Abstract
Cardiac tumors are rare and complex entities. Early assessment and differentiation between non-neoplastic and neoplastic masses, be they benign or malignant, is essential for guiding diagnosis, determining prognosis, and planning therapy. Cardiac sarcomas represent the most frequent primary malignant histotype. They could have manifold presentations so that the diagnosis is often belated. Moreover, considering their rarity and the limitation due to the cardiac location itself, the optimal multimodal management of patients affected by primary cardiac sarcomas still remains highly difficult and outcome dismal. Therefore, there is an urgent need to improve these results mainly focusing on more adequate tools for prompt diagnosis and exploring new and more effective therapies. Knowledge about the molecular landscape and pathogenesis of cardiac sarcoma is even more limited due to the rarity of this disease. In this sense, the molecular characterization of heart tumors could unfold potentially novel, druggable targets. In this review, we focused on genetic aberrations and molecular biology of cardiac sarcomas, collecting the scarce information available and resuming all the molecular findings discovered in each tumor subtype, with the aim to get further insights on mechanisms involved in tumor growth and to possibly highlight specific molecular profiles that can be used as diagnostic tests and unveil new clinically actionable targets in this tricky and challenging disease.
Keywords
Background
Cardiac tumors, both benign and malignant, are rare diseases. They account only for a small fraction of all cardiac masses, mostly represented by pseudo-tumors like thrombi, vegetations, and aneurysms. The majority of initial medical reports on cardiac tumors were based on autopsy findings. With echocardiography increasingly used since the 1960s, successful diagnosis of ante-mortem cardiac tumors has risen. Their estimated frequency ranges from 0.0017% to 0.33%, mostly represented by benign tumors (75%), with cardiac myxomas being the most common accounting for nearly half of them. 1 Among malignant cases, secondary tumors of the heart, either by metastatic spread or by direct invasion, are far more common than primary cardiac tumors. Primary tumors are mainly sarcomatous in nature, whereas metastases could originate from various organs, generally from lung. 2
Location, morphology, and extent of the mass provide important and meaningful information; half of the primary tumors of the right atrium are malignant while most left atrium tumors are benign. Other imaging findings suggestive of malignancy include involvement of more than one cardiac chamber, size >5 cm, hemorrhagic pericardial effusion, broad base of attachment, and extension into the mediastinum. 3 Early assessment and differentiation between non-neoplastic and neoplastic masses is essential for guiding diagnosis, determining prognosis, and planning therapy.
Whilst being extremely rare, sarcomas of the heart represent the most frequent primary malignant histotype (75–95%). Virtually all types of sarcomas may be observed in the heart with a predominance of angiosarcomas (AS) or undifferentiated sarcomas, while other sarcomas are less frequently described (leiomyosarcoma, rhabdomyosarcoma, and myxofibrosarcoma). 4 They could have clinical presentations ranging from cough and general fatigue to thromboembolism, pericardial effusion, arrhythmias, dyspnea, hemoptysis, or symptoms of congestive heart failure depending on tumor location and on pathological subtype. The tumor site can give an indication of the most probable tumor type in some cases, with the vast majority of AS and myxoma developing in the right atrium and left atrium, respectively.
Due to the rarity of the disease, evidence for the optimal multimodal management of primary cardiac sarcomas is limited. There is no standardized approach, which is then often extrapolated from that of soft tissues sarcomas (STS) in other sites, thus adopting a “one size fits all” approach. Moreover, inevitable limits due to the cardiac location itself must be considered. 5 Complete surgical resection provides the greatest chance for survival in primary localized cardiac sarcoma, but it is rarely achievable in practice given the aggressive and infiltrative nature of these tumors. Moreover, radiotherapy, an effective adjuvant tool for sarcomas of the extremities, is barely applicable to cardiac sarcomas considering that the heart itself is usually sensitive to radiation injury leading to substantial risk of cardiomyopathy and chronic pericarditis. Finally, the typical cardiotoxicity of anthracyclines, which represent the preferred chemotherapeutic choice for sarcomas, could limit their use in neoadjuvant/adjuvant or advanced settings in these cases.
Even if based on retrospective, small, and heterogeneous case series, sarcomas of the heart seem to be very aggressive tumors with a dismal prognosis; tumor recurrence and metastases represent frequent and often early events. A high rate of both relapse and distant metastases within the follow-up period is reported in a series of malignant cardiac tumors seen over 15 years (45.5% and 72.2%, respectively) 6 and the median overall survival (OS) is scanty: in older retrospective case series, most patients died within 12–16.5 months after initial diagnosis. 7 In more recent series, survival reaches 38.8 months but only for patients who underwent complete resection in referral centers. Long-term survivals are uncommon and significantly depend on the histotype. 4
Knowledge about the molecular genetics and pathogenesis of cardiac sarcoma is even more limited. Most publications consist of case reports, with only a few reporting clinical/pathological and molecular features of cohorts of cardiac sarcomas (Table 1).
Molecular analysis of primary cardiac sarcoma available in literature.

In this review, we focus on genetic aberrations and molecular biology of cardiac sarcoma, collecting the scarce information available and summarizing molecular findings of each tumor subtype with the aim of highlighting specific molecular profiles that may be used as diagnostic tests or potential targets for therapy in this complex and challenging disease.
Angiosarcoma
AS is the most common differentiated cardiac sarcoma, generally originating in the right atrium. It represents about 25–40% of cases of cardiac sarcoma, occurring more frequently in men within the third and fifth decade of life (Figure 1). 36
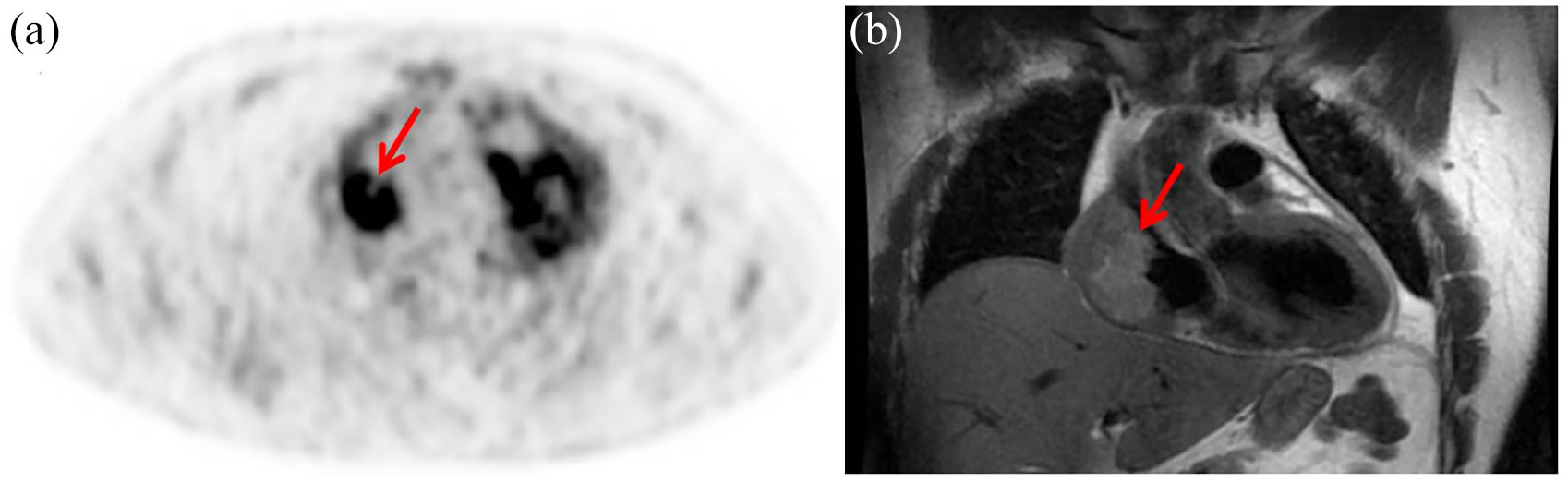
Angiosarcoma of the right atrium in a 38-year old man. 18F-FDG PET/CT shows abnormal uptake (SUVmax = 8.7) in the right atrium (a). FDG uptake corresponds to the voluminous (6 × 4.7 × 3.4 cm) neoplastic mass showed by the coronal image of the cardiac MRI (b). For clarity, the uptake visible on the left refers to the physiological FDG uptake due to the contractile activity of the left ventricle.
Cardiac AS is defined by endothelial differentiation and characterized by a high proliferation rate, permeating and destructive nature, propensity to metastasize, and poor prognosis with patients succumbing to disease within a few months of diagnosis.
37
Molecular aberrations detected in cardiac AS often involve
It has been shown that almost half of soft tissue AS carry recurrent somatic mutations in angiogenic signaling pathways, providing a rationale for investigating targeted therapies. In particular these mutations involve
Indeed,
Another frequent genetic alteration in soft tissue AS is
Alterations of
Alterations of the
Other mutated genes detected in soft tissue AS (
With regards to the latter,
Regarding chemotherapy, doxorubicin-based regimens remain the recommended first-line schemes for AS, as for other histological subtypes of STS. For second and further lines, no specific algorithm of treatment has been established. Among possible strategies for STS, taxanes have shown efficacy specifically in AS. In 1999, Fata
Regarding targeted therapies, the most relevant clinical application in STS was observed in the treatment of gastrointestinal stromal tumors and dermatofibrosarcoma protuberans, revolutionizing the outcome of patients affected by these rare histotypes. For all other principal types of STS, the only approved tyrosine kinase inhibitor (TKI) is pazopanib, a multitargeted tyrosine kinase inhibitor, with activity against vascular endothelial growth factors 1, 2, and 3, and platelet-derived growth factors. Pazopanib demonstrated its activity in a randomized, placebo-controlled, phase III trial, showing a significant improvement in progression free survival (PFS) and a favorable trend in OS in patients with advanced non-adipocytic STS who had previously been treated with anthracycline- or ifosfamide-based chemotherapy. 46 However, it is not specified if any cardiac AS were included in the study.
There are few published data on pazopanib efficacy, specifically in vascular sarcomas. Considering the expression of pro-angiogenic growth factors, AS are inherently a target for antiangiogenic agents, thus the evaluation of vascular-targeted agents in these tumors is of particular interest. Recently, a retrospective study of patients with advanced vascular sarcomas, including AS, treated with pazopanib in real life practice was performed to specifically investigate its activity in these subtypes and provide a benchmark for clinical practice. 47 Results reported no significant difference in efficacy between cutaneous and non-cutaneous AS and radiation-associated and non-radiation-associated AS, showing an overall activity of pazopanib comparable with other STS subtypes. Once again, it is unclear if there were any cases of primary cardiac AS.
Based on the same assumptions, other monoclonal antibodies and TKI, such as bevacizumab, sorafenib and imatinib, mainly characterized by antiangiogenic mechanism of action, have been tested in AS alone or in combination with chemotherapy.48–52 In most trials, it is unclear whether primary cardiac AS are included within the study population, or, if they are, their number is too small to draw credible conclusions in the specific cardiac setting. The results of the principal studies are reported in Table 2. They were sometimes disappointing, sometimes interesting, but none eventually led to approval of these molecules in conventional treatment of AS.
Studies of monoclonal antibodies and tyrosine kinase inhibitors tested alone or in combination with chemotherapy in angiosarcoma.

AS, angiosarcoma; EHE, epithelioid hemangioendothelioma; mOS, median overall survival; mPFS, median progression free survival; pts, patients; RR, response rate; STS, soft tissue sarcoma.
A randomized phase III trial has recently compared the efficacy of pazopanib alone
Undifferentiated pleomorphic sarcoma
Undifferentiated pleomorphic sarcomas are cardiac sarcomas with no diagnostic histologic pattern or specific immunohistochemical profile. According to the most recent classification, undifferentiated pleomorphic sarcoma (UPS) replaces the now obsolete term pleomorphic malignant fibrous histiocytoma and includes intimal sarcoma (IS). 36 UPS generally arise in the left atrium, with a controversial prevalence depending on criteria adopted for diagnosis; in a broad definition, it represents the most common sarcoma type of the heart, accounting for at least 50% of cardiac sarcomas. 36
This classification of UPS will likely evolve in the coming years as more molecular and cytogenetic findings will be accumulated. Some authors have proposed intimal sarcoma as a distinct subtype of cardiac sarcoma, characterized by
Globally, cardiac UPS have a complex genomic profile with alterations reported in all chromosomes. Whilst amplification of
It is worth noting that other druggable mutations involving
In brief, given the clonal heterogeneity of intimal sarcomas and the potential crosstalk between different signaling pathways, resistance to a single-agent tyrosine kinase inhibitor is a common event and durable disease control is very unlikely to be achieved. Simultaneous targeting of more than one relevant pathway is a theoretical approach that should be explored in prospective clinical trials, while paying attention to monitoring toxicity, especially in heavily pre-treated patients.
Other histologies
Several other histologic subtypes of cardiac sarcoma are recognized. However, each of them accounts for some 10% of cardiac sarcomas 36 and the number of tumors analyzed at molecular level reported in literature is extremely limited. For each subtype, we briefly describe the main molecular alterations identified in other tissues and, where available, in the cardiac setting.
Myxofibrosarcomas (MFS) are approximately 10% of sarcoma of the heart and thought to arise from the fibroblasts of the cardiac connective tissue. Very little is known about clinical presentations, pathologic features, treatments, outcome patterns and molecular landscape of MFS (Figure 2).

Local relapse of myxofibrosarcoma of the left atrium in a 74-year old woman. 18F-FDG PET/CT shows faint uptake (SUVmax = 4.8) in the left atrium (a). Cardiac MRI (b) and CT scan (c) confirm the voluminous, solid formation (47 × 24 mm) adhering to the lower wall of the left atrium with trans-mitral development.
Sun
Primary cardiac osteosarcomas account for approximately 10% of cardiac sarcomas.
36
Only a few cases in literature described their clinical course and some cytogenetic analysis data, reporting allelic losses or imbalances involving 1p36, 9p22, 10q23, 17q22, and 22q13.
66
In general, conventional high-grade osteosarcomas are tumors with complex karyotypes. They are characterized by chromosomal instability, with numerous gross genomic mutations and rearrangements. About 33% of cases have been reported to harbor chromothripsis events and one half of all osteosarcomas exhibit patterns of localized hypermutation that are termed kataegis.
67
Hundreds of genomic rearrangements have been identified in osteosarcomas, including recurrent rearrangements of
Aggressive surgery and postoperative polychemotherapy (based on doxorubicin, cisplatin, ifosfamide, etoposide, mitomycin, methotrexate, and/or gemcitabine) are the most important strategies adopted in cardiac osteosarcoma treatment. However, the prognosis of this tumor is still very poor. 69
Rhabdomyosarcomas (RMS) are tumors showing skeletal muscle differentiation and they account for only 0–5% of all cardiac sarcoma. Most cases appear in children at a mean age of approximately 14 years, and the histologic features are usually of the embryonal subtype. 36
Molecular analysis of only one case of cardiac RMS, harboring a
Leiomyosarcomas (LMS) account for less than 10% of cardiac sarcoma. Histologically, they are composed of elongated and closely spaced spindle cells with smooth muscle differentiation. Wang
Synovial sarcomas (SS) can occur anywhere and in any age group, but frequently manifests in the distal extremities of young adults. Primary cardiac SS is an exceptionally rare entity, accounting for approximately 5% of cardiac sarcomas involving either the pericardium or chambers, with a striking male predominance. They can be either monophasic (more common; spindle cells only) or biphasic (less common; epithelial and spindle cells). From the molecular point of view, SS have a relatively stable genome and they are characterized by the pathognomonic reciprocal chromosomal translocation t(X;18) (p11.2;q11.2), which results in the fusion of the
Current multimodal treatment of cardiac SS includes surgical resection, radiation, chemotherapy and heart transplantation, the latter for selected cases (i.e. young patients with lower grade, small size, and less aggressive tumors). In general, numerous studies show that SS tend to have better survival rates and higher chemosensitivity than other STS subtypes, but these results mainly refer to SS of the extremities. Standard first and subsequent-line of chemotherapy for advanced, recurrent, and metastatic SS are represented by doxorubicin and ifosfamide, high-dose ifosfamide, and trabectedin. Investigational trials exploring multi-TKI agents (imatinib, pazopanib, regorafenib) have been conducted with mixed results. More recently, there has been some progress toward immunotherapeutic strategies. In particular, NY-ESO-1 is a cancer testis antigen, which is expressed in a substantial percentage of SS (49–76%). In this sense, treatment of patients with NY-ESO-1-positive tumors with genetically engineered lymphocytes seems promising and some clinical trials are currently ongoing.76–79
In conclusion, several recurrently altered genes have been identified in cardiac sarcomas mainly in AS and UPS, while adequate molecular characterization of rarer histologies is still lacking. Ideally, deeper and wider genetic analysis should be performed to complete our knowledge of the molecular landscape of all type of sarcoma of the heart.
Future perspective
Primary sarcomas of the heart are rare neoplasms. With manifold clinical manifestation, critical location and scarce evidence from literature, diagnosis of these tumors is often belated, clinical management significantly complex and outcome very poor. Therefore, there is an urgent need of more adequate tools for early diagnosis and more effective treatment. The molecular characterization of heart tumors could unfold potentially novel, druggable targets. Most literature on cardiac tumors consist of case reports and few articles provide details on their molecular profile. Even if several recurrently altered genes have been identified in cardiac AS and UPS, deeper and wider analysis is needed to complete the genetic footprints of these tumors and other cardiac sarcomas (Figure 3a). Most of these genes induce proliferation and angiogenesis through the activation of PI3K/AKT, PLCG1/PKC, and RAS pathways. These findings theoretically support the use of several targeted therapies, including TKI or monoclonal antibodies against KDR/EGFR/PDGFR and small molecules against their downstream signaling effectors (Figure 3b). Simultaneous targeting of more than one relevant pathway should be explored.
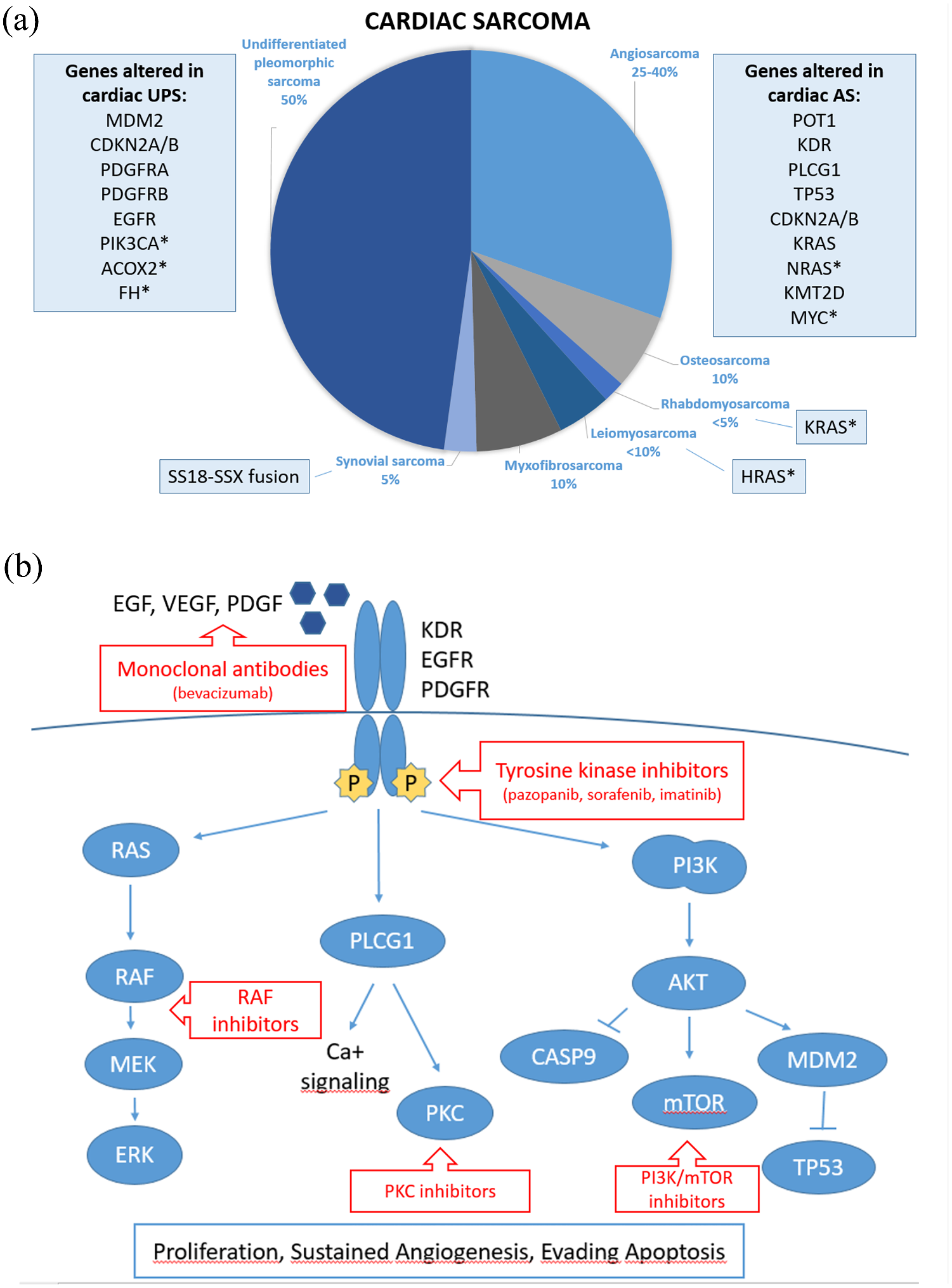
Mutational landscape and potential targeted therapy in cardiac sarcoma. (a) Frequency of each cardiac sarcoma histotype and altered genes identified so far. *indicates mutation reported in a single case. (b) Signaling pathways altered in primary cardiac sarcoma and potential therapeutic targets.
Next generation sequencing has revolutionized the study of genomics and, with decreasing cost per sample and wider accessibility of sequencing platforms, it is likely that more cardiac sarcomas will be characterized in the next future. However, molecular characterization without functional studies is insufficient for the evaluation of novel therapeutic strategies in this setting. The use of tumor models for functional evaluation of alterations identified in cardiac sarcoma is therefore essential and will require the development of adequate
