Abstract
Purpose:
To assess the efficacy and safety of neoadjuvant docetaxel + cisplatin chemotherapy with androgen deprivation therapy for the treatment of locally advanced prostate cancer (PCa) in patients harboring germline DNA damage repair genes (gDDR) defects.
Methods:
We conducted a prospective observational study in patients with locally advanced PCa confirmed with gDDR defects through next-generation sequencing. All patients received either docetaxel + cisplatin (platinum-group) or docetaxel chemo-hormonal therapy (docetaxel group) followed by radical prostatectomy with extended lymphadenectomy. The primary end point was biochemical progression-free survival (bPFS) and secondary end points include postoperative pathological response and safety assessment during the study period.
Results:
A total of 36 patients were included in the study, among whom 14 and 22 patients received docetaxel + cisplatin and docetaxel treatment, respectively. Down-staging of Tumor (T), Nodes (N), and Metastasis (M) stages was observed in 11 (78.57%) and 9 (40.9%) patients (p = 0.041), respectively, in the docetaxel + cisplatin group and docetaxel group. The median bPFS was 7.76 months (95% CI 0.770–14.748) and not reached in the docetaxel group and docetaxel + cisplatin group, respectively. bPFS was significantly longer in the docetaxel + cisplatin group (p = 0.039) with a hazard ratio of 0.386 (95% CI 0.151–0.987, p < 0.05). Furthermore, one patient discontinued docetaxel + cisplatin after second cycle due to severe liver insufficiency which was confirmed as viral hepatitis A and no significant perioperative complications was observed in either group.
Conclusion:
This study suggests that cisplatin may increase docetaxel anticancer activity with tolerable safety profile in patients with locally advanced PCa carrying gDDR defects in the neoadjuvant setting, a hypothesis which will require prospective, randomized confirmation.
Keywords
Introduction
Prostate cancer (PCa) represents the most common cancer in men and patients with locally advanced PCa are at high risk for regional and distant relapse after radical prostatectomy (RP). 1 Hence, the management of locally advanced PCa remains a clinical challenge. Neoadjuvant chemo-hormonal therapy (involving docetaxel in addition to androgen deprivation therapy (ADT)) combined with RP has been reported to achieve pathological response and prolong biochemical progression-free survival (bPFS) in locally advanced PCa patients.2,3 However, the optimal neoadjuvant therapy regimen remains inconclusive, and biomarkers predicting the efficacy of neoadjuvant therapy are urgently needed.
With the advancement of next-generation sequencing (NGS) technology, the prevalence of germline DNA damage repair genes (gDDR) defects in treatment-naïve PCa has been revealed. Deleterious gDDR mutations are found in 11.8% of patients with metastatic PCa 4 and 4.6% with primary localized PCa. 5 Inadequacy in DNA repair due to DDR defects may cause genetic alterations leading to loss of tumor suppressor genes which in turn activates oncogenes and ultimately trigger the development of malignant cells or increases the aggressiveness of tumor cells. Therefore, DDR defects play a critical role in the etiology and progression of PCa. 6
Although not recommended as the standard of care in PCa treatment, platinum-based chemotherapy has demonstrated promising therapeutic effects in the treatment of breast and ovarian cancers with DDR defects, especially in the neoadjuvant setting.7,8,9 Further, the combination of platinum and taxane chemotherapy has been reported to show clinical activity in advanced prostate cancer.10,11 In addition, with emerging data on prevalence of somatic and germline DDR defects in PCa patients, a recent study evaluated the role for platinum-based treatment in molecularly selected patients with advanced PCa and reported that subgroup of patients with DDR defects seemed to derive higher response rates with platinum-based combination treatment (taxanes) than platinum-based monotherapy. 12 Therefore, we hypothesized that locally advanced PCa patients with gDDR defects may benefit from neoadjuvant docetaxel + cisplatin (platinum group) chemo-hormonal therapy and conducted this prospective observational study to compare the efficacy of neoadjuvant docetaxel + cisplatin chemo-hormonal therapy with docetaxel chemo-hormonal therapy in this cohort.
Methodology
Study design and population
In this prospective observational cohort study, patients with locally advanced PCa who were confirmed with gDDR defects and subjected to either docetaxel + cisplatin (platinum group) or docetaxel neoadjuvant chemo-hormonal therapy (docetaxel group) from May 1, 2017, to December 31, 2019, at the Renji Hospital, Shanghai Jiao Tong University, School of Medicine, Shanghai, China, were enrolled (first patient enrolled on May 4, 2017). The choice of the treatment regimen was based on the physicians’ clinical judgment and the patients’ willingness. The study was registered in the Chinese clinical trial registry (Study ID: ChiCTR 2200055704). (URL: http://www.chictr.org.cn/showproj.aspx?Proj=149452). In this article, we report interim analysis results with last follow-up data till August 31, 2021.
Inclusion and exclusion criteria
Male patients aged 18–75 years, histologically or cytologically confirmed to have prostate adenocarcinoma, carrying gDDR (ATR, BLM, ERCC2, ERCC3, ERCC4, ERCC5, FAM175A, FANCA, FANCC, FANCD2, GEN1, HDAC2, MDC1, MUTYH, MRE11, NBN, PARP1, POLD1, POLE, RAD50, RAD51, RECQL4, XRCC4, MLH1, MLH3, MSH2, MSH6, EPCAM, PMS1, PMS2, ATM, BARD1, BRIP1, BRCA1, BRCA2, CDK12, CHEK1, CHEK2, FANCL, PALB2, RAD51B, RAD51C, RAD51D, RAD54L, PPP2R2A) defects detected through NGS with peripheral blood collected at diagnosis, were considered eligible if clinical stage T3a-T4, N0, M0; any T, N1, M0, confirmed by pelvic multiparametric MRI and PSMA PET/CT; was addressed to either docetaxel + cisplatin or docetaxel neoadjuvant chemo-hormonal therapy.
Patients were excluded if the pathology results indicate neuroendocrine PCa including small cell carcinoma; patients with a history of other malignancies; patients with any contraindications to docetaxel or cisplatin use; patients who participated in other clinical studies within a month before the first use of chemotherapy and other circumstances considered inappropriate by investigator.
NGS for assessment of mutations in DDR loci
Initially, with a threshold of minimum coverage of 50× and an allele frequency of over 30%, germline variants called by Genome Analysis Toolkit on white blood cell samples were filtered. Following this, variants that are not on coding regions and synonymous mutations were annotated using Annotate Variation (ANNOVAR) and filtered out. Furthermore, variants with a population minor allele frequency over 0.1% (annotated using the ExAC database) were considered less functional. A log2 ratio more than 0.6 was considered as a copy gain. A log2 ratio less than −0.7 was considered as a copy loss.
Treatment
Patients in both the treatment arms (docetaxel and docetaxel + cisplatin) received continual subcutaneous injection of leuprorelin/goserelin as ADT and intravenous infusion of docetaxel at the dose of 75mg/m2, whereas patients in the docetaxel + cisplatin arm received 75mg/m2 of cisplatin intravenously in addition to docetaxel and ADT for every 3 weeks for a total of four to six cycles. Chemotherapy was commenced within 1 month after the initiation of ADT. After completion of chemotherapy as planned, ADT was discontinued for 2 months before RP with extended lymphadenectomy was performed. ADT was not continued after RP until PSA or clinical recurrence.
Outcomes and end points
The primary efficacy end point was bPFS, defined as the duration from the date of RP to the date of first confirmed PSA recurrence. Secondary end points include postoperative pathological response and safety assessment during the study period. Pathological downstaging was confirmed if the postoperative pathological T stage was lower than the clinical T stage at baseline while the pathological N stage did not rise comparing to the clinical N stage. 13 Pathological complete response was defined as the absence of disease in all removed specimen including the prostate and lymph nodes under pathologic examination. 14
Statistical analysis
The NGS results are represented as heatmap plot and quantitative variables (age, operation time) were represented using values of mean and standard deviation, whereas baseline PSA values and amount of bleeding were represented in median and interquartile range (IQR) as data was not normally distributed. Categorical variables were described in percentages. Proportions of the variables were analyzed using chi-square test or Fisher’s exact test while nonparametric variables were analyzed using Mann–Whitney rank sum test. p < 0.05 was considered as statistically significant. bPFS was assessed by the Kaplan–Meier method, and the median time of bPFS along hazards ratio (HR) and 95% confidence interval (CI) for the comparison of the two treatment groups were provided.
Results
Baseline characteristics
A total of 36 patients were included in the study, among whom 14 and 22 patients received docetaxel + cisplatin and docetaxel treatment, respectively (Figure 1). Patients in the docetaxel + cisplatin group were presented with significantly higher cT stages at initial diagnoses (p = 0.011). There was no significant difference among the other baseline variables between the groups, including age, Gleason score, and primary PSA level (Table 1). The gDDR mutation landscape in the two groups is provided in Figure 2. All the patients in both groups harbored gDDR defects.

Patient disposition flow chart.
Baseline clinical characteristics.

IQR, interquartile range; PSA, prostate-specific antigen.
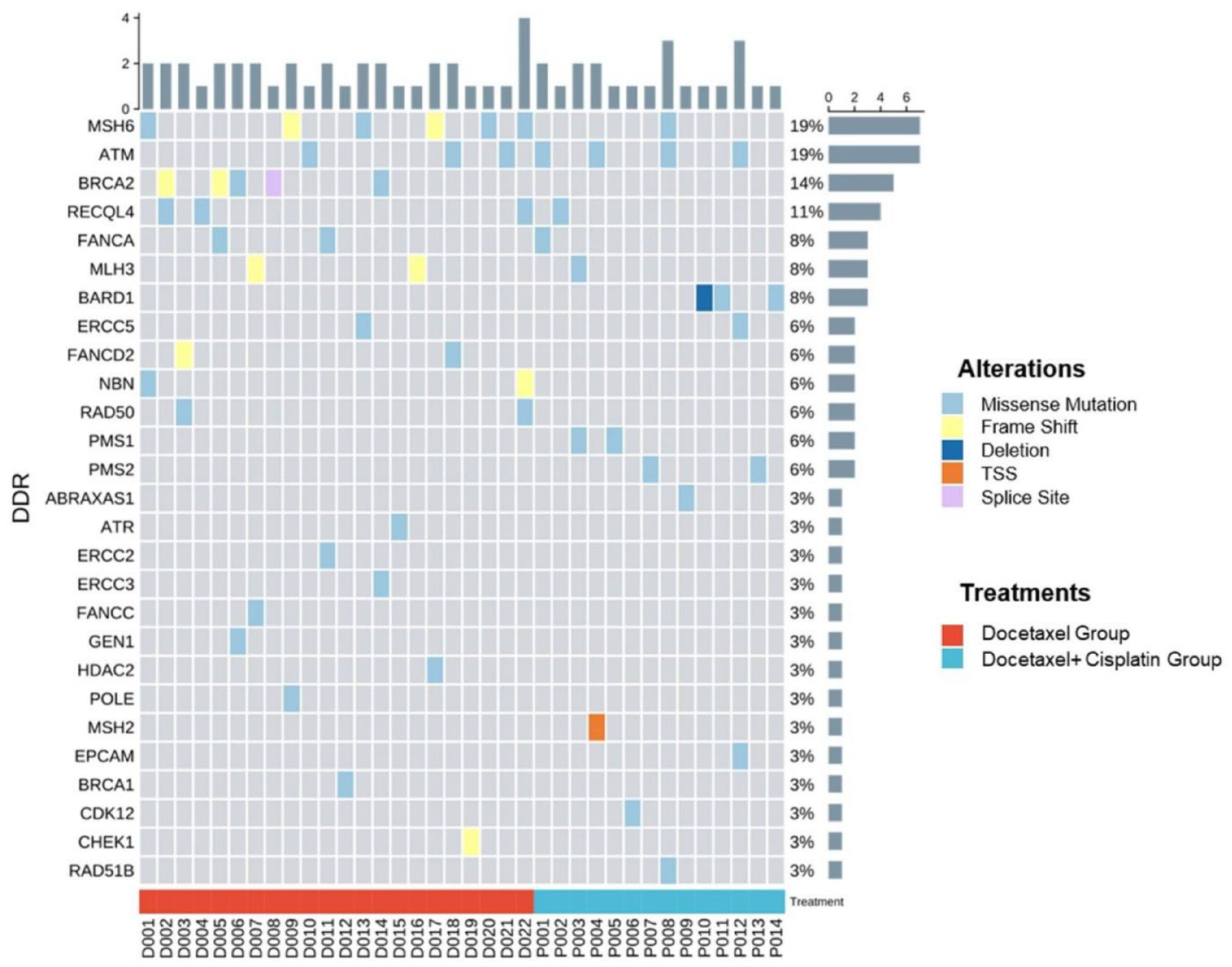
Germline DDR gene alterations.
In the docetaxel + cisplatin group, six and seven patients underwent a total of four and six cycles, respectively, while one patient terminated chemotherapy treatment after two cycles due to severe liver insufficiency caused by hepatitis A. In the docetaxel group, 12 and 10 patients underwent a total four and six cycles, respectively.
Efficacy of docetaxel + cisplatin treatment regimens
Pathological downstaging was observed in 11 (78.57%) and 9 (40.9%) patients in the docetaxel + cisplatin and the docetaxel group, respectively. The rate of pathological downstaging was significantly higher in the docetaxel + cisplatin treatment group (p = 0.041). An uprise in TNM stages was observed in four (18.18%) patients in the docetaxel group but none in the docetaxel + cisplatin group (p = 0.141) (Table 2, Figure 3). Pathological complete response (pT0) was observed in 4/14 (supplementary Figure 1) and 2/22 patients in docetaxel + cisplatin and docetaxel group, respectively.
Postoperative clinical characteristics.
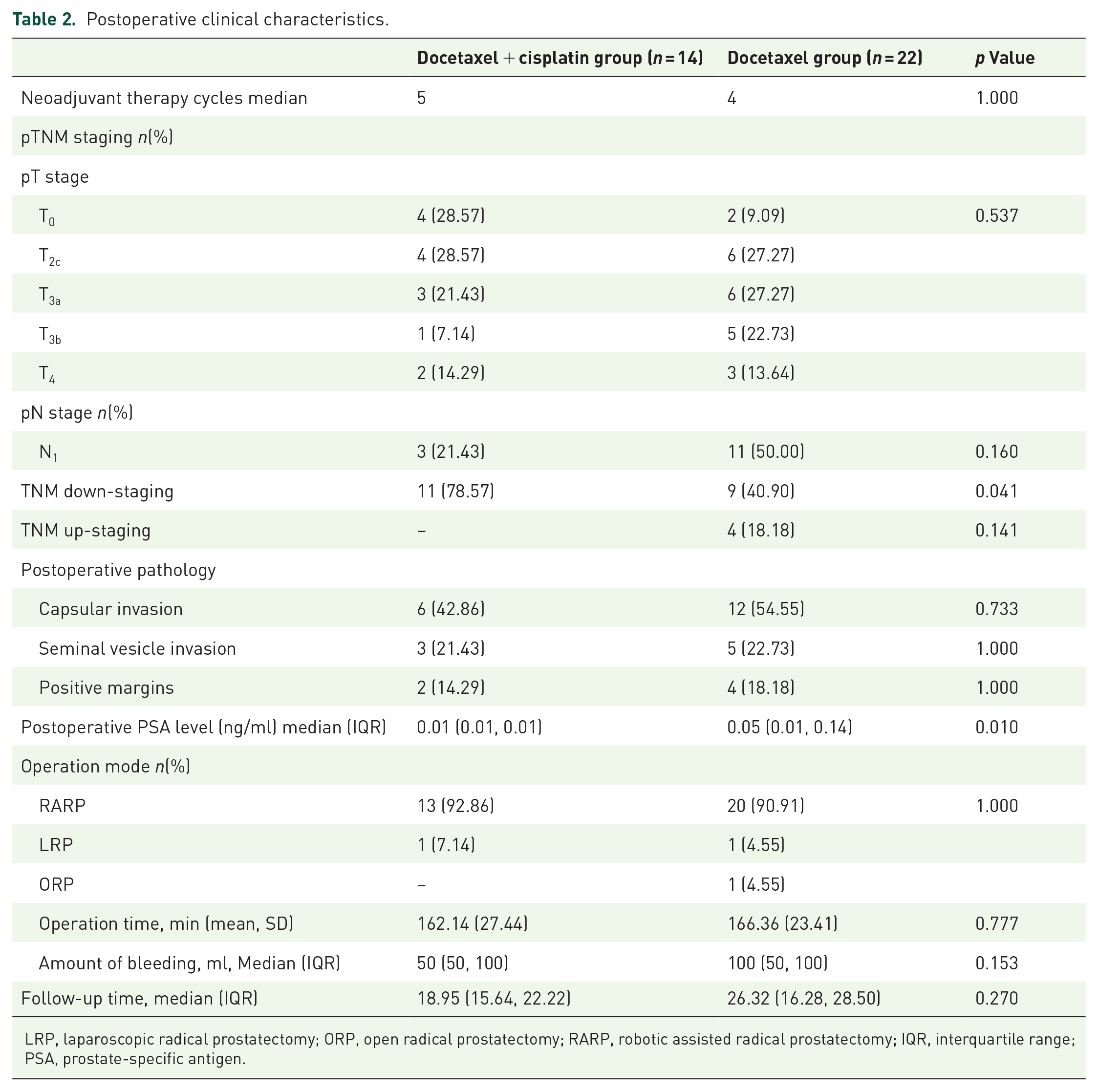
LRP, laparoscopic radical prostatectomy; ORP, open radical prostatectomy; RARP, robotic assisted radical prostatectomy; IQR, interquartile range; PSA, prostate-specific antigen.

Clinical and pathologic T stage in the docetaxel + cisplatin group and docetaxel group.
Apart from postoperative pathology, parameters at the time of RP were not significantly different between the two treatment groups. Operative parameters including operation mode, operation duration, and blood loss showed no difference between the two groups (Table 2). Residual postoperative positive margins were observed in two (14.29%) patients in the docetaxel + cisplatin group and four (18.18%) patients in the docetaxel group. Logistic regression analysis revealed only treatment regimen (docetaxel + cisplatin versus. docetaxel) to be significantly associated with pathological downstaging (odds ratio: 5.296, 95% CI 1.143–24.548, p = 0.033). Other factors including age, preoperative PSA, primary PSA, biopsy Gleason score, clinical TNM stage, and number of chemotherapy cycles were not associated with pathological downstaging (Supplementary Table 1).
Biochemical progression free survival
Median follow-up time for the docetaxel group was 26.32 months (IQR 16.28, 28.50), while median follow-up time for the docetaxel + cisplatin group was 18.95 (IQR 15.64, 22.22). Follow-up time of the two groups showed no significant difference. At follow-up, 6/14 patients in the docetaxel + cisplatin group and 17/22 patients in the docetaxel group developed biochemical recurrence. Median bPFS for the docetaxel group was 7.76 months (95% CI 0.770–14.748) while median bPFS for the docetaxel + cisplatin group was not reached. bPFS was significantly longer in the docetaxel + cisplatin group (p = 0.039). Cox regression analysis revealed postoperative PSA to be a prognostic factor for bPFS (p < 0.05), and the addition of platinum treatment significantly reduced the risk of biochemical progression after surgery (HR: 0.386, 95% CI 0.151–0.987, p < 0.05, Figure 4). All the other factors including age, preoperative Gleason score, preoperative PSA, primary PSA, clinical TNM stage, margin status, and number of chemotherapy cycles were not associated with bPFS (Supplementary Table 2).
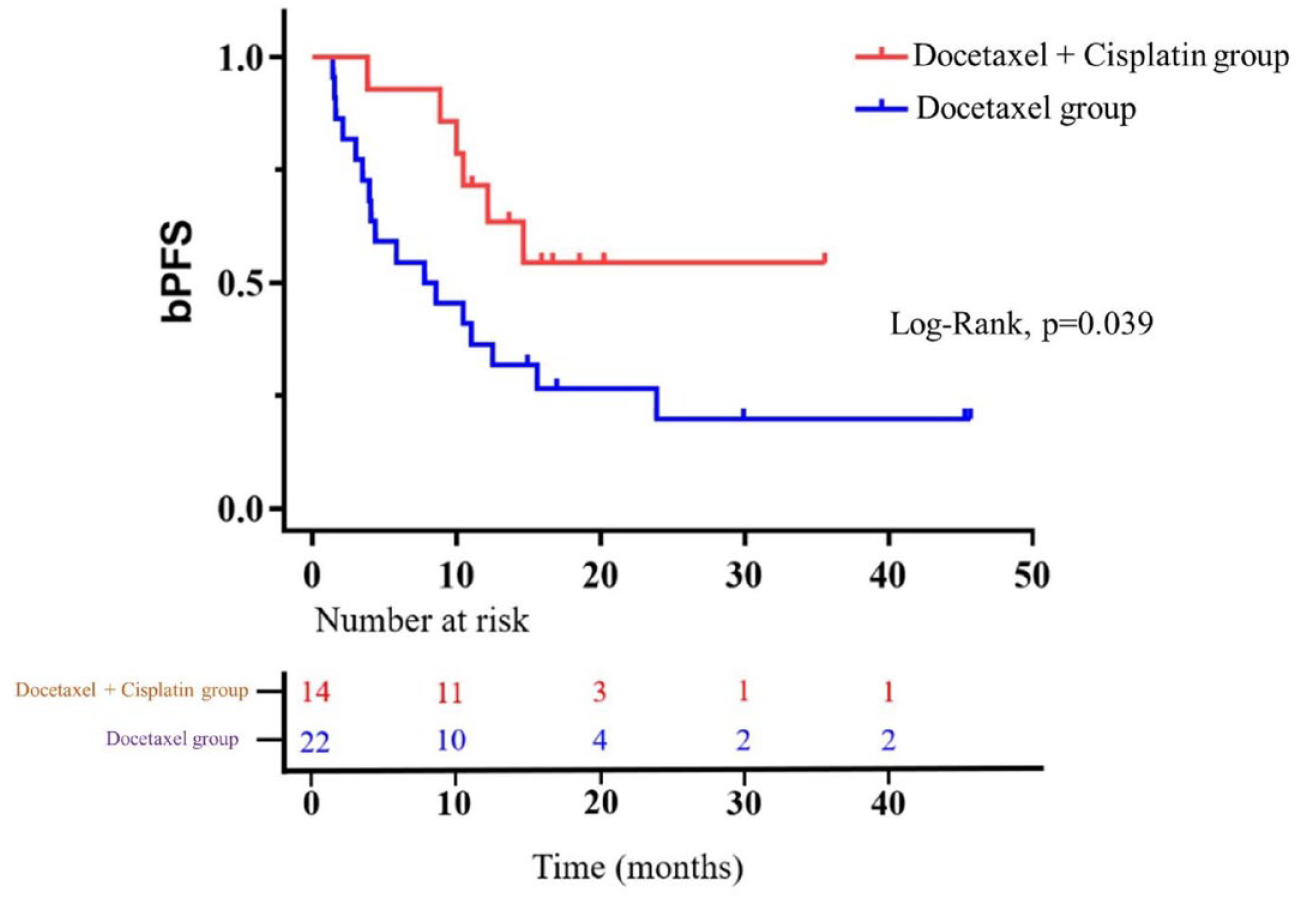
Biochemical progression-free survival.
Safety
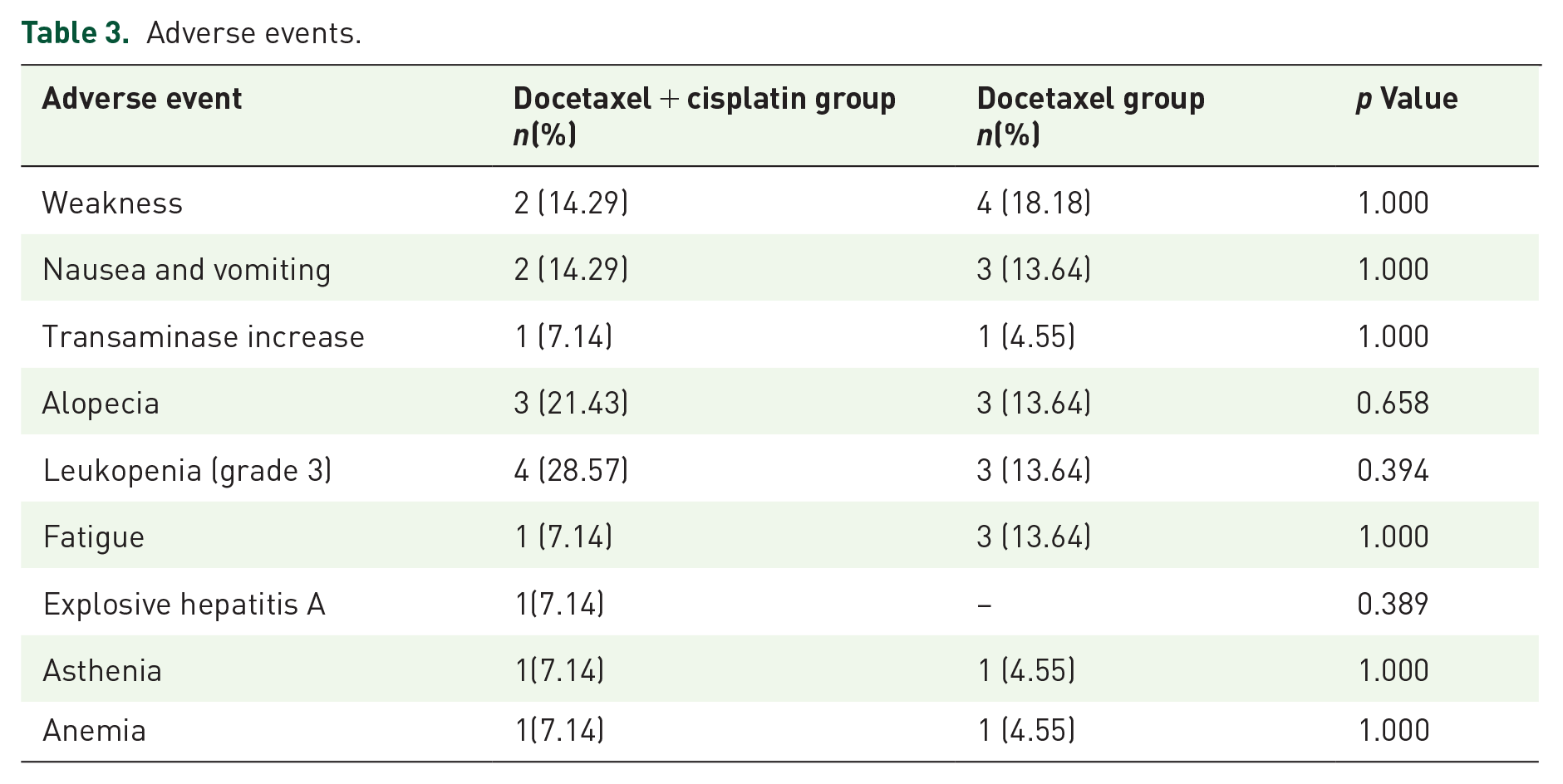
The major adverse events observed were grade 3 leukopenia (four patients in docetaxel + cisplatin and three in docetaxel groups), alopecia (three patients each in docetaxel + cisplatin and docetaxel group), weakness (two patients in docetaxel + cisplatin and four in docetaxel group), and nausea and vomiting (two patients in docetaxel + cisplatin and three in docetaxel group). The other adverse events are provided in Table 3. None of the patients in either treatment group experienced any perioperative complications.
Adverse events.
Discussion
In the current study, we focused on the cohort of locally advanced PCa patients with gDDR defects. Recently, DNA repair deficiency has been recognized as a potential driver of PCa. 15 Wei et al. 16 conducted a sequencing study to clarify the landscape of gDDR mutations in Chinese PCa patients and revealed that 53% of newly diagnosed high to very high risk PCa patients carried gDDR variants of unknown significance, while 8.1% carried deleterious gDDR defects.The clinical significance of gDDR defects in PCa has not yet been completely understood, but associations have been found with adverse clinicopathological characteristics and unfavorable prognosis. Leongamornlert et al. 17 reported PCa patients with gDDR loss-of-function mutations present more advanced disease stages. Germline BRCA mutations have been shown to be highly relevant to nodal involvement, distant metastasis, and poor survival outcomes in PCa patients. 18 For localized/locally advanced PCa patients treated with radical treatment, germline BRCA mutation carriers demonstrated significantly worse 3-, 5-, and 10-year cause-specific survival rates. 19 As for metastatic hormone-sensitive PCa patients, gDDR defects carriers show shorter time to castration resistance. 20 Therefore, more studies are needed to explore the clinical features and optimal treatment of primary PCa patients carrying gDDR defects.
In our study, 22 locally advanced PCa patients with gDDR defects received neoadjuvant docetaxel chemo-hormonal therapy followed by RP. Nine patients (40.9%) experienced pathological down-staging, and two patients (9.09%) showed pT0. Pan et al. 2 conducted a retrospective study enrolling unselected very high risk locally advanced PCa patients, in which 52 patients were treated with neoadjuvant docetaxel chemo-hormonal therapy. In their study, 32 patients (61.5%) achieved pathological down-staging, while 9 patients (17.31%) showed pT0. 1 These results indicate that gDDR defect carriers may still benefit from docetaxel chemo-hormonal therapy. In line with our findings, Gallagher et al. 21 reported that castration-resistant prostate cancer (CRPC) patients carrying germline BRCA mutations remain sensitive to taxanes. Nevertheless, in the present study, patients in the docetaxel group experienced biochemical recurrence at a median time of 7.76 months (95% CI 0.770–14.748) after RP, indicating PCa patients with gDDR defects may be more prone to recurrence after surgery, and the curative intent may not be achieved in this cohort through this treatment regimen.
Herein, we suggested that locally advanced PCa patients with gDDR defects may benefit from additional platinum agents. Platinum-based chemotherapy is generally not recommended in the treatment of PCa, since phase III trial of satraplatin failed to achieve a significant overall survival benefit in unselected CRPC. 22 However, platinum compounds cause DNA damage through distorting the double helix of DNA which are generally repaired by the DDR pathway. Hence, DDR defects could indicate sensitivity to platinum-based chemotherapy.23,24 Pleasingly, platinum-based chemotherapy in selected advanced PCa patients with DDR alterations has shown promising results.25–27 Mota et al. 12 conducted a study of 109 metastatic CRPC patients who underwent germline genomic profiling and revealed that patients with DDR mutations were more likely to achieve a PSA decline of 50% when treated with platinum-based chemotherapy. Fan et al. 28 carried out a study of 55 metastatic CRPC patients who progressed on docetaxel-based chemotherapy and underwent genomic profiling for 14 specific DDR pathway genes and reported that superior PSA progression free survival was observed in patients with gDDR defects. Hager et al. 29 carried out a systematic review involving various clinical trials that have evaluated platinum agents in the treatment of advanced prostate cancer and reported that platinum compounds have moderate antitumor potential in advanced PCa patients and further recommended that translational evidence on the nature of DDR pathway deficiency in individual PCa patients can provide a valuable molecular biomarker for treatment decisions. In the present study, 14 patients received additional platinum compounds (cisplatin) in the neoadjuvant therapy regimen. Eleven patients (78.57%) achieved pathological down-staging and four patients (28.57%) were confirmed with pT0. Although patients in the docetaxel + cisplatin group showed more advanced cT stages at diagnosis, they achieved better pathological down-staging. At a median follow-up time of 18.95 months (IQR 15.64, 22.22), 6/14 patients experienced biochemical recurrence. The addition of platinum compound significantly prolonged bPFS in this cohort (median Not Reached versus 7.76 months, p = 0.039). These findings suggest that neoadjuvant docetaxel + cisplatin chemo-hormonal therapy (docetaxel group) may be a better treatment option in locally advanced PCa patients with gDDR defects.
In the present study, there were no differences observed between the two groups in operation duration, blood loss, and positive surgical margin rates. The most common adverse events reported in CRPC patients treated with platinum-based chemotherapy were hematologic toxicities and gastrointestinal (GI) disorders.22,30 In our cohort, one patient in the docetaxel + cisplatin group discontinued neoadjuvant therapy due to viral hepatitis A. The major adverse event observed in the platinum group was grade 3 leukopenia, which occurred in four patients (28.57%). There were no significant differences in any adverse event rate between the two groups, indicating additional platinum agents was well tolerated in this cohort. Although neoadjuvant chemotherapy is not a standard treatment in prostate cancer, it has been used in breast cancer. Recent studies reported that the addition of veliparib (PARP inhibitor) and carboplatin to paclitaxel followed by doxorubicin and cyclophosphamide improved pathological response in patients with operable triple-negative breast cancer. 31 With this emerging data on activity of poly adenosine diphosphate [ADP]–ribose polymerase(PARP) inhibitors, further prospective trials evaluating the clinical benefits associated with the combination of PARP inhibitors and platinum agents in molecularly selected prostate cancer patients with gDDR defects are much warranted.
Our study is the first to assess the role of docetaxel + cisplatin chemotherapy in treatment-naïve PCa patients carrying gDDR defects. However, there remain certain limitations. Firstly, our study is a non-randomized observational study, where the patients were pragmatically subjected to treatment regimens which could introduce unaccounted selection bias. Since the chemotherapy regimens with docetaxel + cisplatin was not a standard of care for prostate cancer even in late-stage cases and the cisplatin-based chemotherapy regimen might bring more adverse events in elder people, we did not choose to perform a randomized study. Secondly, our study a pilot study. Thus, the number of patients enrolled was less and we did not attempt to perform subgroup analysis. Moreover, due to expensive genetic testing methods, majority of patients may not opt for genetic tests for identification of gDDR alterations, thereby resulting in limited sample size. Further, the follow-up was also relatively short. However, the study is ongoing and the patients are subjected to longer follow-up for obtaining mature data in order to substantiate our current preliminary findings.
Conclusion
In summary, based on postoperative pathological down-staging and bPFS, it is notable that neoadjuvant docetaxel + cisplatin (platinum group) chemo-hormonal therapy in gDDR mutated patients with locally advanced PCa might be efficacious and safe with tolerable toxicity profiles when compared to docetaxel alone. However, further pivotal prospective randomized clinical trials are warranted to leverage the benefits of neoadjuvant docetaxel + cisplatin (platinum group) therapy in patients with locally advanced PCa with gDDR defects.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221128356 – Supplemental material for Efficacy of neoadjuvant docetaxel + cisplatin chemo-hormonal therapy versus docetaxel chemo-hormonal therapy in patients with locally advanced prostate cancer with germline DNA damage repair gene alterations
Supplemental material, sj-docx-1-tam-10.1177_17588359221128356 for Efficacy of neoadjuvant docetaxel + cisplatin chemo-hormonal therapy versus docetaxel chemo-hormonal therapy in patients with locally advanced prostate cancer with germline DNA damage repair gene alterations by Chenfei Chi, Jiazhou Liu, Liancheng Fan, Yinjie Zhu, Yanqing Wang, Jianjun Sha, Yiran Huang, Baijun Dong, Jiahua Pan and Wei Xue in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
