Abstract
Biochemical recurrence is an evolving space in prostate cancer, with increasing multidisciplinary involvement. Androgen deprivation therapy has shown proof of its value in complementing salvage radiotherapy in high-risk biochemical relapsing patients; ongoing trials aim to further refine this treatment combination. As systemic treatments, and notably next-generation androgen receptor targeted agents, have moved towards early hormone-sensitive and non-metastatic stages, the prostate specific antigen (PSA)-relapse disease stage will be undoubtedly challenged by future evidence from such ongoing clinical trials. With the use of modern imaging and newer molecular technologies, including integration of tumoral genomic profiling and liquid biopsies in risk stratification, a path towards a precision oncology-focused approach will become a reality to guide in the future decisions for patients with a diagnosis of biochemical recurrence.
Keywords
The notion and relevance of biochemical recurrence (BCR)
Definition and clinical outcomes
In contrast to other malignancies, prostate cancer (PC) is characterized by a lower mortality and the majority of patients living with PC have a prognosis of many years. Globally, PC accounts for 7.3% of the overall number of new cancer cases (14.1% for males), and 3.8% of deaths, with an all-stage 10-year survival rate at 98%.1,2
Except when it isn’t. Most patients with prostate cancer are diagnosed at a localized or locoregional stage, which explains the better survival rates. However, for men diagnosed with metastatic PC, the 5-year survival rate is estimated to be 31%; patients diagnosed with upfront metastatic disease contribute to half of PC deaths. 3 In the US, owing to the high incidence of the disease (first in males, 21% of all new cancer cases), mortality from metastatic PC ends up being the second highest cause n males (10% of deaths). 4
The critical moment that often points towards a less optimistic prognosis is the failure of primary treatment, be it radical prostatectomy (RP), radiotherapy (RT), brachytherapy, high-intensity focused ultrasound (HIFU), cryosurgery, or other focal therapy options. This failure will most frequently be in the form of a rising prostate-specific antigen (PSA), without macroscopically detectable disease in the first instance, i.e., a biochemical recurrence (BCR). The definition of BCR depends on the type of prior definitive therapy. In patients who have undergone RP, the European Association of Urology (EAU) 2020 guidelines propose that a rising serum PSA level should be considered a BCR. 5 Ultrasensitive PSA levels >0.01 ng/ml, in combination with clinical characteristics such as the International Society of Urological Pathology (ISUP) grade and surgical margin status, may be predictive of PSA progression after RP. In patients treated with RT, the RTOG-ASTRO Phoenix Criteria define BCR as a rise in PSA level of 2 ng/ml or more above the nadir, regardless of androgen deprivation therapy (ADT) use or of the nadir value. 6
Several nomograms have been created for the estimation of BCR risk, such as the CAPRA-S, the MSKCC, and the Walz nomograms.7 –9 A recent update of the latter seems to provide an elegant estimates BCR risk at 12 and 24 months post-RP based on PSA, Gleason score (GS), pT stage, surgical margin, and lymph node status. 10
The rate of BCR within 10 years following definite treatment is 2040% after radical prostatectomy (RP)11 –13 and 30–50% following radiotherapy (RT).14,15 BCR represents a true progression: it is associated with a 24–34% risk of developing metastasis,10,16 with a PSA of 0.4 ng/ml a better predictor of this. The median time from RP to PSA failure is reported to be between 2 years13 (Freedland et al.) and 3 years, 17 with a median metastasis-free survival (MFS) reported at 8–10 years.11,17 After RT, PSA-doubling time (PSA-DT) correlates with the site of recurrence. While patients with local recurrence have a PSA-DT of 13 months, those with a PSA-DT of 3 months present with distant metastases. 18 EAU guidelines utilize PSA-DT (cut-off: 1 year) and GS (cut-off: 8) to define low and high-risk BCR post-RP, and interval to primary therapy (cut-off: 18 months) and GS (cut-off: 8) to define the respective BCR groups post-RP.19,20
Does MFS translate into PC-specific mortality? Overall, in unselected patients, the median time from metastasis to death was historically reported as 5 years. 11 However, this data was validated before the recent therapeutic advances in metastatic castration-sensitive (mCSPC) and castration-resistant (mCRPC) prostate cancer. In a cardinal mCRPC study, at a median follow-up (FU) of 6 years (rather limited for such purposes), the 5-, 10-, and 15-year cause-specific survival from the respective time of biochemical recurrence was 93% [95% confidence interval (CI), 90–96%), 73% (95% CI, 66–79%), and 55% (95% CI, 41–67%), but varied widely between the highest and lowest risk subgroups. 13 PSA-DT (especially if less than 3.0 months), pathological GS (8–10), and time from surgery to biochemical recurrence (⩽3 years) are strong predictors of metastasis and PC-specific mortality,11 –13 although the latter factor has not been retained in more recent analyses. 17 These factors stratify patients at distinct risk groups, with a median MFS ranging from 1.0 year in the highest risk group to 15.0 years in the lowest risk group. 17 For example, the median MFS is 15 years and PC–specific survival is approximately 90% in patients with a PSA-DT of ⩾15 months, whereas these are approximately 1 year and 20% respectively for patients with a PSA-DT of <3 months.13,17 The EAU BCR risk stratification has been externally validated post-RP and found to be significantly predictive of 5-year MFS [hazard ratio (HR): 3.46; p < 0.001) and PC-specific mortality (HR 5.12; p < 0.001). 19
Biology of disease recurrence
The identification of the earliest alterations in PC can give important insights into the relationships among primary and metastatic sites. Across cancer types, metastases have been reported to originate from single clones in the primary tumour (monoclonal seeding), or multiple clones (polyclonal seeding), however the distribution of these patterns across specific tumour types is not fully known. 21 This polyclonal seeding is a frequent event in prostate cancer and has also been found to be associated with oncogenic alterations of ADT resistance, such as MYC amplification or pathogenic AR substitution, suggesting that the tumour cell populations with a significant survival advantage are not confined within the boundaries of an organ site but can successfully spread to and reseed other sites. 22 In fact, in metastatic PC, multiple metastases were found to be more closely related to each other than any of them were to the primary prostate tumour, with sharing of sub clonal alterations by different metastases, indicating possible inter clonal cooperativity or re modelling of metastatic niches by initial colonizing prostate cancer clones, making them attractive habitats that other clones can colonise. 22 Multiple subclones achieve a metastatic potential through early alterations, such as tumour protein 53 (TP53) or phosphatase and tensin homolog (PTEN). TP53 mutations, in particular, appear to be strongly implicated in metastatic spread and can be easily detected with a liquid biopsy even before the appearance of metastatic lesions. 23 Secondary lesions appear to develop in the form of spread between distant sites, rather than single waves coming from the primary tumour. AR alterations are rarely present in the primary tumour but seem to develop in the CRPC setting. AR aberrations are found to be heterogeneous and involve multiple events at different sites.
If such findings are further exploited and confirmed, primary tumour sequencing and liquid biopsies might prove helpful in detecting occult clones even before they become clinically visible. Importantly, the better understanding of the complex patterns of sub clonal differentiation between different metastases from distinct anatomical sites, with individual subclones able to seed polyclonally from one metastasis to another, support the rationale for an approach combining the local control of the primary with ablation of all oligometastatic tumour deposits.22 –25 Implementation of metastasis-directed therapies (MDT), together with systemic therapies, therefore represents an interesting therapeutic option to potentially eliminate sources for metastatic spread and improve oncological outcome in patients with relapsing prostate cancer (see discussion below).
Impact of novel imaging technologies
The notion of BCR is recently challenged by technological advances. Molecular and functional imaging shrink the space of PSA-only relapse by increasing the abilities of detecting and localizing sites of otherwise occult recurrent disease. The most sensitive positron emission tomography (PET) tracers currently available are the class of prostate-specific membrane antigen (PSMA)-targeted radiotracers. Its comparative detection sensitivity in the BCR setting has been assessed against a previous-generation tracer, choline, demonstrating the superiority of Ga-PSMA in terms of detection rates at any PSA level,26 –28 and has now been included as a recommendation in this setting in international guidelines. 29
At the usually low PSA levels BCR features, even with PSMA-PET and more so with conventional imaging, the eventual identification of macroscopic disease is invariably limited to an oligo-recurrence context. Other than enlarging this space as it shrinks the true BCR space, the availability of such imaging has also shifted the treatment paradigm towards metastasis-directed treatment, both in the CSPC and the CRPC setting. This has shown clinically meaningful benefit such as ADT-free survival, CRPC-free survival, or time to treatment escalation.30 –34
Precision medicine and molecular strategies for risk stratification
DNA-repair pathway alterations have gained significance in the therapeutic arena of advanced prostate cancer. The prognostic significance of such mutations, notably for the BRCA2 gene, was noted in the last decade, with identified association with poor survival outcomes, including metastatic relapse and cancer-specific survival post local treatment.35,36 In a relevant meta-analysis, BRCA2 mutations have been shown to predict poor survival outcomes in prostate cancer patients in terms of cancer-specific survival and overall survival (OS), with pooled HRs of 2.53 (95% CI: 2.10–3.06, p < 0.001) and 2.21 (95% CI: 1.64–2.99, p < 0.001), respectively. 37
Specifically, in BCR, overexpression of BRCA1/2 in prostatectomy specimens seems to be independently predictive of biochemical recurrence. 38 No information exists on specific phenotype-genotype prognostic correlations for the BRCA or other DNA repair pathway genes.
Multigene panels, such as the 17-gene Oncotype Dx, 39 the 31-gene Prolaris, 40 and the 22-gene Decipher 41 assess BCR risk at the time of diagnosis or post-RP. Although independently validated, these panels have not yet entirely revolutionized localized and loco-regional prostate cancer management for two main reasons. Firstly, their predictive ability is not uniform across the spectrum; for example, Oncotype DX does not significantly predict BCR in certain subgroups such as patients less than 56 years old, nor are all its components individually predictive. In addition, the impact of such tests in treatment decision-making and on PC mortality is unclear, as is their cost-effectiveness and their influence of BCR surveillance post-definite treatment. The Decipher genomic classifier 41 attempts to predict distant metastasis following RP, as a result assessing PC-specific mortality.42,43 Its predictive ability seems to improve if combined with CAPRA-S. 44 In a prospective evaluation of its clinical utility (PRO-IMPACT trial), the use of Decipher test influenced treatment decisions regarding the choice between adjuvant and salvage treatment, and reduced levels of PC-related anxiety.45,46
An interesting classifier is the 15-gene SigMuc1NW signature, which exploits the biomarker potential of tumour-associated antigens (TAAs) such as mucin 1 (MUC1) and opa interacting protein 5 (OIP5). The signature was validated to strongly predict BCR (HR, 2.44; 95% CI, 1.53–3.87; p = 1.62e-4) after adjusting for clinico-pathological factors such as GS, surgical margin status, age, and stage. 47 These TAAs, although previously investigated in other cancer types, are relatively novel PC-associated TAAs. Despite this, the signature has not been independently validated on other large databases.
In our era of large data and bioinformatics, attempts have been made to assess the predictive, biomarker potential of other signatures involving miRNAs or lncRNAs; however, these are as yet immature for PC.48 –52
Finally, although checkpoint inhibition has not yet proven of meaningful benefit in metastatic prostate cancer, there is some recent limited evidence that increased programmed death-ligan 1 (PD-L1) expression is associated with biochemical recurrence.53,54
Liquid biopsy technology has further enhanced the detection possibilities related to PC recurrence, using more sophisticated techniques compared to measurements of PSA levels, upon which the conventional notion of BCR is based. Such methods include targets such as circulating tumour cells (CTCs), cell-free nucleic acids (such as mRNA), and extracellular vesicles, detected on readily accessible body fluids such as blood, semen, or urine. 55 An example of success in PC is the detection of the androgen receptor (AR) splice variants, such as AR-V7, in CRPC, predicting resistance to systemic treatments such as enzalutamide or abiraterone acetate. This new modality is slowly entering the space of PC diagnostics and accurate distinction from benign conditions. 56 Circulating mRNA technologies and second-generation CTC technologies have also entered the BCR space, with positive retrospective associations with a higher risk for biochemical recurrence following RP, even in the context of negative PSA.56 –58 Although it is still very early for such technologies to translate into clinical practice and guide treatment decisions, also due to the low abundance of circulating biomarkers in localized or early-recurring PC, it nevertheless incites strong interest for the future.
Hormonal therapy
Immediate versus deferred ADT
Androgen-deprivation therapy, as the mainstay for macroscopically recurrent disease, is an obvious option for the management of BCR. Given that the volume of not yet macroscopic disease in BCR is low, the obvious question is whether such management would be beneficial in a BCR setting. Similarly, it is necessary to estimate the potential benefit and optimal timing of any intervention need to be in order for it to be clinically meaningful.
Initial evidence was obtained from two large retrospective studies. The CaPSURE trial (2096 patients with BCR post-RP or RT) demonstrated no significant advantage to immediate ADT versus deferred (at metastatic disease or ⩾2 years after BCR) (HR for mortality: 0.91, 95% CI, 0.52–1.60) at a median FU of 54 months. The estimated 5-year OS (95% CI) was 85.7% versus 87.7%, the 10-year OS was 69.8.1% versus 69.3%. 59 In a larger retrospective study (5804 men), salvage ADT was associated with OS or PC-specific mortality in both the post-RP (HR: 0.35 and HR: 0.43 respectively) or the post-RT cohort (HR: 0.62 and HR: 0.65 respectively) in patients with PSA-DT < 9 months. 60
The prospective phase III TOAD (TROAG 03.06) trial questioned the optimal timing of ADT in patients with rising PSA. 61 Interestingly, however, the trial was not strictly homogeneous in regard with the eligibility criteria; in addition to the pure PSA relapse patients (post-RT or post-RP with or without post-operative RT), the study also included patients with a de novo incurable disease (due to age, comorbidities, or locally advanced disease), the latter amounting to 11% of the total cohort. The study assessed an immediate versus deferred ADT; although the scheduled delay for the trial was at least 2 years, 52% of patients started ADT within 2 years, and patients with poor risk features (such as short PSA-DT) at the time of relapse started with a median delay of 12.3 months. Immediate ADT borderline improved 5-year OS (91.2% versus 86.4%, log-rank p = 0.047; unadjusted HR: 0.55, p = 0.05; adjusted HR: 0.54, p = 0.047; n = 293). Survival curves seem to start to separate after 5 years, with 6-year and 7-year survivals estimated at 76.4% versus 85.6% and 65.5% versus 81% respectively. However, this was not maintained in the PSA relapse only cohort, despite more patients in this group (5-year OS 78.2% versus 83.4%, log-rank p = 0.10; unadjusted HR: 0.58, p = 0.10; adjusted HR: 0.59, p = 0.19; n = 261). The time to local progression was significantly in favor of the immediate ADT arm (adjusted HR: 0.51, p = 0.001), while time to a PC complication did not differ (adjusted HR: 0.78, p = 0.16). No significance could be demonstrated for PC-specific mortality, as the number of events was low. Intriguingly, the time to development of castration resistance from treatment start differed significantly between the two arms in favor of immediate ADT (HR: 0.30, p < 0.001). As the use of continuous versus intermittent ADT was similar between the two arms, this seems to be a true effect. The authors suggest that this may reflect the development of clonal resistance in the untreated patient as the disease progresses, with a reduction of treatment responsiveness and effectiveness in overt metastatic disease. 61 A biological explanation for this reduction in the risk of CRPC development with the use of immediate ADT versus referred observed by Duchesne et al. 61 might be related to the fact that the timing of ADT corresponds to a state of early versus more advanced disease; evidence from mHSPC indicates that disease volume is a predictive factor for progression to CRPC, 62 and in the same direction there seems to be differentiation towards a more ‘drug resistant’ disease in late advanced metachronous disease, often driven by the use of systemic treatment in the localized disease stage. 21 Finally, the higher percentage of high and very high-risk cancers in the delayed ADT arm (42% versus 33% in the immediate ADT arm) could also partly account for the observed shorted time to CRPC in this arm. 61 At the molecular level, an explanation could be that advanced disease has a much higher level of phosphoinositide 3-kinase (PI3K) pathway alterations/PTEN loss, compared to early disease, and this is related to androgen insensitivity, decreased transcription of AR target genes, and CRPC development.63,64
The study also highlighted the difficulties encountered in this space, with slow accrual, high screening failure rate, and early trial termination. The more optimistic outlook on the results of this trial is probably the fact that the immediate arm appears to gain increasing benefit for absolute mortality difference after the 5 first years; approximately 15% by the end of 7 years, and 25% after 8 years, yet these are estimated results, as the median study FU was only 5 years, and very few events occurred in later years. 61
A similar phase trial, ELAAT (Clinicaltrial.gov identifier: NCT00439751), was designed by the Canadian Urological Oncology Group to assess the optimal timing of ADT in men with PSA rise post-RT, but failed to reach its accrual goal. In an attempt to improve significance by increasing the cohort size, a pooled analysis of the TOAD and ELAAT data was performed (n = 261 + 78 = 339). The reasons to start ADT in the two trials were development of symptoms, metastases on conventional imaging, or PSA-DT of ⩽6 months. The combined analysis showed no difference in all-cause mortality (HR 0.75, p = 0.37) or PC-specific mortality, whereas time to local progression, distant progression, and prostate cancer complications differed significantly in favor of immediate ADT. Several explanations may account for the loss of significance for OS in the combined analysis: ELAAT accrued older patients with a higher all-cause mortality risk and a lower relative risk of PC-specific mortality (relative to the overall mortality risk) and had a smaller difference in PSA between the immediate and deferred arms (3.98 and 18.1 ng/ml respectively for the ELAAT group, versus 3.52 and 30.2 ng/dl for the TOAD group); on the other hand, more patients in TOAD had a relapse-free interval of less than 2 years from RT (30% versus 10% for ELAAT), indicating that TOAD had more high-risk patients for which immediate treatment would be potentially more beneficial. 65
Intermittent versus continuous ADT
A large Canadian Cancer Trial Group phase III trial assessed intermittent versus continuous ADT in 1,386 patients with rising PSA (>3 ng/ml) at more than 1 year after RT. 66 Interestingly, RT could be either primary or salvage, hence introducing heterogeneity in terms of prognosis and therefore survival. The trial had a non-inferiority design and met its primary outcome; at a median FU of 6.9 years, the median OS was 8.8 years in the intermittent group versus 9.1 years in the continuous ADT group (HR for death: 1.02, 95% CI 0.86–1.21, p = 0.009), with an estimated 7-year cumulative rates of PC-specific death at 18% versus 15% respectively (HR: 1.18, 95% CI 0.90–1.55, p = 0.24). Other than meeting the non-inferiority threshold, intermittent treatment had a more favorable toxicity profile and resultant quality of life in respect to urinary problems (p = 0.006), hot flashes, (p < 0.001) and libido (p < 0.001), with a trend towards improvement of fatigue (p = 0.07). The authors highlight that the role of predictive factors such as age, GS, and PSA kinetics in the selection of patients for intermittent therapy remains to be defined. They recognize that the known long-term morbidity of ADT, such as in the development of metabolic syndrome and cardiovascular disease. Interestingly, the reported rate of deaths unrelated to PC attributed to a cause other than complication of treatment initiated after castration resistance or other primary cancer was higher in the continuous ADT group (35.9% versus 28%). Overall, any potential benefit on PC–specific mortality of continuous ADT might be balanced by the benefit of avoiding death from other causes, such as cardiovascular disease, using intermittent ADT.
Given the heterogeneity in the long-term response to intermittent therapy, early identification of aggressive diseases would be useful. Higano’s team have shown that the duration of the first off-treatment interval is prognostic for time to CRPC and death in these patients. 67 The same group demonstrated that patients with longer time of PSA rise (⩾60 days) after the first non-castrate level testosterone (>50 ng/dl) during the first off-treatment have good outcomes in terms of risk of developing CRPC, and it could be used for stratification and patient selection in terms of treatment strategies and optimal time for re-starting ADT. 68
Addition of ADT to salvage local treatment and oligometastatic disease
The French phase III GETUG 16 trial 69 assessed the addition of short-term, 6-month ADT to salvage RT (66 Gy in 33 fractions) in patients with rising PSA (between 0.2 and 2 ng/dl), previously undetectable post-RP. At a median FU of 63 months, the addition of ADT resulted in improved 5-year biochemical and clinical PFS (80% versus 62%, HR: 0.50, p < 0.0001) across all patient subgroups, to the price of moderately increased hot flushes and sweats (8% versus 1% of ⩾G2 events), without any serious cardiovascular or other toxicity. The highest benefit was observed for the patients with a baseline PSA of >0.5 µg/l and a PSA-DT of ⩽6 months. The MFS was not estimated in this trial, as only the first progression event was recorded, and this would usually be local progression with or without PSA progression. Given the low baseline PSA levels and the low-risk features of the study cohort (e.g., 90% of the cohort had a Gleason score of up to 7), a longer FU would be necessary to reliably demonstrate any effect on MFS. In this sense, it is impossible to assess the real benefit of the ADT addition to RT on ultimate OS outcomes.
Radiotherapy in the GETUG-16 trial consisted of the prostate bed (including the seminal vesicle area), with a recommendation for pelvic irradiation only in patients who did not have node dissection during radical prostatectomy, and only if the risk of nodal involvement was greater than 15% according to the Partin tables. The phase III NRG Oncology/RTOG 0534 SPPORT trial 70 randomised more precisely, according to the field of salvage RT and the addition of ADT, in a 3-arm fashion: prostate bed RT (64.8–70.2 Gy) versus prostate bed RT (64.8–70.2 Gy) with short-term ADT (4–6 months) versus prostate bed RT (64.8–70.2 Gy) and pelvic RT (45 Gy) with short-term ADT (4–6 months). In the 1792 randomised patients, freedom from progression was 71%, 83%, and 89%, respectively, at a median FU of 6.4 years, with statistical significance demonstrated between each treatment arm, i.e., with each treatment addition. Freedom from distance metastasis also showed a trend for benefit at 5 years (91.7% versus 94.4% versus 95.2%), with a significant reduction for the triple modality arm (p = 0.0140) compared to baseline, but without any significant differences in OS.
Designed prior to the establishment of GnRH analogues as first choice hormonal therapy with RT, the RTOG 9601 trial randomised patients with PSA between 0.2 and 4.0 ng/ml post-RP [(both persistently elevated PSA and rises after an initial complete biochemical response) to salvage RT and 2 years bicalutamide treatment (150 mg daily) versus salvage RT and placebo]. The study has the longest reported median FU (13 years) and showed a significant benefit for 12-years OS (76.3% versus 71.3%, p = 0.04), metastatic PC (14.5% versus 23%, p = 0.005), and PC-specific mortality (5.5% versus 13.4%, p < 0.0001) at 12 yrs. Patients with a lower Gleason score (⩽7), a PSA level of less than 0.7 ng/ml, or negative surgical margins may have less benefit from the addition of antiandrogen therapy. 71 The RTOG 9601 team performed a subsequent subgroup analysis showing a lack of benefit for PSA levels <0.6 ng/ml (HR: 1.16; 95% CI, 0.79–1.70) and increased late grades 3 to 5 cardiac and neurologic toxic effects (odds ratio, 3.57; 95% CI, 1.09–15.97; p = 0.05). In contrast, in patients with PSA > 1.5 ng/ml, there was a 25% 12-year absolute benefit (HR: 0.45, 95%CI: 0.25–0.81). 72
The ongoing British RADICALS-HT trial addresses not only the question of addition of GnRH analogues or bicalutamide to post-operative RT (immediate or salvage), but also of the optimal duration of treatment, using a 3-arm randomization (RT alone versus RT plus 6 months of hormonal treatment versus RT plus 24 months of hormonal treatment) (Clinicaltrials.gov identifier: NCT00541047).
With the implementation of next generation imaging techniques in the clinical workflow, 73 a considerable proportion of relapsing prostate cancer patients are diagnosed with an oligometastatic disease, an intermediate state between a localized relapse, and a widespread metastatic status. In the last years, MDT modalities like stereotactic body radiotherapy (SBRT) or salvage lymph node dissection have emerged as valid treatment options to treat these patients, to postpone systemic therapies, and in some cases to improve outcome.74,75 Although two major prospective studies published so far proposed MDT as alternative to systemic ADT,34,31 emerging evidence suggest a potential synergistic effect of this combined strategy to improve outcome of these patients.76,77 Ongoing trials are prospectively testing the use of SBRT with or without 6 months of concomitant ADT (Clinicaltrials.gov identifier: NCT04302454), the impact of intermittent ADT with or without elective nodal pelvic irradiation (Clinicaltrials.gov identifier: NCT03630666), or elective nodal irradiation versus SBRT, both combined with 6 months of ADT, 78 in patients with an exclusive oligometastatic nodal relapse. Androgen receptor targeted agents are also tested with SBRT in other ongoing clinical trials. 79
Chemotherapy
Unsurprisingly, docetaxel trials have focused on high-risk BCR patients, given their higher risk for distant metastasis. Following a number of encouraging phase II trials,80 –82 two phase III trials have been reported on the use of docetaxel for BCR.
In the phase III TAX 3503 trial, 83 which was terminated early, 413 patients high-risk BCR patients (PSA-DT ⩽ 9 months and PSA ⩾ 1 ng/ml) were randomised to receive 18 months of ADT (leuprolide and bicalutamide), with or without, docetaxel (75 mg/m2 IV 3-weekly, 10 cycles). The final analysis included data from the trial and a subsequent registry created after completion of study accrual to secure the primary endpoint. At a median FU of 33.6 months, the study showed a trend towards improving median PFS (26.2 months versus 24.7 months, HR: 0.80, 95% CI 0.61–1.04, p = 0.09) and in OS for the intention-to-treat population (medians not reached, HR 0.51, 95% CI 0.23–1.10. p = 0.08), with good testosterone recovery in both arms.
Another phase III trial assessed the addition of docetaxel (70 mg/m2 IV 3-weekly, 6 cycles) to ADT in 254 patients with rising PSA and high-risk criteria after primary local therapy. 84 For inclusion in the trial, patients had to present with one or more of the following: node-positive adenocarcinoma, positive surgical margins, GS ⩾ 8, PSA velocity >0.75 ng/ml per year, PSA-DT ⩽ 6 months, and time to PSA recurrence ⩽12 months. At a median FU of 10.5 years, there was no significant difference in PSA-PFS (20.3 months versus 19.3 months, HR: 0.85, 95% CI: 0.62–1.16, p = 0.31), or radiologic PFS (8.9 years versus 9 years, HR: 1.03; 95% CI 0.74–1.43, p = 0.88), while the OS data was not mature (12-year survival rate 60% versus 55%, HR: 0.86, 95% CI 0.56–1.31, p = 0.49). The use of docetaxel had no significant effect on quality of life during the first 12 months of treatment.
A third phase III trial is currently ongoing (Clinicaltrials.gov identifier: NGR-GU002) assessing docetaxel in patients with persistently elevated PSA ⩾ 0.2 ng/ml (Clinicaltrials.gov identifier: NCT03070886). A number of phase II docetaxel-based combination trials have been reported, some of which demonstrated significant toxicity (Table 2).85 –87
It ought to be highlighted that the STAMPEDE trial allowed for previously treated relapsing non-metastatic patients with PSA progression only, although the reported numbers for previously treated M0 (which would include the pure BCR patients) were small both for the control (3%), docetaxel (2%), docetaxel/zoledronic acid (4%), and abiraterone acetate (3%) arms.88,89 The results for these PSA-relapse only patients have not been separately reported, so no relevant conclusions can be made.
Next-generation hormonal agents
Given the impressive impact next-generation hormonal agents (NHA) have had in the mCSPC and mCRPC settings, it is not surprising that there would be attempts to see whether the benefit could extend to their use in the much earlier stage of BCR.
No phase III trial evidence is reported yet. The ongoing 3-arm randomised EMBARK trial (enzalutamide plus leuprolide versus placebo plus leuprolide versus enzalutamide monotherapy) in BCR patients with high-risk features (PSA-DT ⩽ 9 months, PSA ⩾ 2.0 ng/ml post-RP, and ⩾5.0 ng/ml and ⩾nadir + 2.0 ng/ml post-RT) has completed accrual, with results pending. 90
The ongoing EORTC 1532 is a randomised phase II trial evaluating the efficacy of darolutamide versus ADT in men with asymptomatic hormone-naïve PC (non-metastatic or up to 4 non-visceral lesions) and a PSA ⩾ 2 ng/dl (Clinicaltrial.gov identifier: NCT02972060). The hypothesis of this study is that AR antagonists may have a similar efficacy in reducing PSA compared to ADT, with a better tolerability due to the maintenance of normal systemic testosterone levels. The primary endpoint is PSA response at 24 weeks.
The phase II STREAM trial assessed the combination of prostate bed RT (66 Gy), 6 months of ADT and enzalutamide in patients with a GP ⩾ 7, and a PSA recurrence within 4 years of RP, with PSA levels of 0.2–4.0 ng/ml. In a cohort of 38 men, the combination had a good safety profile, without any grade 4/5 or unexpected toxicities. The primary endpoint of 2-year PFS was 65% (95% CI: 47, 78) versus 51% (95% CI: 33, 67) for historic controls. PSA remained at undetectable levels in 69% at 2 years, and the 3-year PFS was 53% (95% CI 37, 68). 91
The ongoing randomised phase II SALV-ENZA trial is designed to assess the combination of salvage prostate bed RT (66.6–70.2 Gy) with 6 months of enzalutamide versus placebo in high-risk BCR patients (GS ⩾ 8 and either pT3 or positive margins). 92 Although it is important that this is a randomised trial, the standard arm omits ADT, which is a de-escalation compared to current standards, evoking the assumption that enzalutamide would sufficiently replace ADT. In addition, the trial includes only patients with PSA up to 0.7 ng/ml, which, based on previous evidence, likely excludes patients with higher PSA levels that are more likely to benefit from a combinatorial approach to RT, notably with ADT.
The ongoing randomised RTOG3506 (STEEL) trial also assesses the addition of 2 years of enzalutamide to salvage RT and 2 years of ADT and stratifies for high-risk factors (GS 8-10, locoregional node involvement at RP, seminal vesicle invasion, persistently elevated PSA after RP, and PSA > 0.7 ng/ml). 93
The phase I CARLHA-GEP1294 trial assessed the combination of RT with 6 months of abiraterone acetate with ADT in patients in BCR post-RP. Abiraterone was started 1 month prior to RT, while ADT was started either together with RT or 1 month prior. The addition of AA did not increase pelvic toxicity, however showed increased G3 liver toxicity at the standard dose of 1000 mg daily. The phase II trial is ongoing at the recommended dose of 750 mg daily.
The South American phase II LACOG-0415 is a trial that assessed two NHA agents, abiraterone acetate and apalutamide, in a 3-arm randomised fashion (apalutamide alone versus apalutamide and abiraterone versus abiraterone and ADT) in patients in BCR (PSA ⩾ 4 ng/dl and PSA-DT < 10 months) post-RP or RT (n = 22 patients), as well as in a node-positive patient cohort (not candidate to local therapy) and an mCSPC cohort. The hypothesis driving the study was that ADT-free alternatives with the use of NHA could provide high efficacy with a favorable safety profile in patients with advanced CSPC. In the total cohort of 128 patients, the double NHA combination as well as the abiraterone/ADT combination achieved high efficacy in terms of the primary endpoint of PSA decline of ⩽0.2 ng/ml at week 25 [70.5% (95% CI: 54.8–83.2%) and 73.8% % (95% CI: 58.0–86.1%) respectively], while apalutamide monotherapy did not [57.1% (95% CI: 41.0–72.3%)]. Radiologic disease control and PSA decline of ⩾80% and ⩾50% at week 25 were similar amongst treatment arms. Interestingly, there were no differences in quality of life between the 3 arms. No results per sub-cohort were presented; it is therefore not possible to know how the results translate for the BCR sub-cohort, which accounted for only 17.2% of the total cohort. 95
The ongoing randomised phase II FORMULA-509 trial assesses the addition of the same combination (abiraterone and apalutamide) to salvage RT and ADT for high-risk BCR patients such as PSA ⩾ 0.7 ng/ml, pathologic N+ disease, or numerous adverse risk factors (e.g., pT3b-T4, primary pattern 5 disease) (Clinicaltrials.gov identifier: NCT03141671).
The phase II BALANCE (Clinicaltrials.gov identifier: NRG GU-006) trial assesses in a placebo-controlled fashion the addition of 6 months apalutamide to salvage RT in high-risk BCR patients, including patients that never negativized their PSA post-RP. As the trial does not allow for ADT, patients with PSA > 1 ng/ml are excluded, aiming to use PSA as a biomarker for the use of NHA (Clinicaltrial.gov identifier: NCT03371719).
The ongoing phase II SPARTAR trial, designed by the team that performed the STREAM trial, assesses the addition of both an NHA and chemotherapy to a hormone-radiation in the high-risk BCR setting (relapse within 4 years of RP, GS ⩾ 8 or GS 7 with high-risk features such as pT3, N+ or positive margins). Trial treatment consists of ADT and apalutamide 120 mg daily, concurrently with RT followed by 6 cycles of 3-weekly docetaxel 75 mg/m2. The ADT/apalutamide treatment is scheduled to continue for 36 weeks or until unacceptable toxicity or disease progression, and the study. The trial primary endpoint is 3-year PFS, with a hypothesis of improving it to 75% from a historic 50%. 96
Immunotherapy
Vaccines
Following on evidence from a smaller phase II trial, 97 the randomised PROTECT phase III trial assessed the use of sipuleucel T in 176 patients with rising PSA post-RP. Although sipuleucel achieved a 48% increase in PSA-DT (p = 0.038), the median time to biochemical failure, the study primary endpoint, did not differ between the two arms (18 months versus 15.4 months, HR: 0.936, 95% CI 0.637–1.376, p = 0.737). 98 Although the study indicated biological activity for sipuleucel T in this setting, including documenting robust and sustained induced immune responses, a much larger cohort number and more robust endpoint with longer FU would be needed to accurately assess the potential of this cellular immunotherapy modality for BCR.
Several early phase trials involving TAA-based vaccines, including the poxviral Prostvac vaccines and PSMA vaccines, have reported moderate anti-tumour specific responses and prolongation in PSA-DT (Table 2).99 –105 Furthermore, the adenovirus/PSA vaccine APP21 is currently assessed as a monotherapy versus combination with ADT in a randomised phase II trial (Clinicaltrials.gov identifier: NCT00583752).
Checkpoint inhibition
Some steps have also been made in the direction of using checkpoint inhibition in BCR. The anti-CTLA4 tremelimumab was assessed in combination with short-term (6 months) bicalutamide treatment in a phase I trial, reporting G3 diarrhoea and rash as dose-limiting toxicities. In 3 out of 11 patients (27%) delayed prolongation of PSA-DT was observed several months after completing treatment. 106 Nivolumab monotherapy for up to 2 years is currently assessed in high-risk BCR patients with PSA-DT < 10 months (Clinicaltrials.gov identifier: NCT03637543).
An ongoing trial assesses the use of nivolumab monotherapy in patients with MMR-deficient/MSI-high and PC with rising PSA (Clinicaltrials.gov identifier: NCT04019964). The trial also includes patients with high tumour mutational burden (>20 mut/Mb) and CDK12-alterations.
Immunotherapy combination trials
A currently ongoing phase II trial (Clinicaltrials.gov identifier: NCT03315871) involves the use of the Prostvac recombinant vaccine in biochemically recurrent PC, in combination with bintrafusp alpha, a bifunctional anti-PD-L1/anti-TGFbetaRII fusion protein in biochemically recurrent PC patients, as well as CV301, a poxviral based TAA vaccine targeting MUC1 and CEA. A combination of the pTVG-HP vaccine with pembrolizumab is also currently ongoing (Clinicaltrials.gov identifier: NCT03600350).
Recently, a phase I/II trial of a combination synthetic DNA therapy of plasmids encoding for PSA and PSMA (INO-5150) and interleukin-12 (INO-9012) in 62 patients with rising PSA reported a good safety profile, with immunogenicity observed in 76% and 85% of patients remaining progression-free at 72 weeks. 107
Targeted therapy and other agents
PARP inhibitors
The PARP inhibitor olaparib is currently evaluated in a phase II trial designed to test the hypothesis that PARPi monotherapy may be active in men with high-risk BCR. The enrichment stage of the trial includes confirmation of presence of a mutation in a gene of the DNA repair pathway. In the reported interim results of the trial, olaparib without ADT showed a satisfactory tolerance profile and interesting activity. It should be noted that 35% of men had a BRCA/ATM alteration, 15% of patients showed a PSA50 response, including two complete PSA responses (all of whom had a BRCA2 mutation), another 20% had minor PSA responses, and median PSA progression-free survival was greater in men with versus without BRCA2/ATM muts (9 versus 4 mo; p = 0.02). 108
The phase II ROAR trial (Clinicaltrials.gov identifier: NCT03533946) evaluates rucaparib in the same setting, requiring a BRCAness signature tested on liquid or soft tissue biopsy, with a primary endpoint of PSA50 response.
A combination of olaparib with the anti-PDL1 agent durvalumab is also evaluated in a phase II trial on BCR patients with alterations in the DNA repair pathway genes (Clinicaltrials.gov identifier: NCT03810105).
Antiangiogenics
A number of anti-angiogenic drugs, including bevacizumab, sunitinib, and lenalidomide have been investigated in phase II trials with moderate results, alone or in the context of multimodality treatment (Table 2).86,87,109 –111 The only phase III trial involved thalidomide, an anti-angiogenic drug with immunomodulatory properties, has been tested in a placebo-controlled phase III trial in association with ADT with a crossover phase, showing an effect on time to further PSA progression (17.1 months versus 6.6 months, p = 0.0002). 112
PI3K/Akt and metabolic pathways
Two phase II trials involving AKT inhibitors have failed to show any meaningful activity as monotherapy or in combination with anti-androgen therapy (Table 2).113,114
Cox inhibitors, notably celecoxib which also inhibits activation of Akt by phosphorylation in PC cells, have also been evaluated in phase II studies showing significant effect on PSA-DT but conflicting results in terms of toxicity (Table 2).115,116
The combination of atorvastatin and celecoxib was also investigated (Table 2). 117 A meta-analysis on the use of statins in prostate cancer found it was associated with a 21% reduction in the risk of BCR among those treated with RT (HR: 0.79, p = 0.01, 10 studies), whereas it was not associated with BCR among those treated with RP (HR: 0.94, 95% CI 0.81–1.09, p = 0.43, 15 studies). In the overall cohort, statin use was associated with a 22% reduction in the risk of metastasis, and a 24% reduction in risk of both all-cause and prostate cancer-specific mortality. 118
Vitamin supplementation has also been investigated in several early trials (monotherapy or combinations) with moderate results (Table 2).119 –123 Finally, in a meta-analysis of 5 retrospective studies, metformin use was marginally associated with reduction in the risk of BCR (HR: 0.82, 05% CI: 0.67–1.01, p = 0.06). 124
Other targeted therapies
A number of phase II trials evaluated difference targeted therapies on pathways involved in prostate cancer development, with results ranging from complete lack of efficacy in term of PSA response to modest improvements to PSA-DT (Table 2).125,126
Discussion
Given the disease evolution of PC, especially with the introduction of new treatments in the mCSPC, nmCRPC and mCRPC settings, a sufficiently long-life expectancy is necessary for BCR to influence mortality. As a result, trials with follow-up longer than 10 years are needed to accurately assess the impact of systemic treatment in BCR. Furthermore, any survival gain should be balanced against the side-effects of treatment and the effects on overall quality of life in these men that feature a long disease-specific expectancy at the time of BCR diagnosis. It is therefore important, when considering initial systemic therapy for BCR, to optimize the timing of therapy and patient selection.
The so far available phase III evidence indicates that early salvage RT provides benefit which is greater at lower PSA levels. In contrast, the absolute benefit from the addition of hormone therapy in such low PSA levels would be less. Higher PSA levels are likely to be associated with increased probability of detecting metastatic disease, notably distant, with molecular imaging. It transpires those patients with higher PSA levels may preferentially derive treatment benefit from hormone therapy for occult distant metastatic disease. It has become progressively clear, therefore, that not all biochemically relapsed men need systemic treatment.
In this direction, the current ESMO guidelines advise that early ADT alone is not recommended for men with BCR unless they have a rapid PSA-DT, symptomatic local disease, or proven metastases and that, in the absence of metastatic disease, such men should be offered intermittent rather than continuous treatment. 127 The cut-off for rapid PSA-DT is not precisely determined nor universally applied. EAU guidelines use a 12-month cut-off to indicate high-risk BCR patients, 19 trials have used a range of 5–15 months, and it has been clearly demonstrated that a PSA-DT of <3 months indicates a poor prognosis and increased risk for metastatic disease.
The absolute PSA value at the time of BCR has gained more evidence for its value as prognostic and predictive factor. The post-hoc analysis of the RTOG 9601 trial probably provides the strongest available evidence in this direction, indicating a lack of benefit from hormonal therapy for PSA levels <0.6 ng/ml, with increased associated toxicity and, conversely, a 12-year absolute benefit of 25% in patients with PSA > 1.5 ng/ml, compared to the 5% absolute benefit in the overall study cohort.71,72 It seems, therefore, that we are starting to sieve through this large space of BCR, making a distinction between those patients for which salvage RT alone would be sufficient as standard treatment, and those who require hormonal therapy, short- or long-term.
When considering docetaxel, the two reported phase III trials reported on PFS, with one failing to show benefit, and the other showing borderline benefit, whilst OS data were not mature in the former and not reported in the latter.83,84 Both studies aimed to include high-risk BCR patients, but their criteria for this were not identical, and they also differed in the docetaxel regimen and number of cycles. This is reminiscent of the landscape of docetaxel trials for locally advanced, non-metastatic disease, with contradicting results amongst trials that differed in the definition of high-risk disease, docetaxel regimen, accrual power and timing of treatment, and failure to show OS survival benefit despite improvements in failure-free survival. 128
Table 1 summarizes the current evidence from phase III and leading phase II trials, as well as the current consensus recommendations based on ESMO and EAU guidelines. Table 2 summarizes the historical trial data for agents that are no longer under active investigation. Currently, international consensus recommends the addition of ADT if high-risk features, such as PSA over a given cut-off (varying between 0.5 and 0.7 ng/ml),129,130 short PSA-DT, symptoms, or macroscopic disease. Regarding duration of this ‘adjuvant’ ADT, while awaiting the results of the RADICALS-HT trial, the best available evidence comes from GETUG-16, in the form of a short-term (6 months) LHRH analogue treatment. The future phase III studies investigating NHAs will undoubtedly shape the field and are eagerly awaited. An interesting angle on the design of available phase II NHA trials is that they looked into the monotherapy option, aiming to assess where ADT use can be spared with (although initial phase II data are rather disappointing), as well as into combination options.
Summary of current phase III evidence and principal phase II trial data on systemic treatment for BCR.

ADT, androgen deprivation therapy; BCR, biochemical recurrence; HR, hazard ratio; mo, months; MFS, metastasis-free survival; OS, overall survival; PC, prostate cancer; PFS, progression-free survival; pGS, pathological Gleason score; PLND, pelvic lymph node dissection; PSA-DT, prostate-specific antigen doubling time; RP, radical prostatectomy; RT, radiotherapy; WPRT, whole pelvis radiotherapy.
Summary of historical early phase trial data on systemic treatment for BCR.
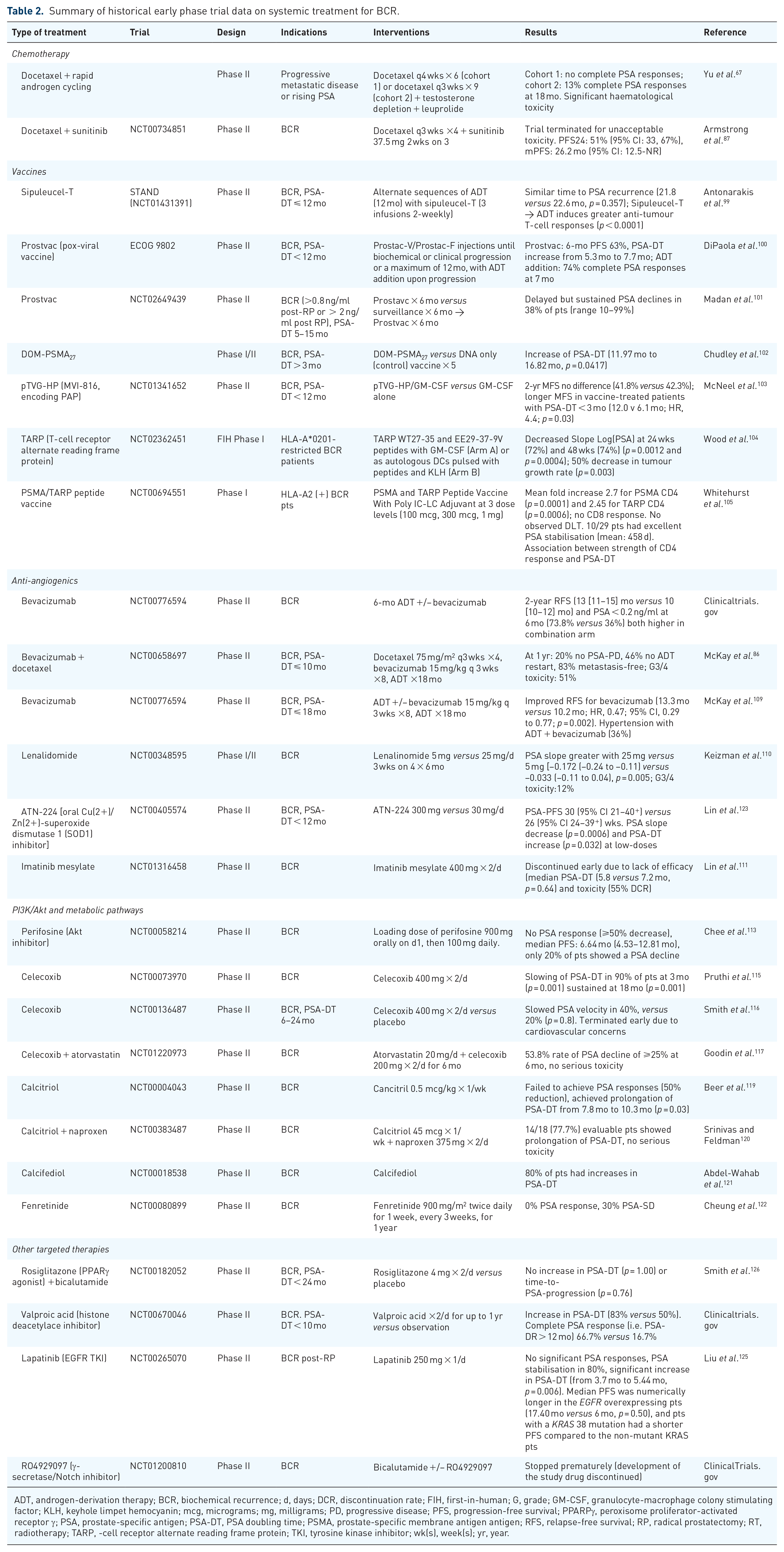
ADT, androgen-derivation therapy; BCR, biochemical recurrence; d, days; DCR, discontinuation rate; FIH, first-in-human; G, grade; GM-CSF, granulocyte-macrophage colony stimulating factor; KLH, keyhole limpet hemocyanin; mcg, micrograms; mg, milligrams; PD, progressive disease; PFS, progression-free survival; PPARPγ, peroxisome proliferator-activated receptor γ; PSA, prostate-specific antigen; PSA-DT, PSA doubling time; PSMA, prostate-specific membrane antigen antigen; RFS, relapse-free survival; RP, radical prostatectomy; RT, radiotherapy; TARP, -cell receptor alternate reading frame protein; TKI, tyrosine kinase inhibitor; wk(s), week(s); yr, year.
Conclusion
Because of the heterogeneous and dynamically evolving BCR stage, and in order for future studies to be more informative and more efficient in directing clinical practice, patients ought to be ideally stratified per high-risk features such as PSA levels and PSA-DT, as well as according to primary treatment modality (RP versus RT). As more sophisticated molecular techniques enter the arena, it is possible to envisage a future for biochemically relapsed PC where liquid biopsies would provide a dynamic molecular mapping that would guide patient selection, optimal treatment modality, and optimal treatment timing.
Footnotes
Conflict of interest statement
KK: Participation to advisory boards for: Amgen, Astellas, Astrazeneca, AAA, Bayer, Clovis, Curevac, ESSA, Genentech, Janssen, MSD, Orion, Sanofi. Honoraria are provided to Gustave Roussy, my institution. TZ: Honoraria (to institution)/travel grants — Janssen, Amgen, Ferring, Debiopharm, Bayer, Astellas. Research Grants — Varian Medical Systems GB: Advisory boards and symposia: Amgen, Janssen Oncology, Sanofi, Astellas-Pharma, Roche, Bayer, Genesta. Travel accomodations, expenses: Amgen, Astellas-Pharma, Astra Zeneca, Ipsen, Janssen Oncology, Sanofi AP: advisory boards: Basilea, Congress participation: Amgen
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
