Abstract
Background:
Inhibitors targeting programmed cell death 1 (PD-1) and programmed death-ligand 1 (PD-L1) have unprecedented effects in cancer treatment. However, the objective response rates (ORRs), progression-free survival (PFS), and overall survival (OS) of PD-1/PD-L1 blockade monotherapy have not been systematically evaluated.
Methods:
We searched Embase, PubMed, and Cochrane database from inception to July 2019 for prospective clinical trials on single-agent PD-1/PD-L1 antibodies (avelumab, atezolizumab, durvalumab, cemiplimab, pembrolizumab, and nivolumab) with information regarding ORR, PFS, and OS.
Results:
Totally, 28,304 patients from 160 perspective trials were included. Overall, 4747 responses occurred in 22,165 patients treated with PD-1/PD-L1 monotherapy [ORR, 20.21%; 95% confidence interval (CI), 18.34–22.15%]. Compared with conventional therapy, PD-1/PD-L1 blockade immunotherapy was associated with more tumor responses (odds ratio, 1.98; 95% CI, 1.52–2.57) and better OS [hazard ratio (HR), 0.75; 95% CI, 0.67–0.83]. The ORRs varied significantly across cancer types and PD-L1 expression status. Line of treatment, clinical phase and drug target also impacted the response rates in some tumors. A total of 2313 of 9494 PD-L1 positive patients (ORR, 24.39%; 95% CI, 22.29–26.54%) and 456 of 4215 PD-L1 negative patients (ORR, 10.34%; 95% CI, 8.67–12.14%) achieved responses. For PD-L1 negative patients, the ORR (odds ratio, 0.92; 95% CI, 0.70–1.20) and PFS (HR, 1.15; 95% CI, 0.87–1.51) associated with immunotherapy and conventional treatment were similar. However, PD-1/PD-L1 blockade monotherapy decreased the risk of death in both PD-L1 positive (HR, 0.66; 95% CI, 0.60–0.72) and PD-L1 negative (HR, 0.86; 95% CI, 0.74–0.99) patients compared with conventional therapy.
Conclusion:
The efficacies associated with PD-1/PD-L1 monotherapy vary significantly across cancer types and PD-L1 expression. This comprehensive summary of clinical benefit from immunotherapy in cancer patients provides an important guide for clinicians.
Background
Immune checkpoint blockade therapy targeting programmed cell death 1 (PD-1) and programmed cell death ligand 1 (PD-L1) has revolutionized cancer treatment in the past decade. 1 Currently, three PD-1 monoclonal antibodies (cemiplimab, nivolumab, and pembrolizumab) and three PD-L1 inhibitors (atezolizumab, avelumab, and durvalumab) are approved by the United States (US) Food and Drug Administration (FDA) for dozens of indications. These PD-1/PD-L1 inhibitors statistically improved the overall survival (OS) of cancer patients and have emerged as the standard therapy for multiple malignancies.1,2 It should be acknowledged that, in many clinical trials, because it usually takes a long time to obtain OS information, the efficacies of immunotherapy were represented by the independently confirmed objective response rates (ORRs) evaluated based on the standard guidelines such as Response Evaluation Criteria in Solid Tumor (RECIST).
Although immunotherapy has unprecedented effects on cancer treatment, only a fraction of patients can benefit from it.1,2 Currently, a major challenge for immunotherapy is to find ideal biomarkers that can identify patients who are susceptible to treatment and avoid serious toxicities and unnecessary costs for non-responders. Previous studies have revealed that PD-L1 immunohistochemistry (IHC), microsatellite instability (MSI), tumor mutational burden (TMB), T-cell receptor clonality, T-cell infiltration levels, gene expression signatures and peripheral blood biomarkers were associated with clinical response in immunotherapy. 3 Among them, PD-L1 expression was treated as biologically plausible biomarker for the prediction of treatment response to immunotherapy. Currently, companion PD-L1 expression diagnostic assays were granted by FDA for use in patients with lung cancer, urothelial cancer, renal cell cancer, and melanoma. 4 In fact, only two biomarkers, namely PD-L1 and MSI, were approved for PD-1/PD-L1 based immunotherapy.1,3 However, it is unclear whether PD-L1 expression status is robustly predictive of clinical benefit across diverse human cancers, or outside of these specific clinical trial populations.
Understanding of these issues may have important public health and clinical implications given the significant increase in the application of immunotherapy is expected in the future. Here, with accumulated evidence, we conducted a comprehensive meta-analysis to assess the clinical efficacies of PD-1/PD-L1 monotherapy in published clinical trials. In addition, we investigated the role of PD-L1 expression status as a predict biomarker, and quantified the potential differences in the incidences of tumor response, progress-free survival (PFS), and OS among a variety of cancer types and treatment strategies.
Methods
Search strategy and study selection
A comprehensive search of PubMed, Embase, and Cochrane databases from inception to July 2019 was performed to identify prospective clinical trials with PD-1/PD-L1 blockade monotherapy. The major search keywords and medical subject headings used were: atezolizumab, avelumab, cemiplimab, durvalumab, nivolumab, pembrolizumab, checkpoint inhibitors, PD-1, programmed cell death receptor 1, PD-L1, programmed cell death 1 ligand 1, clinical trial, and phase. The references of published studies and reviews were also examined for additional eligible trials. All the authors independently carried out the initial search, screened the title and abstract for potentially relevant studies, and identified trials as included, excluded, and uncertain. For uncertain studies, the full-texts were reviewed for the confirmation of eligibility. Any discrepancy was resolved by discussion and unanimous agreement.
Both inclusion and exclusion criteria were pre-specified. To be eligible, trials had to meet the following criteria: (a) population: prospective phase I, II, or III trials recruiting adult subjects (>18 years old) with cancer; for patients with reported PD-L1 expression status, the cutoff values for PD-L1 positivity/negativity and PD-L1 status were extracted from the original studies directly; (b) intervention: patients were treated with PD-1/PD-L1 inhibitor (avelumab, atezolizumab, cemiplimab, durvalumab, nivolumab, and pembrolizumab) monotherapy irrespective of dosage and duration in at least one arm; (c) outcomes: available information on ORR, PFS and OS. Trials published online before print were included, but meeting abstracts were excluded. In addition, studies were excluded if they were retrospective trials. Other publications, including review articles, basic research, case reports, letters, comments, correspondences, editorials, and cost effectiveness analyses were also excluded. When multiple publications of the same trial occurred, only the most recent, and/or most complete, reporting articles was selected. Any discrepancies were settled by discussion until unanimous agreement was reached. All the included articles represented unique studies.
Data extraction
For each study, the following items were exacted: name of study, first author and year of publication, clinical phase, line of treatment, cancer type, PD-L1 detection assay, name of the PD-1 or PD-L1 inhibitor, dosing schedule, number of patients recruited, median progression-free survival (PFS), median OS, median treatment duration (range), median follow up (range), number of patients for efficacy analysis, median time to response (range), median duration of response (range), number of complete response (CR), number of partial response (PR), number of objective response (OR), number of PD-L1 positive patients for efficacy analysis, number of CR in PD-L1 positive patients, number of PR in PD-L1 positive patients, number of OR in PD-L1 positive patients, number of PD-L1 negative patients for efficacy analysis, number of CR in PD-L1 negative patients, number of PR in PD-L1 negative patients, and number of OR in PD-L1 negative patients. All authors independently carried out the data extraction.
Risk of bias
To evaluate the methodological quality of eligible studies, the seven-item Jadad ranking system including randomization, double blinding, and the flow of recruited subjects (withdrawals and dropouts) were applied. 5 As previously described, a controlled study could achieve a Jadad score of between 5 (optimal methodological quality) and 0 (poor methodological quality). Any disagreements were resolved by discussion and unanimous agreement.
Statistical analysis
The primary purpose of this study was to investigate the overall incidence and corresponding 95% confidence interval (CI) of objective response rates (ORRs) in cancer patients treated by PD-1/PD-L1 inhibitors. ORR referred to the percentage of patients whose tumor shrunk (partial response) or disappeared (complete response) after treatment. To calculate the incidence, the number of subjects for efficacy analysis and the number of responses were extracted from every study. We compared the tumor response in PD-L1 positive patients and PD-L1 negative patients by calculating the odds ratios in each trial. To investigate PFS and OS, we derived the hazard ratios (HRs) and their 95% CI from each trial, separately for PD-L1 positive patients and PD-L1 negative patients.
Statistical heterogeneity across studies was evaluated by Cochrane’s Q statistic. The I2 statistic was calculated to assess the extent of inconsistency contributable to the heterogeneity across different studies. 6 The assumption of homogeneity was considered invalid for I2 > 25% and p < 0.05. Summary ORs and incidences were calculated using fixed-effects model or random-effects model depending on the heterogeneity. The heterogeneities of PFS and OS between PD-L1 positive patients and PD-L1 negative patients were assessed by an interaction test and expressed as P for interaction.
Potential publication bias was assessed by visual inspection of a funnel plot, and also evaluated using the tests of Egger et al. and Begg et al.7,8 Two-sided p < 0.05 were considered statistically significant.
Results
Search results
We identified a total of 11,130 potentially relevant publications from the initial search, including 5125 from PubMed, 5362 from Embase, and 643 from Cochrane; 4893 records were removed due to duplication. After screening of the titles and abstracts, 5885 articles did not meet our inclusion criteria. Additional eligibility assessing the full-texts of the remaining studies, 160 prospective clinical trials involving 28,304 cancer patients were included in the final analysis.9–168 Among these patients, 22,165 subjects were treated by PD-1/PD-L1 inhibitors, the rest 6139 patients were included in the control arms. A flow chart presenting the study selection is shown in Figure 1.

Flow-chart diagram of selected trials included in this study.
Study characteristics
The baseline characteristics of the eligible trials included in this study were demonstrated in Supplemental Table S1. The median treatment duration of PD-1/PD-L1 inhibitors lasted between 1.4 months and 11.9 months, and median follow up ranged from 2.6 months to 49.9 months. The median time to response in immunotherapy is relatively stable, with most response occurred within 3 months after treatment (range, 1.4 months to 4.5 months). In some studies, several different types of tumors or different clinical phases were investigated and reported in one article. Accordingly, 185 arms of PD-1/PD-L1 blockade immunotherapy arms were found in the 160 eligible records. Among these 185 arms, the agents administrated were nivolumab (n = 68, 6403 patients), pembrolizumab (67, 8124), atezolizumab (22, 4094), avelumab (17, 1785), durvalumab (9, 1674), and cemiplimab (2, 85). The responses to PD-1/PD-L1 inhibitors were examined in lung cancer (n = 40, 7265 patients), melanoma (21, 3501), urothelial cancer (14, 3443), gastric or gastro-esophageal junction cancer (10, 1265), lymphoma (10, 757), head and neck cancer (9, 1166), renal cancer (9, 915), breast cancer (7, 652), ovarian cancer (6, 573), mesothelioma (5, 200), sarcoma (5, 154), and other types of cancers (49, 2274).
Of all the eligible 185 arms, 108 arms (58.38%) revealed or partly revealed the responses to immunotherapy based on PD-L1 expression status (Supplemental Table S2). In most cases, the cutoff value for PD-L1 positivity or negativity was that PD-L1 stained cells accounted for 1% of cancer cells, or cancer and immune cells, assayed by immunohistochemistry staining techniques. However, the thresholds were 5% in 17 studies,10,17–20,24,26,35,40,47,56,61,69,78,88,96,168 10% in 3 articles,31,127,159 25% in 5 studies,43–45,48,49 and 50% in 3 records.121,131,155 In addition, only PD-L1 positive cancer patients were recruited in 17 studies.19,24,50,110,123,125,128,132,136,137,145,146,152,155,158,161,164 In the present study, we extracted the PD-L1 expression status directly from the original records.
ORR
Overall, the eligible 160 studies recorded 4747 responses to immunotherapy. The summary ORRs in patients receiving PD-1/PD-L1 inhibitors was 20.21% (95% CI, 18.34–22.15%). 26 types of tumors were investigated in more than two trials. Next, we calculated the pooled ORRs in these cancers. The incidences of responses, in decreasing order, were shown in Figure 2. As expected, the overall response rates varied significantly by tumor types, with the highest anti-cancer activities were reported in carcinogen-induced tumors or malignancies driven by viral infections, such as classic Hodgkin’s lymphoma, desmoplastic melanoma, and the virally induced Merkel cell carcinoma of the skin. The second subgroup of tumors with relatively high ORRs were cancer with a relative high immunogenicity, 1 such as melanoma and lung cancer, liver cancer and renal cancer. Some tumors, such as prostate cancer, colorectal cancer, and ovarian cancer, only had limited T-cell infiltration, and were considered immune excluded. 12 Accordingly, the response rates in these tumors were relatively low.

The pooled ORRs in 26 different types of cancers. Vertical line indicates the overall mean incidence of tumor responses calculated from all 22,165 patients in 160 eligible trials.
A total of 862 complete responses were observed in 19,418 cancer patients, and the overall incidence of CRs was 3.85% (95% CI, 3.06–4.73%). Moreover, the incidences of CR in 25 tumors are listed in Supplemental Figure S1. It was noted that the incidences were relatively low in most types of tumors. In addition, 3400 partial responses were reported and the pooled incidence of PR was 15.83% (95% CI, 14.36–17.35%). The incidences of partial responses from 25 tumors were presented in Supplemental Figure S2. The most frequent PRs were found in cutaneous cancer, Merkel cell cancer, and lymphoma.
Impact of clinicopathological characteristics on ORR
We further conducted the efficacy analysis based on PD-L1 expression status (Figure 3). Among 9494 patients that were PD-L1 positive, 2313 responses were observed (ORR, 24.39%; 95% CI, 22.29–26.54%); whereas in 4215 PD-L1 negative patients, 456 responses were recorded (ORR, 10.34%; 95% CI, 8.67–12.14%). Overall, there was a significant difference in ORRs between PD-L1 positive patients and PD-L1 negative patients (odds ratio, 2.65; 95% CI, 2.34–3.00; p < 0.001). We further examined the impact of PD-L1 expression status on tumor responses in 12 types of cancers (Figure 3). The magnitudes of efficacy of immunotherapy were greater for PD-L1 positive patients than for PD-L1 negative patients in all types of tumors. However, the differences were insignificant in breast cancer, renal cancer, Merkel cell cancer, liver cancer, and ovarian cancer.

Comparison of the ORRs between patients that were PD-L1 positive (blue) and patients that were PD-L1 negative (red) in PD-1 or PD-L1 blockade monotherapy.
Next, we investigated the incidences of CR and PR based on PD-L1 expression status, respectively. In total, 276 CRs were reported among 6904 PD-L1 positive cancer patients, and the overall incidence was 4.03% (95% CI, 2.86–5.40%). In contrast, only 84 CRs occurred in 2883 PD-L1 negative patients, and the incidence was 2.37% (95% CI, 1.52–3.40%). PD-L1 positive patients had a higher incidence of CRs compared with PD-L1 negative patients (odds ratio, 2.22; 95% CI, 1.69–2.91; p < 0.001) (Supplemental Figure S3). The pooled incidence of PR in PD-L1 positive patients was 18.48% (95% CI, 16.61–20.43%) as 1310 partial responses were discovered in 6845 subjects. By contrast, 224 PRs occurred in 2732 PD-L1 negative patients, and the incidence was 7.41% (95% CI, 5.91–9.05%). The overall incidence of PR in PD-L1 positive patients and PD-L1 negative patients was significantly different (odds ratio, 2.41; 95% CI, 2.02–2.86; p < 0.001) (Supplemental Figure S4).
We also examined the association between tumor response and some other features including line of treatment, clinical phase, drug target, and numbers of recruited patients in several major tumors (Figure 4). Line of treatment was an important contributor to ORRs in lung cancer and melanoma, but not in urothelial cancer. In all cancers except melanoma, the ORRs in phase I/II trials were similar compared with the ORRs in phase III trials. In GC/GEJC, patients treated with PD-L1-targeted agents showed higher ORRs than those treated with PD-1-targeted agents. The impact of numbers of recruited patients on the efficacies was insignificantly in all examined types of tumors.

The association between tumor responses and several major clinicopathological characteristics in lung cancer (A), melanoma (B), urothelial cancer (C), GC/GEJC (D), head and neck cancer (E), and renal cancer (F).
Immunotherapy versus conventional treatment
Next, we compared the efficacies of PD-1/PD-L1 blockade immunotherapy versus conventional treatment in patients that were PD-L1 positive and PD-L1 negative (Figure 5). In total, 26 randomized, controlled trials (RCTs) including 13,899 patients, were eligible (Supplemental Table S3). Among them, 21 studies were phase III RCTs, four studies were phase II RCT, and one study was phase II/III RCT. In all, 12 studies were conducted in lung cancer, 4 in melanoma, 3 in GC/GEJC, 2 each in urothelial cancer, renal cancer, and head and neck cancer, and 1 in colorectal cancer. All studies were performed in solid tumors, and 6993 (50%) of 13,899 patients had lung cancer. Patients in the intervention arms received nivolumab in nine studies, pembrolizumab in eight studies, atezolizumab in six studies, avelumab in two studies, and durvalumab in one study. The methodological qualities of the eligible trials were generally moderate to good. Randomized treatment allocation sequence generated in all trials. The main issue affecting quality was lack of blinding.
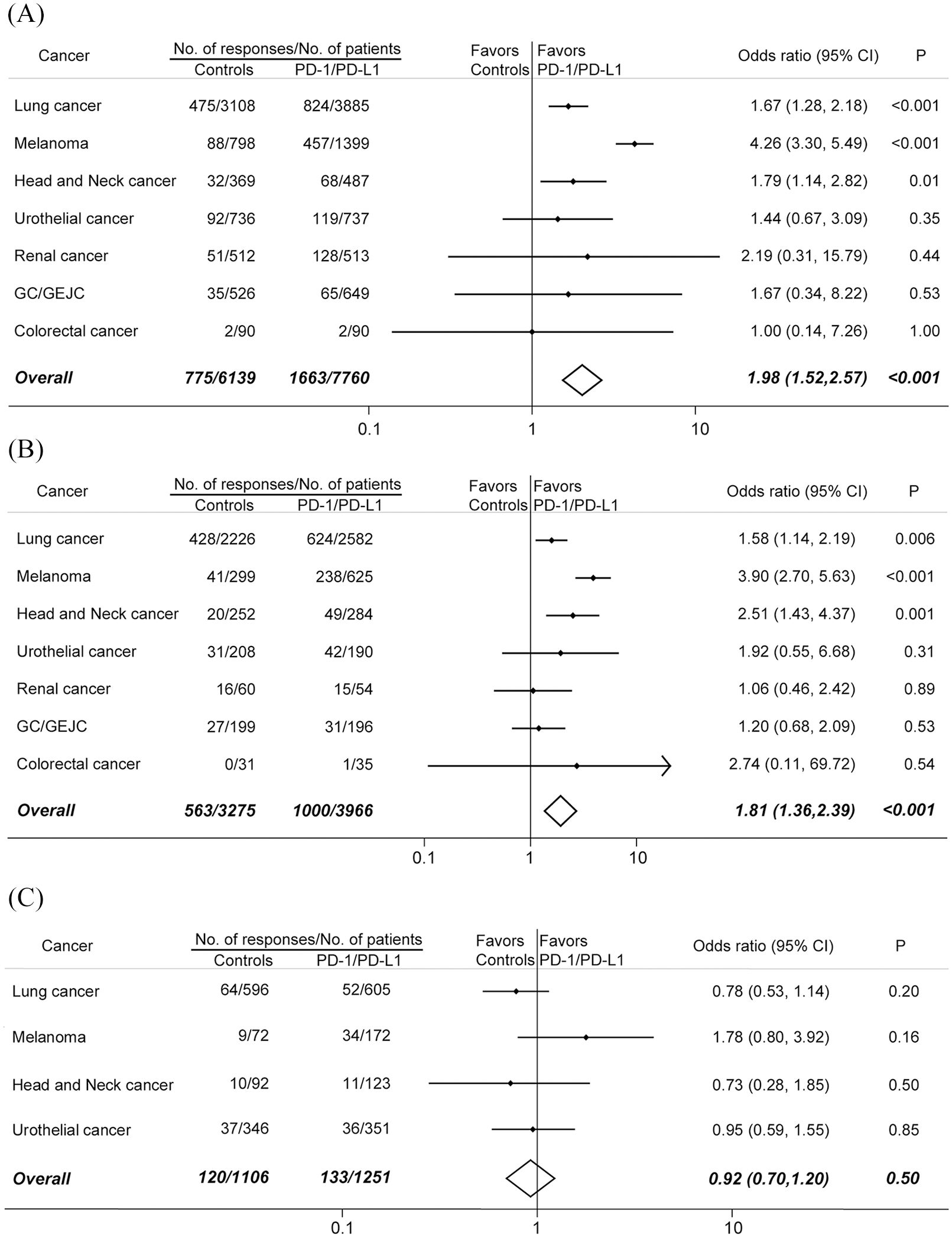
Comparison of the ORRs between PD-1 or PD-L1 blockade monotherapy and conventional therapy in (A) all recruited patients, (B) patients who were PD-L1 positive, and (C) patients who were PD-L1 negative.
Among 7760 patients treated with PD-1/PD-L1 inhibitors, 1663 tumor responses occurred, whereas in 6139 patients treated with conventional agents, 775 responses were recorded. The difference was significant (odds ratio, 1.98; 95% CI, 1.52–2.57; p < 0.001). Comparisons between immunotherapy and controls in seven types of tumors were shown in Figure 5A.
For PD-L1 positive patients,1000 responses were reported from 3966 patients in the PD-1/PD-L1 blockade arms; 563 responses occurred in 3275 patients in the control arms (Figure 5B). More PD-L1 positive patients responded to PD-1/PD-L1 inhibitors than to conventional agents (odds ratio, 1.81; 95% CI, 1.36–2.39; p < 0.001). On the other hand, 2357 patient who had PD-L1 negative disease from 10 RCTs were included in our analysis. PD-1/PD-L1 blockade immunotherapy did not increase the tumor responses compared with conventional treatment (odds ratio, 0.92; 95% CI, 0.70–1.20; p = 0.50) (Figure 5C).
It was well known that some non-responders also derived significant benefit from immunotherapy.1,2 Accordingly, we evaluated PFS and OS in patients treated with PD-1/PD-L1 inhibitors and explored their association with PD-L1 expression status (Figure 6). Seven RCTs with 1944 PD-L1 positive patients and 1788 PD-L1 negative patients were included in PFS analysis (Figure 6A). Compared with controls, PD-1/PD-L1 blockade immunotherapy could significantly improved PFS in patients that were PD-L1 positive (HR,0.76; 95% CI, 0.61–0.95), but not in patients that were PD-L1 negative (HR, 1.15; 95% CI, 0.87–1.51), and the difference between these two groups were statistically significant (Pinteraction < 0.001). Overall, the PFS values were similar in patients treated with immunotherapy and patients treated with conventional therapy (HR, 0.94; 95% CI, 0.76–1.16).
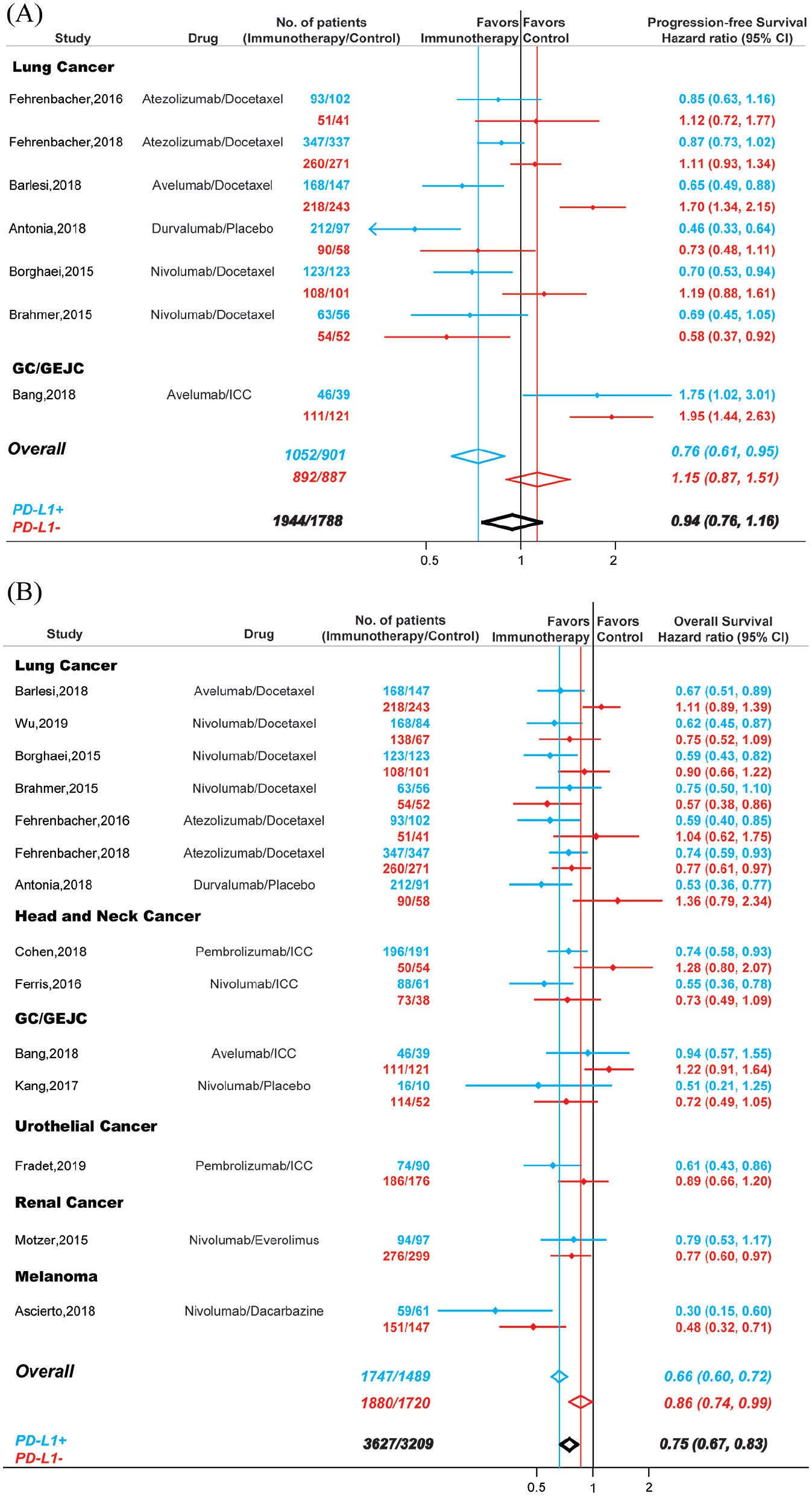
Comparison of PFS (A) and OS (B) between PD-1 or PD-L1 blockade monotherapy and conventional therapy. Blue, patients that were PD-L1 positive; red, patients that were PD-L1 negative; black, all recruited patients.
For OS, 14 studies including 3627 PD-L1 positive patient and 3209 PD-L1 negative patients were investigated (Figure 6B). Compared with conventional therapy, PD-1/PD-L1 blockade immunotherapy decreased the risk of death by 25% in cancer patients (HR, 0.75; 95% CI, 0.67–0.83). However, the OS in patients that were PD-L1 positive (HR, 0.66; 95% CI, 0.60–0.72) and patients that were PD-L1 negative (HR, 0.86; 95% CI, 0.74–0.99) were significantly different (Pinteraction < 0.001).
Publication bias
Potential publication biases were assessed by Begg’s funnel plots and Begg’s rank correlation. Visual inspection of the Begg’s funnel plot did not identify substantial asymmetry in all the analysis conduct in this study. The Begg’s rank correlation test also indicated no evidence of publication bias.
Discussion
To our knowledge, this is the largest and most comprehensive meta-analysis investigating the clinic benefit in cancer patients treated with PD-1/PD-L1 blockade monotherapy. Previous studies included fewer trials and focused mainly on some specific cancers, such as non-small cell lung cancer, 169 advanced urothelial carcinoma, 170 and malignant melanoma. 171 Here, with published data from over 28,000 subjects in 160 perspective clinical trials, we report a meta-analysis evaluating the association between PD-1/PD-L1 blockade monotherapy and tumor response, PFS, and OS in cancer patients. Such a global overview of clinical efficacies is important, as the results constitute a critical reference for clinicians, drug developers, and basic scientists, and complementary information in drafting the clinical practice guidelines.
The development of immunotherapy has revolutionized the treatment of cancer and are increasingly being used in earlier disease settings and in combination with other therapies.172,173 A key unmet need in immunotherapy is the development of predictive biomarkers, which can identify patients that are likely to respond to PD-1/PD-L1 inhibitors and therefore reduce exposure and risk of toxicities for those patients with little potential for benefit of response. Given the high-cost of immunotherapy, in the era of value-based cancer care this becomes even more important. However, predictive biomarkers to PD-1/PD-L1 blockade immunotherapy are difficult to develop. The potential markers are usually functional targets rather than stable oncogenic targets; hence the expression is context-dependent and transient, and can be influenced by various factors in the microenvironment. In addition, clinical issues in evaluating any reliable biomarkers are often associated with some inherent characteristics of immunotherapy such as limitations to evaluate tumor responses with standard imaging methods, clinical response and survival benefit are not necessarily related.
Because PD-L1 participates in suppressing immunogenicity and is a direct or indirect target of PD-1/PD-L1 antibodies, it was the first and most popular biomarker routinely used for cancer patients treated with immunotherapy in clinical trials. Indeed, accumulating evidences has revealed that PD-L1 expression was associated with the efficacy of immunotherapy and clinical outcomes.2,173 Moreover, several companion IHC assays for PD-L1 expression has been approved by FDA (22C3 for pembrolizumab; SP142 for atezolizumab). 174 However, some major pitfalls still remain for PD-L1 expression evaluation such as different antibodies (22C3 for pembrolizumab, 28-8 for nivolumab, SP142 for atezolizumab, and SP263 for durvalumab) and various labelling threshold for positivity/negativity. In 2017, the Blueprint PD-L1 IHC Assay Comparison Project showed that these assays had different performances: PD-L1 expression were comparable for 22C3, 28-8, and SP263, but not for SP142. 175 Furthermore, despite similar analytical performance of 28-8, 22C3, and SP263, interchanging assays and thresholds still led to “misclassification” of PD-L1 status for some patients. Linear epitope mapping experiments revealed different binding features for these four PD-L1 antibody clones. It could cause particular staining patterns depending on PD-L1 conformation or isoform expression. Besides analytical consideration, other issues exist for PD-L1 evaluation. For example, biopsies cannot represent the entire tumor due to spatial heterogeneity. In addition, PD-L1 expression was regulated at transcriptional, post-transcriptional, and protein levels.1,172 In clinical practice, conventional treatments such as chemotherapy and radiotherapy are also considered as potential regulators of PD-L1 expression as well as the antitumor immunity. 176 In the present study, considering the original researchers knows their trials better than anybody else, we extracted and illustrated the IHC assay and cutoff values from every eligible trial, and classified the PD-L1 positivity and negativity based on the results from the original manuscripts. Hence, our study is subject to any biases or errors of the original investigators, and the results are generalizable only to the patient groups eligible for these trials.
Given the limitations of PD-L1 expression evaluation, new strategies are developing to select stable and reliable genomic biomarkers. Research in this field mostly focused on TMB as a surrogate marker for tumor immunogenicity. For most tumors, it is reported that higher TMB was association with improved OS in patients receiving PD-1/PD-L1 inhibitors. 177 Moreover, a study across 27 tumors revealed that a correlation between TMB and immunotherapy response rate by linear regression analysis. 178 However, TMB has not been approved as a companion biomarker because the ideal quantification methods and threshold values have not been defined for clinical application (i.e. sequencing approach, tumor tissue or blood TMB, whole-exome sequencing or selected gene panel, and sequencing depth). On the other hand, tumor with MSI showed impressive clinical benefit to immunotherapy. 179 In addition, mutations in DNA mismatch repair (MMR) genes such as MSH2, MSH6, MLH1, and PMS2 were also associated with durable response to PD-1/PD-L1 inhibitors. 180 These studies led to the first tissue-agnostic approval for PD-1/PD-L1 blockade treatment across tumors with dMMR/MSI-H. 181 However, it should be note that the frequency of dMMR/MSI-H in tumors not within colorectal, endometrial, or gastric cancer is only 0.8% based on one study including 11,139 cancer samples from 39 cancer types. 182 Accordingly, all the available single biomarkers showed some limitations in the real-world clinical application. Currently, it is believed that combinable biomarkers and/or algorithms using multiplex ways and the support of artificial intelligence will be more successful. 174 For example, in OAK trial, 183 a so-called “Teff signature” (IFN-γ, PD-L1, CXCL9) was applied as a surrogate for pre-existing immunity. This signature was correlated with PD-L1 expression but was more sensitive than PD-L1 in predicting OS from atezolizumab.
Consist with previous findings,2,169,184 our analysis here showed that there was a statistically significant difference between PD-L1 positive and PD-L1 negative patients in terms of ORR, PFS, and OS. PD-L1 significantly increased the chance of tumor response in melanoma, urothelial cancer, head and neck cancer, lung cancer, GC/GEJC, mesothelioma, and thymic cancer. In addition, compared with conventional treatment, the ORRs were significantly higher in PD-L1 positive patients but not in PD-L1 negative patients only in lung cancer, melanoma, and head and neck cancer. These results suggested PD-L1 could be a valuable predictive biomarker in selected tumors. However, about 10% of cancer patients who were PD-L1 negative showed tumor responses, and PD-1/PD-L1 monotherapy could decreased the risk of death by 14% for PD-L1 negative patients. These data suggested that PD-L1 expression status neither guaranteed nor precluded response to PD-1/PD-L1 monotherapy in all cases. Recently, Davis et al. also evaluated the role of PD-L1 expression as a predictive biomarker in 45 FDA approvals of immune checkpoint inhibitors across 15 tumor types. 184 It should be noted that there were several differences between these two studies. First, our primary aim was to assess the ORRs in cancer immunotherapy. Many clinicopathological characteristics could impact the tumor responses including tumor type, PD-L1 expression status, clinical phase, line of treatments. We further examined the association between ORRs and these factors in our study, while Davis et al. focused mainly on the predictive value of PD-L1 expression because nine FDA approvals linked to a specific PD-L1 threshold and companion diagnostic biomarker. Second, our study included 160 perspective clinical trials with over 28,000 patients. With the increased statistical power, our meta-analysis was the most up-to-date and comprehensive study, and should be more reliable and solid. In addition, the assessment of the response to immunotherapy is insufficient regarding clinical value, it has been proven that patients treated with immunotherapy have a significant benefit even if they do not respond to treatment. 2 Accordingly, we also examined PFS and OS in both PD-L1 positive patients and PD-L1 negative patients. Our results revealed that PD-1/PD-L1 blockade monotherapy significantly decrease the risk of death by 34% for PD-L1 positive patients and by 14% for PD-L1 negative patients. It should be acknowledged that the survival benefit between cancer patients who were PD-L1 positive and those who were PD-L1 negative were significantly different.
The molecular basis underlying tumor resistance to immune checkpoint inhibitors is not yet clear; it may be due partly to inadequate cancer specific T cells function, 185 insufficient anti-cancer T cell generation, 186 and development of T-cell memory impairments. 186 Lack of sufficient or suitable neoantigens, or impaired neoantigen processing or presentation of neoantigens, can lead to dysfunctional impairments during the development of cancer-reactive T cells. 187 It is well established that efficacious anti-cancer response followed by PD-1/PD-L1 inhibitors need clonal-proliferation and reactivation of antigen-experienced T cells in the tumor micro-environments, and eventually secret the cytolytic effector to kill the cells showing tumor-associated antigen.186,187 Accordingly, it seems more critical to understand whether the PD-1/PD-L1 pathway was active rather than focusing solely on the expression of PD-L1 during cancer immunotherapy. Thereby, predicting tumor responses to PD-1/PD-L1 blockade immunotherapy remains the greatest challenge, and considerable efforts should be made to profile the complex and dynamic factors governing the strength and duration of immune response in the immunotherapy, making treatment decisions on a personalized basis.
From the standpoint of patient counseling, several results derived from this meta-analysis are important. Although approximately one-fifth of patients respond to PD-1/PD-L1 monotherapy in clinical trials, the tumor responses vary significantly across different cancers and PD-L1 expression. These numbers could be critical to share with patients before they began PD-1/PD-L1 blockade immunotherapy. In addition, PD-L1 expression status can inform the proper selection of patients who have a significantly increased chance of tumor responses in lung cancer, melanoma, and head and neck cancer.
This study is restricted by some limitations. First, this meta-analysis relied on published results rather than on individual patients’ data. Accordingly, we cannot exclude the fact that other clinicopathological characteristics can affect the response and influence our results. Second, the majority of studies are phase I/II trials, which add heterogeneity to our analysis. We adjusted this heterogeneity by conducting random-effects models to achieve the overall ORRs. Furthermore, we examined the ORRs in different phases in several major tumors. In most examined cancers, ORRs in phase III are similar compared with ORRs in phase I/II. Even so, it might underestimate the real incidences of tumor responses given that trials with limited numbers of patients received disproportional weight in calculation. Third, some included studies were open-label trials. Even for the double-blinded RCTs, skillful clinicians can identify the tumor responses induced by PD-1/PD-L1 antibodies, which may cause potential bias. Fourth, these included studies are conducted at various medical centers by different researches, and may have some subjectivities in recording clinical outcomes. Our analysis is subject to any errors or biases of the original researchers, and the conclusions are generalizable only to recruited patients included in these trials.
Conclusion
With 28,304 cancer patients from 160 perspective trials, our comprehensive analysis revealed the clinical benefit to PD-1/PD-L1 monotherapy in cancer patients. This global overview can be used as a reference and may guide clinical practice and drug development.
Supplemental Material
supplement – Supplemental material for Efficacy of PD-1/PD-L1 blockade monotherapy in clinical trials
Supplemental material, supplement for Efficacy of PD-1/PD-L1 blockade monotherapy in clinical trials by Bin Zhao, Hong Zhao and Jiaxin Zhao in Therapeutic Advances in Medical Oncology
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Natural Science Foundation of China (No. 31571417), Postdoctoral Science Foundation of China (No. 2018M641862 and No. 2019T120282), and Wenzhou Municipal Science and Technology Bureau (No. Y20180086). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
