Abstract
In patients with renal cancer, brain metastasis is associated with poor survival and high morbidity. Poor life expectancy is often associated with widespread extracranial metastases. In such patients, a multidisciplinary approach is paramount. Brain metastases-specific therapies may include surgery, radiosurgery, conventional radiation and targeted therapies (TT) or a combination of these treatments. Some factors are important prognostically when choosing the best strategy: performance status, the number, size and location of brain metastases, the extension of systemic metastases and a well-controlled primary tumour. Failure of chemical therapy has always been attributed to an intact blood–brain barrier and acquired drug resistance by renal cancer cells. Recent studies have demonstrated objective responses with TT in a variety of cancer types, including renal cancer. In most cases, these agents have been used in combination and in conjunction with whole-brain radiation therapy and radiosurgery. Local control appears to be better with the combined method if the patient has a good performance status and may improve overall survival. This review summarizes current literature data on multidisciplinary approach in the management of renal brain metastasis with radiation, surgery and TT with an emphasis on potential better outcomes with a combination of current treatment methods.
Introduction
Brain metastases (BM) occur in approximately 3.5–17% of patients with renal cell carcinoma (RCC) [Bennani et al. 2014]. Shuch and colleagues reported that the median overall survival (OS) after diagnosis of BM was 10.7 months with 1-year, 2-year and 5-year OS rates of 48%, 30% and 12%, respectively [Shuch et al. 2008]. The development of BM appears to be related to active extracranial metastatic disease [Samlowski et al. 2008]. Bianchi and colleagues found that the rate of BM was correlated with the distribution of other metastatic sites: in the presence of abdominal metastases (gastrointestinal tract, liver, ovaries, genitourinary tract, adrenal glands and abdominal lymph nodes), the rate was 2%; it was 16% in patients with bone and thoracic metastases (respiratory system and thoracic lymph nodes) [Bianchi et al. 2012]. Moreover, an association between the loss of chromosome 9 and metachronous BM from clear cell RCC was described [Gutenberg et al. 2014]. RCC is considered a radioresistant tumour [Chang et al. 2005], but many studies showed similar rate of local control in BM from radioresistant and nonradioresistant cancer treated with radiotherapy [Yaeh et al. 2015]. In cases of solitary brain lesion or surgically accessible sites, surgical resection represents a valid therapeutic option. Meanwhile, in setting of multiple site of intracranical disease or in inaccessible cerebral locations and in presence of extracerebral metastases, brain radiotherapy in combination with targeted therapy shows an improved tumour local control [Hara et al. 2009; Wowra et al. 2002]. Patients with BM from RCC are frequently excluded from clinical trials, and therefore there are no validated treatment guidelines [Heng et al. 2014].
Method
We performed a systematic literature search in electronic databases: PubMed, Embase, Cochrane Library, OVID and Springer. The search was limited to English-language articles. The search terms included: RCC, BM, radiotherapy (whole brain radiotherapy [WBRT], stereotactic radiosurgery [SRS]), targeted therapy (sunitinib, sorafenib, pazopanib, bevacizumab, axitinib, cabozantinib, temsirolimus, everolimus), tyrosine kinase inhibitors (TKI) mammalian target of rapamycin inhibitors, mTOR inhibitors.
Selected studies evaluated the outcome and the safety of treatment of BM from RCC; the treatment included any approved systemic therapy and local therapy (radiotherapy: WBRT and SRS) and the efficacy of the combination of these methods.
Prognostic factors
The number of central nervous system (CNS) lesions was an independent factor of CNS recurrence with median CNS progression-free survival for solitary and multiple BM of 13 months and 4 months, respectively. The rapid CNS progression of patients with multiple BM appears to be related to ‘CNS-homing tumours’ [Sawrie et al. 2008; Shuch et al. 2008]. Volg and colleagues showed that extracranial metastatic disease determines the outcome of patients with BM and that the presence of BM alone was not a risk factor for poorer OS (p = 0.358). The independent risk factors for shorter survival were Eastern Cooperative Oncology Group performance status (ECOG PS) [hazard ratio (HR) 2.74, p = 0.001] and the time from primary tumour to metastases (HR 0.552,p = 0.034) [Vogl et al. 2010]. In patients undergoing metastasectomy, Naito and colleagues identified an association between four prognostic factors [BM (HR 3.73); >1.0 mg/dl C-reactive protein (HR 2.45); incomplete resection by metastasectomy (HR 2.15); highest histological grade in Japanese classification (HR 1.88)] and a worse prognosis [Naito et al. 2013]. According to Diagnosis-Specific Graded Prognostic Assessment, Karnofsky performance status (KPS) and the number of BM represented the two most significant prognostic factors [Sperduto et al. 2010, 2012]. Bennani and colleagues showed three prognostic factors of better survival in patients with BM, that is, the absence of intracranial hypertension (p = 0.01), deep metastases (p = 0.03) and systemic metastases (p = 0.049) [Bennani et al. 2014]. Histology was not associated with survival and local tumour control [Staehler et al. 2011].
Molecular mechanisms of radioresistance
Ionizing radiation induces radioresistance by activation of different pathways, such as the ras/mitogen-activated protein kinase, phosphatidylinositol 3′-kinase (PI3K) and signal transducer and activation of transcription3 (Stat3), promoting angiogenesis and endothelial cell proliferation via Raf and vascular endothelial growth factor receptor (VEGFR) pathways [Ibrahim et al. 2012; Jain et al. 2007; Kasibhatla et al. 2007; Zingg et al. 2004]. Niu and colleagues studied the correlation between vascular endothelial growth factor (VEGF) expression and Stat3 activity in different human cancer cell lines. In hypoxic conditions, the oncogenic tyrosine kinase v-Src, (upstream of Stat 3) induced VEGF upregulation, playing an important role in tumour angiogenesis [Niu et al. 2002; Xin et al. 2009].
In tumour cells, the presence of hypoxia and areas of necrosis improve the development of abnormal tumour vessels and unfavourable tumour microenvironment characterized by low pH, heterogeneous distribution of oxygen concentrations, nutrient delivery and high interstitial fluid pressure [Brown and Wilson, 2004; Carmeliet and Jain, 2011; Morris and Harari, 2014]. Many studies showed that chronic hypoxia resulted in genetic instability, due to suppression of DNA repair mechanisms [altered DNA double strand break repair; suppression of DNA mismatch repair (MMR), nucleotide excision repair (NER) and double-strand break (DSB) repair] resulting in radioresistance. Ionizing radiation induced reactive oxygen species, free radicals and DNA damage. The association between levels of partial pressures of oxygen in tumour cells seems to be related to the response to radiotherapy [Chan and Bristow, 2010; Chan et al. 2009; Um et al. 2004]. Hypofractionated radiotherapy seems to oppose DNA repair mechanisms of radioresistant clonogenes and to damage the tumour vasculature, leading to dynamic gradients of oxygenation in tumour cells [DiBiase et al. 1997].
Local treatment
Surgical resection represents the standard of care for the treatment of solitary BM in accessible locations and in absence of widespread disease. However, WBRT and, most recently, SRS have been shown to improve OS, local tumour control and to stabilize clinical symptoms (Table 1). Many studies have shown the safety and feasibility of such local treatments [Cannady et al. 2004; Chang et al. 2005; Hara et al. 2009; Sheehan et al. 2003; Siebels et al. 2002]. In a retrospective study from M.D. Anderson Cancer Center, the median OS in patients who underwent WBRT, was 4.4 months [Wrónski et al. 1997]. Repeated Gamma knife surgery (GKS) plays an important role in the treatment of multiple BM from RCC, especially in patients with controlled disease and no extra cerebral tumour. The actual local tumour control rate after the initial GKS was 95% and 4-year long-term survival was 91% in patients free from relapse of intracranial disease after repeated radiosurgery [Wowra et al. 2002]. Verma and colleagues showed that local control of BM was superior (p = 0.002 and p < 0.0001, respectively) with surgery and SRS therapy to observation without local treatment [Verma et al. 2012]. Ippen and colleagues conducted a retrospective study of 66 patients with BM. The OS was 13.9, 21.9 and 5.9 months for patients treated with SRS only, additional surgery and WBRT, respectively [Ippen et al. 2015]. SRS with a dose of 20 Gy appears to be associated with better local control, when compared with a standard dose of 16–18 Gy (12-months local control rates: 81% versus 50%, respectively; p < 0.001) [Rades et al. 2015].
Locoregional treatment effect on the outcome of patients with BM from RCC.

BM, brain metastasis; RCC, renal cell carcinoma; SRS, stereotactic radiosurgery; WBRT, whole brain radiotherapy; OS, overall survival; LC, local control, LTS, long-term survival.
SRS alone, surgery + SRS, WBRT + SRS.
Effects of targeted therapy on brain metastases
The supposed main obstacle to the treatment of BM is the blood–brain barrier (BBB)/blood–tumour barrier (BTB) which inhibits drug penetration into cancer cells. The BBB is characterized by tight junctions, which decrease pinocytosis across the endothelium [Pardridge, 2002; Preusser et al. 2012]. Preclinical studies showed the role of the brain microenvironment and endothelial membrane remodelling, particularly in mechanisms of anti-angiogenic radiosensitization [Truman et al. 2010; Wachsberger et al. 2003]. Moreover, BBB expresses the P-glycoprotein (PgP)/multidrug resistance (MDR) protein, a drug efflux pump which transports drugs from the brain, limiting the distribution in the CNS [Comerford et al. 2002; Dai et al. 2003; Eichler et al. 2012; Minocha et al. 2012]. Many studies showed the activity of TT on BM suggesting the diffusion of these drugs through the BBB. Carden and colleagues showed that anti-VEGF therapy was not associated with an increased frequency of intracerebral bleeding [Carden et al. 2008]. In the European Advanced RCC Sorafenib expanded-access study, sorafenib was administered to patients with previously treated advanced RCC. Subjects with cerebral disease were included in the protocol; the incidence and grade of adverse events in this subgroup was similar to the overall population [Beck et al. 2011; Stadler et al. 2010]. The PREDICT (Patient characteristics in RCC and Daily practICe Treatment with sorafenib) study showed the safety and efficacy of sorafenib in different subgroups of patients with advanced RCC [BM, non-clear cell histology, high Memorial Sloan-Kettering Cancer Center (MSKCC) risk score, ECOG PS ⩾2, no previous nephrectomy or no previous systemic therapy] [Jäger et al. 2015]. The median duration of therapy was 7.3 months for the total population and 7.0 months for patients with cerebral disease.
Gore and colleagues demonstrated the activity of sunitinib on BM: 9% of patients achieved objective response rate (ORR); 33% stable disease (SD) ⩾3 months and the clinical benefit (objective response + SD ⩾3 months) was 42%. In this subgroup of patients, the median PFS was 5.3 months [95% confidence interval (CI) 4.4–5.6] and the median OS was 8.2 months (95% CI 7.4–9.6) [Gore et al. 2009, 2011, 2015]. In the Italian cohort of patients with BM from RCC who received sunitinib in the Expanded Access Programme (EAP), the ORR was 4%, with 35% of cases with a SD of at least 3 months and a clinical benefit (objective response + partial response + SD ⩾3 months) of 39% [Sternberg et al. 2015]. Lim and colleagues showed the efficacy of sunitinib in six patients with brain lesions from clear cell RCC without prior local treatment (surgical resection or radiotherapy). Two subjects achieved a near-complete cerebral response [Lim et al. 2013]. Some studies showed the activity of pazopanib on BM [Jacobs et al. 2013; Roberto et al. 2015; Santoni et al. 2015]. Matrana and colleagues also showed the efficacy of pazopanib in a subgroup of patients with BM from RCC, recording stabilization of cerebral disease in 60% of cases and regression of brain lesions in 13% of patients [Matrana et al. 2013].
Recently, in the METEOR trial patients with BM adequately treated and stable were included for therapy with cabozantinib or everolimus; data on this subgroup of subjects is not yet available [Choueiri et al. 2015].
The RECORD1 trial and the REACT study (RAD001 EAP) demonstrated the safety of everolimus in patients with RCC previously treated with VEGFr-TKI therapy, also in a subgroup of subjects with BM [Grünwald et al. 2012; Motzer et al. 2010]. The ARCC (Global Advanced Renal Cell Carcinoma) trial showed the safety of treatment with temsirolimus on BM from RCC previously treated with surgical resection or radiotherapy and neurologically stable [Hudes et al. 2007].
Targeted therapy preventive effect
Verma and colleagues evaluated the impact of the treatment of sunitinib or sorafenib on the incidence of CNS metastases [Verma et al. 2011]. The median OS was 25 months versus 12.1 months (p < 0.0001) in the TT and non-TT groups, respectively. In a multivariate analysis, TKI therapy showed a lower risk of developing brain lesions (HR 0.39; 95% CI 0.21–0.73;p = 0.003) and better OS (HR 0.53; 95% CI 0.38–0.74; p < 0.001). Lung metastases at diagnosis were associated with an increased incidence of BM (HR 9.6; 95% CI 2.97–31.14; p < 0.001). In a retrospective study of 199 patients without BM, treated with or without TT (sunitinib, sorafenib, bevacizumab, temsirolimus or everolimus), Vanhuyse and colleagues found no significant impact of anti-angiogenic agents on the cumulative rate of BM (HR = 0.67; 95% CI 0.45–0.97; p = 0.18) [Vanhuyse et al. 2012]. Meanwhile, in the retrospective analysis of a subgroup of 139 patients enrolled in the TARGET trial [Escudier et al. 2007], an association between sorafenib therapy and a reduction of the incidence of BM was demonstrated (the overall incidence of BM was 3% versus 12% in sorafenib and placebo arm, respectively) [Massard et al. 2010a].
Targeted therapy in combination with radiotherapy
The role of TT in combination with radiation therapy is currently under investigation. In a retrospective study of the University of Rochester Medical Center, the concurrent use of TT (sunitinib, sorafenib, pazopanib or temsirolimus) with radiotherapy treatment (WBRT, STS or both) was not associated with a statistical increased OS [Bates et al. 2015]. Cochran and colleagues showed that TT (TKI, mTOR inhibitors, bevacizumab) improved OS and local control in patients with metastatic RCC (mRCC) treated with GKS when compared with other treatments (chemotherapy or immunotherapy). The median OS was 16.6 months and 7.2 months for patients receiving TT and other agents, respectively (p = 0.04). Local control at 1 year was 93.3% for the TT group versus 60% for subjects treated without TT. There was no statistical difference in neurological death between these cohorts of patients (21.1% and 30.3%, respectively; p = 0.47) [Cochran et al. 2012]. Simultaneous TT based on sunitinib or sorafenib and SRS for patients with BM from RCC has been shown to be safe and effective; the median OS reported was 11.1 months (p = 0.038) and the local control rate was 100% at 12 months and 96.6% at 24 months [Staehler et al. 2011]. Staehler and colleagues showed the safety and efficacy of the combination of high-dose hypofractionated therapy with simultaneous treatment with sunitinib in 22 patients with metastatic mRCC [Staehler et al. 2010]. In the retrospective analysis of 106 patients with mRCC and cerebral metastases treated with TT (sunitinib, sorafenib, bevacizumab and temsirolimus) and local treatment (WBRT, SRS and neurosurgery) conducted by Vickers and colleagues, KPS <80% at the start of therapy (HR 2.07; 95% CI 1.2–3.6), diagnosis to treatment time <12 months (HR 2.6; 95% CI 1.5–4.5) and more than four BM (HR 3.1; 95% CI 1.3–7.5) were associated with worse OS from the time of diagnosis of cerebral disease [Vickers et al. 2013]. In the retrospective series of 65 patients with RCC and BM treated with antiangiogenic agents or mTOR inhibitors and no concurrent local therapy (SRS, WBRT, surgery or both radiotherapy and surgery), Bastos and colleagues revealed that clear cell histology (p < 0.0001), solitary BM (p = 0.004) and favourable risk score according to the MSKCC model (p = 0.001) are favourable prognostic factors. In this study, neurological complications were recorded in five patients, including three patients with haemorrhagic BM and two patients with radiation necrosis [Bastos et al. 2014].
Tables 2 and 3 summarize results of studies discussed above.
Outcome of patients with BM from RCC treated with several TT.

BM, brain metastasis; RCC, renal cell carcinoma; NA, not available; OS, overall survival; PFS, progression-free survival; CR, complete remission; PR, partial remission; SD, stable disease; PD, progressive disease; TT, targeted therapy; Su, sunitinib, So, sorafenib; pazo, pazopanib; Beva, bevacizumab; Eve, everolimus; Tem, temsirolimus.
Toxicities related to a multimodality treatment of patients with BM from RCC.
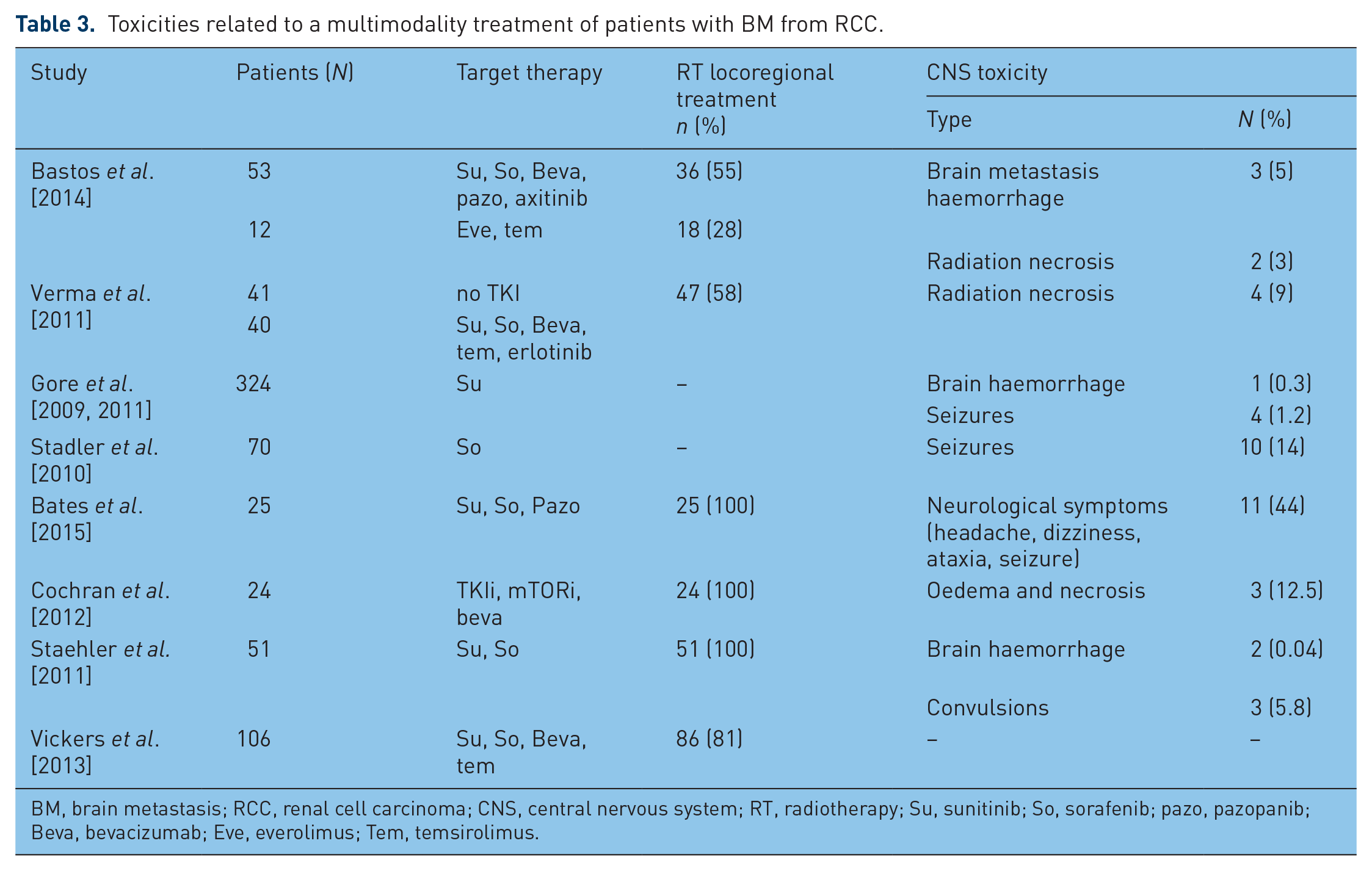
BM, brain metastasis; RCC, renal cell carcinoma; CNS, central nervous system; RT, radiotherapy; Su, sunitinib; So, sorafenib; pazo, pazopanib; Beva, bevacizumab; Eve, everolimus; Tem, temsirolimus.
Conclusion
We know that BM occur in approximately 3.5–17% of patients with RCC. Unless indicated by clinical signs or symptoms, the use of CT or RMN brain scan is not recommended [Escudier et al. 2014]. The prognosis for these patients is poor, with median survival typically measured in weeks [Shuch et al. 2008]. Clearly, KPS and the number of BM represented the two most significant prognostic factors. Multidisciplinary approach is very important in these patients. Definitive antitumour treatment may include radiation therapy, surgery and TT. When selecting the appropriate treatment, individual patient characteristics (KPS status, systemic cancer burden) and the number, size and location of BM are very important considerations.
Radiation therapy includes WBRT and SRS. For decades, WBRT has been advocated for patients with multiple lesions. WBRT is also used for patients with a low KPS or a low life expectancy. SRS is a more preferred treatment modality for radio-resistant lesions, as are those from RCC. SRS improves OS, local tumour control and stabilizes clinical symptomatology. SRS is also increasingly used as adjuvant therapy in patients who have undergone metastatic brain tumour resection. Surgery is considered standard of care for solitary metastases larger than 3 cm and in noneloquent brain areas. Meanwhile, in presence of patients with surgically inaccessible, multiple BM, or extracerebral disease, multimodality treatment may represent a recommended option. The multimodality treatment has showed to improve patients’ outcome and local control, without increasing the frequency of intracerebral bleeding [Carden et al. 2008] or neurological complications [Cochran et al. 2012; Staehler et al. 2011]. However, this analysis derived by retrospective studies, case series and case reports and so we do not know definitely whether or not this therapeutic combination results into an improved survival. Staehler and colleagues noted the safety and efficacy of the combination of high-dose hypofractionated therapy with simultaneous treatment with TT [Staehler et al. 2011]. In other studies, KPS <80% at the start of therapy, diagnosis to treatment time and more than four BM were limiting factors, in terms of worse OS to this approach. Moreover, TKI therapy seems to have a preventive effect on the risk of developing brain lesions [Bernard Escudier et al. 2007; Massard et al. 2010b; Verma et al. 2011].
The role of immunotherapy in patients with RCC is still under investigation. In CheckMate 025 trial, a randomized, open-label, phase III study that compared nivolumab with everolimus in patients with previously treated RCC, subjects with BM metastasis were excluded [Motzer et al. 2015] and then no data are available. However, a recent case report of a patient treated with pembrolizumab, an anti-programmed death receptor 1 (PD1)-blocking antibody, in combination with steroids, showed a regression of BM from RCC [Rothermundt et al. 2016].
Therefore, no conclusive recommendation can be produced and further studies are needed to elucidate the exact role of the combination therapies and the best drugs activity in treating BM. These questions would be answered better by prospective randomized trials.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
