Abstract
Together with the use of novel oral targeted therapies, a multidisciplinary approach can be used to effectively treat patients with advanced pancreatic neuroendocrine tumours (pNETs). Here we review the integration of the oncology nurse to the newly developed oral treatment setting for patients with pNETs. From the outset, the nurse must be involved in various processes, including performance of baseline assessments (e.g. blood pathology, cardiac and lung function testing, patient history) and general medical observations, treatment administration, dietary guidance, evaluation of comorbidities, and review of concomitant medications. Patient education and establishment of a strong partnership in care before the start of pNET therapy ultimately increase treatment adherence and reduce potential toxicities. Regular review of general patient status and disease progression and continuous monitoring of adverse events also help enhance treatment outcomes and subsequently improve quality of life. Nurses’ knowledge of agent-specific toxicities and prompt, proactive management is a critical aspect of care. In essence, as the pNET treatment landscape evolves, the role of the healthcare professional in overall patient care must shift accordingly.
Introduction
Neuroendocrine tumours (NETs) are a diverse group of epithelial neoplasms originating from various anatomical locations throughout the body, including the lung, pancreas, midgut, and appendix; gastroenteropancreatic neuroendocrine tumours (GEP-NETs) account for approximately two-thirds of cases [Klimstra et al. 2010; Anlauf, 2011; Oberg et al. 2010]. Although diagnostic and therapeutic advancements have contributed to declines in both the incidence and the prevalence of many cancer types [Ferlay et al. 2010; Jemal et al. 2010], the number of NET diagnoses appears to be on the rise [Yao et al. 2008; Lawrence et al. 2011]. Because NETs may not manifest specific early symptoms, they are often diagnosed at an advanced stage; approximately half of patients have metastatic disease at the initial diagnosis [Modlin et al. 2008; Yao et al. 2008]. Patients with GEP-NETs may have a history of vague gastrointestinal symptoms for years before the diagnosis; in fact, their symptoms are often misdiagnosed as irritable bowel syndrome [Vinik et al. 2010]. Subsequent prognosis is dependent on cancer stage, primary anatomical disease site, and histological findings [Yao et al. 2008]. Well differentiated NETs are classified as low or intermediate grade. For patients with well differentiated or moderately differentiated pancreatic NETs (pNETs), the median survival time is 27 months [Klimstra et al. 2010; Yao et al. 2008]. Conversely, poorly differentiated NETs are associated with more aggressive disease and poorer prognosis [Klimstra et al. 2010]. Despite advances in our knowledge of NETs, approximately two-thirds of patients with NETs die within 5 years of diagnosis [Yao et al. 2008; Modlin et al. 2008]. The combination of new therapies and practical management strategies administered through a multidisciplinary approach [National Comprehensive Cancer Network, 2012] may afford improved outcomes overall for patients with NETs.
The role of the oncology nurse in neuroendocrine tumours
The optimal care of patients with NETs involves, as it does for other patients with cancer, close cooperation among multiple medical professionals [National Comprehensive Cancer Network, 2012]. During both diagnosis and treatment stages, patients with NETs are likely to interact with a number of healthcare professionals, including primary care physicians, various specialists (e.g. oncologists, endocrinologists, radiologists), surgeons, oncology nurses, and nurse practitioners, as well as with social workers and counsellors. Throughout the course of the disease, patients with NETs are likely to encounter oncology nurses, who play a major role in providing information about the disease, organizing diagnostic tests, administering treatment, and ensuring overall patient wellbeing [Bouvier, 2003; Grande and Haller, 2009; Gardner, 2009]. Therefore, it is important for nurses, whether working in specialist multidisciplinary treatment centres or in a general oncology environment, to be familiar with current NET treatment guidelines, patient assessment parameters, and best practice [Grande and Haller, 2009].
With the emergence of new oral therapies for pNETs, it is vital that oncology nurses continue to play a role in the ongoing education and support of patients. Regular assessments should be undertaken to monitor and manage drug toxicity and to ensure satisfactory symptom control. Before initiating oral therapy, the oncology nurse should perform a baseline assessment of the patient. This includes a general history of the patient, such as current medical conditions and comorbidities, current medications, general observations (blood pressure, pulse, temperature), basic blood test results (full blood count, biochemistry profile, blood sugar level, liver function tests, vitamin B12 and vitamin D levels, if relevant), specific NET markers such as chromogranin A, neuron-specific enolase, 5-hydroxyindoleacetic acid (serotonin) [Yao et al. 2011; O’Toole et al. 2009; Baudin et al. 2011] and other appropriate hormonal markers. Other tests might include cardiac echogram and lung function testing. Pretreatment education should provide specific information regarding drug administration (how and when to take a specific pNET treatment), dietary requirements (foods to avoid, timing of medications in relation to meals), possible interactions with concomitant medications, potential drug side effects, and strategies to manage such adverse events (AEs). A relationship must be established between the patient and the nurse and, if applicable, the family (partnership in care). For patients in the community, experiencing side effects from a new medication but not knowing how to adequately manage these effects or whom to contact with questions can be challenging. Support and guidance from a nurse can equip patients in the practical management of pNETs.
Overview of neuroendocrine tumour treatment and recent advances in pancreatic neuroendocrine tumour treatment
For patients with diagnoses of organ-confined NETs, surgical resection can offer the possibility of durable remission [National Comprehensive Cancer Network, 2012]. However, for those with advanced disease, surgery is unlikely to provide a cure despite potential symptom control through debulking of the tumour burden [Yao et al. 2008]. For patients with functioning tumours (defined as tumours that secrete hormones that cause physiological symptoms) and for those with positive Octreoscan (Mallinkrodt Pharmaceuticals, Dublin, Ireland), short- and long-acting somatostatin analogues (e.g. octreotide, lanreotide) may be used to reduce symptoms associated with hormone hypersecretion, including diarrhoea and flushing, potentially improving quality of life (QoL) [Kulke et al. 2012; Khan and Caplin, 2011; Gardner-Roehnelt, 2012]. Octreotide also has been shown to have antitumour efficacy in patients with advanced mid-gut NETs and has been approved by the European Medicines Agency (EMA) for this indication [Rinke et al. 2009]. Among patients with nonfunctioning tumours, available treatment choices include surgery, chemotherapy, radiopeptide therapy for metastatic disease, liver-targeted therapies for progressive liver metastases, and palliative care to reduce pain and other tumour symptoms [National Comprehensive Cancer Network, 2012]. Local palliative radiotherapy can also be used to control local symptoms [Oberg et al. 2004; Khan and Caplin, 2011]. Historically, systemic chemotherapy has had little impact on pNETs, and its toxicity has precluded widespread use of aggressive combination chemotherapy regimens in patients with pNETs [Burns and Edil, 2012]. Generally, therapeutic options aimed at delaying disease progression in patients with advanced NETs have been limited [Kulke et al. 2012; Oberg et al. 2004; Burns and Edil, 2012). However, there has been a major advance in pNET treatment in recent years as research into the mechanisms behind tumour growth has resulted in the development of novel, targeted, therapeutic agents [Kulke et al. 2011; Capdevila and Tabernero, 2011].
Two such agents have now been approved by the US Food and Drug Administration (FDA) and EMA for the treatment of well differentiated pNETs: the multitargeted vascular endothelial growth factor receptor tyrosine kinase inhibitor (VEGFR-TKI) sunitinib and the mammalian target of rapamycin (mTOR) inhibitor everolimus [Eads and Meropol, 2012; National Cancer Institute, 2011a, 2011b]. In phase III trials, both agents were shown to provide significant gains in progression-free survival (PFS) compared with placebo. In the RADIANT-3 trial, oral everolimus (10 mg/day) plus best supportive care (BSC) was evaluated versus placebo plus BSC in 410 patients with pNETs [Yao et al. 2011]. Everolimus demonstrated a statistically significant and clinically meaningful 6.4-month prolongation of median PFS compared with placebo [Yao et al. 2011]. Furthermore, 73% of everolimus-treated patients experienced stable disease and treatment benefits were durable. Objective tumour response rate was 5% in patients receiving everolimus and 2% in those receiving placebo [Yao et al. 2011]. In a phase III trial, treatment with oral sunitinib (37.5 mg/day) was evaluated in 171 patients with advanced/metastatic pNETs compared with placebo [Raymond et al. 2011]. The original accrual goal was 349 patients. However, this study was terminated early and unblinded, based on recommendations by the independent data monitoring committee [Blumenthal et al. 2012]. At the interim analysis, sunitinib was found to significantly prolong PFS by almost 6 months compared with placebo. Objective tumour response rate was 9.3% in patients receiving sunitinib and 0% in those receiving placebo [Raymond et al. 2011]. Consequently, everolimus and sunitinib were approved by the FDA and EMA in 2011 and are now recommended therapy in the most recently updated NET treatment guidelines for patients with advanced pNETs [Kulke et al. 2012; National Comprehensive Cancer Network, 2012; Falconi et al. 2012; Jensen et al. 2012]. Here we will discuss the administration of these new agents, which patients might benefit most from treatment, and the management of treatment-associated AEs.
Administration of targeted treatments for pancreatic neuroendocrine tumours
Dosage
Dosage and administration details for everolimus and sunitinib are reviewed in Table 1 [Pfizer Inc., 2011; Novartis Pharmaceuticals Corporation, 2012]. The recommended daily dose of everolimus for the treatment of pNETs is 10 mg once daily, although temporary dose reductions or interruptions may be required to manage severe or intolerable adverse reactions (suggested dose 5 mg/day). Specifically, patients with hepatic impairment may require dose reductions to 5 mg for moderate impairment (Child–Pugh class B). For sunitinib, the recommended daily dose for pNETs is 37.5 mg once daily, but increments or decrements of 12.5 mg may be required based on safety and tolerability in an individual patient [Pfizer Inc., 2011; Novartis Pharmaceuticals Corporation, 2012].
Dosage and administration information for everolimus and sunitinib.
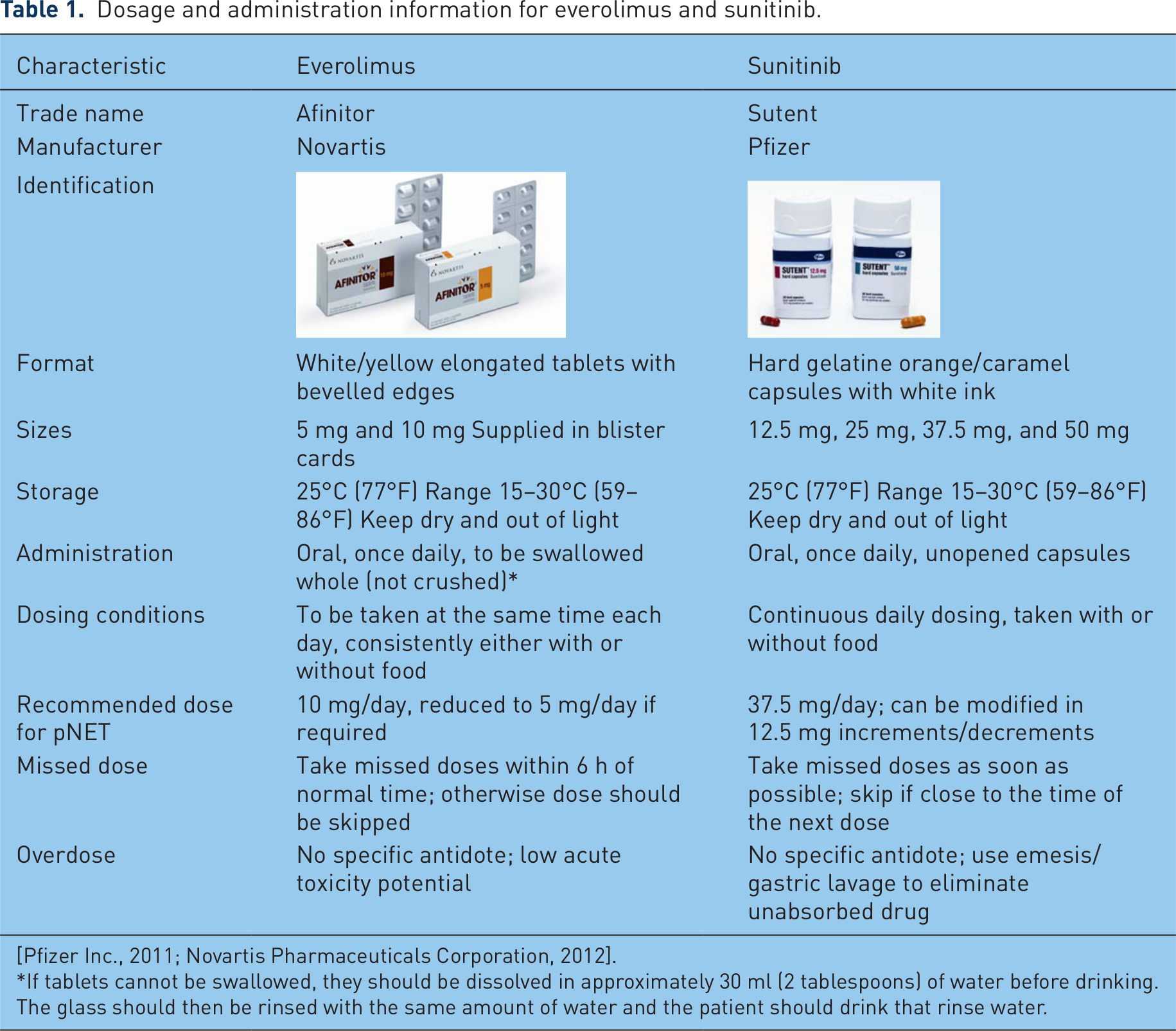
If tablets cannot be swallowed, they should be dissolved in approximately 30 ml (2 tablespoons) of water before drinking. The glass should then be rinsed with the same amount of water and the patient should drink that rinse water.
Drug–drug interactions
Concomitant use of cytochrome P450 3A4 (CYP3A4) inhibitors or inducers should be avoided or they should be administered with caution because they alter the bioavailability of everolimus and sunitinib (Table 2) [Pfizer Inc., 2011; Novartis Pharmaceuticals Corporation, 2012; Moldawer and Wood, 2010; Grande and Haller, 2009; Kovarik et al. 2006]. Grapefruit, grapefruit juice, and other foods that are known to inhibit CYP activity may increase drug exposure and should be avoided during treatment with either agent. St John’s wort (Hypericum perforatum) may decrease drug exposure unpredictably and should also be avoided. Dosing of everolimus or sunitinib may return to previous levels after the discontinuation of such CYP3A4 inhibitors or inducers.
Drug–drug interactions and management with targeted agents in pancreatic neuroendocrine tumours.

[Pfizer Inc., 2011; Novartis Pharmaceuticals Corporation, 2012; Moldawer and Wood, 2010; Grande and Haller, 2009].
CYP3A4, cytochrome P450 3A4; HIV, human immunodeficiency virus; mTOR, mammalian target of rapamycin; VEGFR-TKI, vascular endothelial growth factor receptor tyrosine kinase inhibitor.
Use in specific patient populations
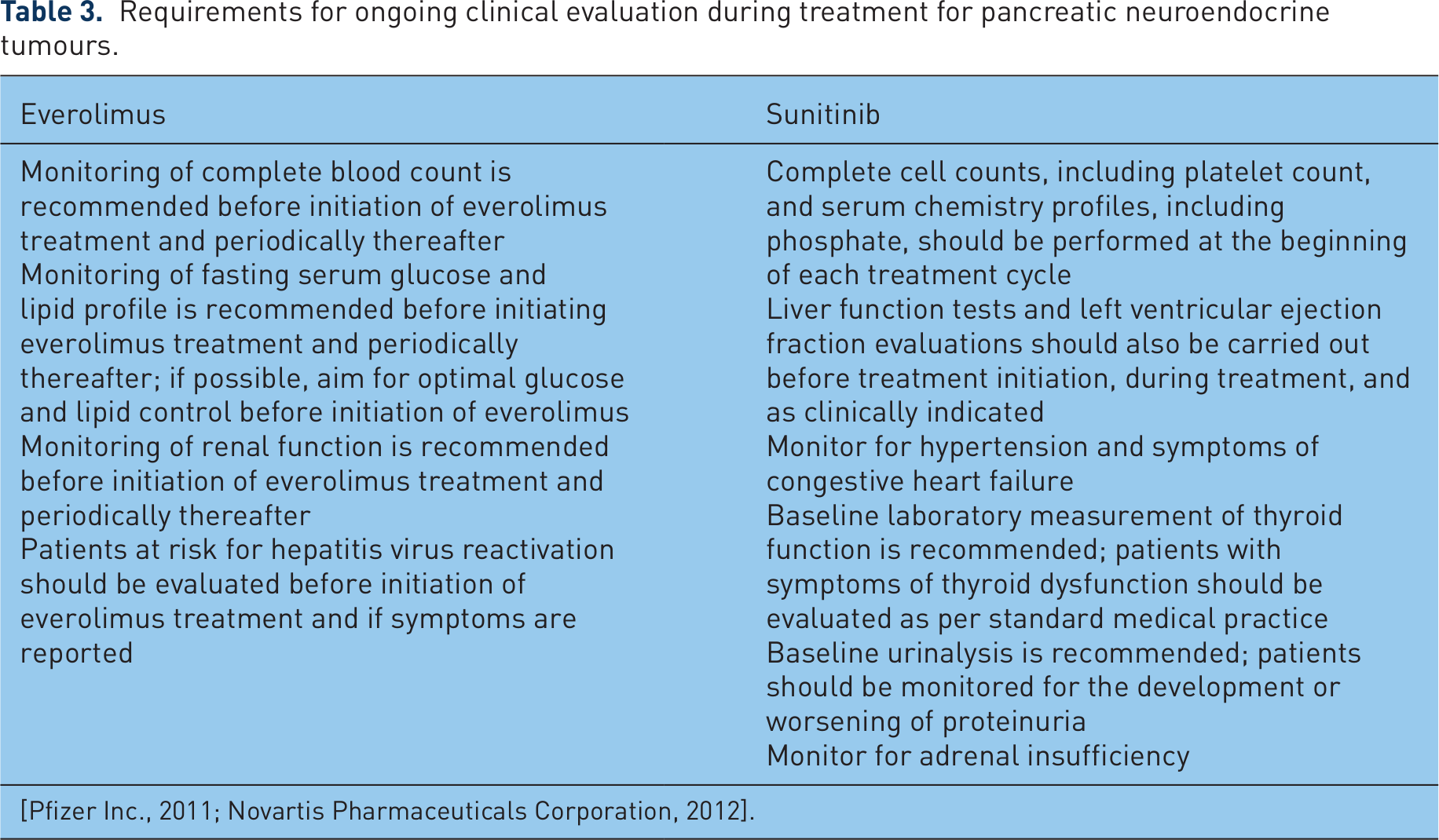
Warnings and precautions should be considered in specific patient populations [Pfizer Inc., 2011; Novartis Pharmaceuticals Corporation, 2012]. Everolimus or sunitinib is not recommended in pregnant women or in children for the treatment of pNETs. No dose alterations are required in older patients or in those with renal impairment [Pfizer Inc., 2011; Novartis Pharmaceuticals Corporation, 2012]. Patients with hepatic dysfunction may require dosage modifications with both agents, and sunitinib (but not everolimus) has been rarely associated with hepatotoxicity resulting in liver failure or death. Of note, for patients with any cardiac insufficiency (e.g. left ventricular dysfunction, arrhythmia), sunitinib may not be a suitable therapy; the perceived benefits should be weighed against the possible risks of treatment. Ongoing clinical evaluations should be conducted during the course of pNET treatment (Table 3) [Pfizer Inc., 2011; Novartis Pharmaceuticals Corporation, 2012].
Requirements for ongoing clinical evaluation during treatment for pancreatic neuroendocrine tumours.
Determining which patients will benefit from treatment
Although both sunitinib and everolimus are recommended in the current pNET treatment guidelines, these targeted agents are not interchangeable, and careful consideration is required to determine which drug is more appropriate for a patient. Patient populations evaluated in phase III studies have varied between agents. Sunitinib has been evaluated only in patients with pNETs with well differentiated disease. However, everolimus has been assessed in patients with a wider range of tumour types, including well differentiated and moderately differentiated tumours [Yao et al. 2011; Pavel et al. 2011; Raymond et al. 2011]. Although outcomes between studies cannot be directly compared, overall findings from the RADIANT-3 trial with everolimus are considered to be more robust and reliable than those from the phase III sunitinib study in patients with pNETs [Phan, 2013]. However, treatment selection should take into account numerous factors, including tumour grade and histology, patient age, comorbidities, and patient compliance.
Choice of therapy should also reflect the heterogeneity of functional pNETs and the symptoms displayed. Given that the inhibition of platelet-derived growth factor receptor by sunitinib can promote pancreatic β-cell survival [Mokhtari and Welsh, 2010], sunitinib treatment may be suitable for patients with glucagonoma but may have detrimental effects on patients with insulinoma. Preclinical studies have shown sunitinib to, in fact, reverse type 1 diabetes [Louvet et al. 2008]. In a patient with metastatic renal cell carcinoma, however, sunitinib treatment induced long-term remission from recent-onset type 1 diabetes mellitus [Templeton et al. 2008]. Conversely, increased blood glucose level is a drug class effect of mTOR inhibitors [Rodriguez-Pascual et al. 2010]; sunitinib is the preferred agent in patients with brittle diabetes [Phan, 2013]. Treatment with everolimus has been shown to benefit patients with insulinoma and in several cases has helped patients successfully achieve normoglycaemia [Ong et al. 2010; Kulke et al. 2009].
For patients with any history of cardiac events, everolimus should be considered an option because sunitinib treatment may result in heart failure [Pfizer Inc., 2011]. Hypertension is reported to be the most common cardiovascular toxicity among sunitinib-treated patients [Gupta and Maitland, 2011]. Sunitinib may exacerbate symptoms of congestive heart failure or left ventricular dysfunction, leading to cardiac death [Bello et al. 2009; Chu et al. 2007; Telli et al. 2008]. Reports indicate that even in patients without cardiac concerns at baseline, sunitinib may induce the development of heart failure shortly after treatment initiation, again highlighting the importance of drug selection in advance and continued patient monitoring [Khakoo et al. 2008].
Other issues of particular importance associated with pNET therapy include compliance (these agents are administered orally at home rather than as infusions in the clinic), retention of information by older patients, and management of symptoms over extended durations [Gardner, 2009; Jacobs, 2009]. Nurses should recognize the rationale for using targeted agents such as everolimus and sunitinib in pNETs and should be able to effectively communicate this information to patients and caregivers. Patients who understand their treatments are more likely to adhere to their therapy regimens and are less likely to discontinue because of toxicity. In the phase III sunitinib study, median treatment duration was 4.6 months. Dose interruption was required in 30% of patients receiving sunitinib and in 12% receiving placebo [Raymond et al. 2011]. In the RADIANT-3 study, median treatment duration was 8.8 months. Dose adjustments were required for 59% of patients receiving everolimus and for 28% of patients receiving placebo [Yao et al. 2011]. Knowing that support staff are knowledgeable about expected tolerability profiles may help the patient be more adherent and may decrease the severity of AEs if potential toxicities are addressed in a timely, efficient manner.
Management of adverse events
Adverse events associated with targeted therapy
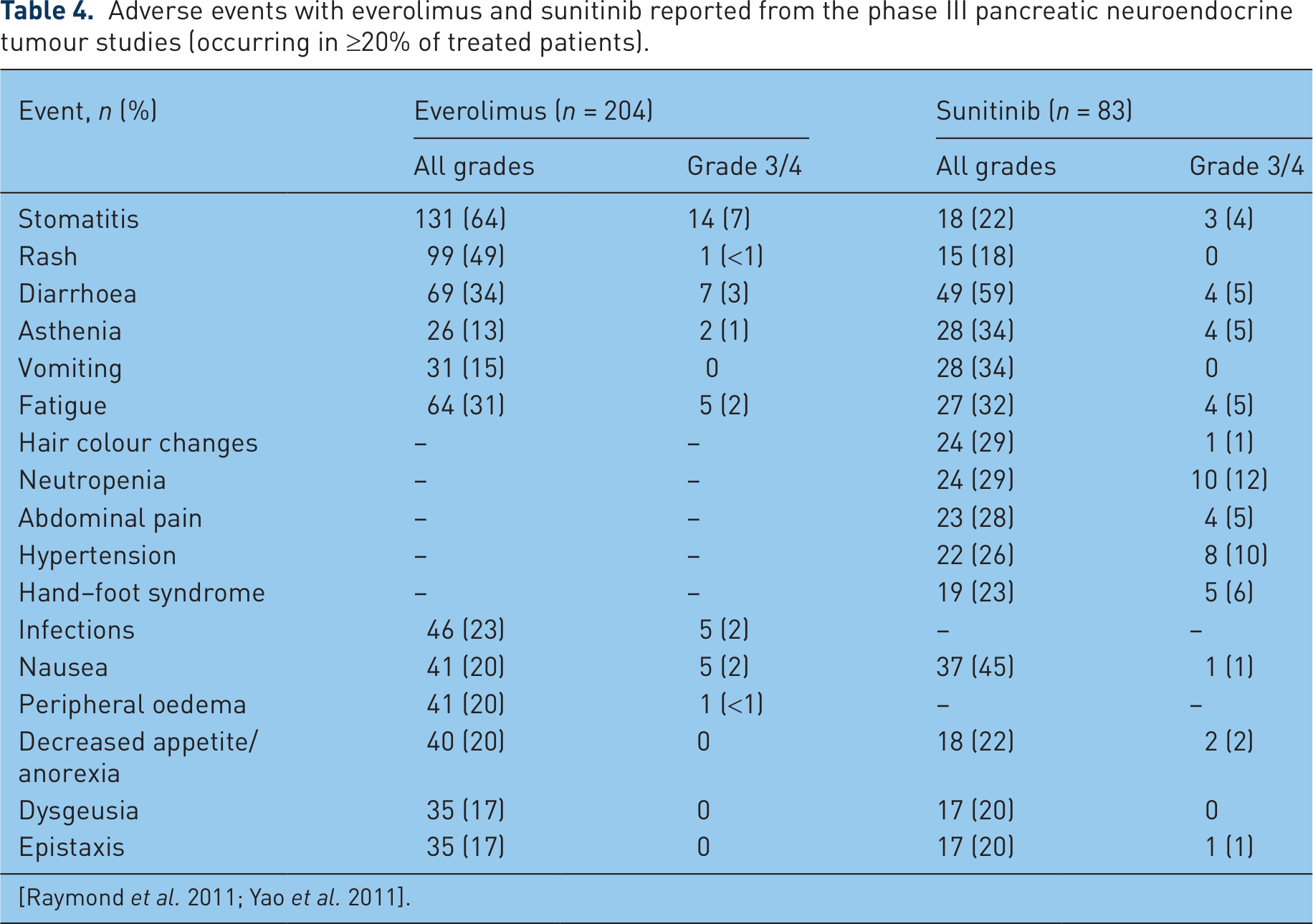
The tolerability profiles of both everolimus and sunitinib are well established from multiple studies in patients with cancer [Novartis Pharmaceuticals Corporation, 2012; Pfizer Inc., 2011], and the most common (≥20% of patients) AEs observed in the phase III pNET trials have been identified (Table 4) [Raymond et al. 2011; Yao et al. 2011]. Noninfectious pneumonitis (including pneumonitis, interstitial lung disease, lung infiltration, and pulmonary fibrosis) occurred in 17% of patients in the everolimus treatment group in the RADIANT-3 trial [Yao et al. 2011]. Although many AEs occur during treatment with both everolimus and sunitinib, most reported toxicities are mild or moderate. Some AEs are particularly associated with one agent. For instance, stomatitis, rash, infections, and noninfectious pneumonitis are associated with everolimus, and hand–foot syndrome (HFS), fatigue, neutropenia, hypertension/cardiac function abnormalities, and depigmentation are associated with sunitinib.
Adverse events with everolimus and sunitinib reported from the phase III pancreatic neuroendocrine tumour studies (occurring in ≥20% of treated patients).
Common adverse events
Nursing assessments are important both to evaluate symptom changes resulting from disease progression and to identify early AEs related to treatment [Grande and Haller, 2009; Jacobs, 2009]. Because many symptoms may be self treated, it is critical to engage patients and caregivers actively in patient care [Grande and Haller, 2009]. Basic components that should be considered throughout the AE management process include thorough medical history, patient awareness and education, and drug treatment recommendations. Nurses should be able to recognize the physical manifestations of specific toxicities [Figure 1(a–c)] and to identify AE management recommendations (Table 5).

Illustration of some common adverse events observed during treatment with everolimus or sunitinib. (a) Stomatitis [Porta et al. 2011]. Reprinted with permission. Copyright © 2011 Elsevier Ltd. A. All rights reserved. (b) Hand–foot syndrome [Tsai et al. 2006]. Reprinted with permission. Copyright © 2006 American Society of Clinical Oncology. All rights reserved. (c) Depigmentation: (top left) patient with gastrointestinal stromal tumour receiving sunitinib in a 4-week-on/2-week-off schedule; (top right) untreated identical twin brother; (bottom) close up of beard hair of treated patient, with characteristic striping from treatment [Hartmann and Kanz, 2008]. Reprinted with permission. Copyright © 2008 American Medical Association. All rights reserved.
Management of common adverse events during treatment with everolimus or sunitinib.

[Novartis Pharmaceuticals Corporation, 2012; Pfizer Inc., 2011; Moldawer and Wood, 2010; Balagula et al. 2012; Ferte et al. 2011; de Oliveira et al. 2011; Boers-Doets et al. 2011; White et al. 2010; Rodriguez-Pascual et al. 2010; Nathan et al. 2009; Grunwald et al. 2010; Cohen and Oudard, 2012; Ravaud, 2011; Di Lorenzo et al. 2011; Eisen et al. 2012; Bhojani et al. 2008; Aparicio-Gallego et al. 2011].
ACE, angiotensin-converting enzyme; QoL, quality of life.
Everolimus-related toxicities and management strategies
Stomatitis, defined as inflammation of the mucous membranes of the mouth distinct from that seen with chemotherapy, may occur anywhere in the oral cavity, the inner surfaces of the lips, or the tongue and is associated with erythema, oedema, burning sensation, and occasional bleeding from the affected area [Porta et al. 2011; Wojtaszek, 2000]. It is most likely to occur during the first months of therapy and can usually be resolved through oral hygiene without treatment interruption [Novartis Pharmaceuticals Corporation, 2012; Ferte et al. 2011; de Oliveira et al. 2011; Boers-Doets et al. 2011].
Dermatological AEs and rash also occur frequently during everolimus treatment; most instances are grade 1 or 2. They generally resolve spontaneously, although topical or oral treatments may offer patient relief. Dose modifications of everolimus are required only for patients with more severe cases; for these patients, short-course oral steroids may also be used [Novartis Pharmaceuticals Corporation, 2012; Moldawer and Wood, 2010; Balagula et al. 2012].
The inherent immunosuppressive properties of everolimus may predispose patients to infection. Pre-existing infections should be resolved before everolimus therapy is initiated; appropriate antibiotic therapy should be initiated immediately if infection is diagnosed, and everolimus dose modifications should be made as necessary. Prophylactic therapy with daily sulfamethoxazole trimethoprim can also be considered in patients at high risk [Novartis Pharmaceuticals Corporation, 2012; Moldawer and Wood, 2010]. Noninfectious pneumonitis may be diagnosed in patients with nonspecific respiratory signs and symptoms, such as breathlessness or cough with associated hypoxemia. Infectious, neoplastic, and other causes should be excluded by appropriate investigations, including lung imaging, sputum and blood cultures, and lung function testing. Many early cases of noninfectious pneumonitis are asymptomatic; therefore, the nurse should be suspicious if a patient has vague breathing symptoms and should implement prompt detection and management. Patients with low-grade or minimal symptoms may continue everolimus therapy without dose alterations; however, if symptoms are moderate, dose reduction or interruption should be considered until symptoms improve. If symptoms of noninfectious pneumonitis are severe, everolimus therapy should be discontinued; steroids may be indicated until clinical symptoms resolve [Novartis Pharmaceuticals Corporation, 2012; Moldawer and Wood, 2010; White et al. 2010; Rodriguez-Pascual et al. 2010]. If steroids are used, glucose levels should be monitored closely because of the increased risk for hyperglycaemia [Clore and Thurby-Hay, 2009].
Additional AEs related to everolimus treatment include anaemia and hyperglycaemia, which should be managed according to the current standard of care/standard consensus guidelines [Novartis Pharmaceuticals Corporation, 2012; Jemal et al. 2010]. Patients may also develop hyperlipidaemia, which should generally resolve without intervention but may require antilipid drug therapy [Novartis Pharmaceuticals Cor-poration, 2012; Moldawer and Wood, 2010; Nathan et al. 2009]. Overall, most AEs observed during everolimus treatment are reversible, many without treatment interruption or modification [Novartis Pharmaceuticals Corporation, 2012].
Sunitinib-related toxicities and management strategies
HFS, characterized by palmar–plantar lesions in areas of friction or trauma (commonly in the hands and feet), may significantly affect a patient’s QoL and physical functioning and often leads to treatment discontinuation. HFS can be minimized by various skin care measures before initiation of sunitinib therapy. These include topical treatments and avoidance of friction, especially in the feet, which may provide some relief; however, severe cases necessitate dose alterations and interruptions [Grunwald et al. 2010; Cohen and Oudard, 2012; Ravaud, 2011; Di Lorenzo et al. 2011; Eisen et al. 2012]. The nurse should regularly evaluate patients with HFS to detect increased effects of toxicity and should follow up with appropriate, prompt treatment. Select management options include alcohol-free moisturizers, use of shock-absorbing shoe inserts and moisture-wicking socks, and steroid creams or skin adhesives [Kollmannsberger et al. 2011; Ravaud, 2011].
Hypertension, defined as consistent systolic blood pressure of at least 140 mmHg and diastolic blood pressure of at least 90 mmHg, is a common occurrence in patients treated with VEGFR-TKIs, a significant proportion of whom develop severe hypertension. Patients should be treated with antihypertensive therapy, and the sunitinib dose should be reduced if the condition is persistent [Grunwald et al. 2010; Cohen and Oudard, 2012; Eisen et al. 2012]. Cardiovascular AEs have been reported in 11–27% of patients treated with sunitinib [Pfizer Inc., 2011]. One patient enrolled in the phase III study died of grade 5 cardiac failure, which was attributed to the study drug [Raymond et al. 2011]. Cardiac damage in the form of cardiomyopathy is manageable, provided patients receive appropriate monitoring and treatment at the first indication of symptoms. Before initiation of sunitinib therapy, the cardiac risk/benefit ratio should be carefully considered, and special emphasis should be placed on regular assessment of left ventricular function. Any patients demonstrating symptoms of congestive heart failure should immediately discontinue sunitinib therapy [Pfizer Inc., 2011; Cohen and Oudard, 2012; Ravaud, 2011].
Fatigue is another AE commonly encountered by patients receiving sunitinib and covers a range of symptoms, including tiredness, exhaustion, and lack of strength, all of which have a high impact on QoL and daily functioning. Fatigue should be treated with supportive care according to guideline recommendations, and a reduction of sunitinib should be considered in patients with severely reduced QoL [Cohen and Oudard, 2012; Grunwald et al. 2010]. Among patients who develop neutropenia during sunitinib therapy, treatment can continue if the severity is less than grade 3, but for severe (grade 3–4) or recurrent neutropenia, treatment should be interrupted until toxicity decreases [Bhojani et al. 2008]. Additional AEs associated with sunitinib treatment include hormone problems (thyroid dysfunction, managed with monitoring or with thyroid hormone replacement in severe cases), gastrointestinal disturbances (managed by antidiarrhoeal medication and dietary modification, with sunitinib dose reductions or interruptions in severe cases), and depigmentation, usually affecting the hair (managed with supportive care) [Cohen and Oudard, 2012; Ravaud, 2011; Aparicio-Gallego et al. 2011; Bhojani et al. 2008; Grunwald et al. 2010; Schwandt et al. 2009]. Overall, several toxicities associated with sunitinib may require treatment interruption or discontinuation; this may affect tumour progression and patient outcomes and should be considered when a patient is evaluated for oral therapy.
Implications of treatment discontinuation
In vitro studies have suggested that even temporary discontinuation of sunitinib may enable tumour advancement through tumour revascularization and renewed proliferation [Ebos et al. 2009], although this has not been confirmed in the clinic. The phase III study of sunitinib in pNETs was discontinued early, possibly resulting in an overestimation of the true treatment effect [Raymond et al. 2011, 2012]. Data from the sunitinib trial were subsequently reanalyzed by the FDA, with calculations producing a median PFS of 10.2 months for sunitinib [Blumenthal et al. 2012] as opposed to 11.4 months, as originally published [Raymond et al. 2011]. In addition, the FDA analysis indicates that the overall survival benefit reported after 18% of events occurred was not maintained as the trial progressed and was nearly equal at the 43% event rate (40% in the sunitinib arm, 46% in the placebo arm; hazard ratio, 0.74; 95% confidence interval, 0.47–1.17) [Blumenthal et al. 2012]. Unfortunately, because of early termination, it was not possible to evaluate the effects of treatment discontinuation within the shortened time scale of the study and in a smaller population than planned.
Implications of disease progression
Despite recent advances in therapy, patients with pNETs have experienced disease progression while receiving everolimus or sunitinib. Thus, these drugs are not curative; underlying factors for lack of response continue to be explored. In a rat model of pancreatic cancer, use of everolimus twice weekly for 4.5 weeks (alone or in combination with 177Lu-DOTATATE, a somatostatin receptor blocker) resulted in occurrence of distant metastasis in 77% of patients. Combination therapy was less effective than treatment with 177Lu-DOTATATE alone [Pool et al. 2013]. To better understand the mechanisms of resistance and treatment failure for everolimus, types of progression events in the placebo and everolimus arms of the RADIANT-3 trial were compared [Yao et al. 2013]. Types of progression included new metastases, pre-existing lesion growth, and new metastases in combination with pre-existing lesion growth. When the everolimus and placebo arms were compared, the fractions of progression events were 21% versus 22%, 54% versus 49%, and 24% versus 27% for the three types of progression, respectively [Yao et al. 2013]. These results indicate that the metastatic phenotypes were similar in the two treatment arms of RADIANT-3, and they suggest that a more aggressive metastatic tumour phenotype does not emerge in patients who experience disease progression during everolimus treatment.
In vitro and clinical studies have also shown that the antiangiogenic activity of sunitinib causes upregulation of proangiogenic molecules and subsequent drug resistance [Teule and Casanovas, 2012]. A mouse model of NETs has shown initial antitumour response to sunitinib and then progression because of reactivation of the VEGF pathway, resulting in sunitinib resistance and subsequent primary tumour growth and metastases [Teule and Casanovas, 2012]. This suggests that the mechanism by which tumours develop resistance to sunitinib may make them a more aggressive phenotype [Teule and Casanovas, 2012]. It is unknown whether this phenotypic shift occurs in patients with pNETs treated with sunitinib who experience progressive disease.
Conclusion
Treatment options for patients with pNETs have significantly improved since the recent approvals of everolimus and sunitinib. Both agents provide clinical benefit for patients with NETs, but they are not interchangeable, and they have distinct AE profiles requiring careful vigilance. Thorough consideration of therapy choice and AE management requirements should be made before therapy is initiated to ensure optimal clinical benefit for each patient. Given that patient compliance with oral therapies may be an issue, oncology nurses should rigorously educate patients about their specific treatment, potential toxicities, and AE management. In conjunction with a supportive environment and a partnership in care (patient, family, nurse), nurses must be able to recognize symptoms of disease progression and must continuously monitor AEs to ensure the best possible treatment and patient wellbeing. The approval of two new oral therapies for the treatment of pNETs represents a paradigm shift away from infusion therapy. In addition, the implementation of multidisciplinary healthcare provider teams for patient management makes it vital for the oncology nurse to provide education to and support for patients with pNETs. Thus, the integration of the oncology nurse to the oral treatment setting is as important as it is to the delivery of intravenous treatment in the clinic. In conclusion, although conversion to oral therapies for pNETs is welcome for optimal patient care, it will require a significant practical shift in the treatment paradigm for patients and their healthcare providers.
Footnotes
Acknowledgements
Writing assistance in the form of copyediting, editorial assistance, and table and figure development was provided by Sally-Anne Mitchell, PhD and Jennifer M. Kulak, PhD of ApotheCom.
Funding
Financial support for writing assistance was provided by Novartis Pharmaceuticals Corporation (Basel, Switzerland).
Conflict of interest statement
MC declares no conflict of interest. NP has served on the NET advisory board for Novartis and has received speaking honoraria from Novartis and Pfizer (New York, USA).
