Abstract
Background:
No clinical trial has directly compared nab-paclitaxel/gemcitabine (nab-P/G) with FOLFIRINOX (fluorouracil/leucovorin/oxaliplatin/irinotecan) in metastatic or advanced pancreatic cancer (mPC or aPC). We conducted a systematic review of real-world studies comparing these regimens in the first-line setting.
Methods:
Results:
Of 818 records initially identified, 35 were duplicates and 749 did not meet the eligibility criteria, mostly because they were either not comparative (
Conclusions:
Although FOLFIRINOX was associated with slightly longer median OS in more studies, the differences, when available, were not statistically significant. Therefore, a randomized, controlled trial is warranted. Toxicity profile differences represent key considerations for treatment decisions.
Introduction
Pancreatic cancer is estimated to be the third leading cause of cancer-related mortality in the United States (US).1,2 As has been the case for many years, the projected number of deaths in 2019 (45,750) is expected to nearly equal the number of new cases (56,770).1,2 While the 5-year survival rate for all stages combined is approximately 9% (lowest among all cancers), more than half of all patients diagnosed with pancreatic cancer present with metastatic disease, which carries a 5-year survival rate of approximately 3%.1,2
Prior to recent treatment advances, single-agent gemcitabine was the standard of care for many years for patients with metastatic pancreatic cancer (mPC).
3
Today, gemcitabine monotherapy remains a therapeutic option for patients with mPC who have poor performance status,4,5 but for patients with good performance status, it has been shown to be inferior compared with two newer chemotherapy combinations. In the PRODIGE/ACCORD trial of patients with mPC, treatment with FOLFIRINOX (fluorouracil, leucovorin, oxaliplatin, and irinotecan) resulted in a median overall survival (OS) of 11.1 months
On the basis of these results, the current National Comprehensive Cancer Network and European Society for Medical Oncology guidelines recommend combination chemotherapy with nab-P/G or FOLFIRINOX as the preferred first-line treatments for patients with mPC who have good performance status.4,5 A recent study assessing the patterns and predictors of systemic therapy choices in mPC reported that the use of first-line gemcitabine monotherapy in the US decreased from 72% in 2006 to 16% in 2015. Conversely, there was a reciprocal increase in the use of either nab-P/G or FOLFIRINOX in this setting. 11 This study also found that patients treated at community practices and by oncologists with lower volumes of patients with mPC were more likely to receive nab-P/G as a first-line treatment, while younger male patients were more likely to receive FOLFIRINOX. 11 However, the rationale for choosing between nab-P/G and FOLFIRINOX remains unclear.
To date, there has been no head-to-head randomized, controlled trial comparing nab-P/G with FOLFIRINOX in patients with mPC. Therefore, a number of retrospective, nonrandomized studies from institutional or healthcare systems have compared nab-P/G and FOLFIRINOX in an attempt to elucidate differences in safety and effectiveness. However, cross-comparisons between trials are not ideal; for example, the numerically higher OS observed with FOLFIRINOX in PRODIGE/ACCORD
6
To address this need, we conducted a systematic review of real-world patient data comparing outcomes, including effectiveness, safety, duration of treatment, supportive care use, and resource utilization, with nab-P/G
Methods
Search strategy
The
Eligibility criteria
Abstracts were separately screened by two independent reviewers and any discrepancies were resolved through discussion and consensus. Studies included were real-world, retrospective analyses of first-line therapy in patients with mPC or aPC that directly compared nab-P/G with FOLFIRINOX. Eligible studies were required to have data on effectiveness [OS, PFS, time to treatment failure (TTF), or overall response rate (ORR)], treatment duration, or resource utilization. Studies with radiotherapy and review articles were excluded, and duplicates were removed.
Data extraction and reporting
Selected studies were reviewed and data on populations, interventions, and outcomes were extracted from abstracts, posters, and full papers into a database. Data included region, treatment period, number of patients (total and in each arm), baseline and clinical characteristics (including ECOG PS), effectiveness and safety outcomes, treatment duration, cost of therapy, resource utilization, and second-line treatment, including any associated outcomes. The quality of the included studies was independently assessed by two reviewers using the Newcastle–Ottawa Scale (NOS). 13 Any disagreements were resolved by a third reviewer. The NOS was developed to assess the quality of nonrandomized case-control and cohort studies based on three parameters, selection, comparability, and exposure/outcome, and assigns a maximum of 4, 2, and 3 stars, respectively, for these domains. A study was considered as high quality if the NOS score was ⩾7 stars.14,15 Because some studies did not report patient numbers, the values reported in this review are noted as being greater than the sum of the patient numbers reported in each particular category. In addition, the data reported varied by study (e.g. decimals/no decimals); we chose to represent all values as they appeared in the original reports for accuracy.
Results
Study selection
The initial search identified 818 records, of which 35 were duplicates; an additional 749 were excluded based on eligibility criteria, leaving 34 studies for further analysis (Figure 1).

Study selection.
Study and population characteristics
The 34 studies included patients treated between 2000 and 2018 in North America, Europe, and Asia (Table 1). Of these, 21 studies (62%) assessed only patients with mPC. Among 13 studies that assessed patients with aPC, 4 did not report the breakdown of the number of patients with aPC and mPC, and in the remaining 9 studies, most patients [1725 of 2205 (78%)] had mPC. Overall, 16,505 patients, including >11,476 (70%) with mPC, were assessed. Of these, >6915 patients, including >6349 (92%) with mPC, received nab-P/G or FOLFIRINOX. The numbers of patients treated with nab-P/G or FOLFIRINOX appear lower than the numbers of all assessed patients because some studies did not report the numbers separately for each regimen, while other studies evaluated additional regimens.
Study and population characteristics.
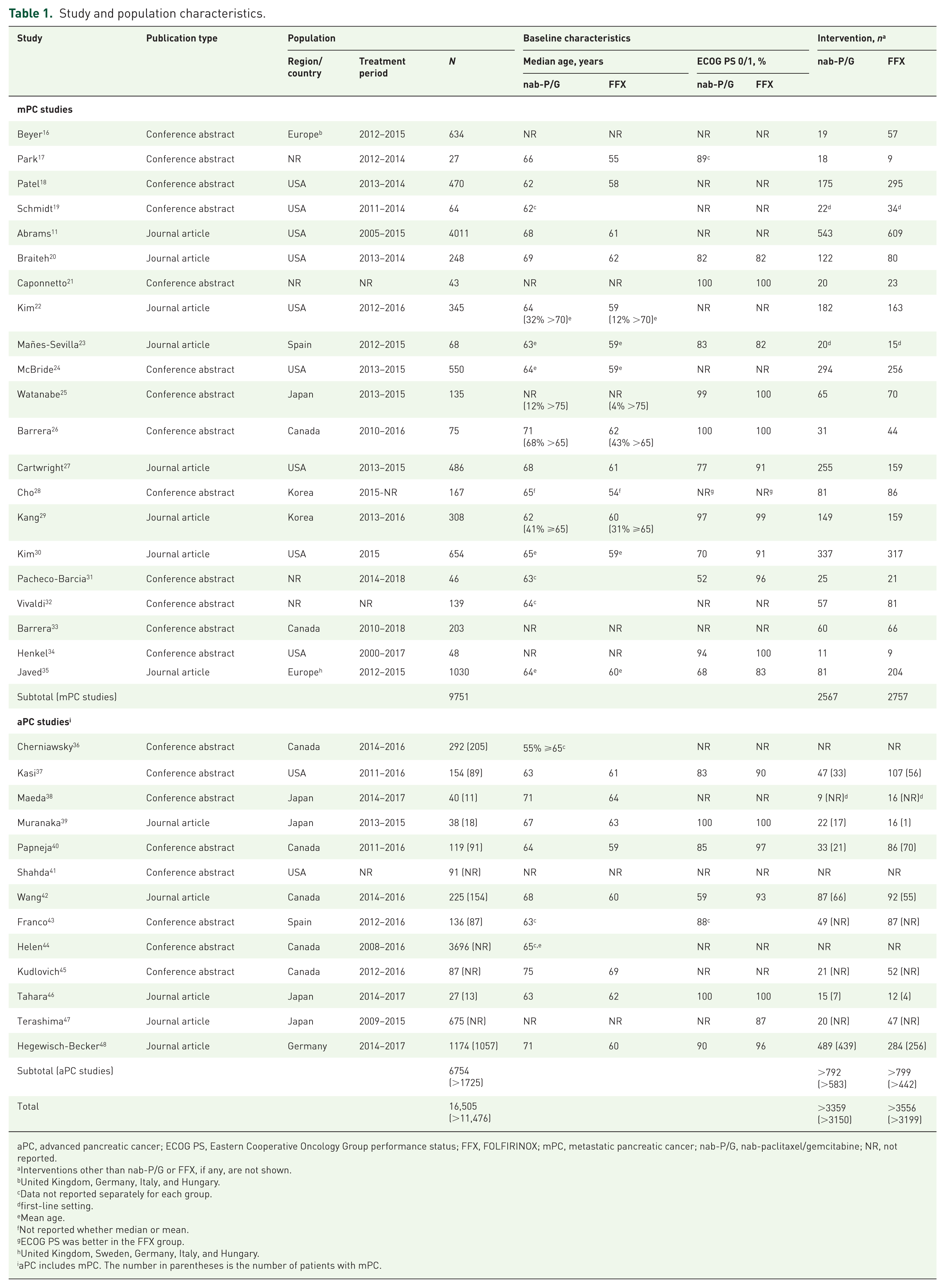
aPC, advanced pancreatic cancer; ECOG PS, Eastern Cooperative Oncology Group performance status; FFX, FOLFIRINOX; mPC, metastatic pancreatic cancer; nab-P/G, nab-paclitaxel/gemcitabine; NR, not reported.
Interventions other than nab-P/G or FFX, if any, are not shown.
United Kingdom, Germany, Italy, and Hungary.
Data not reported separately for each group.
first-line setting.
Mean age.
Not reported whether median or mean.
ECOG PS was better in the FFX group.
United Kingdom, Sweden, Germany, Italy, and Hungary.
aPC includes mPC. The number in parentheses is the number of patients with mPC.
In general, patients who received nab-P/G were older and had worse performance status than those who received FOLFIRINOX. Age was reported separately for the two groups in 21 studies, and the median/mean age of patients who received nab-P/G was numerically greater than those who received FOLFIRINOX (Table 1). In 11 of the 17 studies that reported ECOG PS separately for the two groups, a higher proportion of patients had good performance status (ECOG PS of 0 or 1) in the FOLFIRINOX cohort; 5 studies had equal proportions, and 1 had a higher proportion of patients with good performance status in the nab-P/G group (Table 1).
Of the 32 included studies, 23 (72%) were assessed by the NOS to be of high quality (⩾7 stars; Supplementary Table 1). The remaining nine studies were of moderate quality (seven studies with 6 stars; two studies with 5 stars), primarily due to comparability and outcome biases. The mean NOS score across all studies was 7.47. The most common comparability bias was the absence of study-controlled factors in addition to the compared patient population. The most common outcome bias was the lack of reporting of adequate follow up. Several studies did not receive an NOS star for selection and outcome parameters because they did not report a specific record or database used to ascertain exposures or assess outcomes.
Effectiveness outcomes
Overall, 31 studies (>5237 patients) reported an OS, PFS, or TTF (Table 2). Effectiveness outcomes generally overlapped between patients who received nab-P/G and those who received FOLFIRINOX. A total of 27 studies (>4173 patients) of various populations (aPC, mPC, ECOG PS 0/1) reported a total of 32 median OS values with FOLFIRINOX and 30 with nab-P/G (2 were reported as ‘not reached’; Table 2). Among the 30 direct comparisons, a numerically longer median OS was reported with FOLFIRINOX in 18
Effectiveness outcomes: OS, PFS, and TTF.

1L, first line; aPC, advanced pancreatic cancer; ECOG PS, Eastern Cooperative Oncology Group performance status; FFX, FOLFIRINOX; mPC, metastatic pancreatic cancer; nab-P/G, nab-paclitaxel/gemcitabine; NR, not reported; OS, overall survival; PFS, progression-free survival; TTF, time to treatment failure.
Reported as database persistence, a proxy for OS.
Reported as time to discontinuation.
Modified FFX.
For patients with ECOG PS 0 or 1, median OS was 12.1 and 11.4 months in nab-P/G and FFX groups, respectively
(
For patients with ECOG PS 0 or 1, median TTF was 4.2 and 4.3 months in nab-P/G and FFX groups, respectively.
59% of patients received modified FFX. The OS and PFS were reported to be statistically similar between FFX and mFFX groups.
For patients with ECOG PS 0 or 1, median OS was 14.1 and 13.7 months in nab-P/G and FFX groups, respectively.
aPC includes mPC. The number in parentheses is the number of patients with mPC.
Biomarker study observing homologous recombination deficiency low

Overall survival in patients with advanced or metastatic pancreatic cancer receiving nab-P/G or FOLFIRINOX.
Among the 18 direct comparisons (10 in patients with mPC and 8 in those with aPC; >2388 patients), the median PFS was numerically longer with FOLFIRINOX in 10 studies
Overall, six studies reported response data. In two studies that reported response data for patients with mPC, the ORRs were 34% and 39% with nab-P/G
Treatment duration
A total of 10 studies reported the duration of treatment. Patients who received nab-P/G were treated for a median of 95 to 261 days
Second-line chemotherapy
A total of 10 studies (2184 patients) reported data on the proportion of patients who received second-line chemotherapy. In nine studies that reported data separately for the two regimens, 9–76% of patients treated with nab-P/G as first-line therapy received second-line therapy
Safety outcomes
Safety outcomes were reported in 14 studies (2205 patients), including 8 single-institution studies. The incidence of all grade and grade 3/4 adverse events (AEs) is summarized in Table 3. Among the six single-institution studies to report grade 3/4 neutropenia, the rates were higher with FOLFIRINOX in five studies
Safety outcomes.

AE, adverse event; aPC, advanced pancreatic cancer; DVT, deep vein thrombosis; FFX, FOLFIRINOX; GI, gastrointestinal; mPC, metastatic pancreatic cancer; nab-P/G, nab-paclitaxel/gemcitabine; NR, not reported; PE, pulmonary embolism; WBC, white blood cell.
Reported as polyneuropathy.
Single-institution studies
Reported as sensory neuropathy.
Grade 3 or higher.
Reported as treatment suspension.
Used modified FFX.
Grade ⩾3 neutropenia and GI events were reported to be more common in the FFX group.
aPC includes mPC.
Reported as neuropathy.
Supportive care/resource utilization
A total of five studies (1986 patients) reported data on supportive care or resource utilization.18,20,22,25,27 In two studies (605 patients) that reported frequency of granulocyte colony-stimulating factor (G-CSF) use, fewer patients treated with nab-P/G received G-CSF (0% and 27%)
Cost of care
A total of four studies (1532 patients) reported data on costs associated with healthcare in patients treated with nab-P/G and FOLFIRINOX.18,22,24,28 Overall, two studies reported that the total monthly cost of care for patients treated with nab-P/G was US$16,628 and US$23,605
Discussion
This systematic review reports data from 34 studies that assessed first-line treatment in a real-world setting of 16,505 patients, including at least 6349 patients with mPC who were treated with nab-P/G or FOLFIRINOX (see Table 1). Although the effectiveness of nab-P/G and FOLFIRINOX in patients with mPC or aPC varied among studies, most of the survival data tended to overlap (see Table 2 and Figure 2). Furthermore, in 12 of 14 statistical comparisons, the OS was not significantly different between patients treated with nab-P/G
In most studies reporting safety, the authors concluded that nab-P/G exhibited a more favorable safety profile than FOLFIRINOX. Some studies reported that patients treated with nab-P/G experienced fewer and less severe AEs, needed dose modifications less frequently, and discontinued less often because of AEs. In real-world studies, comparing safety data, especially nonlaboratory-based AEs, is problematic due to the lack of uniform definitions as used in randomized clinical trials; therefore, we focused on comparing grade 3/4 AEs reported for the two regimens within single-institutional studies. In most of these studies, the rates were higher with FOLFIRINOX
In 2014, Gresham and colleagues performed a systematic review and network meta-analysis of randomized clinical trials of chemotherapy regimens in patients with aPC (9989 patients in 23 studies) that included nab-P/G and FOLFIRINOX.
12
Our observation that there was no clear distinction in effectiveness between nab-P/G and FOLFIRINOX is consistent with their report of no significant difference in OS or PFS between the two regimens. In direct comparisons, FOLFIRINOX was associated with significantly higher odds of grade 3/4 neutropenia
In the PRODIGE/ACCORD trial, the most common grade 3/4 AEs with FOLFIRINOX were neutropenia (46%), fatigue (24%), vomiting (15%), diarrhea (13%), thrombocytopenia (9%), sensory neuropathy (9%), anemia (8%), thromboembolism (7%), and febrile neutropenia (5%). 6 In the MPACT study, the most common grade ⩾3 AEs with nab-P/G were neutropenia (38%), fatigue (17%), peripheral neuropathy (17%), thrombocytopenia (13%), anemia (13%), diarrhea (6%), and febrile neutropenia (3%). 7 Post hoc analyses of the MPACT trial also demonstrated that nab-P/G dose reductions and dose delays are additional strategies that can help reduce toxicity without compromising efficacy, and that prolonged first-line treatment with nab-P/G until disease progression can improve survival rates.49,50 Similarly, various modifications in FOLFIRINOX components and doses are often experimented with in clinical practice in efforts to improve outcomes.
The recent results from the PRODIGE 24-ACCORD trial showed significantly longer disease-free survival and OS in patients receiving a modified FOLFIRINOX regimen (without bolus fluorouracil) compared with those receiving gemcitabine in the adjuvant setting. 51 The most common grade 3/4 AEs reported with the modified FOLFIRINOX regimen were neutropenia (28%), diarrhea (19%), increased γ-glutamyltransferase levels (18%), paresthesia (13%), fatigue (11%), sensory peripheral neuropathy (9%), nausea (5%), and vomiting (5%). 51 These results may support tolerability of modified FOLFIRINOX in the adjuvant setting and suggest the potential for use in patients with mPC. The National Comprehensive Cancer Network guidelines were revised recently to include modified FOLFIRINOX as a preferred category 1 recommendation for mPC. 5
The results from the MPACT and PRODIGE trials brought meaningful promise to the treatment landscape for patients with mPC; therefore, the focus now is centered on determining the optimal sequence of these regimens. As a result of the phase III NAPOLI-1 trial, liposomal irinotecan plus fluorouracil and leucovorin is a National Comprehensive Cancer Network category 1 recommendation for patients who received first-line gemcitabine-based therapy.5,52,53 Further, several early-stage clinical trials have investigated the sequencing of nab-P and fluorouracil-based regimens. In GABRINOX, a phase I/II study that assessed treatment with nab-P/G followed by FOLFIRINOX in patients with mPC, a median OS of 17.8 months was reported. 54 However, this regimen resulted in grade 3/4 neutropenia, thrombocytopenia, and diarrhea occurring at higher frequencies compared with nab-P/G or FOLFIRINOX alone.6,7 In SEENA-1, a phase II study of nab-P/G followed by either modified FOLFIRINOX (without bolus fluorouracil) or nab-P/G alternating with FOLFIRI (without oxaliplatin), a median OS of 12.3 months and a safety profile generally similar to that for nab-P/G or FOLFIRINOX alone were reported.6,7,55 A phase II study recently showed that using FOLFIRINOX in a stop-and-go fashion (4-month FOLFIRINOX followed by LV5FU2 maintenance) produced similar efficacy (PFS, 5.7 months; OS, 11.2 months) compared with 6-month FOLFIRINOX (PFS, 6.3 months; OS, 10.1 months). 56 However, this strategy resulted in a greater proportion of patients with grade 3/4 neurotoxicity (19%) compared with standard FOLFIRINOX (10%). Finally, multiple algorithms have been proposed to guide treatment decisions in individual cases.57,58 Future efforts may shed additional light on appropriate sequencing regimens for personalized care.
Study limitations
The sample size of 34 studies may be considered relatively small, and this may impact the ability to draw strong conclusions from the data. In addition, in the absence of standardizing criteria used in randomized clinical trials, interpretation of certain outcomes (e.g. response data) is problematic with real-world evidence from multi-institutional studies. Furthermore, the studies varied in terms of population, treatment duration, study design, and details of specific results. Some of the differences in patient characteristics may also have affected the observed outcomes. For example, healthier/younger patients treated more frequently with FOLFIRINOX
In addition, studies of patients with aPC were included to maximize the inclusion of patients with mPC. The OS, a more reliable indicator of effectiveness than other measures, was reported in only approximately 50% of the patients treated with nab-P/G or FOLFIRINOX. To date, data from most of the included studies were not completely mature, and some studies did not report patient numbers, which may have added to the variability and lower quality of some studies caused by a reporting bias. Overall, the variability did not allow for performance of a robust meta-analysis with specific comparisons of effectiveness and safety outcomes.
Conclusion
To our knowledge, this is the first systematic review examining real-world outcomes with nab-P/G
Supplemental Material
Supplementary_Table_1 – Supplemental material for Real-world comparative effectiveness of nab-paclitaxel plus gemcitabine versus FOLFIRINOX in advanced pancreatic cancer: a systematic review
Supplemental material, Supplementary_Table_1 for Real-world comparative effectiveness of nab-paclitaxel plus gemcitabine versus FOLFIRINOX in advanced pancreatic cancer: a systematic review by Elena Gabriela Chiorean, Winson Y. Cheung, Guido Giordano, George Kim and Salah-Eddin Al-Batran in Therapeutic Advances in Medical Oncology
Footnotes
Appendix
Search terms and strategy.

| Coverage and strategy | Search terms |
|---|---|
| No. 1 (populations) | Pancreatic neoplasms OR pancreas cancer OR pancreas adenocarcinoma OR pancrea* n/3 adenocarcinoma OR pancrea* n/3 neoplasm |
| No. 2 (interventions) | Albumin bound paclitaxel OR nab-paclitaxel OR abraxane OR folfirinox |
| No. 3 (study design) | Observational OR registries OR retrospective* OR population OR prospective OR claims database OR electronic medical OR health records OR single center study |
| No. 4 (outcomes) | Treatment pathway OR overall survival OR progression-free survival OR disease-free survival OR disease progression OR ORR OR CR OR PR OR quality of life OR HRQOL OR QOL utility OR EQ-5D OR SF-6D OR functional assessment of cancer therapy OR FACT OR EORTC QLQ-C30 OR treatment and pathway OR sequence OR duration OR modification OR failure OR patient-reported outcome OR patient-reported outcome measures OR patient-reported outcomes OR health care utilization OR resource utilization OR hospitalization |
| Final search | Nos. 1 AND 2 AND 3 AND 4 |
Acknowledgements
The authors thank Padmini Tandavakrishna, MS, for her assistance with the literature search. Writing assistance was provided by Narender Dhingra, MBBS, PhD, of MediTech Media, Ltd., and funded by Celgene Corporation. MediTech Media also provided editorial support. The authors are fully responsible for all content and editorial decisions for this manuscript.
Funding
This work was supported by Celgene Corporation.
Conflict of interest statement
EGC has served in an advisory role for Pfizer, Novocure, Genentech/Roche, Celgene, AstraZeneca, Eisai, Five Prime, Vicus, Halozyme, Seattle Genetics, Ipsen, and Array; she has received travel, accommodations, expenses from AstraZeneca; and her institution has received research funding from Celgene, Incyte, Stemline Therapeutics, Ignyta, Merck, Lilly, and Boehringer Ingelheim. WYC declares no conflict of interest. GG has received honoraria from Celgene and Sanofi; consulting or advisory role for Celgene; and he has received travel, accommodations, expenses from Celgene. GK has been a consultant and speaker for Celgene and Ipsen. SEAB has been in a consulting or advisory role for Merck, Roche, Celgene, Lilly, Bristol-Myers Squibb, and SERVIER; he has been on the speakers’ bureau for Lilly, Roche, Celgene, and Nordic Bioscience; he has received funding from Celgene, Roche Pharma AG, Lilly, Novartis, Vifor Pharma, Medac, and Hospira.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
