Abstract

Introduction
Female stress urinary incontinence (SUI) remains a highly prevalent, yet undertreated pelvic floor disorder impacting millions of women globally. In the United States, it is estimated that approximately 45% of adult women complain of SUI symptoms, yet fewer than 40% are seeking care.1,2 Management strategies have largely remained constant over recent years, with only a few notable changes, as outlined in the American Urological Association (AUA)/Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction (SUFU) guidelines from 2017 to 2023. 3 Importantly, single-incision mid-urethral slings (SISs) are now considered a viable treatment option for the index SUI case, similarly to prior traditional retropubic and transobturator mid-urethral slings. Other key points addressed during the 2024 First International Functional and Reconstructive Urology Update (IFRUU) conference encompassed diagnostic strategies, shared decision-making when discussing treatment options, and the future of SUI treatment among evolving technologies. Discussion points included surgical selection, evolving reimbursement concerns, and long-term durability of outcomes including patient satisfaction. By addressing patient-centered concerns and tailoring care based on symptom priority, clinicians can better navigate treatment choices for improved female SUI outcomes.
Overview of conference content
This paper is based on a structured review and synthesis of lectures presented by expert faculty during a recent 2024 multidisciplinary conference: IFRUU. This summary focused on Female SUI. The reviewers evaluated videos, slide decks, and discussion summaries to extract clinically relevant information, expert perspectives, and consensus trends. Presentations included here were (Table 1):
List of female SUI presentations at IFRUU.
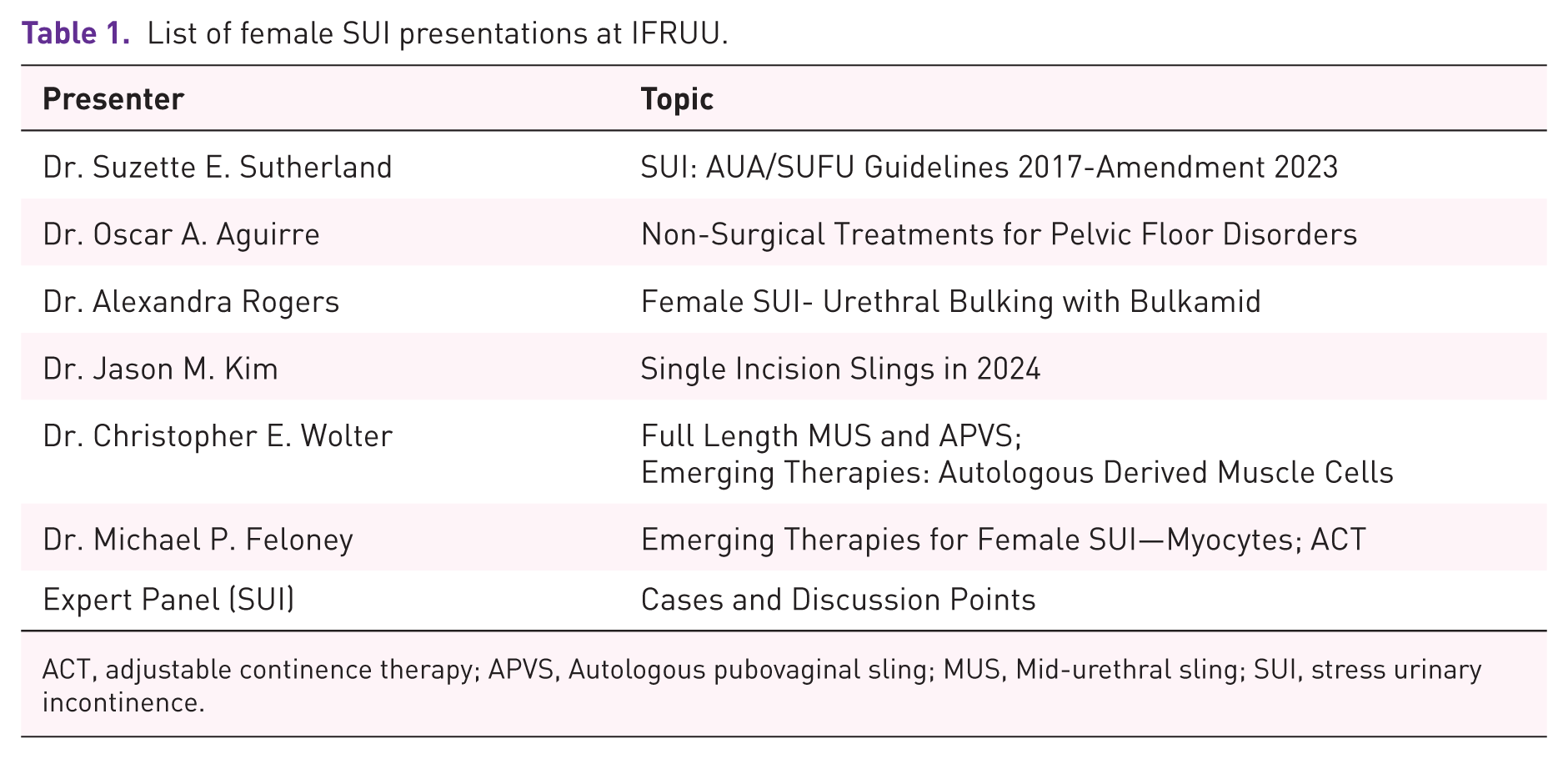
ACT, adjustable continence therapy; APVS, Autologous pubovaginal sling; MUS, Mid-urethral sling; SUI, stress urinary incontinence.
Results
Evaluation and diagnosis
Per AUA/SUFU guidelines, the initial SUI assessment is grounded in a five-point process: medical history, focused physical exam, visual cough stress test, post-void residual (PVR), and urinalysis. Urodynamic studies (UDS) and cystoscopy are reserved for unclear cases only. During the expert panel discussion at IFRUU, the use of UDS was debated. While all panelists agreed that UDS was not needed in cases of clear-cut SUI on history and physical exam, more complicated cases resulted in variable answers from different panelists. For example, in patients with mixed urinary incontinence, there were varying opinions about which to treat first, SUI versus Urge Urinary Incontinence (UUI), and the role and timing of UDS. With readily diagnosable SUI, yet refractory over active bladder (OAB)/UUI (failed OAB meds), some panelists felt proceeding with a sling to treat the SUI first was advisable, with plans to continue to work on the OAB/UUI thereafter; while other panelists felt more comfortable with identifying possible UDS parameters concerning the OAB/UUI prior to proceeding with any surgical intervention. The panel universally agreed that UDS was generally recommended in the setting of SUI if the etiology of incontinence is not clear on history and physical exam, if the SUI cannot be elicited on a visual cough stress test, if the PVR is significantly elevated, and/or in the setting of prior pelvic floor surgery or trauma.
Non-surgical interventions
Traditional conservative management for SUI includes pelvic floor physical therapy, biofeedback, pessaries, and observational management. Options exist for patients who prioritize treatments for vaginal dryness and sexual function. These include CO2 laser therapy, 4 platelet-rich plasma injections,5,6 and radiofrequency therapies; 7 albeit not all are FDA-approved. There are very limited quality data concerning efficacy and safety. Additionally, the AUA/SUFU guidelines for both Female SUI and Genitourinary Syndrome of Menopause do not support their use. Ultimately, more data are needed.
Bulking agents
Urethral bulking is a minimally invasive treatment option for SUI. It is particularly suitable for patients desiring an office-based procedure with quick recovery and/or unwilling to undergo a mesh-based procedure. 3 With the 2020 US introduction of Bulkamid, a more efficacious, durable, and safer bulking agent, the 2023 AUA/SUFU SUI guidelines revision confirmed Bulkamid as both a primary and/or salvage treatment option. This recommendation was made despite Bulkamid’s lower objective efficacy and lower long-term satisfaction rates compared to traditional mid-urethral slings. 3
Recent US data showing a 38% rise in urethral bulking procedures due to Bulkamid coincided with decreased mesh mid-urethral sling placements, owing to the 2008 and 2011 FDA notifications on the use of mesh for vaginal surgery.8,9 Bulkamid meets a critical need, particularly for those who are mesh-averse, want to avoid general anesthesia, and prefer an office-based procedure. With these factors in mind, a UK survey 8 revealed 64% of SUI women preferred the Bulkamid route over a surgical mesh sling, while accepting lower efficacy (cure) rates.
Surgical slings: MUS, SIS, and PVS
Mid-urethral slings (MUSs), including retropubic slings (RPSs) and trans-obturator (TOT) approaches, remain the gold standard treatment for SUI, with over 3 million procedures performed in the United States since their introduction in 1996. It remains the most widely studied anti-incontinence procedure to date, with consistently high cure rates (91% objective, 87% subjective) and very low surgical risk. 10 Albeit rare, concerns about trocar-related injuries and pain in the retropubic space or transobturator space have increased interest in the single-incision slings (SISs). 11
Now supported by robust comparative data, the SIS is included (without a disclaimer about “immature data”) alongside traditional RPSs and TOTs for the primary treatment of SUI; thus, states the most recent 2023 amendment to the AUA/SUFU SUI treatment guidelines. The guidelines are supported by long-term, comparative data and acknowledge that the SIS has demonstrated similar efficacy to RPSs and TOTs, with less mesh burden, enhanced mesh placement with secure anchoring, and fewer anatomical risks. 3 As always, proper surgical technique is key to avoiding injury to adjacent structures, and proper sling positioning and tensioning at the mid-urethra to achieve maximal efficacy (Figure 1).

Illustration of urethral sling options.
Autologous pubovaginal slings, though less commonly used today, remain effective (87–92% success) and appropriate for select patients: those desiring mesh-free options, in non-index cases, in post-mesh-removal cases, and/or as salvage procedures. 12 The surgical approach carries increased morbidity, as it involves an abdominal incision, the need for rectus fascia or fascia lata harvest, and a retropubic trocar passage to properly place the sling about the bladder neck.
Emerging therapies: Myocytes and ACT
Recent innovations in female SUI treatment have expanded beyond traditional sling-based procedures and pivoted toward biologic and implantable therapies, most notably autologous muscle-derived cells (myocytes) and adjustable continence therapy (ACT) periurethral balloons.
Autologous muscle-derived cells
Autologous muscle-derived cells represent a regenerative strategy targeting the malfunction of the sphincter closure mechanism rather than compressing the urethra. Myocytes are extracted directly from the patient, cultured in a lab, and then injected around the urethra. Current studies remain early and lack long-term follow-up at this stage beyond >24 months. While this therapy is still under investigation and not currently FDA-approved, there remains a growing interest in muscle-derived cell (MDC) therapy for female SUI, the current evidence base remains limited and highlights the need for clearer differentiation between reaffirmed practice and true innovation. Studies include a multinational dose-escalation trial and randomized controlled study using a standardized dose of 150 × 106 autologous cells harvested from the vastus lateralis under local anesthesia. 13 In the multicenter RCT, a 2:1 MDC to placebo design with one or two treatment sessions was used, but enrollment stopped early due to an unexpectedly high placebo response. 14 Post hoc analysis showed that a more rigorous follow-up criterion reduced this effect and suggested better outcomes in patients with prior SUI procedures, likely due to sphincter augmentation in a well-supported urethra. 14 These limitations highlight the need for ongoing trials to ensure full efficacy and safety prior to FDA approval and market use.
Adjustable continence therapy
ACT introduces an adjustable, mechanical intervention via dual balloon implants placed adjacent to the bladder neck, mirroring the male ProACT system. ACT provides urethral coaptation while offering postoperative and ongoing adjustability. Initial trials show dose-dependent success, with up to 82% of patients achieving >50% reduction in pad use, though study exclusion criteria like urethral hypermobility may limit its extent of use. 15 However, these results came from small cohorts. Outcomes from European studies of the ACT are promising, with 70–92% symptom improvement and pad reduction in 81% of patients. 16 Procedure time is under 45 min, performed with small bilateral incisions in the outpatient setting. Complication rates of 31%, while present, are generally manageable, with minor long-term morbidity. 17
Importantly, the current evidence base for ACT remains limited to early-phase clinical experience. Ongoing trials and conference-presented data continue to refine patient selection criteria and optimization of balloon volume. At present, ACT may be particularly suited for women seeking a mesh-free adjustable option with minimal morbidity (Figure 2 and Table 2).

Artistic rendering of ACT placement.
Summary of ACT and myocyte therapies.

ACT, adjustable continence therapy.
Shared decision-making and patient-centered care
The growing interest toward myocytes and ACT reflects a broader trend toward individualized and minimally invasive care for female SUI. These therapies may offer valuable alternatives for patients who may have been poor candidates for sling procedures and who have no remaining safe options. Integrating shared decision-making, particularly regarding durability, the need for retreatment, cost considerations, and the maturity of available data, is essential to ensure that innovation translates into meaningful improvement in patient outcomes.
Conclusion
As research and guidelines evolve, long-term outcomes will further refine treatment durability for female SUI. Clear counseling, shared decision-making, and flexible, patient-centered approaches remain key to improving quality of life and functional outcomes.
