Abstract
Background:
Erdafitinib is the first targeted therapy approved for the treatment of patients with metastatic urothelial carcinoma (mUC). Approval was based on a phase II single-arm trial that demonstrated significant activity of erdafitinib in patients with tumors harboring FGFR2/3 alterations. In Brazil, an Expanded Access Program (EAP) provided patients with early access to erdafitinib prior to market authorization. The current report describes characteristics and outcomes of patients with mUC on erdafitinib therapy.
Methods:
Patients with mUC that failed first- and second-line systemic therapies were screened for FGFR2/3 alterations in primary or metastatic tumor tissues. Patients with FGFR2/3 alterations were selected to receive erdafitinib at the standard dosing schedule and were followed prospectively to evaluate the efficacy and safety outcomes.
Results:
From 19 April 2019, through 13 March 2020, 47 patients with mUC from 10 Brazilian centers were tested for FGFR2/3 alterations. Alterations in FGFR2/3 were found in 12 patients (25.5%) and all of them were eligible for the EAP. Four patients (33%) had partial response, while two patients (17%) had stable disease. Progressive disease, the best response, was observed in five patients (42%). At a median follow-up of 16.2 months, the median time to treatment failure (TTF) was 2.8 months. When considering only patients with objective response, the median TTF was 5.3 months. Adverse events (AEs) were reported for any grade and grade 3 or higher in 10 patients (83%) and 5 patients (42%), respectively. The most common AE was hyperphosphatemia.
Conclusion:
This first real-world evidence report of heavily treated patients with mUC confirms the efficacy and safety of erdafitinib in a disease setting with a lack of treatment options.
Introduction
Urothelial carcinoma of the lower and upper urinary tract is the second most common urological malignancy. Patients with advanced disease have a poor outcome with a limited median overall survival (OS), usually less than 1 year. 1 For the past few decades, first-line platinum-based regimens have been the standard of care, and second-line cytotoxic chemotherapy (CT) has shown poor response rate and survival.2,3 In the last few years, immune checkpoint inhibitors (ICIs) have shown significant clinical activity in second-line treatment with an objective response rate (ORR) of about 20%. A total of five ICIs are approved by the US Food and Drug Administration for the treatment of patients with advanced urothelial carcinoma (i.e. pembrolizumab, atezolizumab, avelumab, durvalumab and nivolumab); however, only pembrolizumab has shown to significantly improve OS in the second line compared to standard CT.4,5 Therefore, there is still room for improvement in second-line therapy and beyond. 6
Fibroblast growth factor receptors (FGFRs) are transmembrane proteins normally associated with control of phosphate and vitamin D. When activated, FGFRs regulate differentiation, migration and survival through a complex pathway.7,8 FGFR genetic alterations (mutations and fusions) are associated with neoplastic progression of many tumors including urothelial carcinoma. At least 20% of advanced urothelial carcinoma have been shown to harbor FGFR2/3 alterations and have been associated with worse outcomes. Therefore, targeted therapy with FGFR inhibitors have shown significant ORR in those patients.9–11
Erdafitinib was the first targeted therapy approved for the treatment of patients with metastatic urothelial carcinoma (mUC) based on a phase II single-arm trial, including 99 patients that demonstrated significant activity with ORR of 40% in heavily treated patients with tumors harboring FGFR2/3 alterations.12,13 In Brazil, an Expanded Access Program (EAP) provided patients early access to erdafitinib prior to market authorization. The current report describes the effectiveness and safety of erdafitinib in Brazilian patients with mUC who participated in the EAP of erdafitinib.
Material and methods
Patient profile
Patients with mUC who failed on first- and second-line systemic therapies, including a platinum-based chemotherapy and/or a PD-1/PD-L1 ICI, were screened for FGFR2/3 alterations in primary or metastatic tumor tissue. Patients with FGFR2/3 alterations identified from April 2019 to March 2020 who received erdafitinib at the recommended dosing schedule of 8 mg daily were followed prospectively during the program to evaluate the effectiveness and safety of the treatment. Patients who entered the program were followed as part of routine follow-up by their physician.
This erdafitinib EAP was conducted in accordance with a current national resolution that regulates EAP in Brazil (RDC 38/2013) and also in accordance with the International Council on Harmonisation guidelines for Good Clinical Practice (ICH GCP). 14 Written information was provided, patients agreed to participate in the EAP, and consent was obtained from all participants by investigators before any study procedure.
Sample analysis
The analysis for FGFR2/3 gene alterations was performed in formalin-fixed paraffin-embedded tumor samples using a custom reverse transcriptase polymerase chain reaction (RT–PCR) amplified in real time with the therascreen® FGFR RGQ RT–PCR kit (QIAGEN). The specific gene alterations evaluated were: two-point mutations in exon 7 [p.R248C (c.742C > T) and p.S249C (c.746C > G)], two-point mutations in exon 10 [p.G370C (c.1108G > T) and p.Y373C (c.1118A > G)], and three fusions in the FGFR3 gene (FGFR3-TACC3v1, FGFR3-TACC3v3 and FGFR3-BAIAP2L1) as well as two fusions in the FGFR2 gene (FGFR2-BICC1 and FGFR2-CASP7).
Outcomes and assessments
To describe the objective response rate (ORR) of erdafitinib treatment in patients with mUC in the Brazilian EAP, we used Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 on radiological exams (computed tomography scans or magnetic resonance imaging) performed every 8–12 weeks from treatment initiation. Also, the time to treatment failure (TTF) was evaluated, which consisted of the interval from initiation of erdafitinib to its discontinuation due to disease progression, toxicity, patient choice, or death. Additionally, adverse events (AEs) were evaluated using the Common Terminology Criteria for Adverse Events version 5.0.
Statistical analysis
No formal sample size was calculated. All patients who received at least one dose of erdafitinib were analyzed. Data were summarized by frequency for categorical variables and by median and range for continuous variables.
Results
Patients
From 9 April 2019, through March 13, 2020, 47 patients with mUC from 10 Brazilian centers were tested for FGFR2/3 alterations. Alterations in FGFR2/3 were found in 12 patients (25.5%) and all of them were eligible for erdafitinib treatment in the EAP. The median age was 76 years, nine (75%) were male, and six patients (50%) presented with metastatic disease as first diagnosis. At erdafitinib treatment initiation, three patients (24%), seven patients (59%) and two patients (16%) had one, two and three sites of metastatic disease, respectively. All evaluated patients were found to have tumors harboring FGFR3 alterations, and the most common one was p.S249c, present in nine patients (75%), two patients had FGFR3 p.R248C mutation and one patient had p.Y373C. Neither FGFR2 nor FGFR3 fusions were found in this cohort. Considering the history of prior treatment, all patients had disease progression after at least two lines of prior systemic therapy including platinum-based CT. Ten patients (83%) failed on an ICI, all of them in the second-line setting. The most used ICI was pembrolizumab in six patients (50%). The baseline clinical characteristics, details of prior treatment and FGFR3 alterations of all patients are summarized in Table 1.
Baseline clinical characteristics of patients, prior treatment and FGFR3 alterations.

CT, chemotherapy; ddMVAC, dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin;
FGFR3, fibroblast growth factor receptor 3; ICI, immune checkpoint inhibitor.
Response rate
Eleven patients had RECIST measurable disease and among the ORR was observed in four patients (33%), all of them with partial response (PR). Stable disease (SD) was observed in two patients (17%). Thus, the disease control rate (DCR) was observed in six patients (50%). Progressive disease (PD) as the best response was observed in five patients (42%). Radiological response was not documented in one patient (8%) because there was no measurable RECIST disease; however, clinical improvement was observed as there was clinical reduction of subcutaneous nodules on physical examination. All subjects who had an PR or SD carried the p.S249C mutation on the FGFR3 gene. Regarding the 10 patients previously treated with PD-1 or PD-L1 inhibitors, ORR was evaluated in 9 patients. In this subgroup, PR was observed in two patients (20%), while two patients (20%) had SD. Details of response rate and TTF are summarized in Table 2.
Response rate and time to treatment failure (n = 12).

FGFR3, fibroblast growth factor receptor 3; PD, progressive disease; PR, partial response; SD, stable disease.
Time to treatment failure
At a median follow-up time of 16.2 months, six patients (50%) were alive, and two patients (17%) were still receiving erdafitinib. Median TTF was 2.8 months (range: 1.1–5.7 months) (Table 2). In patients who had PR, the median TTF was 5.3 months (range: 4.6–5.7 months). Four patients (33%) received at least one additional treatment after failure to erdafitinib.
Safety
AEs were reported at any grade in 10 patients (83%). Grade ⩾ 3 AEs were reported in five patients (42%). The most common AE was hyperphosphatemia, which was reported in four patients (33%) as grade 1 or 2, and in one patient (8%) as grade 3. Diarrhea was reported in three patients (25%), two patients (17%) as grade 3. Fatigue also was reported in three patients (25%), two patients (17%) as grade 3. Regarding eye disorders, there were two patients (17%) with blurred vision with complete improvement after temporary suspension of erdafitinib for 7 days and without recurrence of this symptom after restarting erdafitinib with the same previous dose. There were no central serous retinopathy (CSR) events in this population. At least one dose reduction occurred in four patients (33%) due to diarrhea and hyperphosphatemia (in two patients each). One patient (8%) discontinued the treatment due to an AE (diarrhea). There was one death during erdafitinib therapy, not treatment-related, caused by upper gastrointestinal bleeding. Details of AEs are summarized in Table 3.
Adverse events to erdafitinib.

Discussion
This manuscript reports the efficacy and safety of erdafitinib treatment in 12 patients with heavily pretreated mUC from the Brazilian EAP for erdafitinib. All patients included in this analysis had FGFR3 mutations. Considering all patients, the DCR was 50% with an ORR of 33%. Both are slightly lower than in the BLC2001 trial, which could be explained by the physician assessment of the computed tomography results instead of an independent radiological review committee. Another explanation could be that all patients in our study had failed on at least two lines of prior treatment as opposed to the BLC2001 trial where 45% of the patients received erdafitinib as second-line treatment.12,13 Also, prospective phase II and III trials usually have more rigorous inclusion and exclusion criteria to include a homogenous population with fewer comorbidities. Early access programs usually allow accrual of a less selective and a sicker real-world population, potentially impairing the outcome of the population. In addition, only one patient in the current report had erdafitinib dose up-titrated to 9 mg daily compared to 41% in the BLC2001 trial. Physicians’ inexperience with a new medication and/or the characteristics of a real-world population may have played a role in these slightly different results.
Regarding another parameter of erdafitinib effectiveness, when considering patients with PR, TTF was 5.3 months. Despite the difference in definition of TTF (an endpoint that includes toxicity in its definition) and duration of response (DOR), which was similar to the DOR of 5.6 months reported in the BLC2001 trial.12,13
Considering that the majority of patients (83%) in this report received prior PD-1 or PD-L1 inhibitors, we were able to evaluate the efficacy of ICIs. Only 1 out of 10 patients treated with erdafitinib had a response to prior pembrolizumab. The BLC2001 trial included a total of 22 patients that received prior ICI and only 1 patient responded.
Regarding the daily clinical practice and the applicability of erdafitinib, it is worth discussing what types of sample (tumor tissue and/or blood sample) and methods to use to identify the FGFR2/3 alterations. While the evaluation of DNA with next-generation sequence (NGS) techniques seems to be more versatile to identify FGFR2/3 alterations, able to be used in both tumor tissue (tumor DNA) and blood samples (cell-free DNA), the high cost and low availability of the use of these tests and the lack of health insurance coverage limit the use of DNA-based NGS tests. Furthermore, data suggest that DNA-based techniques can miss some FGFR2/3 alterations (fusion and/or mutation). In this context, to evaluate FGFR2/3 alterations in tumor tissue there is a preference for RNA-based techniques. 15 In our study, to assess the FGFR2/3 alterations, we used the same methodology applied in the BLC2001 trial, the RNA-based technique RT–PCR in tumor samples.
Another interesting finding in our analysis was the incidence rate of 25.5% of FGFR2/3 mutations. This is higher when compared to the 18.8% found in the BLC2001 trial, and the 20% described in medical data.11,12 This cohort was probably overestimated as some patients had already been tested and were found to have FGFR mutations before entering the program. Another explanation could be the selection bias considering the small sample size of patients (N = 12). In addition, there are data showing an incidence rate up to 37% of FGFR3 mutations in upper urinary tract tumors. 16 As we did not have this information in our cohort, it might have influenced our findings. Lastly, another explanation may be a different genomic profile in the bladder cancer of Brazilian population that is being addressed in the ongoing Latin American Cooperative Oncology Group 1518 trial. 17
Our study has some limitations. First, only 12 patients in the Brazilian erdafitinib EAP were included in the current analysis, therefore definitive conclusions about erdafitinib use based on this real-world evidence should be taken with caution. In addition, due to the small number of patients we could not evaluate the correlation of response/efficacy and type of FGFR alteration. Second, patients usually required a few weeks to evaluate eligibility with the FGFR alteration and receive erdafitinib, which could have caused tumor progression and decrease in the performance status.
In the context of safety of erdafitinib, severe AEs (grade ⩾ 3) were observed in 42% of the patients, which is similar to the 46% of AEs grade ⩾ 3 reported in the BLC2001 trial. Despite this, only 33% had dose reduction and one treatment discontinuation due to AEs. This was lower than what was reported in the BLC2001 trial, which had 55% dose reduction and 13% treatment discontinuations due to AEs.12,13 It is important to note that considering the possibility of serious eye disorders such as CSR that was described in 21% of patients using erdafitinib at a dose of 8 mg in the BLC2001 trial, it was mandatory that all patients eligible for EAP should undergo a complete ophthalmologic examination before starting treatment with erdafitinib and also if there were any eye symptoms without improvement after 7 days of erdafitinib dose interruption. Also, in our cohort, only one patient was able to titrate the dose to 9 mg, demonstrating the difficulty of managing side effects of a new drug.
The results of our study and the published phase II trial BLC2001 are summarized in Table 4.
Summary of results from Brazilian erdafitinib EAP and BLC2001 trial.
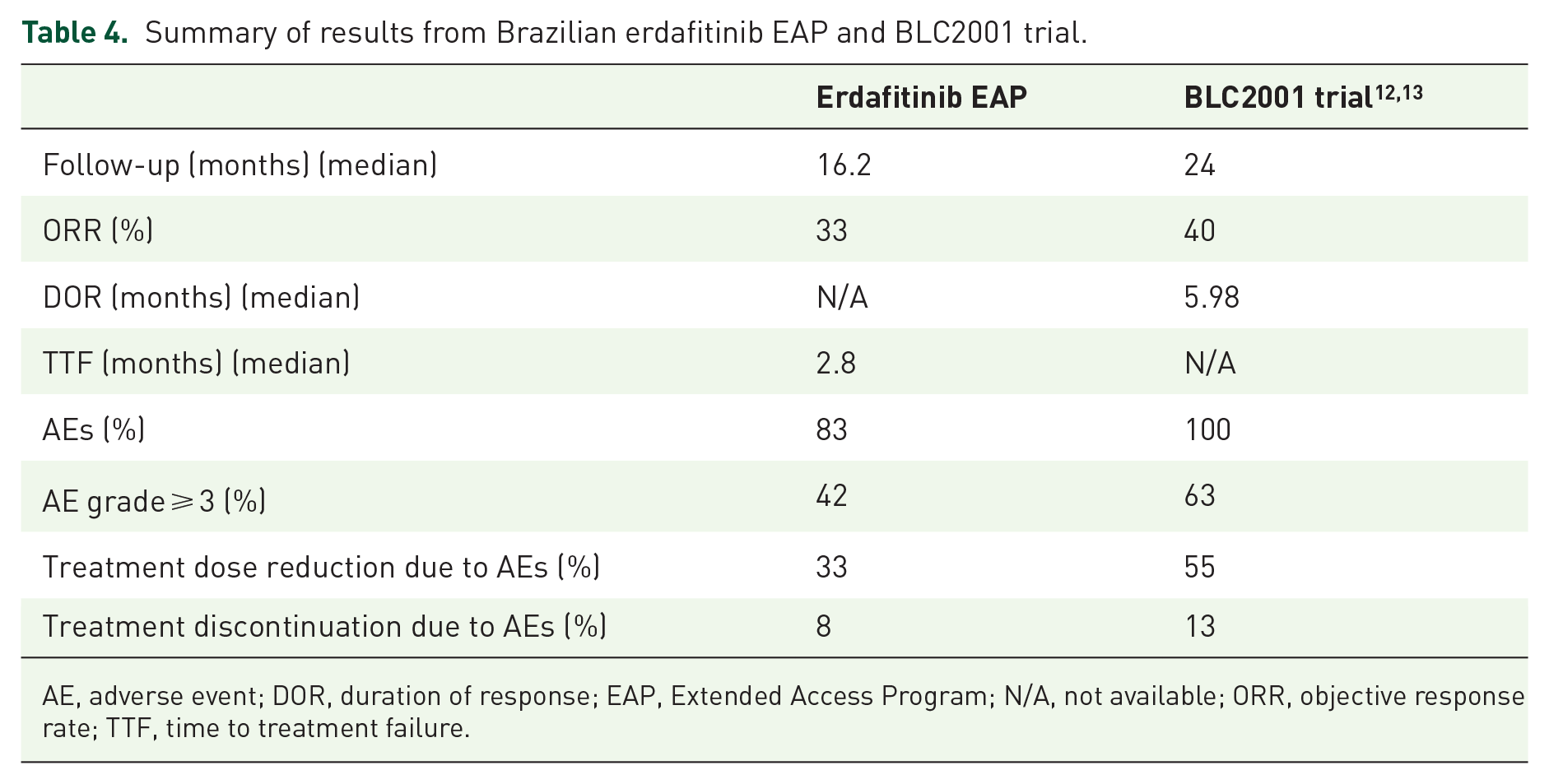
AE, adverse event; DOR, duration of response; EAP, Extended Access Program; N/A, not available; ORR, objective response rate; TTF, time to treatment failure.
Conclusion
To our knowledge, this is the first report with real-world evidence of an erdafitinib EAP for the treatment of patients with mUC. Our data confirm the activity demonstrated in the BLC2001 trial and support the approval of erdafitinib in Brazil for the treatment of patients with mUC harboring FGFR2/3 alterations. Treatment responses were observed in 30–40% of patients, and time to response and DOR were similar to that observed in the erdafitinib pivotal trial. 12
Adverse events are manageable; however, physicians can expect a learning curve to manage AEs and to titrate patients to the higher tolerable dose. These results support erdafitinib as a new treatment option for patients with mUC harboring FGFR alterations in addition to standard treatment options.
Footnotes
Acknowledgements
This erdafitinib EAP is sponsored by Janssen-Cilag Pharmaceutical. The authors, individually and collectively, are responsible for all content and editorial decisions and received no payment from Janssen-Cilag directly or indirectly (through a third party) related to the development of this publication.
Conflict of interest statement
The authors declare research support from Janssen; honoraria from Janssen, Astellas, Roche, MSD and BMS; and advisory board roles for Janssen, BMS and MSD.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations and informed consent
This EAP was conducted in accordance with Brazil’s National Health Surveillance Agency resolution RDC 38/2013, which does not require the evaluation and approval by an ethics committee or institutional review board. All patients who participated in erdafitinib EAP provided informed consent, and agreed to the use of clinical data, keeping personal data confidential.
