association for TSS are for individuals seeking to preserve gonadal function, including those with masses smaller than 2 cm, equivocal ultrasound/physical exam findings, negative tumor markers (hCG and AFP), congenital, acquired, or functionally solitary testis, as well as bilateral synchronous tumors. 4 This case report describes the use of a testis-sparing approach in a 49-year-old male patient who incidentally discovered a testicular mass.
Case presentation
A 49-year-old male patient with no previous medical history was incidentally found to have a right testicular soft tissue mass on ultrasound examination. He was married, and had four children, consisting of two daughters and two sons. The patient denied scrotal or testicular pain and lower urinary tract symptoms. There was no history of weight loss, anorexia, or noticing a scrotal or testicular mass. Physical examination did not reveal a palpable testicular mass, inguinal lymph nodes, or gynecomastia.
Upon referral to our urology tertiary services, a doppler testicular ultrasound confirmed the presence of a hypoechoic, well-defined, vascularized lesion measuring 7 × 7 × 4 mm adjacent to the tunica albuginea but separated from it (Figure 1). Tumor markers were within the normal range (Lactate dehydrogenase: 168 U/L, Alpha fetoprotein: 5.65 IU/ml, and B-HCG: 0.1).
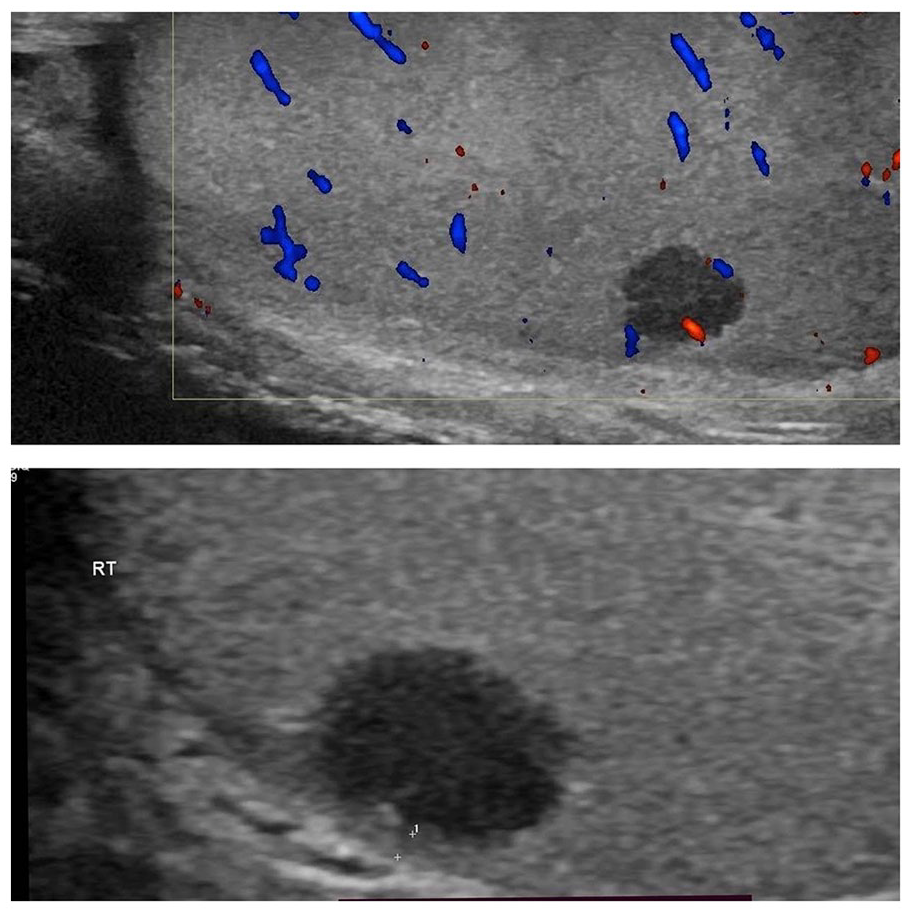
Scrotal Doppler Ultrasound: A right testicular hypoechoic relatively well-defined vascularized lesion measuring 7 × 7 × 4 mm.
During the multidisciplinary meeting, the possibility of a benign tumor was suggested based on the smooth appearance and relatively small size. The urologist and oncologist decided to proceed with TSS. The patient consented for TSS or potential conversion to radical orchiectomy (RO). Furthermore, the patient was counseled about preoperative seminal fluid analysis and sperm banking, yet he opted to proceed without undergoing either, as he fully understood the future risks of infertility and tumor recurrence.
Intraoperatively, a skin incision was made in the right inguinal area, and a subinguinal approach was used to expose the right testicle and spermatic cord while minimizing manipulation and preserving the gubernaculum. The tunica vaginalis was dissected open, revealing the right testicle and overlying tunica albuginea. The spermatic cord was clamped upon extraction of the lesion and surrounding tissue. Intraoperative ultrasound was used to locate the right testicular mass, which was situated in a subtunical location on the inferior-lateral aspect of the right testis. A wedge resection of the mass, including the surrounding tissues (seminiferous tubules), was performed using a knife and a spinal needle to guide the resection. The tunica albuginea and overlying tissues were closed in layers with absorbable sutures, and ultrasound was used to confirm the complete removal of the mass. The postoperative course was uneventful, and the patient was discharged on the same day.
Gross pathology revealed a small brown lesion (Figure 2(a)). Histopathological examination of the resected right testicular tissue showed a well-circumscribed nodule located just beneath the tunica albuginea (Figure 2(b)). The tumor consisted of medium-sized polygonal cells with abundant eosinophilic cytoplasm, rounded nuclei, and prominent central nucleoli. There was no significant cytologic atypia, increased mitotic activity, necrosis, or evidence of vascular invasion (Figure 2(c)). Intratubular germ cell neoplasia was not observed in the adjacent seminiferous tubules (Figure 2(d)). Additionally, the tumor cells exhibited strong and diffuse expression of calretinin immunostain (Figure 2(e)). These findings led to the final diagnosis of a LCT measuring 0.6 cm in its largest dimension and being localized.

(a) A testicular wedge resection measuring 4 × 2 × 1.5 cm, partially covered by tunica albuginea, with round, well-circumscribed brown-colored lesion located just under tunica albuginea measuring 0.6 cm in diameter. (b) This is a section from a partial orchiectomy specimen showing a well-circumscribed nodule located just under tunica albuginea (H+E stain). (c) The tumor is composed of medium-sized polygonal cells with abundant eosinophilic cytoplasm, round nuclei and prominent central nucleoli. There is no prominent cytologic atypia, increased mitotic activity or necrosis (H+E stain). (d) The background shows somniferous tubules (left side of image) with progressive maturation. There is no evidence of germ cell neoplasia in situ (GCNIS) (H+E stain). (e) Tumor cells show strong and diffuse expression of calretinin immunostain.
The patient’s follow-up regimen consisted of semi-annual physical examinations and ultrasound imaging. The first follow-up visit occurred 6 months after the initial surgical intervention, during which no recurrent masses were detected in the right testicle, and vascular integrity remained intact. The multidisciplinary team, comprising urology and oncology specialists, agreed on this follow-up plan, deeming additional investigations or endocrinology follow-up unnecessary.
Discussion
LCT are rare and primarily treated with radical inguinal orchiectomy (RO). TSS for LCT has only been reported sporadically in the medical literature. The clinical presentation of LCT is usually subtle, and there are no specific radiological features to distinguish it from other testicular malignancies, and histopathological diagnosis remains the mainstay of definitive diagnosis. 3
It is generally accepted that masses smaller than 2 cm on ultrasound examination would have a benign nature with less than 10% risk of malignancy, and the preoperative ultrasound diameter of the lesion is considered a significant predictor of malignancy. 5
The management of testicular LCT is mainly surgical, with RO or TSS. 6 Previous studies have found a comparable results between RO, and TSS did not seem to compromise relapse-free survival. 7 TSS also exhibits several other advantages, including improving the quality of life without affect oncological outcome, preserving fertility, and reducing the need for androgenic medications. 8
We informed our patient prior to surgery that TSS can be converted to RO depending on intraoperative findings or frozen section results, if implemented. The patient should be info with additional information about postoperative follow-up, the risk of recurrence, and the potential need for adjuvant therapy, which could impact fertility and androgen production. 8
Our approach does not include frozen section examination (FSE) of the mass to detect the nature as we do believe the size of the mass is small to consider FSE, and the ultimate goal of our approach is to have interventional radiology localization of the mass intraoperatively followed by en-block resection. 5 Several previous reports have shown some advantages of using FSE in such cases;5,8 however, our pathologist believe that FSE in such situations maybe misleading considering the benign pathological feature of LCT.
We present a unique and modified surgical approach for TSS, which includes intraoperative ultrasound-guided localization, en-bloc wedge resection of the lesion with surrounding tunica albuginea and seminiferous tubules, and gubernaculum sparing surgery. These modifications ensure implementation of oncological principles, providing clear margins. The postoperative recovery phase is expected to be less troublesome due to minimal scrotal dissection and sparing of the gubernaculum. 9
Regular follow-up with physical examination and testicular ultrasound is crucial to detect recurrences early, as the reported recurrence rate after TSS is approximately 9.5%. 7 Adjuvant treatment is not offered in our case, as it lacks high-risk pathological features such as tumor size >5 cm, necrosis, nuclear atypia, angiolymphatic invasion, invasive margins, or paratesticular invasion, and >5 mitotic features per 10 high-power fields. Chemotherapy is generally ineffective, and radiotherapy may be used for palliation purposes. 2
Conclusion
TSS offers a valuable alternative to RO for LCT, providing comparable outcomes while preserving fertility and minimizing the need for adjuvant therapy. Our case highlights the importance of meticulous preoperative evaluation and innovative surgical techniques, such as intraoperative ultrasound-guided localization, in achieving optimal oncological outcomes. Regular follow-up is crucial for detecting recurrences early. Overall, TSS represents a safe and effective option for selected patients with LCT, offering the potential for improved quality of life and reproductive outcomes.
Footnotes
Acknowledgements
Thanks to Prof. Sa’ed Zyoud, Clinical Research Center of An-Najah National University Hospital, for his wise pieces of advice.
Authors contribution
F.A., A.A., and R.A. corrected case relevant clinical data, writing the discussion and reviewing the literature, and also involved in the whole case revising and finalizing the final manuscript draft. N.Q. and M.A. revised the pathological features of the case, reviewed the relevant clinical literature and contributed to the whole manuscript writing and revision. M.K. revised the radiological features of the case, involved in writing the manuscript, and revising the final draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The IRB at Najah National University waive approvals for case reports and ask for only informed consent form from the patient.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and the associated images.
