Abstract
Introduction:
For epidermal growth factor receptor mutation-positive (EGFRm) non-small-cell lung cancer (NSCLC), EGFR-tyrosine kinase inhibitors (EGFR-TKIs) are the preferred first-line (1 L) treatment in the advanced setting. Osimertinib, a third-generation EGFR-TKI, received full approval in 2017 for second-line (2 L) treatment of EGFR T790M-positive NSCLC. The REFLECT study characterizes real-world treatment/testing patterns, attrition rates, and outcomes in patients with EGFRm advanced NSCLC treated with 1 L first-/second-generation (1G/2G) EGFR-TKIs before 1 L osimertinib approval.
Methods:
Retrospective chart review (NCT04031898) of European/Israeli adults with EGFRm unresectable locally advanced/metastatic NSCLC, initiating 1 L 1G/2G EGFR-TKIs 01/01/15–30/06/18 (index date).
Results:
In 896 patients (median follow-up of 21.5 months), the most frequently initiated 1 L EGFR-TKI was afatinib (45%). Disease progression was reported in 81%, including 10% (86/896) who died at 1 L. By the end of study, most patients discontinued 1 L (85%), of whom 33% did not receive 2 L therapy. From index, median 1 L real-world progression-free survival was 13.0 (95% confidence interval (CI): 12.3–14.1) months; median overall survival (OS) was 26.2 (95% CI: 23.6–28.4) months. 71% of patients with 1 L progression were tested for T790M; 58% were positive. Of those with T790M, 95% received osimertinib in 2 L or later. Central nervous system (CNS) metastases were recorded in 22% at index, and 15% developed CNS metastases during treatment (median time from index 13.5 months). Median OS was 19.4 months (95% CI: 17.1–22.1) in patients with CNS metastases at index, 24.8 months (95% CIs not available) with CNS metastases diagnosed during treatment, and 30.3 months (95% CI: 27.1, 33.8) with no CNS metastases recorded.
Conclusion:
REFLECT is a large real-world study describing treatment patterns prior to 1 L osimertinib availability for EGFRm advanced NSCLC. Given the attrition rates highlighted in the study and the impact of CNS progression on outcomes, offering a 1 L EGFR-TKI with CNS penetration may improve patient outcomes in this treatment setting.
Introduction
In the last decade, the treatment paradigm for patients with advanced non-small-cell lung cancer (NSCLC) harboring epidermal growth factor receptor mutations (EGFRm) has shifted. For patients with EGFRm, leveraging the sensitivity to EGFR-tyrosine kinase inhibitors (TKIs) introduced EGFR-TKIs as a first-line (1 L) treatment option, 1 and they became the recommended treatment in all international guidelines for patients with locally advanced and/or metastatic NSCLC harboring EGFRm.2–4 The first generation (1G) of EGFR-TKIs, erlotinib, and gefitinib, bind to EGFR with sensitizing mutations in a reversible manner, while second-generation (2G) EGFR-TKIs, such as afatinib, bind irreversibly to EGFR. 5 Response rates to EGFR-TKIs are high (63–83%);1,5–7 however, the majority of patients treated with 1 L EGFR-TKIs will acquire resistance.5,8,9 In around 50% of acquired resistance cases, the T790M resistance mutation in exon 20 of EGFR is observed.8–16 In recent years, the preferred 1 L treatment has shifted toward the third-generation EGFR-TKI, osimertinib.3,4
Osimertinib is an irreversible EGFR-TKI that selectively inhibits the EGFR-TKI sensitizing mutations exon 19 deletion (ex19del) and exon 21 L858R, as well as the T790M resistance mutation and has demonstrated efficacy in NSCLC central nervous system (CNS) metastases.17–23 Osimertinib gained accelerated approval from the US Federal Drug Administration (FDA; November 2015) and European Medicines Agency (EMA; February 2016) for patients with NSCLC that acquired a T790M resistance mutation after EGFR-TKI treatment, with full marketing authorization given in 2017. Based on results from the phase III FLAURA study, in which median progression-free survival (PFS) was significantly longer with osimertinib than comparator EGFR-TKIs, erlotinib, or gefitinib, 19 additional approval was given for 1 L treatment of EGFRm advanced/metastatic NSCLC with osimertinib by the FDA and EMA in April and June 2018, respectively. Median overall survival (OS) in the FLAURA study was also significantly longer with osimertinib versus comparator. 21
Real-world studies (RWS) have confirmed the use of EGFR-TKIs as 1 L standard of care for patients with EGFRm advanced/metastatic NSCLC.24–28 Post-progression testing, subsequent treatment patterns,24,25,29 and survival outcomes25,30,31 are, however, less well reported. Such data may impact clinical decision-making concerning the use of 1 L EGFR-TKIs, particularly in countries where osimertinib is not yet approved in this setting. REFLECT represents one of the largest RWS of a population in Europe and Israel aiming to further characterize the treatment landscape, testing patterns, and attrition rates in patients receiving 1 L 1G/2G EGFR-TKIs treatment in the EGFRm advanced/metastatic NSCLC setting.
Materials and methods
Study design
REFLECT was a retrospective, non-interventional, medical record review in Europe and Israel in patients with EGFRm locally advanced or metastatic NSCLC (NCT04031898). Medical chart review with data collection was conducted from May to December 2019 in Austria, Bulgaria, Greece, Israel, Poland, Romania, Slovenia, and Switzerland.
Participants
Eligible patients were ⩾18 years of age with a confirmed diagnosis of locally advanced unresectable or metastatic NSCLC and laboratory-confirmed EGFRm, with initiation of 1 L 1G/2G EGFR-TKI treatment (gefitinib, erlotinib, or afatinib, as monotherapy or combination therapy) between 1 January 2015 and 30 June 2018. Exclusion criteria included enrollment in an interventional clinical trial related to EGFRm NSCLC; receiving any systemic treatment for advanced disease prior to 1 L EGFR-TKI treatment; and having missing/unknown data on key study dates.
Data sources and collection
Eligible patients were enrolled in a consecutive manner from the earliest date of 1 L EGFR-TKI initiation with data collected electronically from initial diagnosis of NSCLC until death, or the last available medical record at the time of inclusion in the study. All patient records were assigned an anonymized, encrypted identifier.
Standard protocol approvals, registration, and patient consents
This study was performed in accordance with ethical principles consistent with the Declaration of Helsinki, Good Clinical Practice and local legislation on observational studies. The final protocol was approved by the Ethics Committees or Institutional Review Boards involved (see Supplemental Figure S1 for details). Study approvals and waiver of informed consent form were obtained from national and/or local Ethics Committees in participating countries. This retrospective study did not require informed, written consent from patients alive at data collection, except for all sites in Greece and two sites in Switzerland where patient consent was obtained.
Endpoints
The primary endpoints were to describe the type of 1 L 1G/2G EGFR-TKI treatment; the proportion of patients with disease progression on 1 L EGFR-TKIs; and to evaluate real-world PFS (rwPFS) while receiving 1 L EGFR-TKIs. Disease progression was defined as radiological progression, clinical progression, death, or the start of a new line of therapy. To distinguish this from PFS obtained in prospective clinical trials using the response evaluation criteria in solid tumors (RECIST), we refer to it as rwPFS. The proportion of patients who received/did not receive 2 L therapy following progression on 1 L treatment and the type of 2 L therapy received were also primary endpoints.
Secondary endpoints included collection of patient demographics, baseline disease characteristics, and testing procedures for EGFR mutations (type of sample, type of test, and primary/secondary tumor). Other secondary endpoints included T790M testing procedures and results, the proportion of patients with CNS metastases or leptomeningeal (LM) disease at 1 L EGFR-TKI initiation, proportion who developed CNS metastases or LM disease over time, and OS from 1 L EGFR-TKI treatment initiation in patients who had CNS metastases at 1 L EGFR-TKI initiation, in those who developed CNS metastases during treatment, and in those who had no CNS metastases at data collection.
Time to treatment discontinuation (TTD) from initiation of 1 L EGFR-TKI treatment, and the proportion of patients receiving osimertinib at any treatment line were exploratory endpoints.
Statistical methods
Sample size was defined based on the feasibility information from each country, accounting for the number of patients managed in the study-defined period. No formal statistical hypotheses were set and all statistical analyses were descriptive. Median rwPFS, TTD, and OS were estimated by the Kaplan–Meier method and all were stratified by country (TTD and OS by country are not reported here). For the stratified OS analysis, there had to be >20 events and >50% maturity. Patients known to be alive at last date of available follow-up were censored.
Results
Overall, 899 patient records were included from 49 thoracic oncology and pulmonology centers. The per-protocol population was 896 patients (three patients did not meet eligibility criteria). The median (range) duration of follow-up (from initiation of 1 L EGFR-TKI to last known date of follow-up or death) was 21.5 (0.1–58.9) months. Patient demographics and disease characteristics are in Table 1.
Patient demographics and baseline disease characteristics at initial NSCLC diagnosis (per-protocol population).

CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; Exdel19, exon 19 deletion; Ex20ins, exon 20 insertion; NSCLC, non-small-cell lung cancer.
At first diagnosis of metastatic NSCLC.
Other tumor histology included mixed histology n = 8 (1%), large cell carcinoma n = 2 (<1%) and other n = 12 (1%).
One patient was reclassified from the L858R subgroup to the uncommon mutation subgroup based on identification of their uncommon mutational status. This was identified following the main analyses; no impact was seen on the rwPFS and OS results.
Recorded as ‘S768I, exon 20 deletion’.
EGFR testing at baseline
Tissue biopsy at diagnosis was used for EGFR testing in most patients (84%); other specimen types were cytological (12%) and liquid biopsy (4%). Biopsies were taken from the primary tumor (76%), secondary tumor (18%); while liquid biopsy or unknown site comprised 6%. EGFR mutations detected were ex19del in 54% of patients, L858R in 31% of patients, while 14% had uncommon EGFR mutations. The most frequently reported uncommon mutations were G719X (2%), L816Q (2%), and exon 20 insertion (ex20ins; 1%); 6% were reported as other or not specified (Table 1).
1 L treatment and progression
The most frequently initiated 1 L EGFR-TKI was afatinib (45%), followed by erlotinib (27%) and gefitinib (27%). At time of data collection, 765 patients (85%) had discontinued 1 L EGFR-TKI treatment, including 86 patients (10%) who died on 1 L EGFR-TKIs, with 131 patients (15%) still receiving 1 L treatment. Of those who discontinued, 76 patients did so due to an adverse event. In total, 723 patients had 1 L per-protocol progression events: radiological progression in 64%, clinical progression in 16%, death in 12%, and start of a new line of therapy in the absence of documented progression in 9%. Median (95% confidence interval (CI) rwPFS was 13.0 (12.3–14.1) months (Figure 1(a)). Kaplan–Meier estimates of rwPFS suggested some variation between countries (Figure 1(b)). Median (95% CI) TTD of 1 L treatment was 12.6 (11.8–13.3) months (Figure 1(c)).
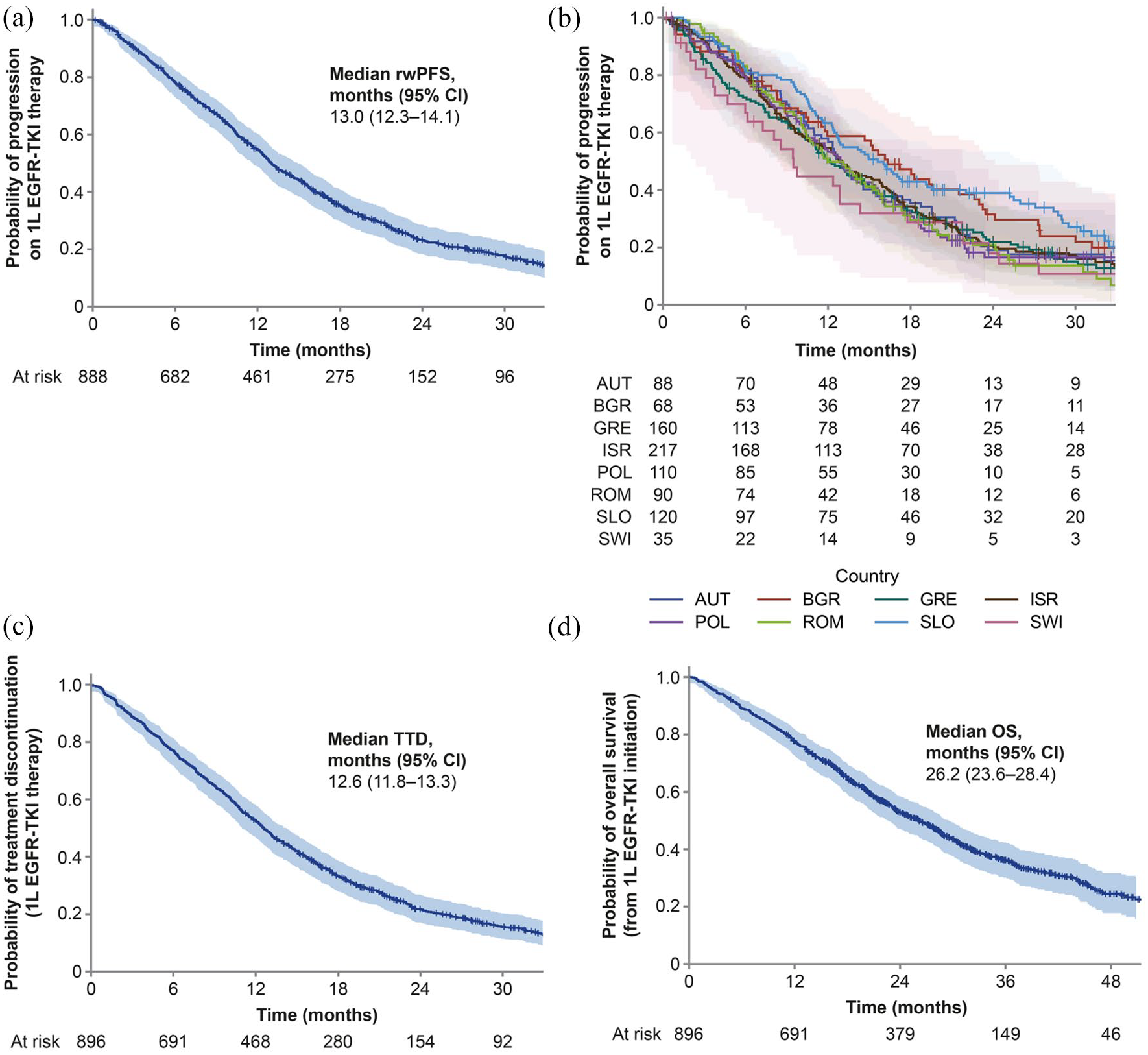
Kaplan–Meier curves: (a) real-world progression-free survival on first-line EGFR-TKI treatment, (b) real-world progression-free survival on first-line EGFR-TKI treatment by country, (c) time to treatment discontinuation of first-line EGFR-TKI treatment, and (d) overall survival from initiation of first-line EGFR-TKI (per-protocol population).
When analyzed by subgroups of EGFR mutation at baseline, median (95% CI) rwPFS was 14.2 (12.8–15.6) months for patients with ex19del, 13.3 (11.7–15.1) months for patients with L858R, and 10.1 (7.7–11.2) months for patients with uncommon mutations.
Survival
At the end of the data collection period, 542 deaths had been recorded (60% maturity). Median (95% CI) OS from initiation of 1 L EGFR-TKI was 26.2 (23.6–28.4) months (Figure 1(d)). Estimated probabilities for OS (95% CI) at 12, 24, and 36 months were 78% (75%–80%), 53% (49%–56%), and 36% (33%–40%), respectively.
Median (95% CI) OS according to EGFR mutation at baseline was 29.0 (27.1–32.5) months for patients with ex19del, 24.0 (20.8–27.5) months for patients with L858R and 18.1 (13.4–22.4) months for patients with uncommon mutations.
CNS metastases and LM disease
At 1 L EGFR-TKI initiation, CNS metastases were recorded in 22% of patients (n = 198/896) and a further 15% (n = 134/896) developed CNS metastases during follow-up (37% of patients (n = 332/896) overall). CNS metastases were most frequently diagnosed using imaging (97%), including magnetic resonance imaging (MRI) and CT; other methods used were tissue biopsy (4%) and cerebrospinal fluid cytology (<1%).
In patients with CNS metastases at initiation of 1 L EGFR-TKI, median OS was 19.4 months (95% CI: 17.1–22.1). The median time (range) from initiation of 1 L EGFR-TKI to CNS metastases diagnosed during treatment was 13.5 (0.1–53.8) months. In patients with CNS metastases diagnosed during treatment, median OS from initiation of 1 L EGFR-TKIs was 24.8 months (95% CIs not available). In patients with no record of CNS metastases at the time of data collection (n = 564), median OS was 30.3 months (95% CI: 27.1–33.8). Treatment for CNS metastases was most commonly whole brain radiation therapy (WBRT; 42%). Other treatments included stereotactic radiosurgery (27%), targeted therapy (22%), and surgical resection (7%); 15% did not receive treatment for CNS metastases.
At initiation of 1 L EGFR-TKI, 1% of patients had LM disease, and another 4% developed LM disease during treatment. Median (range) time to first LM diagnosis during treatment was 18.0 (0.1–52.5) months. The number of deaths in the subgroup analyses were too small to allow OS to be reported. LM disease was diagnosed mainly using imaging, including MRI and CT (88%); the other method used was cerebrospinal fluid cytology (12%).
T790M mutation testing
Among the 723 patients with a 1 L progression event (including 86 patients who died at 1 L), 513 patients (71%) were tested for T790M (at any time). Of these, 29 patients were tested for T790M while receiving 1 L treatment but died on 1 L. 210 patients (29%) had no record of a test.
T790M testing was performed using liquid biopsy (72%), tissue biopsy (21%), cytological specimen (6%), and unknown (1%). Of the 513 patients with 1 L progression who were tested for T790M at any time, 299 (58%) patients were T790M positive and 213 (42%) patients were T790M negative (Figure 2). Therefore, 41% of all patients with 1 L progression had a positive T790M test available.

Testing for EGFR T790M mutations after first-line progression (per-protocol population).
2 L treatment and later
Among the 765 patients who discontinued 1 L, one-third did not receive any 2 L therapy (n = 250; 33%). Of the patients who progressed on 1 L treatment, 515 initiated 2 L therapy (71%). Osimertinib was received by 60% of patients initiating 2 L, which was 43% of all patients with a 1 L progression event. Other 2 L therapies were chemotherapy (32%), targeted therapy (3%), immuno-oncological therapy (3%), and other (2%). Further details on 2 L treatment are in Supplemental Figure S1 and Supplemental Table S2, and on later treatment lines in Supplemental Figure S2 and Supplemental Tables S3 and S4.
Osimertinib treatment and T790M testing
In patients who progressed on 1 L EGFR-TKI treatment, 339 (47%) received osimertinib treatment at any line and irrespective of T790M status, with most patients initiating at 2 L (n = 308; 43%; Figure 3). Among the 299 patients who tested positive for T790M, the majority (95%) received osimertinib at 2 L or later, with 88% receiving osimertinib at 2 L. Surprisingly, a small proportion of patients who progressed on 1 L EGFR-TKIs and were T790M negative received osimertinib at a subsequent line (n = 41; 6%). Similarly, 2% of patients who progressed on 1 L EGFR-TKIs and who were not tested for T790M received osimertinib at a subsequent line.

Osimertinib treatment in patients with first-line progression on EGFR-TKIs (n = 723) by EGFR T790M testing status.
Discussion
The REFLECT study illustrated real-world treatment patterns and outcomes in patients with EGFRm advanced/metastatic NSCLC, who initiated EGFR-TKIs prior to approval of osimertinib for 1 L treatment of EGFRm NSCLC. Progression at 1 L was high, and not all patients with progression were tested for T790M. Among patients who discontinued 1 L EGFR-TKI treatment, one-third never received 2 L treatment: some died while receiving 1 L treatment and for others, the reason for not receiving further treatment was not captured. Of patients who progressed on 1 L EGFR-TKIs, almost half received osimertinib as a subsequent treatment.
Guidelines recommend that patients receiving 1G/2G EGFR-TKIs at 1 L who progress should undergo T790M testing.2–4 Our results showed that only 71% of patients were tested post-progression for T790M (the 513 patients tested included 29 patients who were tested but died on 1 L treatment). Similar testing rates have been reported in other RWS.24,32 Furthermore, the date for 1 L EGFR-TKI initiation for the first patients in REFLECT was from January 2015 and 2 L treatment may have been initiated before, or shortly after, guidelines were updated to recommend T790M testing (EMA approval of osimertinib February 2016). A survey of pathologists from Central and Eastern European countries in 2017 noted that liquid biopsies, typically used for T790M testing, were not reimbursed in Poland, Bulgaria, and Romania. 33 Testing rates and differences in testing strategies of countries may; therefore, have impacted access to osimertinib at 2 L. Although not standard practice, repeated T790M testing via liquid biopsy during EGFR-TKI treatment, rather than a single test at clinical progression as currently recommended,2,4 could allow earlier detection of T790M and a switch to appropriate therapy, potentially leading to improved survival outcomes. This approach is being evaluated in an ongoing trial. 34
REFLECT also showed that 43% of patients progressing at 1 L received osimertinib at 2 L. This was higher than observed in other RWS in the United States, in which only 16% 24 and 25% 32 of patients received osimertinib. In these studies, the low percentages of patients receiving 2 L osimertinib is likely due to the low T790M testing rates; 19% (47/246 patients) and 39% (63/160 patients) were tested, respectively, and consequently low numbers of T790M-positive patients were identified.24,32
While afatinib was the most common 1 L EGFR-TKI received in our study, other real-world retrospective studies have identified gefitinib26,28,29 or erlotinib13,24 as the most commonly initiated 1 L EGFR-TKI. This may have been due to differences in geographic location, dates of data retrieval, reimbursement status, or other inclusion criteria between studies. The data on treatments in REFLECT showed that only 57% of the patients were exposed to 2 L therapy (71% of patients with 1 L progression). These findings are similar to those of other RWS; two retrospective studies in the United States found that only 26% 24 and 44% 25 of patients at progression were exposed to 2 L therapy, respectively.
In our study, median rwPFS on 1 L treatment was 13.0 months. Assessment of progression, however, in RWS is known to be less standardized compared with clinical trials. Consequently, this precludes any meaningful comparison of rwPFS with PFS derived from clinical trials. However, in an RWS in Poland, the median rwPFS was 11.9 months in patients of similar age, gender, and Eastern Cooperative Oncology Group (ECOG) performance status (PS) who received 1 L 1G/2G EGFR-TKIs to our study. 30 By contrast, in an RWS in Italy, where patients were treated with 1 L 1G EGFR-TKIs and 2 L systemic treatment, a low median rwPFS of 4.7 months was observed. 31 Results were likely influenced by dates of data collection prior to 2G EGFR-TKI availability (1 L EGFR-TKI initiation June 2009–May 2013), and the inclusion of patients with ECOG PS 3 and 4. Median OS (26.2 months) in REFLECT was similar to that from an RWS of patients receiving 1 L erlotinib 2011–2016 (23.3 months), 25 and was longer versus patients receiving 1 L afatinib (20.7 months), 25 and in two other RW studies with 1 L EGFR-TKIs 2012–2016 (19.4 months), 30 and 2009–2013 (18.7 months), 31 respectively.
Over one-third of patients had CNS metastases, either at initiation of 1 L EGFR-TKIs or developed during follow-up. Nearly half of patients with CNS metastases were treated with WBRT, which appears high considering the cognitive impairments associated with WBRT. 35 There have been rapid advances in radiotherapy in recent years, so the frequency of WBRT observed may reflect practices which are no longer recommended, the availability of stereotactic radiosurgery, or a lack of reimbursement by the health systems in the participating countries. Current ESMO guidelines suggest that patients with EGFRm NSCLC and CNS metastases are treated with CNS-penetrant next generation EGFR-TKIs to control CNS metastases and delay cranial radiotherapy.2,4 Osimertinib achieves significant exposure in the CNS versus other EGFR-TKIs, 18 and has demonstrated CNS efficacy in patients with untreated EGFRm NSCLC; results suggest reduced CNS progression compared with gefitinib or erlotinib. 20
Limitations of this study included its observational and retrospective nature. The level of detail in reporting the EGFR mutations was therefore variable. The study was descriptive only; it did not have a formal hypothesis on the effectiveness of EGFR-TKIs and was not powered for comparisons between individual drugs/countries. The results reported will, to some extent, reflect the different healthcare systems and the evolution of EGFR testing procedures in each country. Though early access programs for osimertinib in 2 L for T790M-positive patients were available starting from May 2015 to July 2016, reimbursement status may have affected each country’s access to T790M testing and osimertinib; 2 L osimertinib reimbursement began later in Poland, Slovenia, Romania, and Bulgaria (March 2017–March 2018) than in other participating countries (January–July 2016). Also, the study reflects practices at a time when osimertinib was not available for 1 L treatment. Some information, such as patient ethnicity and sites of recurrence at progression was also not collected.
REFLECT is one of the largest RWS in this patient population. It highlights that attrition during 1 L 1G/2G EGFR-TKI treatment is high, with one-third of patients not receiving any 2 L therapy at progression. The study also highlights the negative prognostic impact of CNS metastases. Given the high-attrition rates also reported in many RWS and clinical trials and the impact of CNS progression, it is key to offer the most appropriate 1 L EGFR-TKI treatment with CNS penetration to maximize outcomes, especially as many patients may not receive any subsequent therapies.
Supplemental Material
sj-docx-1-tam-10.1177_17588359211059874 – Supplemental material for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT)
Supplemental material, sj-docx-1-tam-10.1177_17588359211059874 for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT) by Alfredo Addeo, Maximilian Hochmair, Urska Janzic, Elizabeth Dudnik, Andriani Charpidou, Adam Płużanński, Tudor Ciuleanu, Ivan Shterev Donev, Judith Elbaz, Jørgen Aarøe, René Ott and Nir Peled in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359211059874 – Supplemental material for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT)
Supplemental material, sj-docx-2-tam-10.1177_17588359211059874 for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT) by Alfredo Addeo, Maximilian Hochmair, Urska Janzic, Elizabeth Dudnik, Andriani Charpidou, Adam Płużanński, Tudor Ciuleanu, Ivan Shterev Donev, Judith Elbaz, Jørgen Aarøe, René Ott and Nir Peled in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359211059874 – Supplemental material for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT)
Supplemental material, sj-docx-3-tam-10.1177_17588359211059874 for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT) by Alfredo Addeo, Maximilian Hochmair, Urska Janzic, Elizabeth Dudnik, Andriani Charpidou, Adam Płużanński, Tudor Ciuleanu, Ivan Shterev Donev, Judith Elbaz, Jørgen Aarøe, René Ott and Nir Peled in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359211059874 – Supplemental material for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT)
Supplemental material, sj-docx-4-tam-10.1177_17588359211059874 for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT) by Alfredo Addeo, Maximilian Hochmair, Urska Janzic, Elizabeth Dudnik, Andriani Charpidou, Adam Płużanński, Tudor Ciuleanu, Ivan Shterev Donev, Judith Elbaz, Jørgen Aarøe, René Ott and Nir Peled in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-5-tam-10.1177_17588359211059874 – Supplemental material for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT)
Supplemental material, sj-docx-5-tam-10.1177_17588359211059874 for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT) by Alfredo Addeo, Maximilian Hochmair, Urska Janzic, Elizabeth Dudnik, Andriani Charpidou, Adam Płużanński, Tudor Ciuleanu, Ivan Shterev Donev, Judith Elbaz, Jørgen Aarøe, René Ott and Nir Peled in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-6-tam-10.1177_17588359211059874 – Supplemental material for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT)
Supplemental material, sj-jpg-6-tam-10.1177_17588359211059874 for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT) by Alfredo Addeo, Maximilian Hochmair, Urska Janzic, Elizabeth Dudnik, Andriani Charpidou, Adam Płużanński, Tudor Ciuleanu, Ivan Shterev Donev, Judith Elbaz, Jørgen Aarøe, René Ott and Nir Peled in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-7-tam-10.1177_17588359211059874 – Supplemental material for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT)
Supplemental material, sj-jpg-7-tam-10.1177_17588359211059874 for Treatment patterns, testing practices, and outcomes in the pre-FLAURA era for patients with EGFR mutation-positive advanced NSCLC: a retrospective chart review (REFLECT) by Alfredo Addeo, Maximilian Hochmair, Urska Janzic, Elizabeth Dudnik, Andriani Charpidou, Adam Płużanński, Tudor Ciuleanu, Ivan Shterev Donev, Judith Elbaz, Jørgen Aarøe, René Ott and Nir Peled in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Thanks to all the patients and their families. The authors would like to acknowledge Roxana Lupu and Una Rigney, AstraZeneca, for providing support throughout the development of this manuscript; Ana Maria Iordan (MedInteractiv, Bucharest, and Romania) for project management and for support with development of figures, funded by AstraZeneca. Statistical analyses were provided by Planimeter Inc (Budapest, Hungary) and were funded by AstraZeneca. The authors would like to acknowledge Sally Cotterill, PhD, CMPP, of Ashfield MedComms, Macclesfield, UK, an Ashfield Health company for medical writing support in drafting the manuscript and revising to address author feedback that was funded by AstraZeneca in accordance with Good Publications Practice (GPP3) guidelines (![]() ).
).
Author contributions
AA contributed to conceptualization, methodology, validation, formal analysis, investigation, data curation, writing – review and editing and supervision. MH contributed to conceptualization, methodology, validation, formal analysis, investigation, resources, data curation, writing–original draft, writing – review and editing, visualization and supervision. UJ contributed to conceptualization, investigation, data curation and writing – review and editing. ED contributed to conceptualization, methodology, validation, investigation, resources, writing – review and editing, visualization and supervision. AC contributed to validation, investigation and writing – review and editing. AP contributed to conceptualization, methodology, resources, writing – original draft and writing – review and editing. TC contributed to investigation, writing – original draft and writing – review and editing. ISD contributed to investigation, resources, data curation and writing – review and editing. JE contributed to validation, writing – review and editing, supervision and project administration. JA contributed to validation, writing – review and editing, visualization, supervision and project administration. RO contributed conceptualization, resources, writing – review & editing, supervision and funding acquisition. NP contributed to conceptualization and writing – review and editing. All authors approved the final version for submission.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA received honoraria from AstraZeneca, BMS, Eli Lilly, MSD, Pfizer, Roche and Takeda outside the submitted work. MH received honoraria for speaker’s bureau from AstraZeneca, BMS, Boehringer Ingelheim, MSD, Roche and Takeda, and served on advisory boards for AstraZeneca, BMS, Boehringer Ingelheim, MSD, Roche and Takeda. UJ served on advisory councils or committees for AstraZeneca, Boehringer Ingelheim, MSD and Roche, and received honoraria from AstraZeneca, BMS, Boehringer Ingelheim, MSD, Pfizer and Roche. ED served on advisory councils or committees for AstraZeneca, BMS, Novartis, Pfizer, Roche, Sanofi, and Takeda, received honoraria from AstraZeneca, BMS, MSD, Novartis, Pfizer, Roche, Sanofi, Pfizer, and Takeda and received consulting fees from AstraZeneca, BMS, Novartis, Pfizer, Roche, Sanofi, and Takeda, all outside the submitted work. AC served on advisory councils or committees for ASTRA, Boehringer Ingelheim and MSD, received honoraria from ASTRA, BMS, MSD and Roche and received consulting fees from ASTRA and Boehringer Ingelheim, outside the submitted work. AP has received personal fees and non-financial support from AstraZeneca, BMS, Boehringer Ingelheim, Pfizer and Roche, and personal fees from MSD, and Takeda outside the submitted work. TC served on advisory councils or committees for Amgen, Astellas, AstraZeneca, BMS, Boehringer Ingelheim Janssen, MSD. Roche, and Pfizer outside the submitted work. ISD has no disclosures to declare. JE and JA are employees of AstraZeneca. RO is an employee of AstraZeneca AG and owns stocks/shares in AstraZeneca AG. NP served on advisory councils or committees for AstraZeneca, Bayer, Boehringer Ingelheim, BMS, Eli Lilly, FoundationMedicine, Guardant360, Merck, MSD, Novartis, NovellusDx, Pfizer, Roche and Takeda, received honoraria and consulting fees from AstraZeneca, Bayer, Boehringer Ingelheim, BMS, Eli Lilly, FoundationMedicine, Guardant360, Merck, MSD, Novartis, NovellusDx, Pfizer, Roche and Takeda, and received grants or funds from AstraZeneca, Bayer, Boehringer Ingelheim, BMS, Eli Lilly, Merck, MSD, Novartis, Pfizer, Roche and Takeda.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study NCT04031898 was funded by AstraZeneca, the manufacturer of the drugs osimertinib and gefitinib.
Role of the funding source
AstraZeneca played a role in the design and conduct of the study; data collection, and analysis, and interpretation of data; and review and approval of the manuscript. Medical writing support was funded by AstraZeneca.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
