Abstract
Over the last decades, the concept of precision medicine has dramatically renewed the field of medical oncology; the introduction of patient-tailored therapies has significantly improved all measurable outcomes. Liquid biopsy is a revolutionary technique that is opening previously unexpected perspectives. It consists of the detection and isolation of circulating tumor cells, circulating tumor DNA and exosomes, as a source of genomic and proteomic information in patients with cancer. Many technical hurdles have been resolved thanks to newly developed techniques and next-generation sequencing analyses, allowing a broad application of liquid biopsy in a wide range of settings. Initially correlated to prognosis, liquid biopsy data are now being studied for cancer diagnosis, hopefully including screenings, and most importantly for the prediction of response or resistance to given treatments. In particular, the identification of specific mutations in target genes can aid in therapeutic decisions, both in the appropriateness of treatment and in the advanced identification of secondary resistance, aiming to early diagnose disease progression. Still application is far from reality but ongoing research is leading the way to a new era in oncology. This review summarizes the main techniques and applications of liquid biopsy in cancer.
Introduction
In recent years, the results of the Human Genome Project and pharmacogenomics research overcame the old paradigm of ‘one size fits all’ in oncology, providing a large amount of molecular data that generated the concept of ‘precision medicine’ with the aim of tailoring therapies for patients in relation to the personalized patterns of the tumor.1–3
As a consequence of their diagnostic potential, novel biotechnologies highlighted the limitations of current sampling methods: 4 traditional biopsies and surgical procedures are invasive, charged with potential complications, sometimes unrepeatable and cannot be performed when clinical conditions have worsened or when a tumor is inaccessible. 5 Furthermore, the genomic profile of biopsy tissues provides a tumor picture limited to a single point in time, and may also show the genetic heterogeneity of numerous tumor subclones. 6 In fact, many studies have established that the genomic landscape of tumors and metastases dynamically evolve over time in response to selective pressure of therapies that can suppress or promote the growth of different cellular clones. 5 These limitations are particularly evident in the presence of acquired resistance to therapy or in monitoring the disease during follow up. For these reasons, in recent years the new field of oncology research has focused on cancer-derived components that circulate in the bloodstream. 6 Apoptotic or necrotic cancer cells release circulating cell-free DNA fragments, designated as circulating tumor DNA (ctDNA), as well as exosomes (EXOs), namely membrane-encapsulated subcellular structures containing proteins and nucleic acids released by the tumor cells.5–8 Primary tumor and metastatic sites are also able to esfoliate vital cells that, once entered into the bloodstream, are circulating tumor cells (CTCs).
Isolation of these tumor-derived components from peripheral blood and their genomic or proteomic assessment represent a new diagnostic tool that has been called ‘liquid biopsy’ (Figure 1). The initial limitations due to the scarcity of nucleic acid as well as the difficulty in distinguishing between normal and tumoral nucleic acids 9 have been overcome by the increased sensitivity of next-generation sequencing (NGS) techniques, which now may accurately detect genetic and epigenetic aberrations. Liquid biopsy currently offers a high specificity, allowing the collection of robust and reproducible data in a simple and noninvasive way using a blood sample. 7 To date, liquid biopsy is not a routine test in clinical practice, but its potential applications are rapidly growing: from diagnostic genomic profiling to the monitoring of radicality in surgical outcomes, from evaluating either response or resistance to systemic treatments, to quantifying minimal residual disease. 4

Molecular applications of circulating tumor cells (CTCs), circulating tumor DNA (ctDNA) and exosomes as liquid biopsy for personalized medicine.
Here, we describe and compare the characteristics, the current detection techniques and the clinical applications of CTCs, ctDNA, and EXOs in cancer based on our own experience as well as that of others.
Circulating tumor cells
Characteristics and detection techniques
Since the first description in 1869 of tumor cells in peripheral blood, 10 substantial progress has been gained during the past few years from sustained biotechnological applications to isolate CTCs from heterogeneous blood components.
CTCs are shed from either primary or secondary tumor sites; they migrate into the circulatory system and are responsible for the development of distant metastases. 11 CTCs are extremely rare, occurring at a frequency as low as 1 CTC per 106–107 leukocytes, with even lower numbers in early stage diseases. 12 Initially assessed as nonleukocytic, nucleated cells of epithelial origin, CTCs do not have well defined morphological aspects and they may vary according to cancer type and stage. 11 CTCs may also cluster either with parental tumor cells or with fibroblasts, leukocytes, endothelial cells or platelets, forming aggregates with higher propensity to seed distant metastases than single CTCs, thanks to a their survival advantage 13 and to the protection from the immune system and oxidative stress.14,15
However, CTCs provide an ideal approach to molecular cancer diagnosis and treatment options, and their investigation is widespread in cancer research. Based on their properties, several systems have improved their detection and isolation, utilizing their physical differences compared with leukocytes, 16 including antigen expression (Table 1). The major differences are their large size, up to 20–30 µm, mechanical plasticity, and dielectric mobility properties compared with blood cells.17–20 Fruitful methods of isolation include membrane filtration, density gradient stratification, dielectric mobility, photoacustic and microfluidic separation.21–23 However, these techniques are sometimes inadequate in relation to their low specificity. 24 Antibody-based functional assays include cytometric high-throughput imaging, immunomagnetic and adhesion-based separation methods, 28 as well as negative leukocyte depletion and CTC recruitment by specific tumor markers sequentially adopted as depicted in Figure 2. 32
Different technologies to isolate CTCs.

CTC, circulating tumor cell.

Flow chart of combined methods to isolate circulating tumor cells (CTCs). (a) Peripheral blood samples are subjected to density gradient stratification and leukocyte depletion is assessed by an immunomagnetic method using anti-CD45 and anti-glycophorin conjugated microbeads (AUTOMACS (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany)). (b) The CTC-enriched fraction is stained by specific fluorochrome conjugated antibodies and loaded in a dedicated cartridge which then is subjected to dielectric forces (DEPArray (Menarini Silicon Biosystems, Firenze, Italy)). (c) CTCs are visualized by dedicated software and selected by positive fluorescence for tumor-specific markers and negativity for CD45 leukocyte marker. 4’,6-diaminidino-2-phenylindole (DAPI) is used to counterstain nuclei. The CTCs are moved into a parking area and recovered as single or grouped cells in a buffer drop.
However, there is no consensus on the specific antibodies to be used. CTCs express epithelial cell adhesion molecule (EPCAM), which is regarded as a biomarker reflecting a risk factor for tumor recurrence, 33 although cytokeratins, including CK8, CK18, CK19 26 and specific tumor markers (TTF-1, PSA, HER-2, etc.)34–36 are also useful for detection and isolation. On the other hand, EPCAM is usually lost during the epithelial to mesenchymal transition (EMT), the process that sustains both CTC migration and extravasation towards secondary sites, and the resistance to anoikis and apoptosis 37 that has recently been related to poor clinical outcome in breast, 38 colorectal, 39 prostate, 40 ovarian, 41 and non-small cell lung 42 cancers (NSCLCs). Furthermore, CTCs may acquire a stem-cell-like phenotype by expressing typical markers such as CD44, CD133, and aldehyde dehydrogenase (ALDH), and both proliferative and self-renewal properties favoring metastatization in secondary tissues.43,44 It is thus possible to find in the same blood sample CTCs with epithelial, EMT or cancer stem cells phenotype, 38 thus supporting their heterogeneity, and at the same time, limiting their purification and analysis. For this reason, it is necessary to combine differential methods to isolate functionally heterogeneous CTCs.
To date, the CellSearch (Menarini Silicon Biosystems, Firenze, Italy) assay, using the antibody-based immunomagnetic technique and image cytometry, is the only US Food and Drug Administration (FDA) approved CTC diagnostic technology for metastatic breast, prostate, and colorectal cancer, 27 whereas the recent DEPArray system allows both detection and recovery of single CTCs by surface or cytoplasmic markers, as well as size and dielectrophoretic movimentation properties 25 (Figure 3). An alternative method is based on either protein secretion or the migratory properties of CTCs 28 that allows the binding of these cells to synthetic substrates cotreated with specific complementary molecules. However, these methods are under intensive investigation to resolve both their complexity and low specificity.

The DEPArray technology is based on the use of a dielectrophoretic field (DEP) generated by electrodes in a matrix underlying a liquid layer of cells. (a) The DEPArray constellation for the creation of DEP is determined to be a determinant of the entire intrapolarization. (b) The whole cell is isolated from the individual software, and (c) after computational imaging, individual cells or groups of cells are moved and recovered by a drop of buffer in a specific tube.
The challenge of CTC detection is related to the requirement for high sensitivity combined with high specificity, 45 but several factors still hinder standardized clinical application, including the scarcity of CTCs in circulation; the absence of a reliable and efficient marker to distinguish CTCs from other blood-borne cells; 46 and the unaffordability of downstream molecular and genomic characterization in the case of a low number of detected CTCs. 47 Despite different approaches developed to detect CTCs, none of them completely meets the application requirements as loss of CTCs, low purity, and a narrow detection spectrum still need to be addressed. 29 Most of these methods include multiple operative processes, such as erythrocyte lysis, cell centrifugation and washing, which may lead to insufficient capture or cell damage and considerable time-consuming and expensive procedures. In addition, CTC detection and enumeration is still not included in routine tumor staging in clinical practice. These techniques need to be urgently assessed for research purposes, including obtaining viable CTCs for ex vivo expansion and translation to clinical application. 30 In this context, current nanotechnologies may help improve efficiency and specificity in capturing CTCs, 31 since nanomaterials show unique physical properties that can overcome the limitations of traditional CTC detection methods (Table 1).
Clinical applications of CTCs
Although it is necessary to optimize the standard procedure to isolate CTCs, their investigation in modern oncology definitely plays a pivotal role in conjugating basic research with clinical decision-making as a prognostic, diagnostic and predictive dynamic marker in everyday medical practice (Table 2).
Clinical implications of CTCs.

CTC, circulating tumor cell; NSCLC, non-small cell lung cancer.
The primary recognized role of CTCs is in prognosis; in a growing number of tumors, clinical investigators found that the absolute number of CTCs in a 7.5 ml blood sample is significantly associated with prognosis, with a usually neat cutoff value. In fact, in metastatic breast cancer, Cristofanilli and colleagues first demonstrated that patients with more than 5 CTCs in 7.5 ml blood have shorter progression-free survival (PFS) and overall survival (OS) compared with patients with a lower count. 33 In a retrospective analysis of patients with metastatic breast cancer at any line of treatment, either endocrine or chemotherapeutic, pretreatment values of CTCs higher than the established threshold of 5 CTCs/7.5 ml blood correlated with worse prognostic indexes based on an increased baseline number of metastatic sites, newly developed metastatic lesions and sites, shorter time to metastases and worse OS. 48 Similar results were obtained in patients with prostate cancer, in whom OS dropped to 11.5 months, namely one half, in the presence of more than 5 CTCs/7.5 ml versus 21.7 months in patients with less than this cutoff value. 49 The prognostic role of CTCs has also been confirmed in other cancers, including NSCLC, 50 colorectal, 51 gastric, 65 pancreatic, 66 head and neck cancers, 67 neuroendocrine tumors, 68 and sarcomas. 69
CTCs are also useful for diagnosis and could virtually be a substitute for tissue biopsy in cases of inaccessible neoplastic sites or unsuccessful sampling. In advanced neoplastic disease, CTCs may act as a dynamic diagnostic tool since they not only reflect the existence of a neoplasia, but they also run in parallel with the disease, thus increasing or decreasing in relation to the tumor burden, sometimes in a more accurate fashion than the usual soluble biomarkers.33,49,70,71 A recent meta-analysis including 50 studies with 6712 patients with breast cancer clearly demonstrated that CTCs may be a predictor of response to treatment, since the reduction of CTC counts during treatment is associated with longer OS and PFS. 53 Therefore, changes in CTC enumeration in serial assessment during treatment is predictive of therapy response, often at an earlier time than radiologic evidence. 28
Cancer screening is one of the goals of CTC research, but early detection is still an issue. Attempts have been made to use this property in the context of early diagnosis with encouraging results, although in limited cohorts of patients. For example, recent research on a cohort of patients with lung cancer and chronic obstructive pulmonary disease detected CTCs in 3% of patients. All of these patients developed lung cancer within 4 years, as documented by the onset of lung nodules on a spiral computed tomography (CT) scan and histotype diagnosis of early lung cancer after surgical resection of these lesions. 52 According to the same perspective, CTCs may also be helpful in distinguishing malignant from benign lesions. In a recent study, patients with newly diagnosed lung nodules were tested for the presence of CTCs before undergoing CT-guided fine needle aspiration. CTCs were found in 47 out of 67 patients with primary lung cancer, and in 9 out of 12 patients with secondary lung cancer, with a sensitivity of 70%, a specificity of 100% and a positive predictive value of 100%. 72
Once detected, CTCs are available for the analysis of their genomic and proteomic profiles, providing information on the presence of druggable molecular targets (Figure 4). A number of technical bias issues have been resolved, since NGS technologies have increased the sensitivity of mutational state detection and molecular information is now obtainable even from single cells. 32 The amount of DNA recoverable from a single cell corresponds to about 2–7 picograms. This small amount of nucleic acids is subjected to particular preamplification phases by whole genome amplification (WGA), which allows a sufficient amount of the sample to be obtained for molecular screening analysis. 32
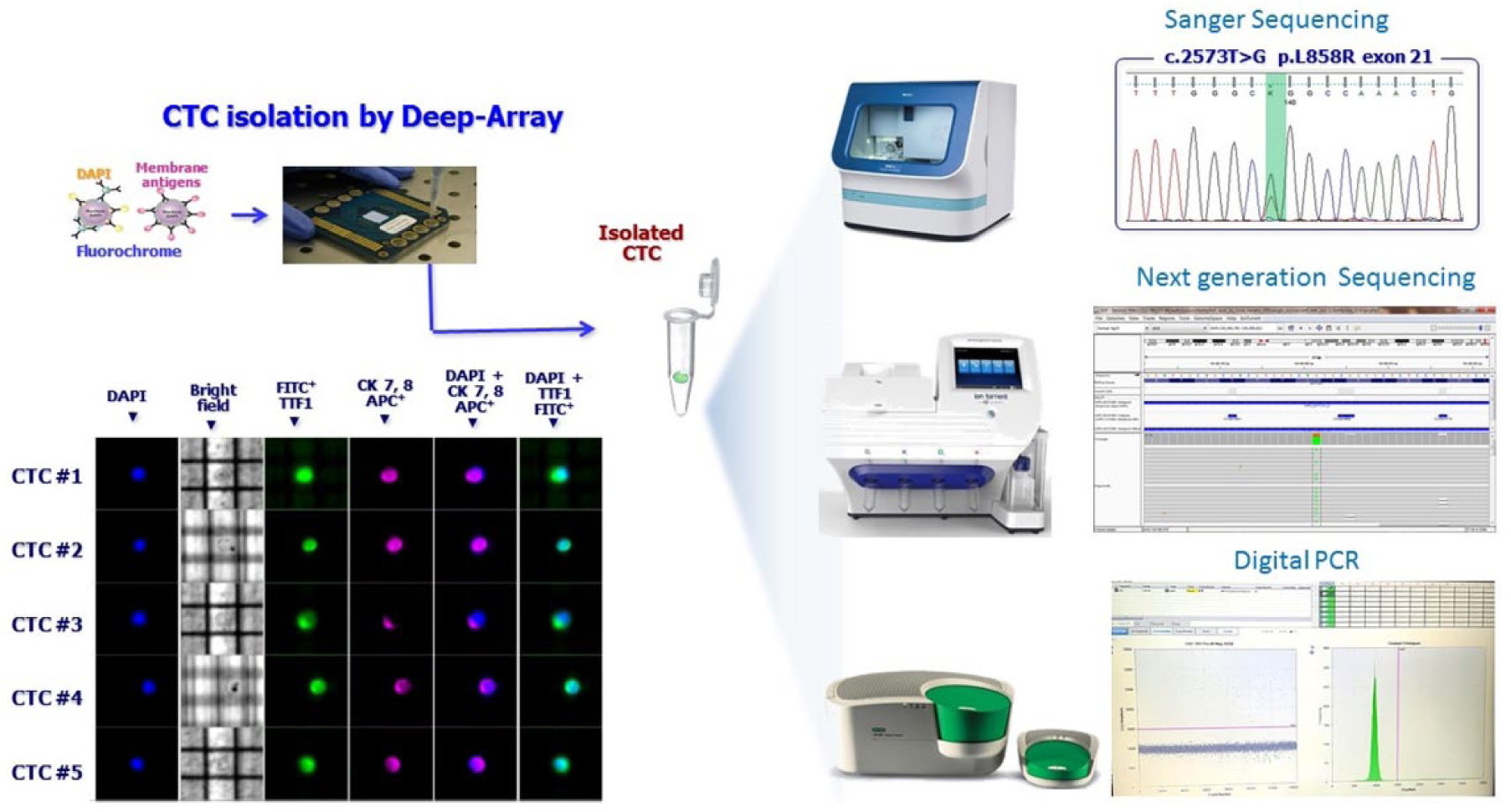
Mutational analysis performed on circulating tumor cells (CTCs) isolated by DEPArray using next-generation sequencing, Sanger sequencing or digital polymerase chain reaction (PCR). DAPI , 4’,6-diaminidino-2-phenylindole.
Combining the expression of ER, BCL2, HER2 and ki-67 on CTCs, a multiparameter endocrine therapy index has recently been proposed as a predictive factor of response to endocrine therapy in breast cancer. 54 However, it has been demonstrated in prostate cancer that both prostate-specific antigen (PSA) and prostate-specific membrane antigen (PSMA) expressed by CTCs are surrogates for androgenic receptor signaling and are considered as possible predictors of response to antiandrogenic therapy in these patients. 56 Data from mRNA in CTCs are also under intensive investigation to evaluate gene expression. In prostate cancer, splicing variants of ARv7 generate truncated forms of the androgen receptor that lack the binding domain and clinical studies showed that this abnormal androgen receptor expression on CTCs may predict the failure of antiandrogen therapy with abiraterone and enzalutamide.57,58 Also, DNA mutations play a primary role in modern cancer therapeutics since they affect driver genes that confer either sensitivity or resistance to therapies. A well known example of gene driver mutations is in melanoma in which BRAF gene derangements are fruitfully targeted by specific inhibitors and the mutational profile of cancer cells may classify different subsets of molecularly different melanomas even in CTCs. 59 The ability of CTCs to reflect the mutational profile of a cancer in a defined clinical sample has recently been explored 60 and suggests that dynamic changes in BRAF mutations may drive anti-BRAF treatment options. Another interesting gene in several tumors is epidermal growth factor receptor (EGFR), whose role is critical in NSCLC in which point mutations are successfully targetable by tyrosine kinase inhibitors (TKIs), hence acting as a positive predictive parameter for treatment response. 61 However, KRAS mutations in colorectal cancer are negative predictors of anti-EGFR response. A recent publication on the expression of KRAS in patients with colorectal cancer showed the high heterogeneity in the KRAS mutational profile, thus explaining the interpatient response variability.62,63
The role of CTCs as a predictive parameter of treatment response is also interesting in liquid biopsy research. Data show that CTC count has a potential role in real-time monitoring of response to therapies. In the first-line treatment of metastatic breast cancer, as part of the SWOG0500 clinical trial, the stratification of patients according to CTC count, at baseline level and during treatment, identified subsets of patients with significantly different survival rates, namely 35, 23, and 13 months respectively. 55 The potential power of CTC count in daily clinical practice is under further investigation, especially when CTC analysis might be used as a decisional treatment parameter. Among all ongoing trials, the METABREAST trial of the first-line treatment of metastatic breast cancer included patients with less than 5 CTCs/7.5 ml in blood treated with endocrine therapy, whereas patients with more than 5 CTCs received chemotherapy [ClinicalTrials.gov identifier: NCT01710605]. Further fields of interest are currently being investigated, with special attention paid to the relevance of CTCs in predicting the outcome of therapies using immune checkpoint inhibitors. It has been reported that patients with stage IV NSCLC treated with nivolumab and patients with programmed death-ligand 1 (PD-L1)-negative CTCs obtained a similar significant clinical benefit, in contrast to patients showing PD-L1-positive CTCs. 64 Given the growing importance of immunotherapy in modern oncology, many other studies are ongoing with the aim of improving this branch of cancer therapy.
CTCs can also provide information on the epigenetic changes in the cancer cells of patients. In particular, DNA methylation in liquid biopsy has been proposed as a potential biomarker for staging, prognosis, and monitoring of response. 73 The epigenetic silencing in promoter regions of tumor suppressor genes can be clearly confirmed in CTCs, as in the case of metastatic breast cancer, when methylation profiles of genes such as CST6 and BRMS1 strongly correlate with higher metastatization capacity and poorer prognosis. 74 Similar data have also been found in metastatic castration-resistant prostate cancer, in which methylation is more frequent in genes related to apoptosis, angiogenesis, and vascular endothelial growth factor (VEGF) signaling pathway. 75 Among all ongoing studies, evidence in colorectal cancer demonstrates that high-methylation profiles significantly correlate with BRAF mutations, hence patients have a poorer prognosis. 76 More than a prognostic tool, methylation profiling of CTCs may also have a predictive role. In metastatic breast cancer, the methylation of the estrogen receptor 1 (ESR-1) gene is associated with resistance to treatment with the combination of everolimus and exemestane. These data may give DNA methylation the role of a potential liquid biomarker for the follow up of patients with cancer undergoing chemotherapeutic treatment. 77 Research on the possible discordance in epigenetic modifications between CTCs and primary site derived cancer cells is still under evaluation, given that such differences may be attributable to tumor heterogeneity 78 and to the specific methylation of EMT-related genes, an evolutionary advantageous feature of cancer cells, which is significantly responsible for tumors spreading through the bloodstream. 79
CTC biology is also characterized by a high heterogeneity at the genetic, transcriptomic, proteomic, and metabolomic levels. In fact, CTCs represent a highly dynamic cell population that may originate from both primary tumor sites and metastases. 80 These cells change their phenotypic and molecular characteristics during the course of the disease under microenvironmental and therapeutic selective pressures. The first evidence of the phenotypic heterogeneity is the differential expression of the EMT markers in the same CTC population. 81 Regarding genomic heterogeneity, different mutations in PIK3CAQ have been demonstrated in single breast cancer CTCs from the same patient. 82 Similarly, in lung cancer, the EGFR gene T790M mutation was found in CTCs from patients with a primary tumor negative for these mutations. 83 Also, heterogeneity of gene expression is described in breast 84 and prostate 85 cancers. To date, these different levels of heterogeneity in CTC samples are proposed as useful tools at the prognostic level to recognize a relapse or to personalize the therapy.
Circulating tumor DNA
Characteristics and detection techniques
The first experimental evidence of cell-free DNA (cfDNA) in the blood was reported by Mandel and Metais in 1948. 86 Thereafter, except for the evidence of cfDNA in patients with systemic lupus erythematosus in 1966, 87 it was only in 1977 that cfDNA entered the field of oncology when Leon and colleagues demonstrated that the concentration of cfDNA was increased in patients with pancreatic cancer, and that in some cases its concentration decreased after therapy. 88 The clinical potential of cfDNA was recognized when Sorenson detected a mutation of the KRAS gene in a sample of plasma from patients with pancreatic cancer. 89 The fraction of cfDNA originated from tumor cells was named cell-free circulating tumor DNA (ctDNA).
To date, different mechanisms for ctDNA release in the bloodstream have been postulated. In normal conditions, cfDNA from apoptotic and necrotic cells is rapidly phagocytosed by macrophages and other scavenger cells90,91 and when the macrophage phagocytosis is exhausted, increased nucleosome amounts are released into the bloodstream, as proved by the evidence that most ctDNA fragments are 180–200 base pairs (bp) in length. 92 This is typical of apoptosis when the degradation of DNA into nucleosomal units generates this bp size of nuclear material. However, ctDNA can also be actively released into the bloodstream by living cells. Bergsmedh and colleagues suggested that this transfer of ctDNA is capable of mediating the metastatization process and generating the genetic instability necessary for the malignant transformation. 93
In normal subjects, the concentration of plasma cfDNA ranges from less than 10 ng/ml to more than 100 ng/ml, 94 with a half life of between 16 min and 2.5 h.95,96 However, high levels of cfDNA have not been correlated with malignant disease, but often associated with other conditions such as inflammation, trauma, or exhaustive exercise. 97 In patients with cancer, ctDNA represents a small proportion of total cfDNA, varying from less than 0.1% to over 10% according to tumor burden, cancer stage, cellular turnover, and response to therapy. 98 Therefore, both quantification and detection of specific variants or a mutational hotspot in ctDNA have a direct impact on the clinical utility of ctDNA. Previous amplicon-based studies have shown differences in fragment lengths between ctDNA and cfDNA, 92 and Underhill and colleagues demonstrated that a specific DNA fraction with a size range that is 20–50 bp shorter than the size of cfDNA of healthy donors is substantially enriched with ctDNA in human cancer. 99 To date, the commercial kits for nucleic acid extraction from different body fluids are based on column-based affinity or magnetic-bead-based methods (Table 3). Due to the lack of standardized procedures in terms of sample collection, isolation and analysis, it is still hard to make a real comparison between different data in the literature. In particular, many critical reviews suggest the need to evaluate preanalytical factors involved in sample collection that may affect ctDNA analysis. Plasma is generally preferred over serum to avoid contamination of cfDNA by genomic DNA derived from the lysis of leukocytes and other hematopoietic cells. 100 However, several studies have demonstrated that other body fluids, such as urine, 101 saliva, 102 and cerebrospinal fluid, 103 may contain a larger amount of ctDNA than plasma. Blood samples should be collected in tubes containing ethylenediaminetetraacetic acid (EDTA), an anticoagulant that inhibits the blood’s DNase activity and is compatible with polymerase chain reaction (PCR) analysis. 104 Furthermore, the time between blood drawing and processing is another critical factor for cfDNA concentration; it is important to remove blood cells that may lyse and release germline DNA, which would dilute ctDNA. 105 Finally, temperature has been shown to have a strong influence on cfDNA levels. In fact, when blood is stored at room temperature before processing there is a massive increase of cfDNA that is presumably released by cells undergoing lysis. 106
Comparison of methods for ctDNA detection and analysis.

ctDNA, circulating tumor DNA; ddPCR, droplet digital polymerase chain reaction; NGS, next-generation sequencing; PCR, polymerase chain reaction.
ctDNA features: integrity, methylation and mutations
As described, in patients with cancer, cfDNA mainly includes germline DNA from normal cells and a minority of a highly variable fraction of ctDNA from cancer cells. Recently, the technological advances have overcome the limitations of the traditional DNA analysis approaches 6 in isolating the low levels of such a minor amount of ctDNA6,107 and two major methods have been optimized: targeted approaches to ctDNA to find specific gene mutations or structural chromosome rearrangements in specific genome regions that are generally mutated in given neoplasias; and untargeted approaches for the detection of de novo ctDNA mutations and somatic copy number variations (CNVs) that do not require any prior knowledge of molecular alteration, for example WGS (whole genome sequencing) or WES (whole exome sequencing) (Table 3).5,107–109 Both of these approaches have advantages and limitations.
To detect somatic point mutations, several PCR-based technologies such as BEAMing (beads, emulsion, amplification, and magnetic) and droplet digital PCR (ddPCR) showing very high sensitivity (range from 1% to 0.001%) are suitable.5,9,107,108 In particular, in BEAMing technologies,110,111 magnetic beads in water-in-oil emulsions are used to perform a single molecule amplification by PCR, followed by a flow cytometry to quantify the genetic variants. In droplet digital PCR, a variant of emulsion-based PCR, the sample is divided into thousands of droplets representing a partition of single molecules, each of them undergoing a PCR analysis by selected primers against known regions of ctDNA. 108 The molecular alterations are then measured by fluorescent probes, which bind to the amplified region.
However, digital PCR technologies are limited by scalability for larger studies and may miss substantial information. To overcome this issue and obtain a larger and more comprehensive analysis of genomic regions, several NGS-based technologies such as TAm-Seq (tagged amplicon deep sequencing), CAPP-Seq (cancer personalized profiling by deep sequencing, Safe-Seq (safe sequencing system), and AmpliSeq appear most useful.5,6,9,107,112
TAm-Seq was the first described in 2012. 113 It works through a parallel amplification of multiple regions (entire genes) using a two-step amplification: an initial preamplification step with a pool of the target-specific primer pairs to obtain a representation of all alleles in the template material; then in order to exclude nonspecific products, the region of interest in the preamplification material is selectively amplified in multiple single-plex PCR. Finally, both adaptors and sample-specific barcodes are attached to the resulting amplicons with another round of PCR. Using this method, the sample is optimized for both the sequence of interest and increased sensitivity compared with standard NGS.5,108,113
CAPP-Seq, developed by Newman and Bratman, 114 is a capture-based NGS method for the detection of ctDNA using biotinylated oligonucleotide selector probes to target specific sequences of DNA. It was assessed in patients with NSCLC with a definite improvement in specificity and detection of mutations in more than 95% of tumor samples.5,107,109
Safe-SeqS was described by Kinde and colleagues in 2011 115 to reduce the NGS error rate to 1% and to increase the sensitivity to rare mutants. It is based on the addition of a unique identifier (UID) to each template molecule, which is then amplified by creating a UID family that is directly sequenced. In this manner, all molecules with the same UID should have the same DNA sequence, and it is possible to identify the ‘supermutants’, namely a UID family in which almost 95% of members show the same mutation. This method yields an error frequency of 1.4 × 10−5. 108
NGS data are analyzed in order to detect different classes of genomic alterations, such as substitutions, insertions and deletions, CNVs or gene fusions, using customized pipelines including different bioinformatics tools (e.g. GATK, Annovar, VarScan2).116,117 First, the FASTQ files, obtained from each sequencing system, undergo quality control (QC) and then the reads from each sample are mapped to the reference sequence hg19 (human genome version 19) with alligners software. Finally, the genomic alterations identified are filtered, annotated, related to clinical relevance according to specific databases (i.e. dbSNP, COSMIC), and reported. 117
The PARE (personalized analysis of rearranged ends) approach was recently introduced to specifically detect somatic structural chromosomal rearrangement. 118
Another field of interest for ctDNA concerns its methylation patterns, since methylation patterns are tissue specific and the methylated ctDNA could be a promising biomarker for early detection of cancer, as a carrier of an epigenetic signature of the parental tissue.
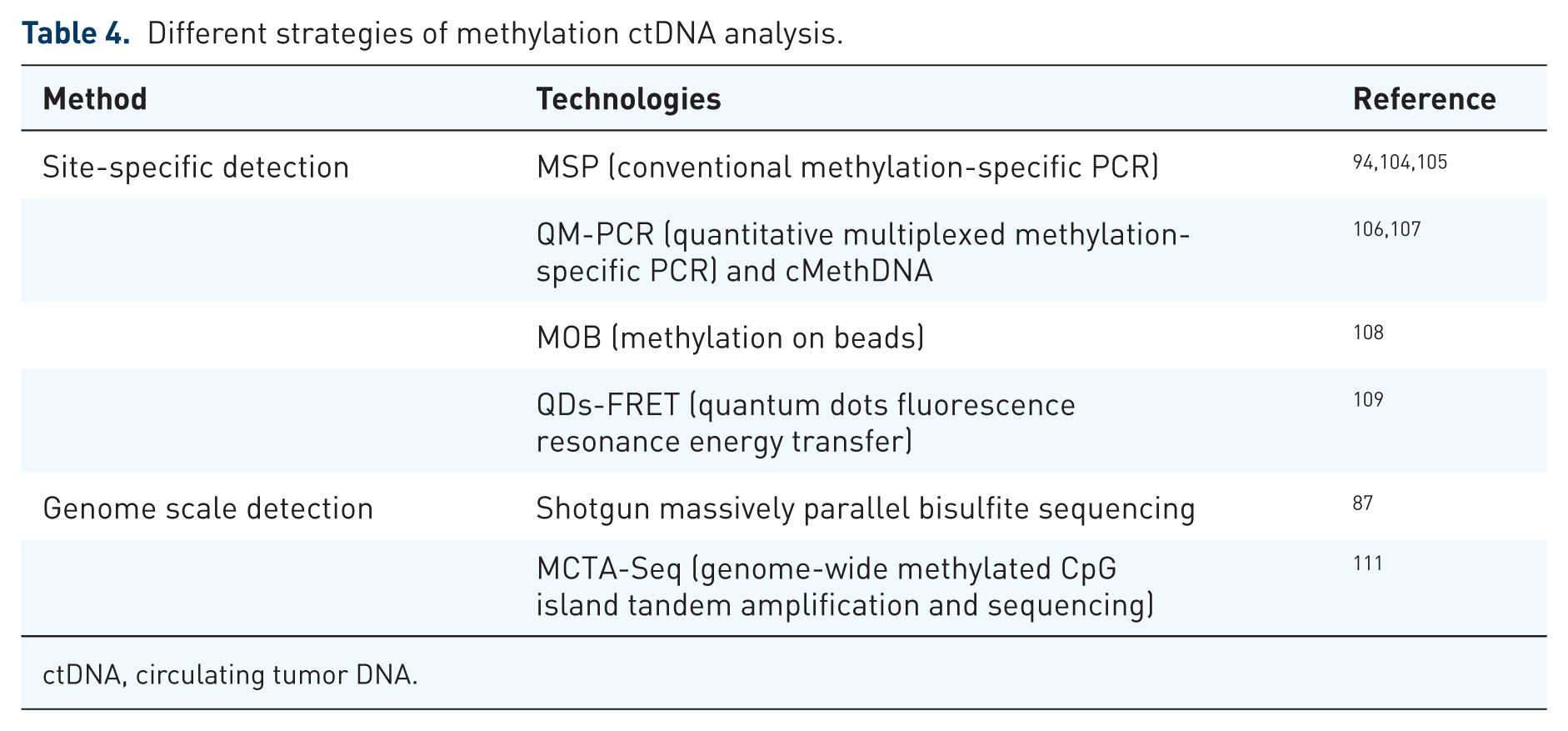
There are different strategies for methylation ctDNA analysis based on bisulfite treatment.5,6,107 These strategies can also be divided into two groups on the basis of detection type: site-specific detection including conventional methylation-specific PCR (Table 4),107,119,120 quantitative multiplexed methylation-specific PCR or cMethDNA,121,122 methylation on beads, 123 and quantum dots-fluorescence resonance energy transfer; 124 genome scale detection, for example shotgun massively parallel bisulfite sequencing 125 or MCTA-Seq (genome-wide methylated CpG island tandem amplification and sequencing), a sensitive method to detect hypermethylated CpG island in ctDNA developed by Wen in 2015. 126
Different strategies of methylation ctDNA analysis.
ctDNA, circulating tumor DNA.
Clinical application of ctDNA
As described, ctDNA may be used as a biomarker with several clinical applications in solid tumors. It is comparable to a maneuverable, easy to find, always updated snapshot of the tumor, capable of reflecting its dimension, molecular heterogeneity, and its evolution over time (Table 5).
Potential key applications of circulating tumor DNA (ctDNA).

cfDNA, cell-free DNA.
Accordingly, its potential applications are numerous, starting from cancer screening. Beck and colleagues investigated the profile of circulating DNA in healthy subjects and found that ctDNA provides useful baseline information regarding the subclinical conditions of patients, including cases of unknown neoplasms that correlate with specific mutations, loss of heterozygosity, or methylation patterns. 127 Subsequently, others assessed the correlation between cancer-related DNA and the development of tumors, However, the results were controversial since ctDNA and cancer-related mutations are also detectable in apparently healthy individuals several years before the clinical evidence of cancer development,128–131 but the same mutations in cfDNA can be detected in healthy volunteers who will never develop a cancer.129,133 Therefore, performing screening tests based on ctDNA in the asymptomatic population could cause overdiagnosis, but the screening of subjects with known risk factors for developing cancer could overtake this problem. 52 However, it is mandatory to perform additional studies to improve the application in this setting of patients.
The quantification of ctDNA to detect the minimal residual disease is a key area of application. It has been demonstrated that the amount of ctDNA is proportional to the residual tumor burden after curative-intent surgery in gastric, lung, and colorectal cancer.132,134,135 A prospective study of 230 patients with early-stage colorectal cancer has demonstrated that the assessment of ctDNA at the first follow-up visit after surgical resection correlates with the recurrence-free survival at 3 years. 136 Similar evidence has emerged in patients with early stage breast cancer; detecting ctDNA after surgery with curative intent indicates a poor prognosis. 137
Moreover, ctDNA can also anticipate the diagnosis of clinical relapse of several months.137,138 Similarly to monitoring the minimal residual disease and describing a tumor’s characteristics, ctDNA can be used to stratify patients at variable risk of recurrence after surgery, selecting who can really benefit from an adjuvant treatment and avoid unnecessary therapies and their relative systemic toxicities. 139
Furthermore, the molecular properties of ctDNA may address treatment options. The characteristics of ctDNA are the mirror of the tumor’s molecular profile. In patients with breast cancer, mutations of TP53 were found both in ctDNA and tumor tissue with a concordance of 43%, 140 and several studies matched the KRAS mutations in primary tumors and in the plasma of patients with pancreatic carcinoma.141–143 This could have important applications in clinical practice allowing the detection of druggable mutations at diagnosis, as well as during treatment to select the therapeutic choices. Treatment with targeted therapies puts the tumor cells under a selective pressure, thus allowing a clonal evolution in a Darwinian manner. Therefore, prolonged treatment with targeted therapies is capable of selecting the cell clone resistant to therapy. Shinozaki and colleagues investigated how the mutated BRAF V600E ctDNA in patients with melanoma correlates with the response to biochemotherapy. They found that 37% of the 103 patients had BRAF-V600E mutated DNA before treatment, while this ctDNA was detectable after treatment in only 1 out of the 10 responders (10%), and in 7 of 10 nonresponders (70%) with a significantly poor association (p = 0.039) between the presence of BRAF-V600E mutated DNA after treatment and OS. 144
Changes in the ctDNA molecular profile can also be detected several months before clinical progression, 145 providing important insights into the mechanisms of resistance and early treatment decision. In addition, data from ctDNA reflect the entire molecular setup of both tumor and metastatic lesions, unlike the tissue biopsy that describes the characteristics of a single specimen in a single lesion. The correlation between KRAS mutations in patients with colorectal cancer and the response to therapy was first investigated by Misale and colleagues, who demonstrated that there is a gradual increase in KRAS-mutated ctDNA in a patient’s serum during treatment with panitumumab or cetuximab. It is therefore conceivable that KRAS mutant clones, present in the primary lesion, undergo a pressure selection that allows the growth of mutant cells. 146 Later, Luis and colleagues also showed that KRAS mutations in cfDNA could be detected in the circulation of 28 patients before treatment with panitumumab, whereas 38% of patients with KRAS wild-type tumors became KRAS mutated after treatment. 147
The trend of EGFR-mutated ctDNA in patients with NSCLC is correlated with the response to treatment with EGFR TKI in 96% of patients, reflecting the sensitivity to treatment. It is also possible to detect the EGFR T790M mutation in ctDNA before clinical progression, thus allowing early intervention. 148 At present, liquid biopsy is approved for clinical practice use only in NSCLC. The Lung-LUX3 trial demonstrated the advantage in terms of PFS of afatinib in EGFR-mutated lung cancer (11.1 months for afatinib versus 6.9 months for chemotherapy; hazard ratio 0.58; 95% confidence interval 0.43–0.78; p = 0.001). 149 This means that it is mandatory to detect the presence of EGFR mutation in patients before treatment.
The discovery of EGFR mutations in ctDNA appears very useful in the presence of difficulties to perform a tumor biopsy. Evidence proved that cfDNA analysis is able to reveal the mechanisms of resistance to EGFR-targeted therapies in NSCLC, as in the presence of EGFR T790M mutation. 148 A phase IV clinical trial proved that the concordance of EGFR mutation in tumor samples and plasma was very high (94.3%), with a specificity of 99.8% and a sensitivity of 65.7%. 150 On 1 June 2016, the FDA approved the use of gefitinib for patients with EGFR mutations detected in ctDNA using the Cobas EGFR Mutation Test v2 (Roche Molecular Systems, Inc., CA, USA) for the detection of exon 19 deletions or exon 21 substitution mutations in the EGFR. However, this is allowed only if it is not possible to perform a tissue biopsy. The approval was based on the phase III, randomized, open-label ENSURE study, which confirmed the efficacy of erlotinib in the first-line treatment of patients with advanced EGFR mutation-positive NSCLC tested with EGFR Mutation Test v2. 151
In conclusion, there is still much to learn about ctDNA, but it is a precious resource in the hands of clinicians. Since 2015 the Therascreen EGFR RGQ PCR Kit (QIAGEN, Hilden, Germany) and the Cobas EGFR Mutation Test v2 (an updated version of the prior Cobas EGFR Mutation Test) have been approved for diagnostic use in the USA. In particular, the Therascreen assay enables the detection of exon 19 deletions and exon 20 and 21 substitutions (T790M and L858R respectively) in the EGFR gene. 148
Exosomes
Exosomes isolation and characterization
EXOs are nano-sized vesicles (40–100 nm) released by cells and detectable in most body fluids, such as plasma, urine, saliva, or ascites. 152 Different from other extracellular vesicles (EVs) which directly bud off from the cell membrane, such as microvesicles (50–1000 nm in diameter) or apoptotic bodies, EXOs are end products of the recycling endosomal pathway and originate from inward budding of the plasma membrane. 153 Although they were previously considered as cellular waste products, 154 it is now well demonstrated that they play a role in intercellular communication, depending on the cargo of functional molecules from donor to distant cells. 155
EXOs take part in many physiological and pathological processes and have been shown to be involved in cancer progression and metastatization. 156 Noteworthy, EXOs released by cancer cells, namely tumor-derived EXOs, promote EMT and affect the proliferation, migration, and invasion of cancer cells, as well as support the angiogenesis and the establishment of an immunosuppressive milieu. 157 EXOs are also emerging as a novel chemoresistance mechanism, primarily depending on drug discharge via vesicle budding, neutralization of antibody-based drugs, and EXO-mediated transfer of micro RNAs (miRNA). 158
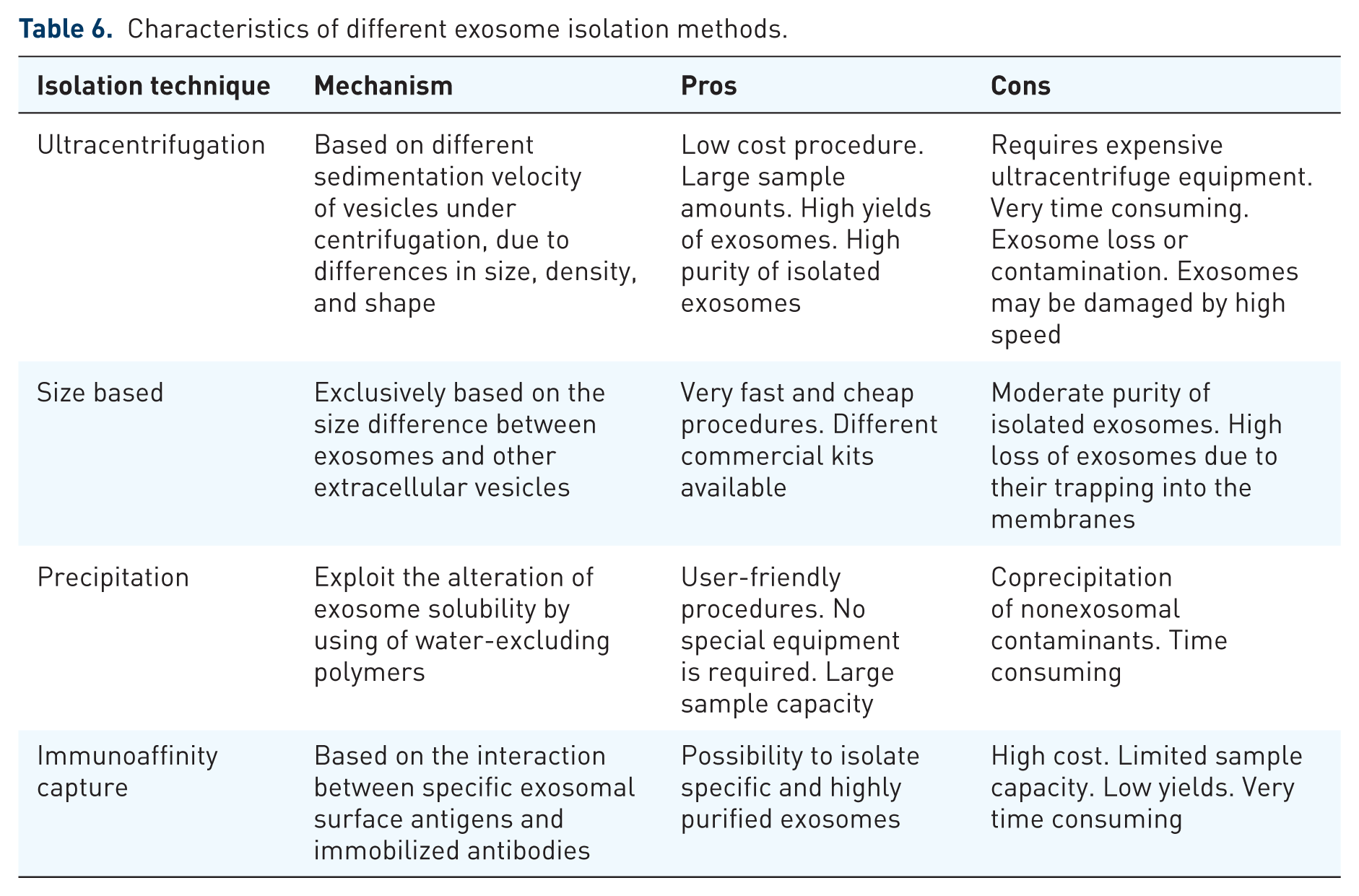
EXOs consist of a lipid bilayer which contains both transmembrane and nonmembrane proteins, as well as noncoding RNAs, mRNAs, and either single-stranded or double-stranded DNA 159 (Figure 5). According to proteomic analyses, EXOs were found to be characterized by a conserved set of proteins independently of their cellular origin, such as CD63, CD81, and CD9 tetraspanins. 153 However, the protein composition of EXOs roughly resembles originating cells, thus suggesting an exosomal cell-type or tissue-specific signature.160,161 It has also been demonstrated that certain RNA transcripts are enriched up to 100 fold in EXOs compared with the donor cells, thus supporting active packaging. 162 Several methods have been developed to efficiently collect EXOs from body fluids (Table 6). The available protocols are generally based on vesicle separation in accordance with their biophysical properties, including size, morphology and density, while others are based on the immunoaffinity capture or altering the EXO solubility to improve their precipitation. 163 To date, a common isolation protocol adopts serial centrifugations with increasing speeds to remove cellular debris and larger plasma membrane-derived vesicles, followed by sedimentation of EXOs by ultracentrifugation (100,000 × g). Despite this method being largely accepted, the ultracentrifugation procedure may result in contaminating events or EXO loss, is time consuming, and does not allow one to selectively isolate tumor-derived EXOs from those originating by other cells. Other isolation techniques exploiting the physical properties of EXOs are density gradient separation by sucrose and size exclusion chromatography. Although both of these methods efficiently yield highly purified EXOs, they are used less often because they are very user intensive. 164 By contrast, immunocapture approaches, such as microplate-based enzyme-linked immuno-sorbent assay or antibody-coated magnetic particles, recognize specific exosomal surface antigens, leading to selective isolation of EXOs and are increasingly used in research. 160 Finally, several commercially available and user-friendly kits cause EXO precipitation in the presence of water-excluding polymers, although lack of a proper selective isolation mechanism may compromise the purity of yields. 165
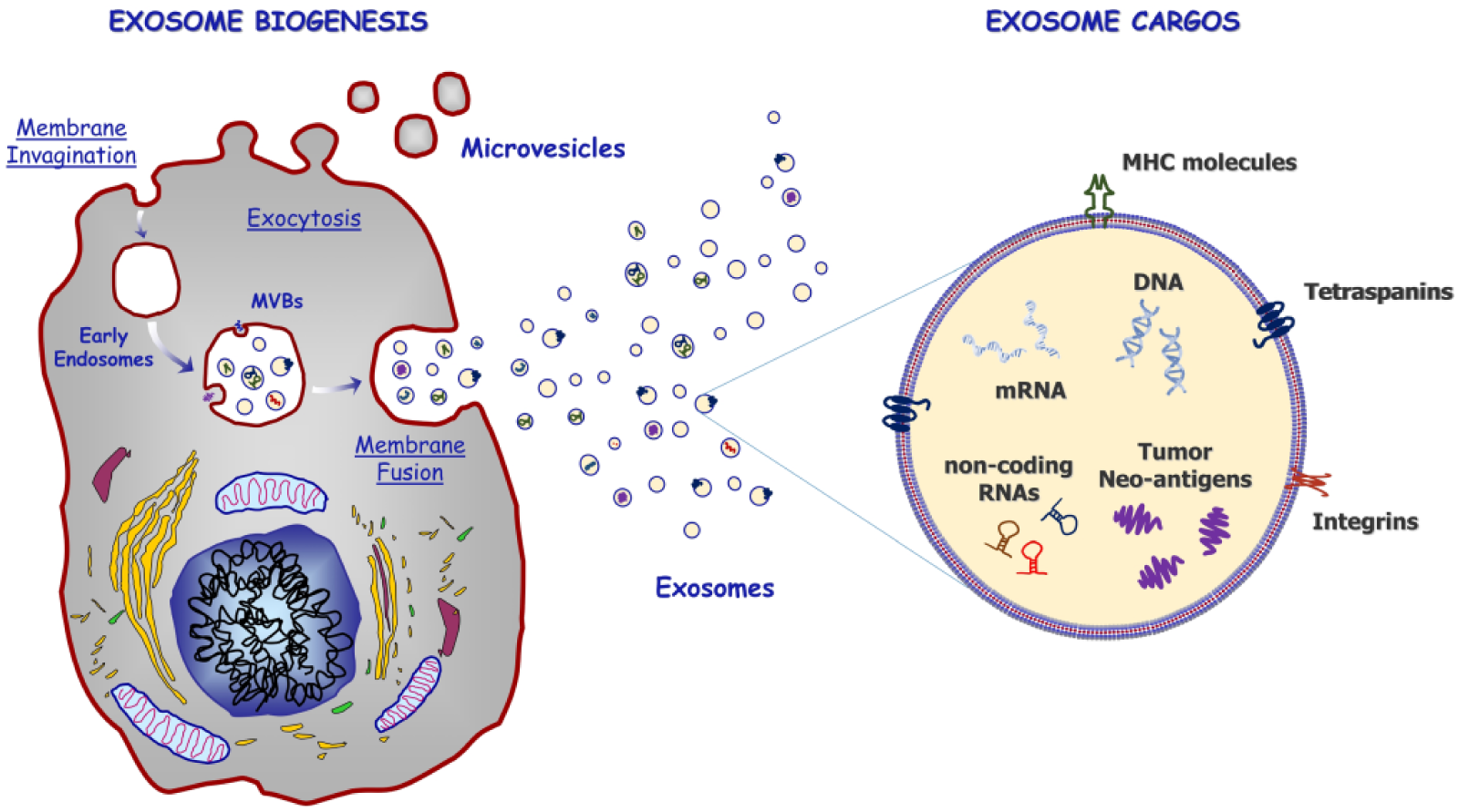
Exosomes biogenesis and content. Exosomes are end products of the recycling endosomal pathway and originate from inward budding of the plasma membrane, leading to the formation of multi vesicular bodies (MVBs). Following active packaging of signaling molecules into MVBs, they fuse with the plasma membrane and release their contents into the extracellular space in the form of exosomes. Exosomes consist of a lipid bilayer which contains both transmembrane and nonmembrane proteins, as well as noncoding RNAs, mRNAs and either single-stranded or double-stranded DNA. They also express a conserved set of proteins independently by cellular origin, including CD63, CD81, and CD9 tetraspanins, while those from cancer cells are rich in tumor-associated antigens. MHC, major histocompatibility complex.
Characteristics of different exosome isolation methods.
Clinical application in oncology
Following their isolation and investigation by electron microscopy or flow cytometry assays, tumor-derived EXOs may be investigated for their protein expression or genetic profile as diagnostic or prognostic markers. 166 To this, CAV1+ EXO have been proposed as potential diagnostic markers of melanoma, 160 while the identification of serum EXOs enriched with high levels of migration inhibitory factor (MIF) can identify patients with pancreatic ductal adenocarcinoma more likely to develop liver metastasis, thus potentially representing an unfavorable prognostic factor. 167 Similarly, EXOs from prostate cancer cells, namely prostasomes, can be detected by prostate-specific membrane proteins, including PSMA, prostate-specific transglutaminase, and prostate stem cell antigen, 161 whose serum and urine levels correlate with tumor burden.168,169
Moreover, since RNAs and DNAs packaged within EXOs (exoRNA and exoDNA, respectively) are protected by the phospholipid bilayer from degradation due to serum ribonucleases and DNases, 170 their analysis has provided additional diagnostic and prognostic information, as well as usefulness for monitoring the treatment response. In this context, specific exosomal miRNA signatures have been described, such as the miR-1246, miR-4644, miR-3976, and miR-4306 that were found upregulated in patients with pancreatic cancer 171 or the overexpression of miR-211 in patients with BRAFV600 melanoma that correlated with reduced sensitivity to BRAF inhibitors. 172 Finally, EXOs are also enriched with single- or double-stranded DNA fragments from all chromosomes, leading to the identification of gene mutations, such as KRAS and EGFR mutations, as already proven in patients with pancreatic cancer.159,173
Another important issue regarding EXOs is the possibility of isolating and analyzing nanovesicles originating from immune cells. This has clear and direct clinical applicability as the phenotypic profile of immune cell derived EXOs may reflect the status of immune system activation, and give useful information to predict response to immune-therapeutic drugs. In this context, Tucci and colleagues demonstrated that a high expression of programmed cell death 1 and CD28 molecules by T-cell derived-EXO (TEX) at baseline predicts the response to ipilimumab, a cytotoxic T-lymphocyte antigen 4 (CTLA4) inhibitor, in patients with metastatic melanoma. 174 Similarly, CD80 and CD86 levels on dendritic cell derived EXO (DEX) reflect the restoration of antimelanoma activity from the immune system, thus supporting both TEX and DEX as reliable prognostic biomarkers in melanoma.
Although promising, data from these studies refer only to small cohorts of patients and are inadequate to support definite conclusions. Further clarifications from large clinical trials are needed to confirm the applicability of EXOs as tumor biomarkers for monitoring cancer progression or driving treatment decisions. Moreover, other possible clinical applications of EXOs in oncology include the possibility of either drug or miRNA delivery within the tumor cells, the identification of newly therapeutic targets to inhibit the molecular mechanisms implicated in cancer progression, as well as the stimulation of an immunological response against cancer cells. These innovative approaches are now under intensive investigation, both in preclinical and clinical trials, and have been recently revisited. 175
Conclusions and future perspectives
The modern procedure of liquid biopsy has been a breakthrough in clinical oncology and still has the powerful potential of a forthcoming revolution; screening, diagnosis, prognosis, and treatment in patients with cancer might be dramatically changed. Similar to traditional biopsies, liquid biopsy of CTCs, ctDNA, and EXOs offers the entire range of information that allows the performance of functional studies. Yet, unlike traditional techniques, liquid biopsy is capable of embracing the spatial and temporal heterogeneity that stands at the biological basis of cancer. 176 Despite such high potential, a significant gap must actually be filled, since the systematic application of liquid biopsy in real practice is still hindered by many hurdles, such as unsatisfactory specificity and sensitivity, lack of standardization, and elevated economic and human resource costs, and still offers many challenges.4,7 In fact, due to the low concentration of CTCs, ctDNA, and EXOs currently recoverable from the patient, the analytical results sometimes suffer from unsatisfactory specificity and sensitivity.25,177–179 The use of different high-throughput analytical platforms often results in difficulty in reproducing results, and highlights the need for standardization and analytical validation of the method used for liquid biopsy.180,181 In the same way, computational analysis needs new tools that can elaborate complex algorithms of data interpretation and clinical correlation of molecular data. Finally, it is desirable that the current high personnel and infrastructure costs necessary for these methods are reduced in response to progress in biotechnology (Table 7).
Overview of challenges and future prospects of liquid biopsies.

CTC, circulating tumor cell; ctDNA, circulating tumor DNA; PCR, polymerase chain reaction.
Advances in technologies, particularly the introduction of NGS techniques, are contributing to increases in the appropriateness of liquid biopsy. However, large-scale and multicenter trials are also ongoing to confirm all the potentialities that are now being studied in order to fully define the exact settings and conditions for the application of liquid biopsy and confirm the comparison of performance with current solid biopsy methods. Data from liquid biopsy might then become novel blood-based tumor markers. Among ctDNA, CTCs, and EXOs, some remarkable differences can be identified (Table 8).4,6,9,182 Even if charged with preanalytical variability, all of these techniques are capable of detecting cancer genomic abnormalities as point mutations, insertions and deletions, amplifications, translocations and copy number alterations, and epigenetic alterations. Given the short half life of free mRNA in circulation, RNA analysis, including miRNA and RNA expression, is preferable on CTCs and EXOs, while only CTCs allow analysis of cell morphology, as well as in vivo studies or functional ex vivo studies. At present, the information obtained from the study of EXOs, CTCs, and ctDNA highlights the complementarity of all information to best define tumor status and prognosis. Several simultaneous analytical approaches are also needed to evaluate minimal residual disease after surgery or chemotherapy.6,139
Comparison between the applications of ctDNA, CTCs, and exosomes.

CTC, circulating tumor cell; ctDNA, circulating tumor DNA; PCR, polymerase chain reaction.
Finally, the preservation of biologic materials in biobanks is currently possible only for ctDNA and EXOs, and results from large-scale trials will shortly provide evidence-based elements for the application of liquid biopsy in clinical practice in the era of precision medicine in clinical oncology.
